Back to Journals » Research and Reports in Urology » Volume 13
Polymorphism of PvuII, Xba1, and SNP 12 Estrogen Receptor 1 (ESR1) in Hipospadias Patients at Tertiary Hospital Center
Authors Siregar S, Sibarani J , Noegroho BS, Firmansyah I, Maskoen AM
Received 16 December 2020
Accepted for publication 18 January 2021
Published 25 February 2021 Volume 2021:13 Pages 105—110
DOI https://doi.org/10.2147/RRU.S296941
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jan Colli
Safendra Siregar,1 Jupiter Sibarani,1 Bambang Sasongko Noegroho,1 Irfan Firmansyah,1 Ani Melani Maskoen2
1Department of Urology, Faculty of Medicine Universitas Padjadjaran, General Hospital Hasan Sadikin Bandung, Indonesia; 2Department of Molecular Genetic Laboratory Universitas Padjadjaran, Bandung City, West Java Province, Indonesia
Correspondence: Safendra Siregar; Bambang Sasongko Noegroho
Department of Urology, Faculty of Medicine Universitas Padjadjaran, General Hospital Hasan Sadikin, Jl. Pasteur No. 38, Bandung, Indonesia
Tel +62811227180
; +62811215058
Email [email protected]; [email protected]
Introduction: The prevalence of hypospadias ranges between 1: 250 to 300 per live birth. Estrogen exposure has been associated with the increasing incidence of hypospadias in humans and a significant relationship between Estrogen Receptor 1 (ESR1) polymorphisms and hypospadias was determined from the previous study. This study aims to determine the proportion of ESR1 polymorphism.
Methods: This was a descriptive study aimed to find the incidence of ESR1 gene polymorphism in hypospadias patients visiting the Urology Outpatient Unit of the Hasan Sadikin Bandung Hospital who will undergo hypospadias surgery. Deoxyribonucleic acid (DNA) was performed using foreskin of hypospadias patient during the surgery then being extracted and will be analyzed using polymerase chain reaction (PCR) sequencing.
Results: Thirty eight samples of hypospadias were identified, 5 samples were unable to sequence and 33 samples were successfully sequenced using the PCR method. The Pvull ESR1 gene was dominated by the T allele which is a wild-type allele and the genotype containing the T allele, namely TT + TC (57,2%). Normal genotype (TT) were more frequent in distal hypospadias, and Heterozygous polymorphisms (TC) was higher in proximal hypospadias. The ESR1 Xba1 gene polymorphism was dominated by the A allele which is a wild-type allele and the AA + AG genotype (76,1%). Normal genotype (AA) was more frequent in distal hypospadias, and both heterozygous hypospadias (AG) and homozygous hypospadias (GG) were found only in proximal hypospadias. The ESR1 SNP 12 gene polymorphisms were found in the combination of genotypes that played a role, namely GA + AA (81%) and the G allele which is a wild-type allele. Heterozygous polymorphisms (GA) was the most finding genotype and more frequent in proximal hypospadias.
Conclusion: ESR1 gene polymorphisms (PvuII, XbaI, and SNP 12) were found in hypospadias patients. ESR1 polymorphisms may correlate with the severity of hypospadias. Further research with a larger sample and better hypospadias grouping is needed to confirm.
