Back to Journals » Pediatric Health, Medicine and Therapeutics » Volume 12
Prevalence and Predictors of Anemia Among Children Aged 6–23 Months in Dodota District, Southeast Ethiopia: A Community-Based Cross-Sectional Study
Authors Sorsa A , Habtamu A , Kaso M
Received 21 November 2020
Accepted for publication 10 February 2021
Published 6 April 2021 Volume 2021:12 Pages 177—187
DOI https://doi.org/10.2147/PHMT.S293261
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Roosy Aulakh
Abebe Sorsa, Ashenafi Habtamu, Muhammedawel Kaso
Arsi University College of Health Science, Asella, Ethiopia
Correspondence: Abebe Sorsa Email [email protected]
Background: Globally, anemia is a public health problem affecting children living in both developed and developing countries with bad consequences on children’s cognitive, social, and economic development.
Objective: To assess the prevalence and predictors of anemia among children aged 6– 23 months residing at Dodota district, Southeast Ethiopia.
Methods: A community-based cross-sectional study was conducted from January–July 2019, at Dodota district, in Southeast Ethiopia. Multistage, random, and systematic sampling techniques were employed to recruit households and study participants. Trained community health extension workers were involved in the data collection. Data were entered into Epi_info 7.2.2 for clean-up and exported to SPSS 21 for analysis. Frequency and proportion were used to describe nominal and ordinal variables. Mean with SD were used to describe continuous variables. Pearson correlation coefficient was used to assess correlation between numeric variables. Regressional analysis was used to assess factors predicting the occurrence of anemia. P< 0.05 with 95% CI was considered significant.
Results: A total of 917 children were included and the prevalence of anemia was found to be 407 (44.4%, 95% CI=41.1– 47.4). When stratified to age category, the prevalence of anemia was highest among the age group of 6– 12 months. Lack of ANC visits, lack of iron supplementation during pregnancy, untimely initiation of complementary feeding, and initiation of cow’s milk before 1 year of age were factors associated with anemia.
Conclusion: The prevalence of anemia was at the level of major public health problems. Antenatal and obstetric factors as well as child feeding practices were risk factors associated with anemia; and utilizing obstetric and childcare practices is highly recommended to mitigate this public health problem.
Keywords: anemia, prevalence, community based, children aged 6– 23 months
Background
'Anemia' refers to a condition in which the hemoglobin content of the blood is lower than normal. Globally, anemia is a public health problem affecting people in both developed and developing countries, with bad consequences to human health as well as social and economic development.1,2 Globally, about 47.4% of children under 5 years are suffering from anemia and, when it comes to developing countries, it affects 46–66% of children aged under 5 years.3,4 About 67.6% and 65.5% of children under 5 years in Africa and Southeast Asia are suffering from anemia, respectively.5 The highest overall prevalence of anemia in children aged under 5 years is recorded in the Western and Central African Region, as high as 75%, and reports from Ethiopia indicated about 60.9% of children under 2 years are suffering from anemia.6,7
Anemia during the first 2 years of life adversely affects cognitive performance, behavior, developmental milestones, and subsequent preschool and school performance of the child, as more than 90% of brain growth and most of development is complete by the age of 2 years.8 Anemia during the childhood period is strongly associated with poor health and physical development. It is also associated with mild-to-moderate mental retardation and poor motor development, leading to reduced academic achievement and work capacity, thereby reducing earning potential and damaging national economic growth in the future.3
On the other hand, anemia has the potential to affect the immune system, hence predisposing the child for additional morbidity from infectious diseases.4,5
Anemic infants had significantly lower z-scores of weight for age than non-anemic ones.6,9 Iron deficiency anemia (IDA) commonly develops after 6 months of age if complementary foods during the weaning period do not provide sufficient absorbable iron. Breast milk is relatively low in iron, although the iron in breast milk is much better absorbed than that in cows’ milk.3,5 Iron deficiency, in turn, is largely due to an inadequate dietary intake of bioavailable iron, inadequate dietary iron during periods of increased iron requirements in infancy, and increased blood loss due to hookworm infestation.9 Iron supplementation during pregnancy, initiation of iron supplementation in infants at 4–6 months of age, effective counseling on supplementation, subsequent compliance, support of breastfeeding, and effective training of healthcare personnel are effective strategies for the prevention of anemia in the community.6,10 Thus far, a number of studies have been conducted on the prevalence of anemia and associated factors indicating that the significance of the problem is at the level of major public health importance, particularly in developing countries, and most of the studies were addressing those under the age of 5 years. As the cognitive and physical impact of anemia is more critical during the first 2 years of life, it is of paramount importance to have extensive shreds of evidence of anemia and associated factors among this age group to mitigate this debilitating disease at the earliest possible stages.4,7,9 A study done in the North east of Ethiopia which was conducted at an institution, elucidated the sex of the child, age, history of diarrhea before 2 weeks, maternal educational status, dietary diversity, introduction of complementary foods, stunting, and household wealth quintile were significantly associated with the anemia. Firstly, very few studies were conducted to assess the prevalence of anemia and associated factors at the community level. Secondly, there was no community level study which assessed the correlation between anemia and child growth and developmental milestones. The objective of this study was to assess the prevalence of anemia and associated factors among children aged 6–23 months at Dodota district.
