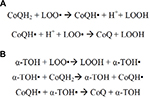Back to Journals » Nutrition and Dietary Supplements » Volume 10
Potential role of coenzyme Q10 in health and disease conditions
Authors Rodick TC, Seibels DR , Babu JR, Huggins KW, Ren G, Mathews ST
Received 23 September 2017
Accepted for publication 30 November 2017
Published 14 February 2018 Volume 2018:10 Pages 1—11
DOI https://doi.org/10.2147/NDS.S112119
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chandrika J Piyathilake
Taylor C Rodick,1 Donna R Seibels,2 Jeganathan Ramesh Babu,1 Kevin W Huggins,1 Guang Ren,3 Suresh T Mathews2
1Department of Nutrition, Dietetics, & Hospitality Management, Auburn University, Auburn, 2Department of Nutrition and Dietetics, Samford University, 3Medicine-Endocrinology, Diabetes & Metabolism, University of Alabama at Birmingham, Birmingham, AL, USA
Abstract: Coenzyme Q10 (CoQ10), an endogenously produced compound, is found in all human cells. Within the mitochondria, it plays a substantial role in energy production by acting as a mobile electron carrier in the electron transport chain. Outside the mitochondria, it acts as an excellent antioxidant by sequestering free radicals and working synergistically with other antioxidants, including vitamin E. Dietary contribution is limited, making endogenous production the primary source for optimal function. Now widely available as an over-the-counter supplement, CoQ10 has gained attention for its possible therapeutic use in minimizing the outcomes of certain metabolic diseases, notably cardiovascular disease, diabetes, neurodegenerative disease, and cancer. Research has shown positive results in subjects supplemented with CoQ10, especially in relation to upregulating antioxidant capability. Emerging research suggests beneficial effects of CoQ10 supplementation in individuals on statin medications. CoQ10 supplementation in individuals participating in strenuous exercise seems to exert some beneficial effects, although the data are conflicting with other types of physical activity. This broad review of current CoQ10 literature, while outlining its physiological/functional significance in health and disease conditions, also offers a dietitian’s perspective on its potential use as a supplement in the promotion of health and management of disease conditions.
Keywords: coenzyme Q, antioxidant, oxidative stress, dietary supplement, statin
Introduction
Coenzyme Q10 (CoQ10), also known as ubiquinone or ubidecarenone, is a vitamin-like nutrient and lipid-soluble compound. As its name implies, it is ubiquitous and present in all human cells. It is primarily located in the mitochondria and also found in cell membranes and lipoproteins. The primary function of CoQ10 is its role in cellular energy production, where, along the inner mitochondrial membrane, the electron transport chain (ETC) uses CoQ10 as a component in oxidative phosphorylation converting products of metabolism (carbohydrates, fats, and proteins) into energy as ATP.1 Ubiquinol (CoQH2), the fully reduced form of CoQ10, has the unique ability to act as a potent fat-soluble antioxidant free radical scavenger by affecting the initiation and propagation of reactive oxygen species (ROS), which cause oxidative stress leading to detrimental effects in damaging lipids, proteins, DNA, and the overall functional status of the mitochondria.2–4 CoQ10 levels decrease with aging, and this loss of antioxidant power coupled with increased ROS production can result in an age-related oxidative stress that can influence the development of other metabolic conditions.3 It has been speculated that CoQ10 supplementation may be effective as an antioxidant in treating certain pathological conditions.5 The purpose of this review was to provide an understanding of CoQ10’s biological functions in relation to its potential use as a supplement for health and in the management of chronic diseases.
Historical significance
CoQ10 was first isolated and characterized in 1955 by a research group headed by Professor RA Morton at Liverpool University, Liverpool, UK.6 This same group later went on to fully elucidate the chemical structure of CoQ10.7 In 1958, Karl Folkers (Merck Sharp & Dohme Inc., Kenilworth, NJ, USA) and Otto Isler (Hoffman-La Roche Inc., Little Falls, NJ, USA) developed the first process for synthesizing CoQ10.8 Two years later, Crane et al raised the possibility that CoQ10 may function within the mitochondrial electron transport chain (ETC).9 In the following years, CoQ10 research gained much attention when it was found to be deficient in patients suffering from cardiovascular conditions.10 In 1975, Dr Peter Mitchell developed the theory that ATP synthesis was driven by an electrochemical gradient across the inner mitochondrial membrane. This characterization of biological energy transfer, in which CoQ10 was necessary, won him the Nobel Prize in 1978.11
Structure, synthesis, and absorption
CoQ10 is a naturally occurring benzoquinone with a 10-unit isoprenyl tail. Although structurally similar to vitamin K, CoQ10 cannot be labeled a vitamin due to the body’s ability to synthesize it de novo.12 It has a molecular weight of 865 g/mol and a melting point of 49°C and exhibits limited solubility in fats and oils. Upon exposure to light and temperature (>55°C), CoQ10 becomes progressively unstable, and its yellow gold color darkens to a deep gold hue. A solidified state is more stable, especially in terms of photo degradation, than a liquefied state. Furthermore, the addition of other antioxidants, such as vitamins E and C, will enhance the stability of CoQ10. Kommuru et al showed that combinations of ascorbic acid and EDTA, both powerful antioxidants, with CoQ10 resulted in a more stable formulation when exposed to light and heat.13
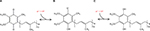
CoQ10 exists in both the oxidized (ubiquinone) and reduced (ubiquinol) forms. The semiquinone intermediate radical, CoQH., plays a role in antioxidant activity (Figure 1). Both ubiquinone and ubiquinol are lipid soluble due to the presence of the 10-unit isoprene tail. They act as a redox pair where the conversion of one form to the other can be readily achieved depending on when and where their functions are needed in the body. For example, tissues that involve high aerobic activity contain more of the oxidized form (ubiquinone) than the reduced form.12 In blood circulation, ~95% of CoQ10 is present in the ubiquinol form. The ratio between ubiquinone and ubiquinol in the blood has shown to be a measure of oxidative stress. Factors such as aging and certain disease conditions have shown to minimize ubiquinol levels in the body, possibly due to increased oxidative and nitrosative stress and also because the body loses the ability to reduce ubiquinone, thus resulting in a lower amount of total CoQ10.11
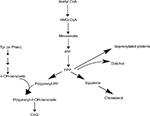
CoQ10 is ubiquitous in human tissues with the highest levels in the heart, liver, and kidney, participating in aerobic cellular respiration generating ATP. The synthesis of endogenous CoQ10 is highly complex, involving multiple compounds including B-vitamins and vitamin C. The quinone structure is derived from tyrosine, the methyl groups are supplied by methionine via S-adenosylmethonine, and the isoprenoid side chain originates from the mevalonate pathway (Figure 2). This pathway links CoQ10 production to the development of cholesterol. Thus, the deficiency of any of these compounds can affect the endogenous production of CoQ10.12
Multiple ways to commercially produce CoQ10 are available. There are two isomeric forms of CoQ10, which determine whether a supplement is a natural or synthetic version of the compound (Figure 3). Natural CoQ10 is an all-trans isomer, while the synthetic compound is a mixture of both the trans- and cis-isomers.14 Yeast or bacterial fermentation remains the most widely used production method due to its specificity toward the natural all-trans isomer. The advantage of an all-trans isomeric form is that it is structurally identical to CoQ10 that is endogenously made in the human body, making it biologically active.15 Alternatively, there is a lower-cost semichemical synthesis that uses a tobacco by-product, solanesol, which contributes tyrosine to the ring structure and the phytyl side chain. This production method contains cis- and trans-isomers that give the isoprenoid tail a bent shape rather than a straight shape. Because the cis-form is not produced in the body and has a different structure, the efficacy and safety of supplementing this form of CoQ10 are yet to be fully understood compared with the all trans-isomeric supplements.