Keywords: estrogen receptor 1, hypospadias, polymorphism
Introduction
Hypospadias is a congenital abnormality of the urethral opening which is located on the ventral aspect of the penis proximal to the tip of the glans penis due to the incomplete development of urethral fusion.1–3 The prevalence of hypospadias ranges between 1: 250 to 300 per live birth. Hypospadias prevalence varies among the world such as Europe which was reached up to 19.9%, North America 34.2%, South America 5.2%. Australia 17.1–34.8%, and Asia 0.6–69% per 10,000 live births.4 The prevalence of hypospadias in Indonesia is not certain. Research conducted by Aritonang et al at RSUP Dr. Cipto Mangunkusumo Jakarta found 324 cases of hypospadias in the period 2002–2014, while Kusuma et al revealed 591 cases between June - September 2018 in 12 teaching hospitals in Indonesia.5,6
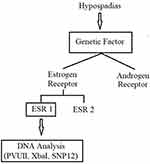 |
Figure 1 Research flow. |
The development of external genitalia in men was influenced by genetic factors, environmental factors, or a combination of both. The genetic factor was believed to be more significant because the process of formation and development of male phenotypes was regulated and controlled by several genes so that many experts believe that changes in DNA composition, either mutations or polymorphisms in these genes, could lead to hypospadias.7–9
Estrogen exposure has been associated with the increasing incidence of hypospadias in humans. There are 2 types of receptors involved in hypospadias, estrogen receptor 1 (ESR1) and estrogen receptor 2 (ESR2). Estrogen binds to ESR1 or ESR2 activates the estrogen-responsive gene and stimulates cells (Figure 1). These receptors are expressed in most of the male urethra cells. Choudhry et al found there were 33 ESR1 gene polymorphisms in 108 samples, these results obtained a significant relationship between ESR1 polymorphisms and hypospadias.7–11 However, this result contradicts the study of van der Zanden et al, which did not find a significant relationship between ESR1 polymorphisms and hypospadias.7,9,10,12
There was still an inconsistent result of ESR1 gene polymorphism association to hypospadias, therefore this study was aimed to know the proportion of ESR1 gene polymorphism in hypospadias patients in our region.
Methods
Patients
This was a descriptive study aimed to find the incidence of ESR1 gene polymorphism in hypospadias patients visiting the Urology Outpatient Unit of the Hasan Sadikin Bandung Hospital who underwent hypospadias surgery. The DNA sample was taken from the foreskin of preputium which was collected during the surgery. DNA extraction was done by using the Homebrew method at the Genetics Laboratory of Eyckman, Padjadjaran University, Bandung. Polymorphism analysis was carried out by ESR1 coding-exon sequencing by using the standard direct sequencing method at the Genetics Laboratory of Eyckman, Padjadjaran University, Bandung. Proximal hypospadias, defined in which the meatus is back on the shaft of the penis, near or within the scrotum. In distal hypospadias, the urethral opening (the meatus) is on or near the head of the penis (glans). A downward bending of the penis, commonly referred to as chordee, may occur. Chordee is found in 10% of distal hypospadias and 50% of proximal hypospadias cases at the time of surgery. Also, the scrotum may be higher than usual on either side of the penis (called penoscrotal transposition). All type of hypospadias was diagnosed at the time of surgery. We define the type of hypospadias based on operative finding. This study has been received Ethics Approval from the Ethics Committee, Faculty of medicine, Universitas Padjadjaran, Bandung, Indonesia with ethical number LB.02.01/X.6.5/71/2020. The patient has received informed consent before procedure that their information will be used in our study. All of participants gave consent to have their data published.
Genetic Analysis
The sample was taken from 38 patients. Five samples were excluded due to failed sequencing. DNA is taken from the foreskin of the penis when the sample is undergoing surgery. DNA extraction using the Homebrew method in the Genetics Laboratory of the Eykman Building, Bandung. The Biorad T100 Thermal Cycler PCR machine primers used were programmed for 2 hours. The PCR cycle used was denaturation 94 ° C, annealing 59 ° C, elongation 72 ° C for 30 cycles.
Electrophoresis was carried out by using 1.5% agarose media processed in an electrophoresis machine for 30 minutes with a voltage of 90 v. The results of electrophoresis were seen by using a UV lamp with a wavelength of 300 nm and photos using the GelDoc program. If the PCR results show that there is a suitable gene, DNA direct sequencing will be continued using the Applied Bioscience 3500 genetic analyzer. (Table 1)
 |
Table 1 Primer Sequence of ESR1 |
The results of measuring DNA levels with nanodrop were obtained at 260/280 in the value range 1.8–2 which shows a good and viable sample. This study was conducted following the Declaration of Helsinki.
Results
Patients Characteristics
There were 38 samples obtained from hypospadias patients. The characteristics of hypospadias in this study were presented in Table 2.
