Back to Journals » Clinical Ophthalmology » Volume 10
Preservative-free bimatoprost 0.03%/timolol 0.5% fixed combination in patients with glaucoma in clinical practice
Authors Pfennigsdorf S, Eschstruth P, Häsemeyer S, Feuerhake C, Brief G, Grobeiu I, Shirlaw A
Received 10 February 2016
Accepted for publication 2 June 2016
Published 20 September 2016 Volume 2016:10 Pages 1837—1846
DOI https://doi.org/10.2147/OPTH.S106159
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Stefan Pfennigsdorf,1 Peter Eschstruth,2 Stefan Häsemeyer,3 Cord Feuerhake,4 Gerrett Brief,5 Ioana Grobeiu,6 Andrew Shirlaw6
1Polch Ophthalmology Practice, Polch, 2Ophthalmology Practice, Kiel, 3Ophthalmology Practice, Wiesloch, 4Ophthalmology Practice, Lehrte, 5Ophthalmology Practice, Dortmund, Germany; 6Allergan Holdings Ltd., Marlow, UK
Purpose: The aim of this study was to evaluate the efficacy and tolerability of, and compliance to, preservative-free (PF), fixed-combination (FC) bimatoprost 0.03%/timolol 0.5% in patients with primary open-angle glaucoma or ocular hypertension in a clinical practice setting.
Patients and methods: This open-label study observed patients switched to PF FC bimatoprost 0.03%/timolol 0.5% due to insufficient intraocular pressure (IOP) control on previous therapies. IOP was measured at baseline and at ~12 weeks. Tolerability and continuation of therapy were also assessed.
Results: A total of 1,553 patients were included in the study, and the per-protocol population comprised 1,391 patients. There were some minor deviations from protocol: some patients with no prior therapy and some who switched for reasons other than insufficient IOP control were included in the analysis. The mean IOP was reduced by 27.4%, from 22.2 mmHg to 16.1 mmHg. In subgroup analyses, the mean IOP was significantly reduced from baseline, irrespective of whether previous treatment was monotherapy or combination therapy, and preserved or PF therapy. Physicians mostly (88.1%) reported the IOP-lowering efficacy of PF FC bimatoprost 0.03%/timolol 0.5% to be as expected or better than expected. Switching to PF FC bimatoprost 0.03%/timolol 0.5% resulted in reductions from baseline in the number of patients reporting ocular symptoms. Adverse events were reported by 6.2% of patients, the most common being eye irritation (1.6%) and eye pruritus (1.0%). Physicians reported treatment compliance as better or unchanged compared with prior treatment in almost all patients (93.9%). Most patients were expected to continue PF FC bimatoprost 0.03%/timolol 0.5% after the end of the study.
Conclusion: Switching to PF FC bimatoprost 0.03%/timolol 0.5% was associated with significant IOP reductions from baseline over 12 weeks. Adverse events were uncommon, and compliance was high compared with previous therapy. PF FC bimatoprost 0.03%/timolol 0.5% may be a suitable treatment for patients with inadequately controlled IOP or who are sensitive to preservatives.
