Back to Journals » Neuropsychiatric Disease and Treatment » Volume 18
Plasma SIRT3 as a Biomarker of Severity and Prognosis After Acute Intracerebral Hemorrhage: A Prospective Cohort Study
Authors Yan T, Wang ZF, Wu XY, Du Q, Yu WH, Hu W, Zheng YK, Wang KY, Dong XQ
Received 30 May 2022
Accepted for publication 17 September 2022
Published 26 September 2022 Volume 2022:18 Pages 2199—2210
DOI https://doi.org/10.2147/NDT.S376717
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jun Chen
Tian Yan,1 Ze-Fan Wang,1 Xiao-Yu Wu,1 Quan Du,2 Wen-Hua Yu,2 Wei Hu,3 Yong-Ke Zheng,3 Ke-Yi Wang,4 Xiao-Qiao Dong2
1The Fourth School of Clinical Medicine, Zhejiang Chinese Medical University, Hangzhou, People’s Republic of China; 2Department of Neurosurgery, Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China; 3Department of Intensive Care Unit, Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China; 4Clinical Laboratory Center, Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China
Correspondence: Xiao-Qiao Dong, Department of Neurosurgery, Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China, Email [email protected]
Objective: SIRT3 may act as a brain-protective factor. We measured the plasma SIRT3 levels of patients with intracerebral hemorrhage (ICH) and further determined the relationship between plasma SIRT3 and clinical outcome plus severity of ICH.
Methods: In this prospective cohort study, we quantified plasma SIRT3 levels in 105 ICH patients and 72 healthy controls. Glasgow Coma Scale (GCS) score and hematoma volume were used to assess severity. Poor prognosis was defined as a Glasgow Outcome Scale (GOS) score of 1– 3 at 90 days after ICH.
Results: Plasma SIRT3 levels were markedly lower in patients than in controls (median, 10.19 versus 13.17 ng/mL; P< 0.001). Among all patients, plasma SIRT3 levels were independently correlated with hematoma volume (beta, − 0.098; 95% confidence interval, − 0.158--0.039; t, − 3.282; P=0.001) and GCS score (beta, 0.465; 95% confidence interval, 0.107– 0.823; t, 2.576; P=0.011). A total of 46 cases had a poor prognosis at post-stroke 90 days. The plasma levels of SIRT3 significantly decreased in patients with a poor prognosis, compared with those with a good prognosis (median, 6.1 versus 11.2 ng/mL; P< 0.001). Plasma SIRT3 was an independent predictor for 90-day poor prognosis of patients (odds ratio, 0.837; 95% confidence interval, 0.708– 0.990; P=0.038). Plasma SIRT3 levels distinguished the development of poor prognosis with area under receiver operating characteristic curve at 0.801 (95% confidence interval, 0.711– 0.872) and plasma SIRT3 levels ≤ 7.38 ng/mL predicted poor prognosis with 63.04% sensitivity and 93.22% specificity.
Conclusion: Declined plasma SIRT3 levels are highly associated with hemorrhagic severity and poor 90-day outcome, thus suggesting that plasma SIRT3 may serve as a potential prognostic biomarker for ICH.
Keywords: intracerebral hemorrhage, prognosis, severity, SIRT3, outcome
Introduction
Intracerebral hemorrhage (ICH) is a serious public health problem with high rates of death and disability, and it accounts for 10–20% of stroke worldwide.1,2 Secondary brain injury induces perihematomal edema and therefore increases intracranial pressure, finally leading to a poor prognosis after ICH.3,4 The initial hematoma induces glutamate release and then leads to oxidative stress and mitochondrial dysfunction. Mitochondrial dysfunction may contribute to the deficiency of adenosine triphosphate generation and then result in the failure of cellular pumps, which causes cytotoxic edema and neuronal apoptosis.5–8 Thus, inhibiting oxidative stress may be an effective method in decreasing ICH injury. In addition, some biomarkers, which are related to inflammation, oxidative stress, neuronal apoptosis and brain edema, have been extensively studied with respect to outcome prediction and severity assessment.9,10 Recently, several systematic reviews and clinical studies of multiple biomarkers have shown that those biochemical markers, eg, E-selectin, P-selectin, matrix metalloprotein-9, homocysteine and C-reactive protein, are strongly associated with clinical outcomes of ICH, but they have not been used in clinical work because of their low clinical predictive significance.11 Thus, studies in search of new biomarkers are under way for aiding in ICH prognostic prediction.
