Back to Journals » Cancer Management and Research » Volume 11
Phyllodes tumor of the breast: diagnosis, management and outcome during a 10-year experience
Authors Ditsatham C , Chongruksut W
Received 9 May 2019
Accepted for publication 12 August 2019
Published 19 August 2019 Volume 2019:11 Pages 7805—7811
DOI https://doi.org/10.2147/CMAR.S215039
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chien-Feng Li
Chagkrit Ditsatham,1 Wilaiwan Chongruksut2
1Division of Head, Neck, and Breast Surgery, Department of Surgery, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand; 2Research Unit, Department of Surgery, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
Correspondence: Chagkrit Ditsatham
Department of Surgery, Faculty of Medicine, Chiang Mai University, 110 Intavaroros Road, Amphoe Muang, Chiang Mai 50200, Thailand
Tel +66 5 394 5532
Fax +66 5 394 6139
Email [email protected]
Background: Phyllodes tumor is a very rare tumor of the breast, incidence being 0.3–0.9% of all breast tumors. Phyllodes tumors are classified into three grades: benign, borderline or malignant. The aim of this study was to investigate patient characteristics, imaging characteristic findings, surgical treatment and outcome of phyllodes tumor.
Materials and methods: This is a retrospective review of phyllodes tumor patients who had undergone surgical management between 1 January 2006 and 31 December 2015 in Head, Neck and Breast Unit, Department of Surgery, Chiang Mai University Hospital.
Results: A total of 188 phyllodes tumor patients were included in this study. The average age of the patients was 35.6 years. The majority of the tumors were of lobulated shape. Tumor morphology from imaging: lobulated shaped tumor 112 cases, oval-shape 37 cases, round shape 8 cases and irregular 5 cases. Surgical management is the standard procedure for treatment of phyllodes tumors. A wide local excision was performed in 147 cases, mastectomy in 20 cases (modified radical mastectomy in 2 cases) and wide excision followed by mastectomy 21 cases. The final pathological diagnoses of the phyllodes tumors were: benign 118 cases (62%), borderline 33 cases (18%) and malignant in 37 cases (20%). The margin did not affect recurrence in all types.
Conclusion: The most frequent is a benign phyllodes tumor. Surgical treatment to remove the tumor with a free margin is acceptable with no recurrence difference from a wider margin. However, younger age and malignant phyllodes tumor are the two highest risk factors for recurrence.
Keywords: phyllodes tumor, breast, surgical margin
Introduction
Phyllodes tumor is a very rare tumor of the breast, incidence being between 0.3% and 0.9% of all breast tumors.1 They are most commonly found in women aged between 40 and 50 years.2 The World Health Organization (WHO) has published guidelines classifying this tumor into benign, borderline or malignant according to the histological features such as stromal cellularity, stromal overgrowth, stromal atypia, mitoses/high power field and tumor margin. The most common grade of phyllodes tumor is benign, benign tumor occurrence being between 60% and 75% of the cases.3 In the past, several literatures recommended that the standard treatment for phyllodes tumor is surgery to remove tumor with a free margin of at least 1 cm to reduce local recurrent.4–6
In this paper, we aimed to investigate patient characteristics, characteristic imaging findings, histopathological diagnostics, surgical treatment and therapeutic outcome of phyllodes tumor patients at Chiang Mai University Hospital over a 10-year period. We also investigated factors related to recurrence.
Materials and methods
This is a retrospective review of patients who were diagnosed with a phyllodes tumor and underwent surgical management. Data were collected from electronic medical records from 1 January 2006 to 31 December 2015 in the Head, Neck and Breast Unit, Department of Surgery, Chiang Mai University Hospital.
Patients data were collected from physical examination. These included age, menopausal status, location of tumor and tumor size from physical examination. Mammography and ultrasonography images were used to evaluate each mass. Details pertinent to tumor size, shape and margin were collected. Three operation procedures for surgical management included wide excision, wide excision followed by mastectomy if the margin was inadequate and mastectomy. The final pathology diagnoses following the operation were divided into benign, borderline and malignant phyllodes tumors in accordance with the WHO criteria. All patients were followed up at least 3 years after operation to check for recurrence. Any recurrence was reported as local (new tumor was found in ipsilateral breast) or distant metastatic recurrence.