Materials and Methods
Study Design and Study Setting
The study was a community-based cross-sectional study which was conducted from January 1–July 31, 2019 at Dodota district, which is found about 100 km southeast of the capital city, Addis Ababa. The district is a low land area with a projected population number close to 40,000 and estimated households of 6,000. The district is clustered under 15 rural administrative kebeles. In each kebeles there are about 200–600 households. The district was selected because high numbers of children were admitted to the nearby hospital with the diagnosis of severe acute malnutrition and anemia. The government of Ethiopia has had a primary health care service delivery at community level for more than a decade by deploying community health extension workers (HEW). There are two health extension workers (HEW) for each kebele. The objective of this study was to assess the prevalence and predictors of anemia at the community level.
Study population: All children within the age group of 6–23 months residing at Dodota district.
Sample size: The prevalence of anemia among children in the age group between 6–23 months was 40–60% according to the EDHS report of 2011 and other study reports.3,9 Using a single proportion formula by taking an average prevalence of 50% with a 5% margin of error, design effect 2, and non-response rate of 15%, the final household sample size became 883.
Sampling technique: Mixed sampling, which included multistage to reach at the household level, systematic and simple random sampling techniques were employed to recruit the house-holds and study participants. The kebeles were selected using simple random technique after obtaining lists of all kebeles from the district administrative office, and seven kebeles were selected. From the seven selected kebeles, numbers of households to be included in the study were proportionally allocated. Using the health extension workers’ household registrar lists as a sampling frame, household study units were selected using systematic sampling technique (Figure 1). If two or more children in the age range of 6–23 months were living in the household units, all were included. Hence, during the study period, there were two eligible children in 17 households, giving the total number of children included to be 917.
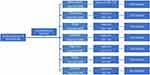 |
Figure 1 Flow chart showing sampling scheme and sampling frame of children living in Dodota district, Southeast Ethiopia (n=883), 2019. |
Those children who had anemia of moderate-to-severe level were given treatment with iron syrup supplementation.
Data Collection, Management, and Analysis
A standardized tool was prepared from previous studies to capture demographic and clinical data, Sociodemographic and clinical data were collected by face-to-face interviewing of the mother/guardian using a structured questionnaire, and the age of the child was determined by using an EPI immunization card when available and by extracting information from the mother. Developmental milestones were assessed using simple, easily noticeable and memorable childhood developmental achievement that the mother can remember. If the mother has older children, she was assisted to compare the child’s developmental milestones with older siblings and sometimes with peer groups in the neighbors.
Health extension workers (HEW) from respective kebeles were recruited to take a 2-day intensive practical training. The training majorly focused on how to collect demographic and clinical data consistently as per the questionnaire tool. Hands-on training was given on how to operate with the machine and how to perform hemoglobin testing as per the standard operation procedure. The hemoglobin level of the child was measured from capillary blood, and one drop of capillary blood was carefully collected from the middle finger of the child by finger prick. A strict aseptic technique was applied by cleaning the finger using 75% alcohol, and a separate lancet was used for each child. Automated HemoSmartGOLD from manufacture in Spain with model C€-0197 and serial number C-168-model was used to determine the hemoglobin concentration and the result was expressed in g/dL.
Regular quality control and supervision were made by laboratory technologist from Asella teaching and referral hospital on a weekly basis.
Data Entry and Analysis
The data were double entered to EPI-info 7.2.2 for clean-up and exported to SPSS version 21 for analysis. Frequency and proportion were used to describe nominal and categorical variables and presented using tables and charts. Normally distributed and continuous variables were expressed as mean±SD, and non-normally distributed variables were presented as interquartile range (IQR). Pearson correlation was used to evaluate the correlation between hemoglobin values and selected maternal and child numeric variables. Linear regression was used to assess the association between hemoglobin value and age at which the child achieved some developmental milestones. Bivariate regression was used to assess factors associated with anemia, and factors with a p-value of <0.2 were further analyzed using multivariable backward regression analysis. Odds ratio (OR) at 95% CI and p-value of <0.05 were taken as statistically significant. Binary logistic regression with corresponding COR and AOR were used to assess factors predicting the occurrence of anemia.