The sirtuins, as a family of highly conservative NAD+-dependent enzymes, participate in transcriptional silencing and regulation of mitochondrial functions.12 As one of the known seven members of the sirtuin family, SIRT3 is distinguished by its main localization in mitochondria.13–15 Of note, SIRT3 obviously attenuated doxorubicin-induced reactive oxygen species (ROS) output in H9c2 cardiomyocytes.16 Intriguingly, SIRT3 expression was significantly down-regulated with ROS generation in cortical neurons of rats with subarachnoid hemorrhage.17 In addition, SIRT3 could effectively lessen oxidative stress and mitochondrial dysfunction after ICH in diabetic rats.18 Thus, SIRT3 might play an important neuroprotective role in acute brain injury. However, up to now, no studies have focused on clinical significance of circulating SIRT3 in ICH patients. In the present study, we performed a prospective cohort study to investigate the prognostic role of plasma SIRT3 in ICH patients.
Materials and Methods
Study Design and Participants
A prospective cohort study was performed of patients with spontaneous ICH, who were admitted to the Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine (Hangzhou, China) between November 2018 and December 2020. All patients were consecutively enrolled according to the inclusion criteria: 1) first-ever stroke, 2) time from the onset of symptoms to admission of 24 h and 3) age of ≥18 years. We further excluded those patients with 1) hematoma resulting from congenital or acquired coagulation abnormalities, hemorrhagic transformation of cerebral infarction, moyamoya disease, intracranial aneurysm, arteriovenous malformation or tumors; 2) a surgical evacuation of hematoma; 3) primary intraventricular bleedings; 4) other previous neurological diseases, like ischemic or hemorrhagic stroke, intracranial tumors and severe head trauma; and 5) other specific diseases or conditions (eg, infection within recent a month, pregnancy, malignancies, uremia, liver cirrhosis, and chronic heart or lung disease). Simultaneously, a group of healthy individuals constituted controls. All procedures in this study were performed in accordance with the 1964 Helsinki declaration. This study was approved by the Institutional Review Board at the Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine (Opinion No: Medical Ethics Review No. 058–01. Written informed consent was acquired from patients’ relatives and controls themselves.
Recorded Variables
Upon arrival at the emergency center, demographics (age and gender), vascular risk factors (hypertension, diabetes mellitus and hyperlipidemia), medication history (use of statins, antiplatelet and anticoagulation drugs) and medical history were inquired. Glasgow Coma Scale (GCS) was utilized to evaluate stroke severity at admission. Each patient underwent a baseline head computerized tomography scan. ICH volumes were calculated using the formula 0.5 × a × b × c.19 The presence of intraventricular and subarachnoid extension of hematoma were also recorded. Non-invasive blood pressure determinations were performed as well as systolic and diastolic arterial blood pressures were recorded upon arrival at emergency center. A Glasgow Outcome Scale (GOS) score of 1–3 at 90 days after stroke onset was assessed as a poor outcome.
Immune Analysis
Peripheral blood samples of ICH patients were collected from 1.0 to 13.0 h (median, 6.0 h; 25th−75th percentiles, 4.5–8.3 h) after stroke and those of controls were acquired at enrollment. Blood glucose and leucocyte count at admission were determined using conventional methods. For measurement of plasma SIRT3 levels, blood samples were separated by centrifugation within 30 minutes and stored at −80°C for subsequent assay. Plasma SIRT3 concentrations were gauged by enzyme-linked immunosorbent assay with commercially available kits (Shanghai Kexing Trading Co., Ltd) in accordance with the manufacturer’s instructions. The measurements were completed by the same technician who was inaccessible to clinical information.