Statistical analysis
Categorical data are presented as frequency and percentage. Fisher’s exact test was used as a means of comparing the categorical variables. Continuous data are presented as mean and standard deviation. Differences between the three groups of phyllodes tumor were assessed using a one-way analysis of variance (ANOVA) test. Multivariable Cox’s proportional hazard model with a step backward elimination method was used to identify the prognostic factors of recurrent phyllodes tumor. The overall survival curves were estimated using the Kaplan–Meier method. All statistical analyses were performed using STATA program version 11.0 (Serial number: 40110561832)
Results
Patient characteristics
The study was submitted to the ethics review board of the Faculty of Medicine Chiang Mai University and approved it. Our retrospective study was scrutinized without requiring consent from patients, because this study was low risk and we collected data as anonymous.
In this study, 188 patients with phyllodes tumor who had been diagnosed and treated in our hospital were included. All patients presented at the hospital with a palpable breast mass. The average age was 35.6 years old (youngest 13 and oldest 77). One hundred and sixty-five women were pre-menopausal and 23 post-menopausal. The phyllodes tumor was in the right breast in 113 cases and the left breast in 75 cases. The mass was located in the upper outer quadrant in 78 cases, the upper inner quadrant in 48 cases, the lower outer quadrant in 19 cases, the lower inner quadrant in 11 cases and centrally in 32 cases (Table 1).
 |
Table 1 Patient and tumor characteristics |
Imaging characteristics
Mammogram and ultrasonography were used to evaluate 182 cases. Ultrasonography alone was used in 74 cases and no imaging took place in 31 cases (Table 1).
Tumor consistency was divided into two main groups: solid 139 cases and mixed solid-cystic 15 cases.
Tumor shapes were: lobulated 112 cases (Figure 1); oval 37 cases; round 8 cases and irregular in 5 cases.
The groupings of the margin border of the tumor were: well defined 149 cases and ill defined 5 cases.
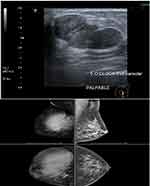 |
Figure 1 Mammogram and ultrasonography showed large lobulated patient who was diagnosed phyllodes tumor. |
Types of operation
In benign phyllodes tumor group, wide excision was performed in 116 cases, mastectomy in 1 case and excision followed by mastectomy 1 case. In borderline phyllodes tumor group, wide excision was performed in 23 cases, mastectomy in 6 cases and excision followed by mastectomy 4 cases. In malignant phyllodes tumor group, wide excision was performed in 8 cases, mastectomy in 13 cases (modified radical mastectomy in 2 cases; 1 patient was negative for lymph node metastasis and 1 patient had metastatic lymph nodes 1 in 13 nodes, indication for 2 patients was axillary lymph nodes palpable without preoperative proven metastasis) and excision followed by mastectomy 16 cases. A surgical free margin (at least 1 cm from tumor) was recorded in 84 cases. A surgical positive margin after the first operation was found in 53 cases in benign, 21 cases in borderline and 29 cases in malignant phyllodes tumor (Table 1). In case of positive margin, 21 cases divided into 1 case of benign, 4 cases of borderline and 16 cases were malignant undergone re-operation and a free margin was finally achieved. Location of the tumor did not affect the free surgical margin (Table 2). In univariate analysis, we found that the type of operation and margin status did not affect recurrence. (Table 3)
 |
Table 2 Relation of the location of the tumor (cm) |
 |
Table 3 Prognostic factors of disease recurrence analyzed by univariable and multivariable Cox proportional hazard model |
Final pathology
Out of the 188 cases of phyllodes tumors, 118 cases were benign (62%), 33 (18%) were borderline and 37 cases (20%) were malignant (Table 1). If the size of the tumor is more than 6 cm, it should be suspected for malignant phyllodes tumor (Table 1).
Adjuvant treatment
In our hospital, we considered radiotherapy as an adjunctive treatment for malignant phyllodes tumors and some borderline phyllodes tumors. Twenty-three cases received adjuvant radiotherapy: 5 of these were borderline and 19 were malignant. No systemic therapy was given in any patient.