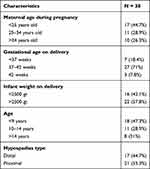 |
Table 2 Patients Characteristics |
Thirty eight samples of hypospadias were identified, 5 samples were unable to sequence and 33 samples were successfully sequenced using the PCR method Analyzes were performed for each primer on the incidence of distal and proximal hypospadias. (Table 3) The Pvull ESR1 gene was dominated by the T allele which is a wild-type allele and the genotype containing the T allele, namely TT + TC (57,2%). Normal genotype (TT) were more frequent in distal hypospadias, and Heterozygous polymorphisms (TC) was higher in proximal hypospadias. The ESR1 Xba1 gene polymorphism was dominated by the A allele which is a wild-type allele and the AA + AG genotype (76,1%). Normal genotype (AA) was more frequent in distal hypospadias, and both heterozygous hypospadias (AG) and homozygous hypospadias (GG) were found only in proximal hypospadias. The ESR1 SNP 12 gene polymorphisms were found in the combination of genotypes that played a role, namely GA + AA (81%) and the G allele which is a wild-type allele. Heterozygous polymorphisms (GA) was the most finding genotype and more frequent in proximal hypospadias. This may conclude that we found polymorphisms more in proximal hypospadias rather than distal hypospadias.
 |
Table 3 PvuII, Xba1, SNP12 Gene ESR1 |
We also investigated PvuII and XbaI haplotypes with hypospadias (Table 4). The T-A haplotype were found in both type of hypospadias, but the C-A, C-G, T-G, and CC/AA genotype combination were found only in proximal hypospadias. We may conclude that polymorphisms maybe associated with the severity of hypospadias.
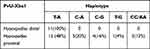 |
Table 4 ESR1 Haplotype in All Types of Hypospadias |
Discussion
Over the past few years, there have been hypotheses that prenatal estrogen exposure in male fetuses contributes to hypospadias, and until now, only a few studies have examined the association between ESR1 gene polymorphisms and hypospadias.13,14
We have evaluated the genotypes of 33 patients to find out whether there are ESR1 gene polymorphisms in hypospadias patients with Sundanese ethnicity. The results obtained were that there were ESR1 gene polymorphisms in PvuII, Xbal, and SNP12 primers so that the effect of the ESR1 gene polymorphism may have contributed to the occurrence of hypospadias.10,12 Research for ESR 1 polymorphism was previously conducted by Watanabe et al mentioned that there is a strong association between the AGAGA Haplotype (SNP 10–14) and hypospadias.12 Another study conducted by Tang et al and Choudry et al found that the ESR1 polymorphism significantly increases the risk of hypospadias.7
In our study, it was found that in the PvUII polymorphism, the T allele was 55%, the C allele 45%, and the TT (36%), TC (36%), and CC (28%) genotypes. This is similar to the study by Ban et al In the Japanese population where the T allele was the most and the TC genotype was the largest.10
XbaI polymorphism obtained A allele (66%) and G allele (33%). The polymorphism genotype was found in AA (48%), AG (36%), GG (15%). This is similar to Ban et al study in the Japanese population where the A allele was the largest and the AA genotype polymorphism was the largest.10
In our study, SNP 12 obtained allele G (41%), A (59%), GG genotype (15%), and AA (33%). This is similar to the study conducted by Deng et al Conducted a meta-analysis of the 4 studies above with a multiethnic population (Caucasian, Japanese, Chinese, and Hispanic), and it was found that the AA genotype was a risk factor for hypospadias.15
Research conducted by Ban et al, to look for ESR1 polymorphisms in Pvull 2 and Xba 1 from 59 hypospadias cases, it was found that the CA-Haplotype of ESR 1 Pvull - Xbal in hypospadias subjects was found to be more than controls.10 In our study we conducted, CA Haplotype is a polymorphism that occurs more frequently than the other Haplotypes. ESR1 PvuII/XbaI is also associated with the degree of hypospadias. In the polymorphism that occur in ESR1 PvuII/XbaI, it is known that a mild degree of hypospadias occurs. In our study, ESR1 Pvull and Xbal polymorphisms were more found in proximal (severe) hypospadias. This suggests the possibility of different racial factors that may play a role or risk factors for different xenoestrogen exposure in each country.
In a multiethnic study of the ESR1 gene conducted by Choudhry et al, it was found that several SNPs and haplotypes were associated with the risk of hypospadias in Hispanics but not in non-Hispanic whites. The Hispanic population itself consists of 50% of American descent and 50% of European descent. ESR1 polymorphisms in hypospadias patients are mostly found of American descent.7 In a study in Japan conducted by Ban et al and Watanabe et al, it was found that there was an association of ESR1 gene with primary PvuII-XbaI and SNP 12 in a population in Japan. In our study, the findings were also the same as studies conducted in Japan, but the prevalence of severe hypospadias was more than mild. The Japanese population is similar to the Indonesian population, possibly because it still has an Asian family, but further research on this must be proven using a larger and more accurate sample.