Statistical Analysis
SPSS 25.0 and MedCalc 9.6.4.0 were used for statistical analysis. Figures were plotted using GraphPad Prism version 9.0. Using the Kolmogorov–Smirnov test or Shapiro–Wilk test, the normal distribution of quantitative data was assessed. They were reported as mean ±standard deviation or medians with 25th–75th percentiles. Qualitative data were reported as counts (proportions). Data were compared among multiple groups using the Kruskal–Wallis H-test. Data comparisons between two groups were performed using the χ2 test, Fisher's exact test, t-test, or Mann–Whitney U-test as appropriate. Bivariate correlations were analyzed using Spearman correlation coefficients. Afterwards, the multivariate linear regression model was built to ascertain independently correlated factors with plasma SIRT3 levels, and results were reported as beta and t values, which reflect the point estimate of the linear association. To identify predictors independently related to 90-day clinical outcomes, we constructed a binary logistic regression model, which contained the variables with P < 0.10 in the univariate analysis. Age, intraventricular hemorrhage and infratentorial hemorrhage were not confirmed to be significantly associated with prognosis of ICH in this study, but those factors have been considered as the conventional prognostic determinants of ICH in a variety of clinical studies; thus, the preceding three factors were forced into the multivariate logistic regression model. Results were presented as odds ratios and 95% confidence intervals (CI). Receiver operating characteristic (ROC) curve analysis was performed to assess the prognostic predictive accuracy of the plasma SIRT3, and the area under the curve (AUC) was estimated. A value of P < 0.05 was considered statistically significant.
Results
Participant Selection
During the study, we first analyzed 143 adults with first-ever spontaneous ICH admitted within 24 hours after the onset of stroke symptoms. Then, we excluded 38 patients because of reasons outlined in Figure 1. Finally, 105 ICH cases were included for further analysis. In addition, 72 healthy controls were recruited. There were no significant differences in age, sex percentage, current smoking and drinking between patients and control groups; and the proportions of hypertension, diabetes and hyperlipidemia of patients were significantly higher than those of the control group (Table 1).
 |
Table 1 Comparisons of Demographic Data and Vascular Risk Factors Between Controls and Patients with Acute Intracerebral Hemorrhage |
Patient Characteristics
ICH patients consisted of 69 males and 36 females, as well as their age ranged from 23 to 90 years (median, 65 years; 25th–75th percentiles, 51–76 years). In the aggregate, 27 patients smoked cigarettes and 28 patients consumed alcohol. History of past illness included hypertension (73 cases), diabetes mellitus (19 cases) and hyperlipidemia (25 cases). The patients were admitted at a median value of 5.0 h after symptom onset (range, 0.5–12.0 h; 25th−75th percentiles, 3.0–7.0 h). As regards hemorrhagic clinical severity, the median value of GCS scores was 12 (range, 4–15; 25th-75th percentiles, 10–14). With respect to some radiological parameters, hematomas of 21 cases were located at cerebral lobes; those of 19 cases were found at infratentorial cavity; intraventricular bleedings were present in 19 cases; subarachnoid hemorrhage occurred in 3 cases; and the median value of hematoma volume was 17.88 mL (25th–75th percentiles, 8.62–28.80 mL; range, 1.18–96.70 mL). Via non-invasive arterial pressure measurements, admission systolic arterial pressure and diastolic arterial pressure ranged from 91 to 234 mmHg (mean, 152.69 mmHg; standard deviation, 25.44 mmHg) and from 55 to 137 mmHg (mean, 89.08 mmHg; standard deviation, 16.49 mmHg) respectively. Laboratory tests showed that blood leucocyte count ranged from 3.0 to 18.3×109/l, with a median value of 7.1 ×109/l (25th–75th percentiles, 5.6–9.4 ×109/l), and there was a median value of 6.34 mmol/l at serum glucose levels (range, 3.92–19.10 mmol/l; 25th-75th percentiles, 5.24–8.18 mmol/l). At 90 days after stroke, GOS scores 1, 2, 3, 4 and 5 were found in 3, 14, 29, 5 and 54 patients, respectively; and GOS scores ranged from 1 to 5 (median, 5; 25th–75th percentiles, 3–5). According to the definition of poor outcome (GOS scores 1–3), a total of 46 patients suffered from a poor outcome at 90 days after stroke.