Keywords: bimatoprost 0.03%/timolol 0.5%, intraocular pressure, prostaglandin, preservative free, glaucoma, fixed combination
Introduction
Glaucoma is a leading cause of blindness, accounting for ~12% of cases globally and 18%–20% of all cases in Europe.1 Primary open-angle glaucoma (POAG) is the most common form of this disease, with prevalence ranging from 0.03% to 20%, as determined by an epidemiological analysis of studies spanning 42 years on glaucoma prevalence.2 Visual impairment due to glaucoma significantly reduces patients’ quality of life; individuals with glaucoma are three times more likely to report difficulty with activities of daily life (such as reading, walking down steps, and driving) compared with individuals without glaucoma.3,4
Glaucoma therapies frequently contain preservatives, such as SofZia®, Purite®, Polyquad®, and benzalkonium chloride, that can negatively affect the ocular surface, particularly in patients with preexisting ocular surface disease (OSD), such as dry eye disease and meibomian gland dysfunction.5 The risk of OSD increases with the treatment duration and the number of antiglaucoma medications taken; 30% of patients under long-term treatment for glaucoma or ocular hypertension (OHT) exhibited signs of mild OSD, while a further 21% exhibited signs of moderate-to-severe OSD.6,7 An observational study of 516 patients determined that 40% of patients with glaucoma experienced treatment modifications due to ocular surface intolerances and demonstrated that intolerance to antiglaucoma treatment can interfere with glaucoma management and outcome.7 Ocular discomfort, burning, stinging, and foreign body and dry eye sensations are significantly less frequent in patients using preservative-free (PF) glaucoma therapies compared with those containing preservatives.8 Factors indicating that a patient may benefit from PF therapy include a known allergy to the preservative, Sjögren’s syndrome, severe dry eye disease, atopic dermatitis and rosacea, a need for concomitant topical therapy, long life expectancy, blepharitis or meibomian gland dysfunction, intolerance to preservative, and a high risk of needing surgical intervention.5
Prostaglandin analog (PGA) monotherapy is a first-line treatment option for many patients with glaucoma owing to its intraocular pressure (IOP)-lowering efficacy, lack of systemic side effects, and once-daily dosing requirement.9 While monotherapy may be sufficient in some cases, many patients have inadequately controlled IOP on monotherapy and require combination treatment, which may be associated with increased adverse events (AEs) and poor compliance.10–12 Fixed-combination (FC) therapies are associated with better efficacy, compliance and tolerability, reduced risk of preservative-related ocular surface symptoms, elimination of washout associated with insufficient time between instillations, and improved quality of life compared with separate products.12–14 Considering the high quality of life and financial costs of an increasing population of patients with glaucoma, effective and tolerable glaucoma therapies are necessary to accommodate all patients.
In patients whose IOP was inadequately controlled with prior therapy, FC bimatoprost 0.03%/timolol 0.5% (GANFORT®; Allergan, Inc., Irvine, CA, USA) has been shown to significantly improve IOP in randomized controlled trials and observational studies.15,16 In an analysis of a large population of patients with glaucoma (n=5,556) from five combined studies, FC bimatoprost 0.03%/timolol 0.5% achieved consistent IOP lowering in both patients who were previously treated and those who were naïve to treatment.17
PF FC bimatoprost 0.03%/timolol 0.5% (GANFORT® UD; Allergan, Inc.) was developed to address the need for PF glaucoma treatment in patients who are intolerant to preservatives and who require additional IOP lowering above that provided by PGA monotherapy. In a randomized, double-blind clinical trial, PF FC bimatoprost 0.03%/timolol 0.5% was associated with reductions in IOP comparable with those achieved with the preserved formulation over 12 weeks of treatment.18
The objective of this study was to evaluate the efficacy and tolerability of PF FC bimatoprost 0.03%/timolol 0.5% in patients with POAG or OHT and compliance to therapy in a clinical practice setting.
Patients and methods
Participants
Patients were required to have a diagnosis of POAG or OHT and an IOP of ≥18 mmHg in at least one eye. The eye with higher IOP at baseline was deemed to be the study eye for the purpose of data analysis. If IOP at baseline was the same in both eyes, then the right eye was selected as the study eye for data analysis. Patients were required to have been previously prescribed topical IOP-lowering therapy but with insufficient IOP control prior to the start of the study. There were no formal exclusion criteria for this study, and patients were prescribed PF FC bimatoprost 0.03%/timolol 0.5% at the discretion of their treating physician, following their usual clinical practice. Treatment was guided by the PF FC bimatoprost 0.03%/timolol 0.5% Summary of Product Characteristics, which recommends that one drop is administered once daily to each affected eye.19 Investigators were guided by the contraindications, warnings, and guidance on pregnancy and lactation specified in the Summary of Product Characteristics for PF FC bimatoprost 0.03%/timolol 0.5%.