This study has several limitations, among others, this study did not use a control group because, in our center, the circumcision case is rare and not done in a tertiary hospital. This study also has a limited sample of 38 samples due to a limitation of hypospadias surgery due to COVID-19 regulation. Other possible influencing factors such as lifestyle characteristics, exposure to the environment, and work history have also not been studied. Further studies to determine the incidence of hypospadias using a control group are strongly recommended.
Conclusion
The PvUII, XbaI, and SNP12 ESR1 gene were found in all samples of hypospadias patients. This was proven by gene analysis using three primers which show polymorphisms both heterozygous and homozygous. ESR1 gene polymorphisms were more frequent in proximal hypospadias. It may be concluded that polymorphisms correlated with the severity of hypospadias, but further study is required to confirm.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wein AJ, Kavoussi LR, Partin AW, Peters CA. Campbell-Walsh Urology.
2. Tanagho E, McAninch J. Disorders of the penis and male urethra. Smith’s General Urol. 2007;40:625.
3. Baskin L. What is hypospadias? Clin Pediatr (Phila). 2017;56(5):409–418. doi:10.1177/0009922816684613
4. Springer A, van den Heijkant M, Baumann S. Worldwide prevalence of hypospadias. J Pediatr Urol. 2016;12(3):152e1–7. doi:10.1016/j.jpurol.2015.12.002
5. Aritonang J, Rodjani A, Wahyudi I. Relationship between complicating factors of hypospadia and complications after TIP: a retrospective study. Indonesian J Urol. 2016;23(2):103–107. doi:10.32421/juri.v23i2.239
6. Kusuma GW, Wisnu PM, Daryanto B, et al. Common practice of hypospadias management by pediatric urologist in Indonesia: a multi-center descriptive study from referral hospitals. Open Access Macad J Med Sci. 2019;7:2242. doi:10.3889/oamjms.2019.628
7. Choudhry S, Baskin LS, Lammer EJ, et al. Genetic polymorphisms in ESR1 and ESR2 genes, and risk of hypospadias in a multiethnic study population. J Urol. 2015;193(5):1625–1631. doi:10.1016/j.juro.2014.11.087
8. van der Zanden LF, van Rooij IA, Feitz WF, et al. Aetiology of hypospadias: a systematic review of genes and environment. Hum Reprod Update. 2012;18(3):260–283.
9. van der Zanden LF, van Rooij IA, Feitz WF, et al. Genetics of hypospadias: are single-nucleotide polymorphisms in SRD5A2, ESR1, ESR2, and ATF3 really associated with the malformation? J Clin Endocrinol Metab. 2010;95(5):2384–2390. doi:10.1210/jc.2009-2101
10. Ban S, Sata F, Kurahashi N, et al. Genetic polymorphisms of ESR1 and ESR2 that may influence estrogen activity and the risk of hypospadias. Hum Reprod. 2008;23(6):1466–1471. doi:10.1093/humrep/den098
11. Wang Z, Liu BC, Lin GT, et al. Up-regulation of estrogen responsive genes in hypospadias: microarray analysis. J Urol. 2007;177:1939–1946. doi:10.1016/j.juro.2007.01.014
12. Watanabe M, Yoshida R, Ueoka K, et al. Haplotype analysis of the estrogen receptor 1 gene in male genital and reproductive abnormalities. Hum Reprod. 2007;22:1279–1284. doi:10.1093/humrep/del513
13. Steinhardt GF. Endocrine disruption and hypospadias. Adv Exp Med Biol. 2004;545:203–215.
14. Vidaeff AC, Sever LE. In utero exposure to environmental estrogens and male reproductive health: a systematic review of biological and epidemiologic evidence. Reprod Toxicol. 2005;20:5–20. doi:10.1016/j.reprotox.2004.12.015
15. Deng C, Dai R, Li X, Liu F. Association between SNP12 in estrogen receptor α gene and hypospadias: a systematic review and meta-analysis. SpringerPlus. 2016;5(1). doi:10.1186/s40064-016-2288-0
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