Change of Plasma SIRT3 Levels
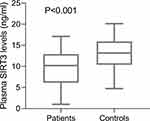
Among this group of ICH patients, plasma SIRT3 levels ranged from 1.02 to 17.11 ng/mL, with a median value of 10.19 ng/mL (25th-75th percentiles, 6.15–12.93 ng/mL). There was 13.17 ng/mL at the median value of plasma SIRT3 levels of healthy controls (range, 4.74–20.13 ng/mL; 25th−75th percentiles, 10.38–15.93 ng/mL). Using Mann–Whitney U-test, it was clear that plasma SIRT3 levels were markedly lower in ICH patients than in healthy controls (Figure 2).
Correlation of Plasma SIRT3 Levels with Hemorrhagic Severity
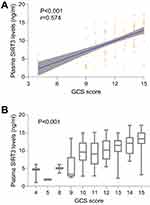
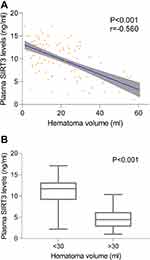
In order to verify correlations of plasma SIRT3 levels with clinical severity indicated by GCS score, GCS score was identified as a qualitative or quantitative variable. Figure 3A shows that there was a significant elevation in plasma SIRT3 levels, with an increase in GCS score. In Figure 3B, there were statistical differences in plasma SIRT3 levels among subgroups with different GCS score, with highest levels in patients with GCS score 15 and lowest levels in those with GCS score 4. Alternatively, in order to demonstrate the relation of plasma SIRT3 levels to radiological severity reflected by hematoma volume, hematoma volume was considered as a quantitative variable and also was dichotomized based on the cutoff value of 30 mL. Figure 4A displays that the significantly negative correlations existed between plasma SIRT3 levels and hematoma volume, and Figure 4B shows that patients with hematoma volume ≥30 mL had substantially lower plasma SIRT3 levels than those with hematoma volume <30 mL.
In Table 2, GCS score, hematoma volume, plasma glucose levels and intraventricular hemorrhage were highly correlated with plasma SIRT3 levels. Furthermore, the preceding four significantly correlated variables were forced into the multivariate linear regression model and thereafter, it was revealed that there was an independent correlation between plasma SIRT3 levels and GCS score (beta, 0.465; 95% CI, 0.107–0.823; t, 2.576; P=0.011) as well as between plasma SIRT3 levels and hematoma volume (beta, −0.098; 95% CI, −0.158 to −0.039; t, −3.282; P=0.001).