Operational Definition
Anemia: defined according to age-based WHO cut-off point criteria, and children with Hb level <11 g/L were considered anemic.5,7
Anemia severity classification: mild anemia (Hb values 10–10.9 g/dL), moderate anemia (Hb value 7–9.9 g/dL), and severe anemia (Hb value <7 g/dL).7
Anemia as Public Health Significance
As per WHO definition: anemia prevalence of over 40% was considered as a major public health problem, between 20 and 40% as a medium-level public health problem, and between 5 and 20% as a mild public health problem.7
Exclusive breast feeding is defined as feeding child-only breast milk.
Complementary feeding: Addition of complement feedings in addition to breast milk.
Ethics Consideration
The research proposal was initially submitted to Arsi University College of health science institutional review board (IRB) and ethical approval was secured to pursue the study. Letter of support was written to Dodota district administration office. After briefing about the purpose and benefit of the study, written consent for participation was taken from the mother/guardian of the child before commencing data collection.
All children who were found to be anemic were given appropriate treatment and for those who were not anemic, the families were counseled on preventive methods of anemia. The study was conducted in accordance with the Declaration of Helsinki. Confidentiality of information about the patients’ medical problem was secured with maximum effort and was not exposed to any other third party.
Results
A total of 883 households were surveyed, from which 917 children were included in the study. Males accounted for 463 (50.3%) and mean age was 15.2±5.74 months, and close to half of the study participants were in the age range of 18–23 months. Regarding the residence and occupation category, as the study was conducted at the community level of rural district, all are rural dwellers and the majorities were farmers by occupation. About 417 (45.7%) mothers/caregivers did not attend formal education, and about 835 (91%) mothers or caregivers were in the age range of 20–34 years (Table 1). Close to three-quarters of mothers had one or more ANC visit during the pregnancy of the child and about two-thirds received iron supplementation during pregnancy. Despite relatively better ANC visit utilization, a significant proportion of mothers gave birth at home, 371 (41.5%). Regarding child feeding practice, about 804 (87.7%) were on exclusive breast feeding (EBF) for 6 months, and the majority of children started on complementary feeding between the age of 6–8 months, where 526 (57.2%) started on cow’s milk as complementary feeding (Tables 1 and 2).
 |
Table 1 Socio-Demographic Characteristics of Children and Mothers Living in Dodota District, Southeast Ethiopia (Number of Children, N=917), 2019 |
 |
Table 2 Unadjusted Binary Logistic Regression: Factors Associated with Anemia Among Children Aged 6–23 Months, Dodota District, Southeast Ethiopia (N=917), 2019 |
The prevalence of anemia in this community-based study was found to be 407 (44.4%), CI=41.1–47.4, and the prevalence varies with the age group with the highest prevalence recorded among the age group between 9–12 months which was 54.6% (Figure 2), and the lowest prevalence reported among the age group of 18–24 months.
 |
Figure 2 Line graph depicting hemoglobin category at specific age in months, Dodota district, Southeast Ethiopia, 2019. |
Using the WHO severity classification of anemia, most cases of anemia were in the moderate range and the severe anemia category contributed the least (Figure 3).
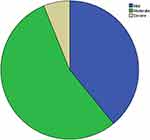 |
Figure 3 Pie chart demonstrating the severity classification of anemia in children in the age group 6–23 months, Dodota district, Southeast Ethiopia, 2019. |
Factors Associated with Anemia
Using linear logistic regression, maternal parity, maternal age, amount of cow’s milk started, age of patient at diagnosis of anemia, and duration of exclusive breast feeding (EBF) were evaluated. Hence, maternal parity is inversely associated with the child’s level of hemoglobin value; as the parity of the mother was increasing, the child hemoglobin level was decreasing. Similarly, the amount of cow’s milk consumed and duration of EBF were also inversely related with the child’s hemoglobin level. Conversely, as the age of the child was increasing, the value of hemoglobin was also increasing, but there was no correlation between the age of the mother and hemoglobin value of the child (Table 3).
 |
Table 3 Pearson Correlation Coefficient Assessing the Correlation Between Hemoglobin and Maternal and Child Numeric Variables, Dodota District, Southeast Ethiopia (N=917), 2019 |
Moreover, using bivariate regression, maternal educational background, ANC visit during pregnancy, iron supplementation, age at initiation of complementary feeding and history of diarrhea over the last 2 weeks were associated with anemia at a p-value of <0.2 (Table 2).