Study design
This was a prospective, observational, open-label, 12-week study (NCT01999348) in which data were collected during routine clinical practice. The study was conducted in accordance with the Good Clinical Practice Guidelines issued by the International Conference on Harmonization, and German requirements for conducting an observational study; the study was approved by the independent ethics committee (Landesärztekammer Baden-Württemberg, Stuttgart, Germany). All patients provided written informed consent.
At the first visit, patients were assessed for baseline characteristics, including IOP and ocular symptoms, prior to commencing PF FC bimatoprost 0.03%/timolol 0.5%. Visits 2 and 3 occurred ~2–4 weeks and 8–12 weeks after initiation of PF FC bimatoprost 0.03%/timolol 0.5%. Because of the observational nature of this study, there was no washout period between treatment with prior therapy and PF FC bimatoprost 0.03%/timolol 0.5%, and there were no formal discontinuation criteria (treatment was discontinued at the investigator’s discretion). The primary objective of the study was to assess the efficacy of PF FC bimatoprost 0.03%/timolol 0.5% as measured by change in IOP from baseline to final visit (Goldmann tonometer). Other study assessments included physician assessment of efficacy and tolerability, patient assessment of tolerability, discontinuations during the study period, continuation of therapy beyond the end of the study period, treatment compliance, and AEs. All medical care and diagnostic procedures were provided at the discretion of the study investigators according to their clinical judgment and local standards of care.
Statistical analysis
A target population of 2,000 patients was planned to be recruited from ~200 centers across Germany. Data entry and analysis used software from Syndeed Medidata GmbH (Konstanz, Germany) and the statistical software package SAS (Version 9.3; SAS Institute Inc., Cary, NC, USA). Medications were analyzed on the basis of original entries and international nonproprietary names. Data analyses were performed descriptively and included preparation of data listings and summary statistics (extreme values, range, mean and median values, and SD) or frequency distribution tables, as appropriate for each item. Data for demographic characteristics at baseline, efficacy and tolerability assessments, discontinuation and continuation of therapy, and compliance compared with prior therapy were provided for the per-protocol population (patients with complete data and IOP ≥18 mmHg in at least one eye at baseline). AE data were provided for the safety population (all patients recruited). Hyperemia was graded using the standard bulbar conjunctival hyperemia scale: 0, none; +0.5, trace; +1, mild; +2, moderate; +3, severe (Allergan bulbar hyperemia grading guide). Patients with complete data were included for statistical analysis of change in IOP. A paired t-test at the two-sided 5% level was used to compare IOP at baseline with IOP at the final visit. A subgroup analysis to compare the reduction of IOP after switching from a prior therapy to PF FC bimatoprost 0.03%/timolol 0.5% was also performed.
Results
The PF FC bimatoprost 0.03%/timolol 0.5% monitoring period took place between November 2013 and December 2014 at 264 centers in Germany. A total of 1,553 patients (safety population) were included in the study. The per-protocol population (patients with IOP ≥18 mmHg in at least one eye at baseline) comprised 1,391 patients. There were no formal exclusion criteria for this study, and patients were treated according to the local standard of care by their physician. Therefore, there were some deviations from protocol: 39 treatment-naïve patients were included in the study and analysis, as were patients with reasons for switching treatment other than insufficient IOP control on previous therapy.
Patient demographics
Baseline patient characteristics are presented in Table 1. The mean (SD) age was 66.2 years (±12.0). The majority of patients had a diagnosis of open-angle glaucoma (OAG) (89.6%), and the median time since first diagnosis of OAG or OHT was 2.9 years (range 1.0–6.8 years). Relevant prior and/or concomitant medical history was recorded in 28.8% of patients, with cardiovascular disease and metabolic and nutritional disorders being most frequent.