 |
Table 2 Bivariate Correlation Analysis Between Plasma SIRT3 Levels and Other Variables in 105 Intracerebral Hemorrhage Patients |
Relationship Between Plasma SIRT3 Levels and Outcome
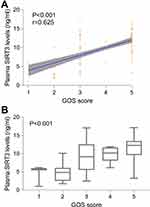
Just as displayed in Figure 5A, plasma SIRT3 levels were remarkably increased with ascending GOS scores. And in Figure 5B, substantial differences existed among subgroups with GOS scores, with highest levels in patients with GOS score 5 and lowest levels in those with GOS score 1. In Table 3, plasma SIRT3 levels were substantially lower in patients with poor outcome than in those with good outcome; as compared to patients with good outcome, those presenting with poor outcome had significantly higher hematoma volume and plasma glucose levels; and GCS score were markedly lower in patients with poor outcome than in other remainders. The variables in Table 3 with P<0.10 in univariate analysis (namely, GCS score, hematoma volume, plasma SIRT3 levels and plasma glucose levels) and other conventional prognostic determinants (namely, age, intraventricular hemorrhage and infratentorial hemorrhage) were incorporated into the binary logistic regression model, and subsequently, it was demonstrated that hematoma volume (odds ratio, 1.071; 95% CI, 1.002–1.144; P=0.042) and plasma SIRT3 levels (odds ratio, 0.831; 95% CI, 0.702–0.984; P=0.031) retained as the two independent predictors of poor outcome.
 |
Table 3 Demographic, Clinical, Radiological and Biochemical Factors for 90-Day Poor Outcome After Acute Intracerebral Hemorrhage |
Moreover, under ROC curve, plasma SIRT3 levels remarkably predicted post-stroke 90-day poor outcome among this group of ICH patients (AUC, 0.801; 95% CI, 0.711–0.872); and plasma SIRT3 level ≤7.38 ng/mL distinguished patients with development of poor 90-day outcome with specificity and sensitivity values of 93.22% and 63.04% (Youden index J, 0.5626) respectively (Figure 6A). Moreover, Figure 6B shows that its discriminatory ability was equivalent to those of GCS score (AUC, 0.846; 95% CI, 0.763–0.909; P=0.318) and hematoma volume (AUC, 0.849; 95% CI, 0.766–0.911; P=0.302).
Discussion
To the best of our knowledge, no data have been available regarding change of SIRT3 levels in the peripheral blood of patients with acute brain injury and its association with severity and prognosis. In the present study, we demonstrated that plasma SIRT3 levels were substantially decreased in ICH patients and were highly associated with disease severity and patient prognosis. Collectively, plasma SIRT3 may be a biochemical marker of acute brain injury.
The accumulating data have shown that SIRT3 may be brain-protective via anti-oxidative activity. In an in vitro model subjected to acute glucose deprivation or acute oxygen–glucose deprivation, the overexpression of SIRT3 significantly diminished oxidative and apoptotic injury to PC12 neurons via reducing ROS and maintaining ATP production.20 Also, using SIRT3 overexpressing plasmid, cortical neurons were free of excitotoxic damage induced by N-methyl-
In normal cerebral cortex of rats, SIRT3 was prominently expressed in neurons.26 Expression of SIRT3 protein was upregulated in in vitro-cultured neurons pretreated with N-methyl-
Serum SIRT3 levels were associated with 1-month mortality in patients with severe community-acquired pneumonia.30 However, there is a paucity of data available regarding the relationship between circulating SIRT3 levels and illness severity in addition to clinical outcomes after acute brain injury. In this study, we found that there was an independent correlation between plasma SIRT3 levels and GCS scores, as well as between plasma SIRT3 levels and hematoma volume; in addition, plasma SIRT3 levels of ICH patients were highly correlated with GOS score. Moreover, besides hematoma volume, levels of plasma SIRT3 were independently related to the 90-day adverse prognosis (GOS score 1–3) of ICH patients. Intriguingly, plasma SIRT3 levels showed similar prognostic predictive ability, as compared to GCS scores and hematoma volume. In conclusion, plasma SIRT3 may be a promising biomarker for the prognosis of ICH.