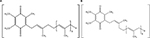  | Figure 3 Isomeric forms of CoQ10. (A) Natural all-trans form, (B) synthetic (-cis and -trans) form. Abbreviation: CoQ10, coenzyme Q10. |
As a lipophilic substance, CoQ10 absorption is enhanced by the presence of lipids, similar to vitamin E.12 Secretions from the pancreas and bile initiate the emulsification process and micelle formation that enhances CoQ10 absorption. Due to its molecular weight and hydrophobic nature, CoQ10 must be dissolved to single molecules in order to be facilitated by lipid carriers in a passive diffusion process along the small intestine.16 There is no specific location within the small intestine where absorption takes place. Increased consumption leads to a lower absorption rate of CoQ10, as roughly 60% of oral dosage forms are excreted in the feces.12 Plasma CoQ10 concentrations increase with increasing doses of CoQ10 and plateau at 2,400 mg, with a decreased efficiency of absorption at higher dosages.17 Either during absorption or after absorption, CoQ10 is reduced to ubiquinol and incorporated into chylomicrons and transported to the liver.16 Eventually, these are packaged into lipoprotein particles and released into circulation.12 Plasma CoQ10 is mainly packaged into very low-density lipoprotein (LDL)/LDL particles, with a small amount incorporated into high-density lipoprotein (HDL) particles. Among its many functions, it is this transport mechanism of CoQ10, along with α-tocopherol, that protects lipoproteins from lipid peroxidation.16
Food sources, supplement, and assessment of status
Widely distributed in nature, CoQ10 is found in both plants and animals. However, animal products tend to have the greatest amount of CoQ10 compared with other sources. Tissues with the highest energy demands typically contain high amounts of CoQ10, including the heart, liver, and kidney.18,19 Nonanimal products also contain fair amounts of CoQ10, the most being in broccoli, spinach, soybean/canola/palm oils, nuts, and legumes (Table 1).20 Contribution from dietary sources is limited, and in the absence of a supplement, endogenous production is left as the major source of CoQ10 in the body for regulation and maintenance. Levels obtained from the diet are estimated to be only 3–5 mg per day (~1%), while the total body pool is 0.5–1.5 g with a half-life varying from 35 to 100 hours, depending on the activity in a given body tissue.2 CoQ10 supplements were initially based on ubiquinone powder in the form of tablets, capsules, and oil-based softgels. The bioavailability of these supplements tended to be poor due to their hygroscopic nature.21 However, newer, ubiquinol-based formulations with increased stability and high bioavailability, including Q-Gel, Q-Nol, and Kaneka QH, are available commercially.21,22 Supplemental dosages in different strengths generally range from 15 to 100 mg. Larger doses for patients are well recognized under pathological conditions such as heart disease, neurodegenerative disorders, cancer, and diabetes.16 CoQ10 supplementation appears to be safe, found in both human and animal studies, with minimal adverse effects. The most common symptoms range from abdominal discomfort to slight nausea. Dosages in the 600–1,200 mg range, seen in Parkinson’s disease (PD) and Huntingdon’s disease trials, have shown no adverse effects. In a pilot study conducted by Shults et al, doses at 3,000 mg were tolerated in PD patients, and any adverse events were labeled unrelated to CoQ10 supplementation.23
  | Table 1 Comparison of dietary food sources with contributive values for CoQ10 per single serving17,18 Abbreviation: CoQ10, coenzyme Q10. |
CoQ10 status is assayed by high-performance liquid chromatography–ultraviolet detection, postextraction from the tissue or plasma.24 Electrochemical detection, which measures both ubiquinone and ubiquinol in the same sample, can also be used.1 However, pathologically deficient levels of CoQ10 present in specific tissues, such as the heart and muscle, may not be detected in the plasma at a given time. A skeletal muscle biopsy is the preferred tissue to determine the endogenous CoQ10 status of patients.25
Role in health
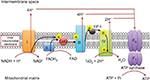
Due to its location in the mitochondria, the predominant function of CoQ10 is in energy production. CoQ10 is positioned between the flavoprotein complexes I, II, and III, where it acts as a mobile electron carrier.26 Using its redox capabilities, CoQ10 shuttles electrons from Complex I (reduced nicotinamide adenine dinucleotide [NADH]–ubiquinone reductase) and Complex II (succinate –ubiquinone reductase) to Complex III (ubiquinol cytochrome c reductase) (Figure 4). Because human life cannot be sustained without this process, the need for ubiquinone is high. Subsequently, ubiquinol is rapidly converted into oxidized ubiquinone, creating a cycle for the generation of ATP.1
In the mid-1970s, Peter Mitchell developed a theory that the redox ability of CoQ10 may play an additional role than electron transport. He found that protonation inside the mitochondria during reduction and deprotonation outside the membrane during oxidation built an electrical gradient between the membranes. Ultimately, this results in a driving force for adenosine diphosphate to be converted to ATP.11 Furthermore, uncoupling proteins (UCPs) are situated along the inner mitochondrial membrane that pulls protons from the outside to the inside of the membrane. Mitchell’s theory predicted that any proton leak not “coupled” with ATP synthesis would provoke uncoupling of respiration and thermogenesis.2,27 As the production of ROS in mitochondria is caused by respiratory activity and, in some instances, semi-ubiquinone, a decrease in the mitochondrial proton gradient caused by UCP activity could lower this potential. In the presence of oxidized CoQ10, Echtay et al found that fatty acids could deliver protons to UCP in the inner membrane more efficiently, which then get translocated into the mitochondrial matrix.28