Prior therapy
Most patients (97.2%) were previously treated with an IOP-lowering therapy. Of those patients previously treated, the majority (73.7%) had been prescribed a preservative-containing IOP-lowering therapy and 16.2% had received prior PF therapy; data on this aspect were missing for 10.1%. Prior therapies reported in ≥5% of patients are shown in Figure 1. Latanoprost and timolol were the most frequent prior therapies. In total, 54.1% of patients taking therapy prior to the study were on a monotherapy regimen; however, 24.4% of patients were taking two therapies, and 21.5% of patients were taking three or more therapies (Figure 2). Insufficient IOP control on earlier IOP-lowering therapy was the main reason for switching to PF FC bimatoprost 0.03%/timolol 0.5% from a prior IOP-lowering therapy in 83.1% of patients (Table 2).
  | Figure 1 Prior intraocular pressure-lowering therapy reported in ≥5% of patients (per-protocol population). |
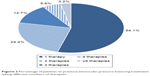  | Figure 2 Percentage of patients on previous intraocular pressure-lowering treatment taking different numbers of therapies. |
Study treatment
The median duration of treatment with PF FC bimatoprost 0.03%/timolol 0.5% was 12.9 weeks (range 9.7–16.7 weeks). Almost all patients (96.9%) started therapy with one drop per day in the study eye and were receiving one drop per day at the final visit (94.3%).
Effect on IOP
Data are presented for the study eye only. For patients with complete data at all visits, switching from a prior IOP-lowering therapy to PF FC bimatoprost 0.03%/timolol 0.5% decreased IOP from the baseline value by a mean of 6.1 mmHg (27.4%; Figure 3): mean ± SD IOP of 22.2±3.6 mmHg at baseline vs 16.1±2.9 mmHg at the final visit (n=1,321). The small number of treatment-naïve patients (n=34) included in the study experienced the largest reduction in baseline IOP at study end (11.2 mmHg, P<0.0001).
For all prior therapy subgroups analyzed, PF FC bimatoprost 0.03%/timolol 0.5% significantly decreased IOP from the baseline value (Figure 3). For patients on a prior IOP-lowering monotherapy, mean ± SD IOP was 22.2±3.3 mmHg at baseline and 16.0±2.8 mmHg at the final visit, a reduction of 6.2 mmHg (−27.9%; P<0.0001, n=791). Similar reductions in IOP from baseline to final visit were seen in patients who received prior PGAs (n=435; −6.0 mmHg), beta blockers (n=210; −6.4 mmHg), and carbonic anhydrase inhibitors (n=108; −6.3 mmHg) (all P<0.0001).
Patients who switched from combination therapies to PF FC bimatoprost 0.03%/timolol 0.5% also experienced significantly reduced mean IOP: from a baseline value of 21.7±3.1 mmHg to 16.2±2.7 mmHg at the final visit, a reduction of 5.4 mmHg (25.0%; P<0.0001, n=329). Reductions in baseline IOP were significant (P<0.0001) for all types of prior combination therapy, ranging from 4.7 mmHg (PGA/beta blocker, n=170) to 5.9 mmHg (carbonic anhydrase inhibitor/beta blocker, n=138).
There was no significant between-group difference for patients switching to PF FC bimatoprost 0.03%/timolol 0.5% from either a preserved or a nonpreserved prior therapy. For patients on a prior preserved IOP-lowering therapy, mean ± SD IOP was 22.1±3.2 mmHg at baseline and 16.1±2.8 mmHg at the final visit, a reduction of 6.0 mmHg (−27.3%; P<0.0001, n=918). For patients on a prior PF IOP-lowering therapy, mean ± SD IOP was 21.8±3.1 mmHg at baseline and 16.2±2.5 mmHg at the final visit, a reduction of 5.6 mmHg (−25.8%; P<0.0001, n=202). For all other prior IOP-lowering therapy subgroups analyzed, PF FC bimatoprost 0.03%/timolol 0.5% significantly decreased mean IOP compared with baseline (Figure 4), including in patients switching from PF bimatoprost (a reduction of 5.6 mmHg; P<0.0001, n=20). At the final study visit, the majority of physicians reported that the IOP-lowering efficacy of PF FC bimatoprost 0.03%/timolol 0.5% was better than or as expected (88.1%).