However, there are several limitations in this study. First, this study recruited almost 100 patients and therefore there is a characteristic of small sample size. Maybe, this is a preliminary study for demonstrating the role of plasma SIRT3 in prognosis of ICH. In future, a larger cohort study is warranted to validate whether plasma SIRT3 may be a biochemical marker of acute brain injury. Second, the main goal of this study is to investigate the impact of admission plasma SIRT3 levels on ICH prognosis. Admittedly, it may be significant to observe dynamic change inf plasma SIRT3 levels after stroke and further to discern the evolution of the plasma levels of SIRT3 during the follow-up period. Thus, such an investigation of circulating SIRT3 levels is warranted. Third, operation must influence postoperative levels of plasma SIRT3 levels. However, its preoperative levels are not affected by operation. Undoubtedly, operation may be related to prognosis of ICH patients. Thus, operation is a confounding factor of prognosis. In this study, we have excluded those patients undergoing a surgical evacuation of hematoma. However, it should be a good topic to determine how a surgical evacuation of hematoma may change the plasma SIRT3 levels. Fourth, accumulating evidence has shown that the role of plasma SIRT3 in acute brain injury may be protective via anti-inflammation and anti-oxidation.20–22 Nevertheless, acute brain injury may be caused by a variety of underlying conditions, that reflect a wide spectrum of related pathogenetic mechanisms. Thus, further animal experiments and clinical studies are needed to discover the specific mechanisms of the protective effect of SIRT3 in acute brain injury. Fifth, clinically, many patients do not undergo a surgical operation, so direct extraction of hematoma samples is impossible. Nevertheless, plasma measurement is more convenient. However, it is undeniable that direct extraction of hematoma samples may have some clinical implications. Last, generally, GOS is frequently used to assess functional outcome of ICH, and 90-day follow-up is a conventional modality for assessing ICH prognosis.31,32 Clearly, modified Rankin scale is also an alternative for function assessment of ICH.33 Nevertheless, it may be of clinical value that follow-up period is extended to 6 months and even 1 year.34
Conclusions
To the best of our knowledge, this study, for the first time, demonstrated that there is a significantly lower plasma SIRT3 levels in ICH patients than in healthy controls; and plasma SIRT3 levels had close correlations with disease severity reflected by GCS scores and hematoma volume, and independently predicted poor outcome of ICH patients. Thus, it is hypothesized that SIRT3 may be implicated in secondary brain injury after ICH and circulating SIRT3 may have the potential to represent a prognostic biochemical marker of ICH.
Abbreviations
GCS, Glasgow Coma Scale; ICH, intracerebral hemorrhage; CT, computerized tomography; ROC, receiver operating characteristic; AUC, area under curve; 95% CI, 95% confidence interval; ROS, reactive oxygen species.
Data Sharing Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Acknowledgments
The authors thank all staffs in Department of Neurosurgery, the Affiliated Hangzhou First People’s Hospital, Zhejiang University School of Medicine (Hangzhou, China) for their technical support.
Funding
This work is financially supported by Key Research and Development Projects of Zhejiang Province (No. 2020C03071) and the Construction Fund of Medical Key Disciplines of Hangzhou (No. OO20200485, No. OO20200055).
Disclosure
The authors have no conflicts of interest in this work.