In the reduced form as ubiquinol, CoQ10 has the ability to act as a potent fat-soluble antioxidant outside the mitochondrial membrane. Not only can it recycle and regenerate other antioxidants such as vitamins E and C, but it can also uniquely affect the initiation and propagation of ROS.2 Mitochondria are major producers of O2− and H2O2 as oxygen consumption is heavily involved in the production of ATP. Depending on the state of respiration, the leakage of electrons coupled with free radical formation can induce oxidative stress in DNA, lipids, and proteins.2 Lipid peroxidation, as the most studied and reviewed, involves the loss of hydrogen from a polyunsaturated fatty acid, resulting in a peroxyl radical. Reduced CoQH2 (ubiquinol) loosely holds electrons and acts to eliminate lipid peroxyl radicals by either directly producing semiquinone radical (CoQH.) or scavenging other peroxyl radicals (Figure 5). As vitamin E also plays a role in free radical attainment, CoQH2 has the ability to regenerate vitamin E from subsequent α-tocopherol radical formation. In animal models, depleted levels of α-tocopherol followed by the subsequent oxidation of ubiquinol suggest that α-tocopherol and ubiquinol act in concert to quench free radicals.12
Another notable function of CoQ10 would be the ability to interact with dihydrolipoic acid, a powerful radical scavenger, by transferring a pair of electrons. This helps to keep CoQ10 in the reduced state, thereby maximizing antioxidant capacity in other extra-mitochondrial membranes.27 CoQ10 has also been linked to improving superoxide dismutase (SOD) function. Endothelial nitrous oxide, an important factor in vascular homeostasis, works to inhibit platelet aggregation and inflammation. ROS produced via oxidative stress in heart failure and diabetic patients could increase NO− production by forming peroxynitrite, a potent oxidizing species, which can exacerbate the oxidative stress. CoQ10, as an antioxidant, protects NO− from forming a pro-oxidant species to preserve endothelial activity. This, in turn, could enhance SOD activity.29
CoQ10 status in disease states
Cardiovascular disease (CVD)
CVD accounts for the death of >2150 Americans each day, averaging one death every 40 seconds. This includes hypertension, heart failure, and coronary artery (heart) disease (CAD/CHD), which alone caused one of six deaths in 2010.30 Patients with CVD have been shown to be deficient in CoQ10. Those with CAD have been reported to have lower amounts of ubiquinol compared with healthy subjects, suggesting that a decreased ratio of reduced to oxidized CoQ10 instigates more oxidative stress.31
Extracellular SOD (ecSOD) serves to lower oxidative damage by catalyzing the dismutation of superoxide to oxygen and hydrogen peroxide.29 Littarru and Tiano found that patients with CAD had lowered amounts of ecSOD.32 Supplementation with 100 mg of CoQ10 three times a day in a controlled, randomized study resulted in a significant increase in ecSOD activity in those patients who were deficient in ecSOD.33 As previously mentioned, a possible scenario could be that CoQ10 salvages NO− by scavenging free radicals. In addition, NO− can interact with cellular targets, can participate in intracellular signaling, and can increase the expression of ecSOD.29,34 A higher ecSOD activity could further protect NO− from becoming inactivated by O2−.33 The mechanism in lowering blood pressure for hypertensive patients can also be attributed to this preservation of the endothelium. Rosenfeldt et al reviewed eight different trials, four placebo-controlled and four non-placebo-controlled trials, in which patients were given 100–200 mg of CoQ10 for 8–12 weeks. The results of all studies combined averaged a decrease of 16 mm Hg systolic and 10 mm Hg diastolic blood pressure.35 Similar results were found even at higher dosages. Littarru et al reported 50% of patients being able to quit using other antihypertensive medications after an administration of 225 mg CoQ10 in the trial group.29 In 1993, Morisco et al performed one of the largest controlled trials (n=641) on cardiomyopathy/heart failure. Patients received either 2 mg CoQ10 per kg/day or placebo for 1 year. Improvement in arrhythmias and episodes of pulmonary edema/asthma were documented in the supplemented group.36
CoQ10 may be effective in cosupplementation with other compounds for CAD patients. A case–control study in Taiwan studied the effects on coadministering CoQ10 with vitamin B6, a precursor for the biosynthesis of CoQ10 from tyrosine.37 They found a positive correlation with CoQ10 and vitamin B6 levels and a reduced risk in CAD. Vitamin B6 was suggested not only to promote the synthesis of CoQ10 but also to minimize homocysteine levels.37 In conjunction with this finding, Lee et al also investigated the effect of CoQ10 supplementation on inflammatory markers in patients with CAD.38 Forty subjects supplemented with higher levels of CoQ10 (150 mg/day) had higher ecSOD levels with lower inflammatory markers (IL-6 and C-reactive protein) and malondialdehyde.38 Although homocysteine levels were not affected by CoQ10 supplementation in this case, the results suggest CoQ10 and vitamin B6 supplementation could possibly work synergistically in maximizing antioxidant potential and minimizing inflammatory response.