Effect on ocular symptoms
Switching to PF FC bimatoprost 0.03%/timolol 0.5% resulted in improvements from baseline in the number of patients reporting ocular symptoms including moderate or severe hyperemia (78.8% reduction), dry eye (26.8% reduction), and moderate/severe eye pain (65.8% reduction) (Figure 5). Of the 148 patients reporting eye pain at the final visit, the improvement in symptoms was considered clinically significant in eleven (7.4%) patients.
  | Figure 5 Effect of PF FC bimatoprost 0.03%/timolol 0.5% on ocular symptoms (per-protocol population). |
AEs and tolerability
Most patients (1,457/1,553, 93.8%) reported no AEs during the study period. AEs were recorded for 96 patients (6.2%), the most common being eye irritation (1.6%) and eye pruritus (1.0%) (Table 3). Physicians considered these to be possibly related to study medication in 55 patients and unrelated in 41 patients (not assessed in one patient; one patient reported >1 adverse event). No serious adverse drug reactions were reported. One fatal outcome was recorded in a patient aged 78 years. No further information regarding cause of death was available, but it was considered unrelated to the study medication. At the final study visit, the tolerability of PF FC bimatoprost 0.03%/timolol 0.5% was reported as being either “very good” or “good” by 89.8% of patients and 92.2% of physicians.
  | Table 3 Incidence of adverse eventsa reported in ≥0.5% of patients (safety population) |
Continuation and compliance with PF FC bimatoprost 0.03%/timolol 0.5%
Physicians rated patients’ compliance compared with previous therapy as better in 53.8%, equal in 40.1%, and worse in 2.5% of patients (data missing for 3.6% patients). A minority of patients (6.5%) discontinued PF FC bimatoprost 0.03%/timolol 0.5% therapy. For the 6.5% who discontinued treatment, the most common reasons for discontinuation were unacceptable ocular tolerability (43 patients, 34.1%) and insufficient IOP control (24 patients, 19.1%). Most patients (89.1%) were expected by their physicians to continue with PF FC bimatoprost 0.03%/timolol 0.5% therapy beyond the end of the study period.
Discussion
In this observational, open-label study, a PF formulation of FC bimatoprost 0.03%/timolol 0.5% was associated with an overall reduction in IOP of 6.1 mmHg (−27.4%) in patients with glaucoma after 12 weeks of treatment. This demonstrates that in clinical practice, PF FC bimatoprost 0.03%/timolol 0.5% can provide clinically meaningful improvements in IOP for patients who were not sufficiently responsive to prior therapy (83.1% of patients included in this study). Previous studies have demonstrated that reductions in IOP of 1 mmHg are associated with a 10%–19% decrease in the risk of glaucomatous progression.20,21
In this study, reductions from baseline IOP were seen in all prior therapy subgroups; patients on a prior monotherapy regimen experienced IOP reductions of 6.0–6.4 mmHg (27.3%–28.6%), while patients previously on combination therapy experienced reductions of 4.7–5.9 mmHg (22.2%–26.7%). Patients also reported substantial improvements in ocular symptoms with PF FC bimatoprost 0.03%/timolol 0.5%, including improvements in moderate/severe hyperemia, dry eye, and moderate/severe eye pain. PF FC bimatoprost 0.03%/timolol 0.5% was rarely associated with AEs (6.2% of patients), and no unanticipated adverse reactions were reported.