References
1. Al-Shahi Salman R, Frantzias J, Lee RJ, et al. Absolute risk and predictors of the growth of acute spontaneous intracerebral haemorrhage: a systematic review and meta-analysis of individual patient data. Lancet Neurol. 2018;17(10):885–894. doi:10.1016/S1474-4422(18)30253-9
2. van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 2010;9(2):167–176. doi:10.1016/S1474-4422(09)70340-0
3. Beez T, Steiger HJ, Etminan N. Pharmacological targeting of secondary brain damage following ischemic or hemorrhagic stroke, traumatic brain injury, and bacterial meningitis - a systematic review and meta-analysis. BMC Neurol. 2017;17(1):209. doi:10.1186/s12883-017-0994-z
4. Murthy SB, Moradiya Y, Dawson J, Lees KR, Hanley DF, Ziai WC; VISTA-ICH collaborators. Perihematomal edema and functional outcomes in intracerebral hemorrhage: influence of hematoma volume and location. Stroke. 2015;46(11):3088–3092. doi:10.1161/STROKEAHA.115.010054
5. Singh S, Singh TG. Emerging perspectives on mitochondrial dysfunctioning and inflammation in epileptogenesis. Inflamm Res. 2021;70(10–12):1027–1042. doi:10.1007/s00011-021-01511-9
6. Brunswick AS, Hwang BY, Appelboom G, Hwang RY, Piazza MA, Connolly ES. Serum biomarkers of spontaneous intracerebral hemorrhage induced secondary brain injury. J Neurol Sci. 2012;321(1–2):1–10. doi:10.1016/j.jns.2012.06.008
7. Prentice H, Modi JP, Wu JY. Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid Med Cell Longev. 2015;2015:964518. doi:10.1155/2015/964518
8. Duan X, Wen Z, Shen H, Shen M, Chen G. Intracerebral hemorrhage, oxidative stress, and antioxidant therapy. Oxid Med Cell Longev. 2016;2016:1203285. doi:10.1155/2016/1203285
9. Troiani Z, Ascanio L, Rossitto CP, et al. Prognostic utility of serum biomarkers in intracerebral hemorrhage: a systematic review. Neurorehabil Neural Repair. 2021;35(11):946–959. doi:10.1177/15459683211041314
10. Lusk JB, Troy J, Nowacki N, et al. An exploratory analysis of biomarkers of perihematomal edema in the CN-105 in participants with acute supratentorial intracerebral hemorrhage (CATCH) trial. J Stroke Cerebrovasc Dis. 2022;31(8):106600. doi:10.1016/j.jstrokecerebrovasdis.2022.106600
11. Sagar R, Kumar A, Verma V, et al. Incremental accuracy of blood biomarkers for predicting clinical outcomes after intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2021;30(3):105537. doi:10.1016/j.jstrokecerebrovasdis.2020.105537
12. Villalba JM, Alcaín FJ. Sirtuin activators and inhibitors. Biofactors. 2012;38(5):349–359. doi:10.1002/biof.1032
13. Ahn BH, Kim HS, Song S, et al. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc Natl Acad Sci U S A. 2008;105(38):14447–14452. doi:10.1073/pnas.0803790105
14. Bell EL, Guarente L. The SirT3 divining rod points to oxidative stress. Mol Cell. 2011;42(5):561–568. doi:10.1016/j.molcel.2011.05.008
15. Papa L, Germain D. SirT3 regulates the mitochondrial unfolded protein response. Mol Cell Biol. 2014;34(4):699–710. doi:10.1128/MCB.01337-13
16. Cheung KG, Cole LK, Xiang B, et al. Sirtuin-3 (SIRT3) protein attenuates doxorubicin-induced oxidative stress and improves mitochondrial respiration in H9c2 cardiomyocytes. J Biol Chem. 2015;290(17):10981–10993. doi:10.1074/jbc.M114.607960
17. Huang W, Huang Y, Huang RQ, et al. SIRT3 expression decreases with reactive oxygen species generation in rat cortical neurons during early brain injury induced by experimental subarachnoid hemorrhage. Biomed Res Int. 2016;2016:8263926. doi:10.1155/2016/8263926
18. Zheng J, Shi L, Liang F, et al. Sirt3 ameliorates oxidative stress and mitochondrial dysfunction after intracerebral hemorrhage in diabetic rats. Front Neurosci. 2018;12:414. doi:10.3389/fnins.2018.00414