In its role in decreasing oxidative stress, CoQ10 supplementation has had successful results in shorter hospital stays for patients undergoing surgical procedures pertaining to cardio blood vessels. Under ischemic conditions, returning blood flow through a vessel can cause inflammatory rather than restorative responses, known as ischemic reperfusion (IR) injury.30 Patients given 150–180 mg of CoQ10 a day for 1 week prior to coronary artery bypass surgery in a trial were shown to have fewer reperfusion arrhythmias, blood requirements, and mediastinal drainage in comparison with control subjects.32 The ability of CoQ10 to inhibit subsequent IR damage during the surgical time frame may suggest benefits for cardiac patients in whom medication may not be enough to treat their condition.
Mitochondrial diseases
Mitochondrial respiratory chain (MRC) disorders develop as a result of mutations in either nuclear or mitochondrial DNA. CoQ10 has been shown to offer potential benefits in the treatment of these heterogeneous disorders. Both in patients with a defect in CoQ10 biosynthesis and in those with MRC disorders, not associated with CoQ10 deficiency, CoQ10 and its synthetic analogs demonstrate some therapeutic benefits, potentially working to improve electron flow through the MRC and increase antioxidant capacity.39
Diabetes
Attributing to ROS formation, hyperglycemia can play a major role in causing complications pertaining to vascular health. CoQ10 status in individuals with diabetes has led to conflicting results due to the nature of the disease. A diabetic may be deficient or adequate in plasma CoQ10 in comparison with nondiabetic individuals. Individuals with type 2 diabetes (T2D) showed lower levels of total CoQ10 and a decreased ratio of ubiquinol to total CoQ10.4 Hasegawa et al observed changes in antioxidant status in T2D patients who displayed higher amounts of ubiquinone compared with ubiquinol.40 However, plasma CoQ10 levels in individuals with type 1 diabetes were increased, suggesting that severity and type could influence CoQ10 status.41
Individuals with T2D may benefit from the antioxidant action of CoQ10 supplementation, as levels are reduced in these patients. Few clinical studies have shown an improvement in glycemic profile with supplementation of ubiquinone. This may be due to the impaired conversion of ubiquinone to ubiquinol.41 With the increasing availability of ubiquinol supplements in recent years, T2D patients have responded positively in terms of increased status. Mezawa et al supplemented nine patients, who continued to take their hypoglycemic medications, with 200 mg of ubiquinol for 12 weeks.42 Results showed significant improvements in hemoglobin A1c (HbA1c) and prevention of the formation of advanced glycation end products, but with no effect on lipid profiles or blood pressure. Hodgson et al, in addition to improved HbA1c, found that blood pressure was improved in a randomized double-blind placebo-controlled study with uncomplicated T2D patients.43 One case study reported that diabetics with congestive heart failure displayed an improvement in systolic and left ventricular function upon supplementation.44 In combination with anti-hyperglycemic drugs, CoQ10 may benefit T2D patients without adverse effects.
Neurodegenerative disease
In a similar manner where ROS attacks lipids and proteins, nucleic acids also succumb to the effects of oxidative stress. Mitochondrial DNA, as a consequence of being located in proximity to ROS formation, is more prone to subsequent damage. Any dysfunction to the mitochondria could result in irreversible mutations of genes that synthesize ATP. Thus, oxidative damage has the ability to cause permanent changes in how our body produces energy, which could lead to cell apoptosis.45 CoQ10 concentrations have been shown to be decreased in Alzheimer’s disease (AD) and PD patients. In an antioxidant assessment, Mischley et al proposed that CoQ10 becomes conditionally essential in PD patients and that supplementation may be clinically beneficial.46 Because of its involvement in the ETC, an elevation in CoQ10 becomes relevant because of its potential in restoring function to damaged mitochondria.25 Wadsworth et al thus concluded that it may be attributed to CoQ10 antioxidant and/or activation of UCP molecules, which lower membrane potential and stress in the mitochondria.47 Although CoQ10 levels in the brain and mitochondria were unchanged in this study, further research in vivo may be necessary to determine the mechanism specific to CoQ10 neuroprotective effects.
Cancer
In the 1970s, Karl Folkers postulated that CoQ10 was needed for normal cell respiration and that any loss or decrease in its bioavailability could alter cell function.48,49 This change in function, leading to the increase in lipid peroxidation, inflammation, and oxidative damage, could lead to the possible development of cancer. An observational study reported that individuals with lung, pancreas, or especially breast cancer were more likely to have low plasma CoQ10 levels compared with healthy controls.49 A study by Bliznakov illustrated that injected doses of CoQ10 (150–750 µg) in mice significantly delayed the onset of 3,4,9,10-dibenzpyrene-induced tumor formation, decreased tumor size, and reduced mortality rates compared with mice that were not given a supplemental injection.50 A more recent trial showed similar results in male Sprague–Dawley rats, where CoQ10 supplementation (0.4 mg/kg/day) demonstrated therapeutic potential against trichloroacetic acid–induced hepatocellular carcinoma, mediated by the suppression of lipid peroxidation, the prevention of reduced glutathione depletion, and decreased elevations of tumor necrosis factor-α and nitric oxide in liver tissue.51 CoQ10 use with chemotherapy was shown to be effective in patients suffering from breast cancer. Tamoxifen, an estrogen-specific receptor antagonist, is used for therapy in all stages of breast carcinomas, which may result in possible side effects leading to oxidative stress. Supplementing CoQ10, riboflavin, and niacin (100, 10, and 50 mg/day doses, respectively) was shown to counteract derangements in blood profiles in combination with Tamoxifen, including hyperlipidemia.52 The antioxidant and anti-inflammatory properties of CoQ10 are suggested to be responsible for the anticancer effect found in such studies.