Children whose mothers did not have formal education were at higher risk of developing anemia [AOR=1.442 (1.096–1.897), p=0.009]. On the other hand, home delivery [AOR=1.339 (1.042–1.719), p=0.022], lack of ANC follow-up [AOR=4.35 (1.460–8.330), p=0.010], missing iron supplementation during pregnancy [AOR=6.45 (3.610–11.491), p<0.001], delayed initiation complementary feeding [AOR=3.838 (2.010–7.326), p<0.001], cow’s milk introduction as complementary feeding before 1 year of age [AOR=1.428 (1.064–1.918), p=0.018], and the presence of diarrhea in the preceding 2 weeks [AOR=3.265 (2.119–5.031), p<0.001] were factors identified to significantly predispose the child for developing anemia (Table 4).
 |
Table 4 Multivariable Regression: Factors Associated with Anemia Among Children Aged 6–23 Months, Dodota District, Southeast Ethiopia (N=917), 2019 |
Children who started on cow’s milk before 1 year had more than a 40% additional risk of developing anemia compared to those who did not starton cow’s milk. Likewise, children who had diarrheal disease 2 weeks before the time of evaluation had more than a 3-fold risk of having anemia (Table 4).
Hemoglobin Level Correlation with Selected Childhood Developmental Milestones
The developmental milestones we evaluated were gross motor development and language development. Accordingly, the mean age at which the child sat alone, crawled, walked unsupported, and started running were 6.84±1, 8.77±1.1, 12.1±1.7, and 18.25±3.7 months, respectively. On the other hand, the mean age for language milestones, achievements that produce first word and meaningful words were 11.5±3 and 20.2±3.5, respectively (Table 5).
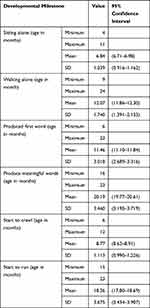 |
Table 5 Describing the Age at Which Selected Developmental Milestones Were Achieved Among Children Aged 6–23 Months, Dodota District, Southeast Ethiopia (N=917), 2019 |
Association Between Hemoglobin Value and Age at Selected Developmental Milestones Achieved
Using multivariate linear regression, we evaluated the correlation between hemoglobin values and selected developmental milestones. Child’s age at developmental achievement was inversely related with time to crawl, sit alone, and walk unsupported, but there was no correlation with the age of the child when starting running. With regards to language developmental milestones, there was no association between hemoglobin and selected language milestones (Table 6).
Discussion
The prevalence of anemia in our study was found to be 44.4%, which was at the level of major public health problem according to the WHO classification, and comparable with studies by Gebreweld et al and Petrucka et al which reported 48.2% and 41.1%, respectively.1,2 However, it is lower than the reports from studies conducted at the Northeast of Ethiopia by Wolde et al, EDHS-2016, and others which revealed prevalence ranging from 57–66.5%.3 –5 This difference could be related to the study design and study setting, as our study was community-based while the former studies were institution-based.3,4 For an obvious reason, an institution-based anemia prevalence study is likely to be higher than community-based studies as patients who are coming to the health institution do have some medical ailments in which disease-related anemia could directly or indirectly contribute for the higher prevalence. The other reason could be related to the technique of hemoglobin determination in which hemoglobin from capillary vessels is higher than the venous blood by 1.5–2 g/dL.
When the prevalence of anemia was stratified to different age groups, the highest prevalence of anemia was reported among the age group of 6–12 months while the lowest was reported among the age group of 18–24 months which is in parallel with the study report from other parts of Ethiopia.5,9 The justification for this could be associated with difficulties and struggles related with the introduction of complementary feeding during the age of 6–12 months, and most families do not know when and what to start as a complementary feeding.