19. Kothari RU, Brott T, Broderick JP, et al. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996;27(8):1304–1305. doi:10.1161/01.str.27.8.1304
20. Shulyakova N, Sidorova-Darmos E, Fong J, Zhang G, Mills LR, Eubanks JH. Over-expression of the Sirt3 sirtuin protects neuronally differentiated PC12 cells from degeneration induced by oxidative stress and trophic withdrawal. Brain Res. 2014;1587:40–53. doi:10.1016/j.brainres.2014.08.066
21. Kim SH, Lu HF, Alano CC, Combs C. Neuronal Sirt3 protects against excitotoxic injury in mouse cortical neuron culture. PLoS One. 2011;6(3):e14731. doi:10.1371/journal.pone.0014731
22. Wang Q, Li L, Li CY, Pei Z, Zhou M, Li N. SIRT3 protects cells from hypoxia via PGC-1α- and MnSOD-dependent pathways. Neuroscience. 2015;286:109–121. doi:10.1016/j.neuroscience.2014.11.045
23. Kurundkar D, Kurundkar AR, Bone NB, et al. SIRT3 diminishes inflammation and mitigates endotoxin-induced acute lung injury. JCI Insight. 2019;4(1):e120722. doi:10.1172/jci.insight.120722
24. Wang Y, Li C, Gu J, et al. Celastrol exerts anti-inflammatory effect in liver fibrosis via activation of AMPK-SIRT3 signalling. J Cell Mol Med. 2020;24(1):941–953. doi:10.1111/jcmm.14805
25. Kim D, Park W, Lee S, Kim W, Park SK, Kang KP. Absence of Sirt3 aggravates cisplatin nephrotoxicity via enhanced renal tubular apoptosis and inflammation. Mol Med Rep. 2018;18(4):3665–3672. doi:10.3892/mmr.2018.9350
26. Guo J, Zhang XL, Bao ZR, et al. Gastrodin regulates the notch signaling pathway and Sirt3 in activated microglia in cerebral hypoxic-ischemia neonatal rats and in activated BV-2 microglia. Neuromolecular Med. 2021;23(3):348–362. doi:10.1007/s12017-020-08627-x
27. Wu X, Luo J, Liu H, Cui W, Feng D, Qu Y. SIRT3 protects against early brain injury following subarachnoid hemorrhage via promoting mitochondrial fusion in an AMPK dependent manner. Chin Neurosurg J. 2020;6:1. doi:10.1186/s41016-019-0182-7
28. Wang T, Cao Y, Zheng Q, et al. SENP1-Sirt3 signaling controls mitochondrial protein acetylation and metabolism. Mol Cell. 2019;75(4):823–34.e5. doi:10.1016/j.molcel.2019.06.008
29. Xu XJ, Yang MS, Zhang B, Niu F, Dong JQ, Liu BY. Glucose metabolism: a link between traumatic brain injury and Alzheimer’s disease. Chin J Traumatol. 2021;24(1):5–10. doi:10.1016/j.cjtee.2020.10.001
30. Zhu W, Chen P, Hu L, Deng L. Serum levels of SIRT3 and other inflammatory factors are associated with clinical outcomes and prognosis in severe community-acquired pneumonia in adults: a prospective study. Medicine. 2021;100(32):e26721. doi:10.1097/MD.0000000000026721
31. Cai Y, Zhuang YK, Wu XY, et al. Serum hypoxia-inducible factor 1alpha levels correlate with outcomes after intracerebral hemorrhage. Ther Clin Risk Manag. 2021;17:717–726. doi:10.2147/TCRM.S313433
32. Leasure AC, Kuohn LR, Vanent KN, et al. Association of serum IL-6 (Interleukin 6) with functional outcome after intracerebral hemorrhage. Stroke. 2021;52(5):1733–1740. doi:10.1161/STROKEAHA.120.032888
33. Law ZK, Dineen R, England TJ, et al. Predictors and outcomes of neurological deterioration in intracerebral hemorrhage: results from the TICH-2 randomized controlled trial. Transl Stroke Res. 2021;12(2):275–283. doi:10.1007/s12975-020-00845-6
34. Lun R, Yogendrakumar V, Ramsay T, et al. Predicting long-term outcomes in acute intracerebral haemorrhage using delayed prognostication scores. Stroke Vasc Neurol. 2021;6(4):536–541. doi:10.1136/svn-2020-000656
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.