Smoking and CoQ10 status
As a biomarker of oxidative stress, antioxidants have been shown to be decreased in smokers, particularly in patients at a risk of CVD and/or chronic heart failure or with altered cholesterol metabolism.53 However, CoQ10 research on smokers with unremarkable medical history is lacking. Al-Bazi et al53 studied 55 young healthy adult smokers (30 males and 25 females) and 51 nonsmokers (26 males and 25 females) by measuring CoQ10 plasma concentrations and lipid levels. They found that smokers had significantly lower CoQ10 concentrations than those who did not smoke. Furthermore, female smokers had lower total cholesterol, HDL, LDL, and CoQ10/LDL ratio than male smokers. On the contrary, Zita et al found that baseline CoQ10 status was increased in healthy men who smoked 1–10 cigarettes a day versus nonsmoking men.54 Ninety percent of the men in this study showed an increase in CoQ10 status regardless of smoking frequency upon CoQ10 supplementation with 30 and 100 mg doses daily for 2 months. Large-scale studies on smokers with no previous medical condition may aid in clarifying whether smoking is an independent risk factor for low CoQ10 status.
Exercise and CoQ10 Supplementation
Regular exercise is known to exert exceptional benefits in lowering the risk for the development of chronic disease and overall mortality pertaining to these diseases.55 Conversely, increased intensity and duration can result in episodes of fatigue and muscle injury due to ROS produced during an exercise session. As a scavenging antioxidant, it has been theorized that CoQ10 supplementation before exercise could protect muscle cells and subsequently improve the aspects of physical performance. Östman et al tested this theory in a 8-week, double-blind controlled trial in 23 healthy men who followed a moderate-exercise regimen, half of whom (n=12) were given a daily dose of 90 mg CoQ10.56 No significant effects were demonstrated in terms of decreased oxidative stress or increased physical capacity. However, studies on the influence on CoQ10 supplementation in high-intensity exercise are scarce, yet promising. Strenuous exercises, such as marathon running, cycling, and intense aerobic training, have become increasingly prominent in the society. The concern is that damage to muscle cells in this type of workout can act as a catalyst for progressive oxidative stress and inflammation in the muscle. Díaz-Castro et al55 compared two groups composed of high-effort runners to distinguish any possible changes in inflammatory and oxidative stress markers after CoQ10 administration. The CoQ group (CG) received five capsules of 30 mg CoQ10 tablets beginning 2 days before the trial, with one capsule taken at every meal leading up to the trial day. After a 50-km mountainous run, the placebo group witnessed a significant increase in pro-inflammatory cytokines as well as in isoprostanes and hydroperoxides, which characterize free radical damage. However, the supplemented CG did not demonstrate increases in oxidative stress markers and witnessed a spike in catalase activity, an indication of antioxidant enhancement.55 These results provide a rationale for further insight into the mechanism by which CoQ10 ameliorates ROS damage.
Drug interactions
Statins
Statins inhibit 3-hydroxy-3-methylglutaryl–coenzyme A reductase (HMG-CoA), a rate-limiting step that converts HMG-CoA to mevalonate in the production of cholesterol.57 Considered a gold standard for the treatment of hypercholesterolemia, statins have been linked to the development of certain myopathies ranging from myalgia to rhabdomyolysis.12 While the incidence of myopathy is usually low, myopathy and rhabdomyolysis are more frequent when statins are combined with other drugs that inhibit cytochrome P450-dependent metabolism of statins in the liver (eg, itraconazole and erythromycin), thereby potentially increasing statin bioavailability. Research speculates that myopathy may be the result of HMG-CoA inhibition, which shuts off the production of mevalonate constituents, such as CoQ10.32 Approximately 60% of CoQ10 is carried by LDL cholesterol, which could explain why levels are decreased in the absence of a transport carrier.3 High-dose statin use has been shown to reduce plasma CoQ10 levels in hypercholesterolemia patients in comparison with healthy controls and has also resulted in an increase in lactate/pyruvate ratios.58 It has also been reported that high-statin use decreased muscle CoQ10 levels and MRC activity, possibly related to a lowered mitochondria volume.32,59 Mitochondrial dysfunction, in response to low CoQ10 and subsequent inefficiency of the respiratory chain, could thus be the end result for patients on long-term statin therapy.4 The mechanism on how statins cause a decrease in CoQ10 levels independent of lowering lipid levels still remains unclear.
The consequence of statin use may be counteracted by the supplementation of CoQ10. However, this issue to this day remains controversial and debated in terms of efficacy.31 Molyneux et al1 supplemented patients with prior statin-induced myalgia (n=44) with 200 mg/day of CoQ10 or placebo for 12 weeks in combination with a statin at 10 mg/day. Although plasma levels of CoQ10 were increased, supplementation had no significant effects in patients who tolerated the statin (73% vs 59%) or in their myalgia score change between statin and statin/CoQ10 administration (6.0 vs 2.3, respectively).1 On the contrary, Caso et al found that muscle pain was diminished by 40%, and daily activities were increased by 38% in myalgia patients receiving statins after 100 mg CoQ10 per day for 30 days.60 As future study is needed to form a definitive resolution with statin and CoQ10 therapy, the repercussions of CoQ10 deficiency should be known to clinicians for at-risk patients. Monitoring CoQ10 levels in patients who are currently on a statin can help control its potential adverse effects. It may also be beneficial to prescribe the lowest dosage needed to minimize cholesterol levels as to avoid a decrease in CoQ10 and induce myopathy.4 The excellent safety record of CoQ10 also makes it reasonable for patients to supplement this compound as a safeguard against any negative effects, if any, of statin therapy.
Other medications
Some other commonly prescribed drugs taken with CoQ10 have been reported to be influenced by the supplement’s activity. Along the same lines of statin use, β-blockers are culprits in lowering endogenous production and depletion of CoQ10. These compounds, especially propranolol, are used to treat hypertension. In some patients, the drug adversely affected myocardial function by hindering important CoQ10 enzymes.61 Concurrent therapy with CoQ10 may be beneficial to compensate for the inhibitory effect.