Most patients with a diagnosis of anemia had moderate levels as per the WHO severity classification, accounting for 54.7% of all anemic cases, which is in line with studies conducted at Jimma town, Southwest Ethiopia which reported a moderate degree of anemia to cover 52% of all anemic patients. Severe anemia was diagnosed in about 6% of anemic cases, which was in contrast with the aforementioned Jimma study and other studies from other settings where there were very few or no cases of severe anemia reported. This could again be explained by the study setting, study design, and study population, as our study was conducted in a rural district while the Jimma study was carried out among urban dwellers.6,7,10,11
Children whose mothers did not have any ANC visit during pregnancy and who gave birth at home were at significant risk of developing anemia, which could be explained by the fact that mothers who had ANC visits and gave birth at institutions are likely to be counseled on the wellbeing of the pregnancy, maternal nutrition, get optimal obstetric care at delivery, and more importantly on well guided on child care and feeding practice after delivery.11–14 Iron supplementation during pregnancy effectively protective against acquiring anemia among children of 6–24 months, which was also demonstrated in a study findings from Turkey and other settings.11–13 Similarly, children born to mothers who did not have any form of education were at about 1.5-fold odds of developing anemia, which is in line with studies conducted in a Southwest and Northeast Ethiopian health demographic survey and other study reports which reported a strong association between lack of maternal education and the occurrence of anemia.13–16 This could be justified by the fact that educated mothers are more likely to adhere to good child feeding practices.
Children who had a history of diarrhea during the last 2 weeks were at a 3-fold risk of acquiring anemia compared to those who did not have a history of diarrhea, which is congruent with the study findings by Pasricha et al and Habte et al, and others which reported a 2–5-fold risk of anemia among children who had a history of diarrhea.13,17 The reasonings for this finding include children with diarrhea disease are likely to have decreased nutrition intake, diarrhea related malabsorption of micronutrients, and increased catabolic demand during the illness. Likewise, children who were initiated on cow’s milk as complementary feeding before the age of 1 year were having about 40% additional risk of developing anemia compared to the counterpart which has been enlightened in different literature as the iron absorption from cow’s milk is only 10% and most children starting cow’s milk before 1 year at high risk of milk induced colitis which could contribute for poor absorption and chronic occult/overt blood loss through stool.18 Timely and optimal initiation of complementary feeding is one of the critical factors in preventing the occurrence of anemia. This has been substantiated by our study findings which indicated a child who started complementary feeding at the recommended age (6–8 months) was at significantly lower risk of getting anemia, which has been supported by other previous reports.1,8,19–21
We also evaluated the correlation between hemoglobin and age of the child at which some selected developmental milestones were achieved. The gross motor developmental milestones achieved during the first 1.5 years were negatively correlated with hemoglobin level, ie, the lower the hemoglobin value the older the child will be to achieve the expected developmental milestone. However, there was no statistically significant correlation between language development and hemoglobin value. A systematic review and metanalysis by Sant-Rayn and colleagues indicated no association between hemoglobin level and age to achieve developmental milestones.11,12,22,23
Conclusion
The prevalence of anemia in our finding was within the range of major public health problems. ANC follow-up, iron supplementation during pregnancy, institution delivery, optimal breast feeding, and timely initiation of complementary feeding and delaying cow’s milk initiation after 1 year were recommended preventive strategies against acquiring anemia.
Limitation of the Study
The study was a cross-sectional survey where it was difficult to demonstrate a causal relation of associated factors. The hemoglobin determination was made using capillary blood, which could give relatively higher hemoglobin levels, hence could to some extent undermine the prevalence of anemia among this age group.
Abbreviations
ANC, antenatal care; AOR, adjusted odd ratio; CI, confidence interval; COR, crude odd ratio; dL, deciliter; EBF, exclusive breast feeding; EDHS, Ethiopia demographic and health survey; HEW, health extension worker; HH, household; g, gram; WHO, World Health Organization.
Data Sharing Statement
Almost all data are included in the paper and if the need arises it is possible to make all data publicly available.
Acknowledgment
We are very much grateful for health extension workers working at respective kebeles of Dodota district for their unreserved cooperation and commitment during data collection. Secondly, we would like to extend heartfelt gratitude to Arsi University for allowing us to carry out the research.
Funding
The authors did not receive any external funding and it was performed as part of the employment of the Arsi University College of Health Science.
Disclosure
The authors declare that they do not have any conflicts of interest for this work.
References
1. Gebreweld A, Ali N, Ali R, Fisha T. Prevalence of anemia and its associated factors among children under five years of age attending at Guguftu health center, South Wollo, Northeast Ethiopia. PLoS One. 2019;14(7):1–13.