In many cases, patients require more than one therapy to control their IOP; however, this may affect efficacy, tolerability, frequency of dosing, and cost.9 It is, therefore, preferable to combine agents into an FC as this reduces the number of drops and preservative instilled each day, decreases cost, improves tolerability and compliance, and avoids a washout effect from instilling multiple agents in rapid succession.22 The efficacy and tolerability of FC therapies compared with latanoprost, travoprost, bimatoprost, and timolol monotherapies, and unfixed combinations have been analyzed in a meta-analysis of 18 studies involving 6,141 patients.14 Five studies included in the analysis demonstrated that FCs were less effective at reducing IOP than their respective unfixed combinations.14 When compared with timolol monotherapy, the latanoprost/timolol FC led to a greater reduction in IOP than the bimatoprost/timolol or travoprost/timolol FCs; however, each FC led to a statistically significant reduction in IOP (P<0.001). The analysis of FC compared with PGA monotherapy determined that the difference in IOP lowering was significantly greater between the FC and travoprost monotherapy (P<0.0001) than that between the FC and latanoprost or bimatoprost monotherapy.14 Tolerability (as measured by conjunctival hyperemia) was consistently better with FCs compared with monotherapy or unfixed combinations, and the relative risk was lower with bimatoprost compared with travoprost or latanoprost.14 Thus, this meta-analysis determined that FCs were more efficacious and led to a lower risk of hyperemia than their individual components.14
The significant reductions in IOP observed in this study concur with the finding that PF FC bimatoprost 0.03%/timolol 0.5% was associated with high levels of compliance in comparison with previous therapy. The majority of physicians (93.9%) rated patients’ compliance with PF FC bimatoprost 0.03%/timolol 0.5% as better than or equal to that with their prior therapy. This level of compliance, reported in comparison with previous therapy, is interesting given that patients were prescribed self-administered medications. Typically, patients only take approximately half of their prescribed doses, and few data are available to provide insight into how adherence can be improved to realize the full health benefits of medicines.23,24
The efficacy and tolerability findings reported here are consistent with those from previous studies assessing the preserved formulation of FC bimatoprost 0.03%/timolol 0.5%, indicating that the PF formulation provides a suitable option for patients who may be sensitive to preservatives. A large combined study reported significant IOP lowering over 12 weeks from baseline with FC bimatoprost 0.03%/timolol 0.5% in patients who were switched from prior therapies of PGAs, beta blockers, and FCs (IOP reduced by 24.5%, 25.9%, and 21.4%, respectively).17 The same study also reported excellent tolerability, with 9.7% of patients on FC bimatoprost 0.03%/timolol 0.5% reporting AEs. The findings from our observational study are also consistent with the efficacy and safety findings from a randomized controlled trial of PF FC bimatoprost 0.03%/timolol 0.5% conducted in patients with OAG or OHT, without signs of OSD. The principal conclusion was that PF FC bimatoprost 0.03%/timolol 0.5% demonstrated noninferiority and equivalence in IOP lowering compared with FC bimatoprost 0.03%/timolol 0.5%, with differences between the treatments in IOP lowering consistently favoring PF FC bimatoprost 0.03%/timolol 0.5%.18
This study also provided insight into the types of patients initiating PF FC bimatoprost 0.03%/timolol 0.5% in everyday clinical practice in Germany. Patients tended to have a diagnosis of OAG (89.6%) and to have been diagnosed with glaucoma/OHT for ~3 years (median 2.9 years). Patients were commonly prescribed PF bimatoprost 0.03%/timolol 0.5% because of insufficient IOP control (83.1%) or intolerability to prior IOP-lowering therapy (34.9%). Timolol and latanoprost were the most common prior therapies. PF FC bimatoprost 0.03%/timolol 0.5% was generally used in accordance with the prescribing information; almost all patients were prescribed the recommended dose of one drop per day.19
Taken together, the results of this study demonstrate significant IOP-lowering efficacy for PF FC bimatoprost 0.03%/timolol 0.5% in everyday clinical practice. PF FC bimatoprost 0.03%/timolol 0.5% represents an effective treatment option for patients whose IOP was insufficiently controlled with prior preservative-containing or PF therapies, as well as those sensitive to preservatives.