There have been speculations on CoQ10 and its effect on patients taking blood-thinning medications, such as warfarin, by negating their anticoagulant activity. However, clinical evidence is still lacking. In fact, one controlled study showed that supplementation of 100 mg CoQ10 in 24 warfarin patients had no long-term effects on prothrombin time over a 4-month period.62 A recent study has shown that amitriptyline, anti-depressant drug, downregulated CoQ10 biosynthesis and that CoQ10 supplementation offered protection against amitriptyline toxicity.63,64 Hypoglycemic drugs have also been shown to deplete CoQ10 status in vivo, and supplementing diabetic patients with ubiquinol has been linked to an improved glycemic profile. It is thus recommended that clinicians adjust the dosage of common diabetic medications, such as sulfonylureas and biguanides, to get the most out of both compounds.16
A dietitian’s perspective
From a patient care perspective, the registered dietitian/nutritionist (RDN) is interested in the effect of supplementation of CoQ10 for patients diagnosed with CVD, diabetes, cancer, and other conditions affected by the loss of CoQ10 due to lower production levels in the body, in prescribed drug use for the treatment of various disorders, such as use of statin drugs to reduce cholesterol, or in conditions where increased need for antioxidants may prove beneficial to health.
Since the availability of CoQ10 from dietary sources is limited, and dependent on additional nutrient intake, including vitamin B6, vitamin C, other B vitamins, and amino acids, a priority of the RDN is the assessment of current diet intake and subsequent recommendation of modifications in food consumption to meet recommended dietary reference intakes and needs. Adequate intakes of B vitamins and vitamins C and E as well as other antioxidants, fiber, and nutrients can also be an important step in the modification of symptoms in many disease conditions.
Supplementation of CoQ10 has been suggested as a means to increase body levels when production of CoQ10 is decreased and/or if an increased level of CoQ10 is needed in certain disease states. Evidence that the supplement use of CoQ10 modifies symptoms of disease has varying levels of effectiveness. In the USA, supplement use of CoQ10 has been rated “possibly effective” for conditions such as congestive heart failure, age-related macular degeneration, diabetic neuropathy, high blood pressure, migraine headache, and muscular dystrophy.65 According to the Evidence Analysis Library of the Academy of Nutrition and Dietetics, its effectiveness when used to treat muscle pain associated with the use of statin drugs to treat high blood cholesterol is not yet conclusive.66 The use of CoQ10 as a supplement should be evaluated for clients/patients on an individual basis along with an assessment of current dietary intake, drug prescriptions, and other medicines and supplements used.
Conclusion
CoQ10 is a vitamin-like compound synthesized in every cell in the body. Mitochondria contain the highest concentration of CoQ10 and play a significant role in the production of ATP. Thus, tissues such as the heart and muscle are in need of substantial amounts due to their high energy requirement. Outside the respiratory chain, CoQ10 also acts as a lipophilic antioxidant to reduce the damaging effects of ROS in the body. Because CoQ10 is made de novo within the body, contribution through the diet is not necessary under normal circumstances. However, factors such as increased age and oxidative stress via metabolic discrepancies can make this nutrient conditionally essential. It has been shown that age-related changes are most evident in the mitochondria and that decreases in CoQ10 content may be specific to certain tissues rather than ubiquitous.67 Age and disease have also shown to influence increased oxidized CoQ10 compared with the reduced form, which may result in decreased levels of total CoQ10.2 In such cases, resupply through diet alone may not be a conclusive answer. Absorption of CoQ10 from dietary contribution is slow and inefficient because of its weight and hydrophobic nature. With the increasing availability of the over-the-counter forms, CoQ10 supplementation may be the best therapeutic source when endogenous CoQ10 synthesis is impaired. Ultimately, CoQ10 may aid in preventing and/or minimizing metabolic conditions that may lead to CVD, diabetes, neurodegenerative disorders, and cancer. Clinical studies encompassing these disease states where improvements in mitochondrial and antioxidant function are shown provide implications for therapeutic CoQ10 supplementation. The safety record of CoQ10 also makes it an excellent compound for human trials. Limitations in CoQ10 supplementation include its high cost and absorption efficiency, as both ubiquinone and ubiquinol are now available for purchase in products with varied solubility formulations. In addition, most of the clinical studies on CoQ10 supplementation reviewed had a relatively small number of participants and short-term duration. Future long-term studies on its use with larger amounts of subjects suffering from conditions caused by oxidative stress would lead to a more defined understanding of CoQ10 action in alleviating the resulting symptoms of deficiency.
Acknowledgments
Research support from the National Institute of Food and Agriculture of the US Department of Agriculture is acknowledged (ALA043-1-14033).
Disclosure
The authors report no conflicts of interest in this work.
References