2. Petrucka PM, Martin H, Kimanya ME, Mosha TCE. Prevalence and predictors of anemia among children under 5 years of age in Arusha Tanzania. Pediatr Health Med Ther. 2018;9:9–15. doi:10.2147/PHMT.S148515
3. Woldie H, Kebede Y, Tariku A. Factors associated with anemia among children aged 6 – 23 months attending growth monitoring at Tsitsika Health Center, Wag-Himra Zone, Northeast Ethiopia. J Nutr Metab. 2015;2015:1–9. doi:10.1155/2015/928632
4. Semedo RML, Santos MMAS, Baião MR, et al. Prevalence of Anaemia and Associated Factors among Children below Five Years of Age in Cape Verde, West Africa. J Health Popul Nutr. 2014;32(4).
5. Central statistics agency (CSA); Federal Democratic Republic of Ethiopia; ICF. Ethiopia Demographic and Health Survey, 2016.
6. WHO. Guideline: Updates on the management of severe acute malnutrition in infants and children. Geneva: WHO 2019.
7. Nambiema A, Robert A, Yaya I. Prevalence and risk factors of anemia in children aged from 6 to 59 months in Togo: analysis from Togo demographic and health survey data, 2013 – 2014. BMC Public Health. 2019;19:1–9. doi:10.1186/s12889-019-6547-1
8. Stoltzfus RJ, Dreyfuss ML. Guidelines for the Use of Iron Supplements to Prevent and Treat Iron Deficiency Anemia. International Nutritional Anemia Consultative Group; 2009.
9. Maduka DU, Anazoeze J, Uzoamka A. Parental perception of childhood anaemia and efficiency of instrument assisted pallor detection among mothers in Southeast Nigeria. Int J Pediatr. 2019. doi:10.1155/2019/7242607
10. UNICEF, WHO, UNU. Iron deficiency anemia: assessment, prevention, and control, a guide for programme manager. Geneva: WHO 2001.
11. Alelign T, Degarege A, Erko B. Prevalence and factors associated with undernutrition and anaemia among school children in Durbete Town, northwest Ethiopia. Arch Public Health. 2015;1–7. doi:10.1186/s13690-015-0084-x
12. Assefa S, Mossie A, Hamza L. Prevalence and severity of anemia among school children in Jimma Town, Southwest Ethiopia. BMC Hematol. 2014;14(3):1–9. doi:10.1186/2052-1839-14-3
13. Pasricha S, Hayes E, Kalumba K, Biggs B. Effect of daily iron supplementation on health in children aged 4 – 23 months: a systematic review and meta-analysis of. Lancet Glob Health. 2013;1(2):e77–86. doi:10.1016/S2214-109X(13)70046-9
14. Yalçın SS, Tezel B, Yurdakök K, et al. A community-based iron supplementation program, “ Iron- like Turkey ”, and the following prevalence of anemia among infants aged 12–23 months. Turk J Pediatr. 2013;55:16–28.
15. Parbey PA, Tarkang E, Manu E, et al. Risk factors of anaemia among children under five years in the Hohoe Municipality, Ghana. Anemia. 2019;2019:1–9. doi:10.1155/2019/2139717
16. Marly AC, Ke´zia KGS, Pascoal TM. Underlying factors associated with anemia in amazonian children: a population-based, cross-sectional study. PLoS One. 2012;7(5).
17. Habte D, Asrat K, Magafu MGMD, Ali IM, Benti T. Maternal risk factors for childhood anaemia in Maternal Risk Factors for Childhood Anaemia in Ethiopia. Afr J Reprod Health. 2013;17(3):110–118.
18. Mcdonagh MS, Blazina I, Dana T, Cantor A, Bougatsos C. Screening and routine supplementation for iron deficiency anemia: a systematic review. Pediatrics. 2015;135(4):723–734.
19. Pasricha S, Drakesmith H, Black J, Hipgrave D, Biggs B. Review Article Control of iron deficiency anemia in low- and middle-income countries. Blood. 2013;121(14):1–3. doi:10.1182/blood-2012-09-453522
20. Kliegman RM, St. Geme J. Nelson Textbook of Pediatrics.
21. Stoltzfus RJ. Iron interventions for women and children in low-income countries. J Nutr. 2011;15:1–7. doi:10.3945/jn.110.128793
22. Feleke BE, Derbie A, Zenebe Y, et al. Burden and determinant factors of anemia among elementary school children in northwest Ethiopia. Afr J Infect Dis. 2018;12(1):1–6. doi:10.21010/ajid.v12i1
23. Fiona K, Yeka A, Rhoda KW. Prevalence and factors associated with anaemia among children aged 6 to 59 months in Namutumba district, Uganda. BMC Pediatr. 2017;17(25). doi:10.1186/s12887-017-0782-3
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