Study limitations
This open-label, observational study looked at the efficacy and AE profile of, and compliance to, PF FC bimatoprost 0.03%/timolol 0.5% therapy in routine clinical practice. It was not possible to compare the IOP-lowering efficacy and safety of PF FC bimatoprost 0.03%/timolol 0.5% directly with that of prior IOP-lowering therapies. A standardized procedure for recording data was not used; therefore, factors such as the time at which the measurement was taken and the method of recording free-text entries may affect the results. Data on pseudoexfoliation glaucoma and pigmentary glaucoma were not requested by the protocol; however, due to the observational nature of the study, some such data were provided. There was no washout period between any prior IOP-lowering therapy and beginning PF FC bimatoprost 0.03%/timolol 0.5% therapy, so any residual effects of prior therapies could have influenced the results. Where therapy was rated as better than or equal to the previous therapy, it is not known whether patients had good or bad compliance previously, as this was rated subjectively by physicians. Patients in clinical studies often demonstrate improved adherence, which may affect the results. The 12-week duration of the study did not allow for longer term evaluation of safety and efficacy. Patients were switched to PF FC bimatoprost 0.03%/timolol 0.5% therapy from a previous therapy, so the low frequency and severity of side effects reported in this study could have been due, in part, to the fact that they were not treatment naïve. Patients were selected and data recorded at the discretion of the treating physician.
Conclusion
Based on the results of the current study, PF FC bimatoprost 0.03%/timolol 0.5% was effective in lowering IOP and improving ocular symptoms in a large cohort of patients. AEs were uncommon, no unanticipated side effects were reported, and compliance was good when compared with previous treatment.
In clinical practice, patients with insufficiently controlled IOP by previous topical glaucoma therapy may benefit from treatment with PF FC bimatoprost 0.03%/timolol 0.5%. Further clinical studies with a longer study duration and increased patient numbers would allow further evaluation of the safety, efficacy, and compliance of PF FC bimatoprost 0.03%/timolol 0.5% in clinical practice.
Acknowledgments
This observational study was funded by Allergan Pharmaceuticals Ireland. The authors thank Newton Healthcare Communications for writing and editorial support, funded by Allergan Pharmaceuticals Ireland.
Disclosure
Pfennigsdorf and Häsemeyer are consultants for Allergan, Inc. Eschstruth is involved in projects with Allergan, Inc., Novartis, and AstraZeneca. Feuerhake and Brief have no disclosures. Grobeiu and Shirlaw are employees of Allergan, Inc. The authors report no other conflicts of interest in this work.
References
Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82(11):844–851. | ||
Cedrone C, Mancino R, Cerulli A, Cesareo M, Nucci C. Epidemiology of primary glaucoma: prevalence, incidence, and blinding effects. Prog Brain Res. 2008;173:3–14. | ||
Freeman EE, Muñoz B, West SK, Jampel HD, Friedman DS. Glaucoma and quality of life: the Salisbury Eye Evaluation. Ophthalmology. 2008;115(2):233–238. | ||
Hyman LG, Komaroff E, Heijl A, Bengtsson B, Leske MC; Early Manifest Glaucoma Trial Group. Treatment and vision-related quality of life in the early manifest glaucoma trial. Ophthalmology. 2005;112(9):1505–1513. | ||
Stalmans I, Megevand SG, Cordeiro F, et al. Preservative-free treatment in glaucoma: who, when, and why. Eur J Ophthalmol. 2013;23(4):518–525. | ||