Molyneux SL, Young JM, Florkowski CM, Lever M, George PM. Coenzyme Q10: is there a clinical role and a case for measurement? Clin Biochem Rev. 2008;29(2):71–82. | ||
Turunen M, Olsson J, Dallner G. Metabolism and function of coenzyme Q. Biochim Biophys Acta. 2004;1660(1–2):171–199. | ||
Lance J, McCabe S, Clancy RL, Pierce J. Coenzyme QIO – a therapeutic agent. Medsurg Nurs. 2012;21(6):367–371. | ||
Sohet FM, Delzenne NM. Is there a place for coenzyme Q in the management of metabolic disorders associated with obesity? Nutr Rev. 2012;70(11):631–641. | ||
Crozier A, Borges G. (Poly)phenolic constituents and the beneficial effects of moderate red wine consumption. J Wine Res. 2011;22(2):131–134. | ||
Cain JC, Morton RA. Some minor constituents of liver oils. Biochem J. 1955;60(2):274–283. | ||
Fahmy NI, Hemming FW, Morton RA, Paterson JYF, Pennock JF. Ubiquinone. Biochem J. 1958;70(1P). | ||
Ramasarma T. A touch of history and a peep into the future of the lipid-quinone known as coenzyme Q and ubiquinone. Curr Sci. 2012;102(10):1459–1471. | ||
Crane FL, Hatefi Y, Lester RL, Widmer C. Isolation of a quinone from beef heart mitochondria. Biochim Biophys Acta. 1957;25(1):220–221. | ||
Crane FL. Discovery of ubiquinone (coenzyme Q) and an overview of function. Mitochondrion. 2007;7(Suppl):S2–S7. | ||
Crane FL. The evolution of coenzyme Q. Biofactors. 2008;32(1–4):5–11. | ||
Potgieter M, Pretorius E, Pepper MS. Primary and secondary coenzyme Q10 deficiency: the role of therapeutic supplementation. Nutr Rev. 2013;71(3):180–188. | ||
Kommuru TR, Ashraf M, Khan MA, Reddy IK. Stability and bioequivalence studies of two marketed formulations of coenzyme Q10 in beagle dogs. Chem Pharm Bull (Tokyo). 1999;47(7):1024–1028. | ||
Ikematsu H, Nakamura K, Harashima S, Fujii K, Fukutomi N. Safety assessment of coenzyme Q10 (Kaneka Q10) in healthy subjects: a double-blind, randomized, placebo-controlled trial. Regul Toxicol Pharmacol. 2006;44(3):212–218. | ||
Cluis CP, Pinel D, Martin VJ. The production of coenzyme q10 in microorganisms. Subcell Biochem. 2012;64:303–326. | ||
Bhagavan HN, Chopra RK. Coenzyme Q10: absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic Res. 2006;40(5):445–453. | ||
Bhagavan HN, Chopra RK. Plasma coenzyme Q10 response to oral ingestion of coenzyme Q10 formulations. Mitochondrion. 2007;7(Suppl): S78–S88. | ||
Lester RL, Crane FL. The natural occurrence of coenzyme Q and related compounds. J Biol Chem. 1959;234(8):2169–2175. | ||
Overvad K, Diamant B, Holm L, Holmer G, Mortensen SA, Stender S. Coenzyme Q10 in health and disease. Eur J Clin Nutr. 1999; 53(10):764–770. | ||
Weber C. Dietary Intake and Absorption of Coenzyme Q. Boca Raton, FL: CRC Press; 2001. | ||
Chopra RK, Goldman R, Sinatra ST, Bhagavan HN. Relative bioavailability of coenzyme Q10 formulations in human subjects. Int J Vitam Nutr Res. 1998;68(2):109–113. | ||
Zaghloul AA, Gurley B, Khan M, Bhagavan H, Chopra R, Reddy I. Bioavailability assessment of oral coenzyme Q10 formulations in dogs. Drug Dev Ind Pharm. 2002;28(10):1195–1200. | ||
Shults CW, Flint Beal M, Song D, Fontaine D. Pilot trial of high dosages of coenzyme Q10 in patients with Parkinson’s disease. Exp Neurol. 2004;188(2):491–494. | ||
Duncan AJ, Heales SJ, Mills K, Eaton S, Land JM, Hargreaves IP. Determination of coenzyme Q10 status in blood mononuclear cells, skeletal muscle, and plasma by HPLC with di-propoxy-coenzyme Q10 as an internal standard. Clin Chem. 2005;51(12):2380–2382. | ||
Steele PE, Tang PH, DeGrauw AJ, Miles MV. Clinical laboratory monitoring of coenzyme Q10 use in neurologic and muscular diseases. Am J Clin Pathol. 2004;121(Suppl):S113–S120. | ||
Ziegler DM. The role of quinones in the mitochondrial electron transport system. Am J Clin Nutr. 1961;9(4):43–49. | ||
Kozlov AV, Gille L, Staniek K, Nohl H. Dihydrolipoic acid maintains ubiquinone in the antioxidant active form by two-electron reduction of ubiquinone and one-electron reduction of ubisemiquinone. Arch Biochem Biophys. 1999;363(1):148–154. | ||
Echtay KS, Winkler E, Klingenberg M. Coenzyme Q is an obligatory cofactor for uncoupling protein function. Nature. 2000; 408(6812):609–613. | ||
Littarru GP, Tiano L, Belardinelli R, Watts GF. Coenzyme Q10, endothelial function, and cardiovascular disease. Biofactors. 2011;37(5):366–373. | ||
Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics – 2014 update: a report from the American Heart Association. Circulation. 2014;129(3):e28–e292. | ||
Singh RB, Niaz MA, Kumar A, Sindberg CD, Moesgaard S, Littarru GP. Effect on absorption and oxidative stress of different oral Coenzyme Q10 dosages and intake strategy in healthy men. Biofactors. 2005;25(1–4):219–224. | ||
Littarru GP, Tiano L. Clinical aspects of coenzyme Q10: an update. Nutrition. 2010;26(3):250–254. | ||
Tiano L, Belardinelli R, Carnevali P, Principi F, Seddaiu G, Littarru GP. Effect of coenzyme Q10 administration on endothelial function and extracellular superoxide dismutase in patients with ischaemic heart disease: a double-blind, randomized controlled study. Eur Heart J. 2007;28(18):2249–2255. | ||
Bogdan C. Nitric oxide and the regulation of gene expression. Trends Cell Biol. 2001;11(2):66–75. | ||
Rosenfeldt F, Hilton D, Pepe S, Krum H. Systematic review of effect of coenzyme Q10 in physical exercise, hypertension and heart failure. Biofactors. 2003;18(1–4):91–100. | ||
Morisco C, Trimarco B, Condorelli M. Effect of coenzyme Q10 therapy in patients with congestive heart failure: a long-term multicenter randomized study. Clin Investig. 1993;71(8 Suppl):S134–S136. | ||
Lee BJ, Yen CH, Hsu HC, Lin JY, Hsia S, Lin PT. A significant correlation between the plasma levels of coenzyme Q10 and vitamin B-6 and a reduced risk of coronary artery disease. Nutr Res. 2012;32(10):751–756. | ||