Rossi GC, Pasinetti GM, Scudeller L, et al. Risk factors to develop ocular surface disease in treated glaucoma or ocular hypertension patients. Eur J Ophthalmol. 2013;23(3):296–302. | ||
Baudouin C, Renard JP, Nordmann JP, et al. Prevalence and risk factors for ocular surface disease among patients treated over the long term for glaucoma or ocular hypertension. Eur J Ophthalmol. 2013;23:47–54. | ||
Jaenen N, Baudouin C, Pouliquen P, et al. Ocular symptoms and signs with preserved and preservative-free glaucoma medications. Eur J Ophthalmol. 2007;17(3):341–349. | ||
European Glaucoma Society. Terminology and Guidelines for Glaucoma. 4th ed. Savona: PubliComm; 2014. | ||
Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701–713. | ||
Patel SC, Spaeth GL. Compliance in patients prescribed eyedrops for glaucoma. Ophthalmic Surg. 1995;26(3):233–236. | ||
Holló G, Hommer A, Antón López A, Ropo A. Efficacy, safety, and tolerability of preservative-free fixed combination of tafluprost 0.0015%/timolol 0.5% versus concomitant use of the ingredients. J Ocul Pharmacol Ther. 2014;30(6):468–475. | ||
Dunker S, Schmucker A, Maier H; Latanoprost/Timolol Fixed Combination Study Group. Tolerability, quality of life, and persistency of use in patients with glaucoma who are switched to the fixed combination of latanoprost and timolol. Adv Ther. 2007;24(2):376–386. | ||
Quaranta L, Biagioli E, Riva I, et al. Prostaglandin analogs and timolol-fixed versus unfixed combinations or monotherapy for open-angle glaucoma: a systematic review and meta-analysis. J Ocul Pharmacol Ther. 2013;29(4):382–389. | ||
Centofanti M, Oddone F, Gandolfi S, et al. Comparison of travoprost and bimatoprost plus timolol fixed combinations in open-angle glaucoma patients previously treated with latanoprost plus timolol fixed combination. Am J Ophthalmol. 2010;150(4):575–580. | ||
Brief G, Lammich T, Nagel E, et al. Fixed combination of bimatoprost and timolol in patients with primary open-angle glaucoma or ocular hypertension with inadequate IOP adjustment. Clin Ophthalmol. 2010;4:1125–1129. | ||
Pfennigsdorf S, de Jong L, Makk S, et al. A combined analysis of five observational studies evaluating the efficacy and tolerability of bimatoprost/timolol fixed combination in patients with primary open-angle glaucoma or ocular hypertension. Clin Ophthalmol. 2013;7:1219–1225. | ||
Goldberg I, Gil Pina R, Lanzagorta-Aresti A, Schiffman RM, Liu C, Bejanian M. Bimatoprost 0.03%/timolol 0.5% preservative-free ophthalmic solution versus bimatoprost 0.03%/timolol 0.5% ophthalmic solution (Ganfort) for glaucoma or ocular hypertension: a 12-week randomised controlled trial. Br J Ophthalmol. 2014;98(7):926–931. | ||
Allergan Ltd. Ganfort UD Preservative-Free: Summary of Product Characteristics. Parsippany, NJ: Allergan Ltd.; 2013. | ||
Chauhan BC, Mikelberg FS, Balaszi AG, et al. Canadian glaucoma study: risk factors for the progression of open-angle glaucoma. Arch Ophthalmol. 2008;126(8):1030–1036. | ||
Heijl A, Leske MC, Bengtsson B, et al. Reduction of intraocular pressure and glaucoma progression: results from the early manifest glaucoma trial. Arch Ophthalmol. 2002;120(10):1268–1279. | ||
Khouri AS, Realini T, Fechtner RD. Use of fixed-dose combination drugs for the treatment of glaucoma. Drugs Aging. 2007;24(12):1007–1016. | ||
Alhewiti A. Adherence to long-term therapies and beliefs about medications. Int J Family Med. 2014;2014:479596. | ||
Nieuwlaat R, Wilczynski N, Navarro T, et al. Interventions for enhancing medication adherence. Cochrane Database Syst Rev. 2014;11:CD000011. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