Lee BJ, Huang YC, Chen SJ, Lin PT. Effects of coenzyme Q10 supplementation on inflammatory markers (high-sensitivity C-reactive protein, interleukin-6, and homocysteine) in patients with coronary artery disease. Nutrition. 2012;28(7–8):767–772. | ||
Hargreaves IP. Coenzyme Q10 as a therapy for mitochondrial disease. Int J Biochem Cell Biol. 2014;49:105–111. | ||
Hasegawa G, Yamamoto Y, Zhi JG, et al. Daily profile of plasma %CoQ10 level, a biomarker of oxidative stress, in patients with diabetes manifesting postprandial hyperglycaemia. Acta Diabetol. 2005;42(4):179–181. | ||
Menke A, Orchard TJ, Imperatore G, Bullard KM, Mayer-Davis E, Cowie CC. The prevalence of type 1 diabetes in the United States. Epidemiology.2013;24(5):773–774. | ||
Mezawa M, Takemoto M, Onishi S, et al. The reduced form of coenzyme Q10 improves glycemic control in patients with type 2 diabetes: an open label pilot study. Biofactors. 2012;38(6):416–421. | ||
Hodgson JM, Watts GF, Playford DA, Burke V, Croft KD. Coenzyme Q10 improves blood pressure and glycaemic control: a controlled trial in subjects with type 2 diabetes. Eur J Clin Nutr. 2002;56(11):1137–1142. | ||
Salles JE, Moises VA, Almeida DR, Chacra AR, Moises RS. Myocardial dysfunction in mitochondrial diabetes treated with Coenzyme Q10. Diabetes Res Clin Pract. 2006;72(1):100–103. | ||
Mancuso M, Orsucci D, Volpi L, Calsolaro V, Siciliano G. Coenzyme Q10 in neuromuscular and neurodegenerative disorders. Curr Drug Targets. 2010;11(1):111–121. | ||
Mischley LK, Allen J, Bradley R. Coenzyme Q10 deficiency in patients with Parkinson’s disease. J Neurol Sci. 2012;318(1–2):72–75. | ||
Wadsworth TL, Bishop JA, Pappu AS, Woltjer RL, Quinn JF. Evaluation of coenzyme Q as an antioxidant strategy for Alzheimer’s disease. J Alzheimers Dis. 2008;14(2):225–234. | ||
Hodges S, Hertz N, Lockwood K, Lister R. CoQ10: could it have a role in cancer management? Biofactors. 1999;9(2–4):365–370. | ||
Folkers K, Osterborg A, Nylander M, Morita M, Mellstedt H. Activities of vitamin Q10 in animal models and a serious deficiency in patients with cancer. Biochem Biophys Res Commun. 1997;234(2):296–299. | ||
Bliznakov EG. Effect of stimulation of the host defense system by coenzyme Q 10 on dibenzpyrene-induced tumors and infection with Friend leukemia virus in mice. Proc Natl Acad Sci U S A. 1973;70(2):390–394. | ||
Fouad AA, Al-Mulhim AS, Jresat I. Therapeutic effect of coenzyme Q10 against experimentally-induced hepatocellular carcinoma in rats. Env Toxicol Pharmacol. 2013;35(1):100–108. | ||
Yuvaraj S, Premkumar VG, Shanthi P, Vijayasarathy K, Gangadaran SG, Sachdanandam P. Effect of Coenzyme Q(10), Riboflavin and Niacin on Tamoxifen treated postmenopausal breast cancer women with special reference to blood chemistry profiles. Breast Cancer Res Treat. 2009;114(2):377–384. | ||
Al-Bazi MM, Elshal MF, Khoja SM. Reduced coenzyme Q(10) in female smokers and its association with lipid profile in a young healthy adult population. Arch Med Sci. 2011;7(6):948–954. | ||
Zita C, Overvad K, Mortensen SA, Sindberg CD, Moesgaard S, Hunter DA. Serum coenzyme Q10 concentrations in healthy men supplemented with 30 mg or 100 mg coenzyme Q10 for two months in a randomised controlled study. Biofactors. 2003;18(1–4):185–193. | ||
Díaz-Castro J, Guisado R, Kajarabille N, et al. Coenzyme Q supplementation ameliorates inflammatory signaling and oxidative stress associated with strenuous exercise. Eur J Nutr. 2012;51(7):791–799. | ||
Östman B, Sjödin A, Michaëlsson K, Byberg L. Coenzyme Q10 supplementation and exercise-induced oxidative stress in humans. Nutrition. 2012;28(4):403–417. | ||
Singh U, Devaraj S, Jialal I. Coenzyme Q10 supplementation and heart failure. Nutr Rev. 2007;65(6 Pt 1):286–293. | ||
De Pinieux G, Chariot P, Ammi-Said M, et al. Lipid-lowering drugs and mitochondrial function: effects of HMG-CoA reductase inhibitors on serum ubiquinone and blood lactate/pyruvate ratio. Br J Clin Pharmacol. 1996;42(3):333–337. | ||
Paiva H, Thelen KM, Van Coster R, et al. High-dose statins and skeletal muscle metabolism in humans: a randomized, controlled trial. Clin Pharmacol Ther. 2005;78(1):60–68. | ||
Caso G, Kelly P, McNurlan MA, Lawson WE. Effect of coenzyme q10 on myopathic symptoms in patients treated with statins. Am J Cardiol. 2007;99(10):1409–1412. | ||
Kishi T, Watanabe T, Folkers K. Bioenergetics in clinical medicine XV. Inhibition of coenzyme Q10-enzymes by clinically used adrenergic blockers of beta-receptors. Res Commun Chem Pathol Pharmacol. 1977;17(1):157–164. | ||
Engelsen J, Nielsen JD, Winther K. Effect of coenzyme Q10 and Ginkgo biloba on warfarin dosage in stable, long-term warfarin treated outpatients. A randomised, double blind, placebo-crossover trial. Thromb Haemost. 2002;87(6):1075–1076. | ||
Moreno-Fernandez AM, Cordero MD, Garrido-Maraver J, et al. Oral treatment with amitriptyline induces coenzyme Q deficiency and oxidative stress in psychiatric patients. J Psychiatr Res. 2012;46(3):341–345. | ||
Cordero MD, Moreno-Fernandez AM, Gomez-Skarmeta JL, et al. Coenzyme Q10 and alpha-tocopherol protect against amitriptyline toxicity. Toxicol Appl Pharmacol. 2009;235(3):329–337. | ||
NATURAL MEDICINES COMPREHENSIVE DATABASE. http://www.naturaldatabase.com. Accessed January 17, 2018. | ||
EVIDENCE ANALYSIS LIBRARY. Available from: http://www.andeal.org.Accessed January 17, 2018. | ||
Sohal RS, Forster MJ. Coenzyme Q, oxidative stress and aging. Mitochondrion. 2007;7(Suppl):S103–S111. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.