Follow up and recurrence
All patients were followed up with physical examination at outpatient department (OPD) every 3 months in first year, every 6 months in second year and then every 1 year follow-up, and imaging was performed every year with mammogram and ultrasonography. Benign phyllodes tumor patients had a median follow-up time of 68.8 months, borderline 86.1 and malignant 70.6 months (Table 1). Out of 181 cases, 9 cases experienced recurrence: 5 benign phyllodes tumor (local recurrence), 1 borderline and 3 malignant (1 case developed pulmonary metastasis). All recurrence was no upgrade rate found. We found that younger age (≤45 yrs) malignant phyllodes tumor and size of the tumor are the highest risk factors for recurrence after operation (Table 3).
The standard treatment of phyllodes is surgical excision with a margin of at least 1 cm to decrease the rate of recurrence.7–9 In our patients (all grades of phyllodes tumor), a surgical margin of less than 1 cm did not affect recurrence (Figure 2, P-value=0.39). Subgroup analysis was done by phyllodes grade. There was statistical difference in margin of malignant phyllodes tumor to risk for recurrence (P-value=0.022) but not affected in benign and borderline phyllodes tumor (P-value=0.334 and 0.668, respectively) (Figure 3).
 |
Figure 2 Kaplan–Meier curve showed recurrence by surgical margin of all phyllodes tumors (P=0.737). |
 |
Figure 3 Kaplan–Meier curve showed recurrence of phyllodes tumor in each classification between free surgical margin and positive margin (P-value). |
Discussion
Phyllodes tumors are rare fibroepithelial tumors found in about 0.5% of the breast neoplasm incidence.10 The most common age range for development is 45–49 years old which is older than the fibroadenoma highest risk group by 20 years;11 however, in our patient sample, the average age is 35 years old. Clinical presentation is usually a painless rapid growing mass. Images from mammography and ultrasonography should indicate suspect phyllodes tumor if the tumor is a well-circumscribed oval or lobulated mass with rounded borders.4 Most tumors, 112 cases, in our hospital records, involved a lobulated shaped mass. Most of the imaging characteristic is difficult to distinguish fibroadenoma from phyllodes tumor.12,13 The size of the tumor should be considered. The average size of fibroadenoma was 2 cm while phyllodes tumor was 4–7 cm.4,10,14,15 In our study, the average size of phyllodes tumor is centimeters and more increase in size followed by the type of phyllodes (3.5 cm in benign, 6.6 cm in borderline and 8.2 cm in malignant). Surgical management to remove the tumor is the mainstay for treatment of all classifications of phyllodes tumors with the aim of achieving a negative margin of at least 1 cm to reduce local recurrence.7,8,10,16 If the surgical margin does not reach 1 cm, further management is different in each classification of phyllodes. Benign phyllodes tumors should be watched carefully because positive margin in benign did not relate to local recurrence.17 But in cases of borderline and malignant phyllodes tumors, a re-excision or mastectomy to achieve an adequate margin should be carried out.4,18 In our study, we found that borderline phyllodes tumors did not differ recurrence rate between negative and positive margin (P-value=0.669) while the malignant phyllodes tumors were to recur more frequently in cases of an inadequate margin significantly (P-value=0.022) (Figure 3). This result was similar to Lu et al.18 Adjuvant radiotherapy (RT) in cases of phyllodes is still controversial. National Comprehensive Cancer Network (NCCN) and the other studies suggest the use of RT in recurrent cases of malignant phyllodes.19 Some authors suggest adjuvant RT to decrease the possibility of local recurrence in both borderline and malignant phyllodes tumor patients who are treated with breast-conserving surgery.20 However, RT did not affect overall survival (OS) and disease-free survival (DFS).20,21 In our institution, we consider the use of RT in the case of malignant phyllodes tumor patients undergoing surgical management with wide excision.
In conclusion from our study, the surgical margin did not significantly affect the risk of local recurrence in all subtypes and resulted in a good outcome in benign and borderline cases. However, malignant phyllodes patients are at a more increased risk of recurrence than other types, so a wide margin should always be considered in this type.
Acknowledgment
This work was supported by the Faculty of Medicine Research Fund, Chiang Mai University (Grant no. 046/2561), Chiang Mai, Thailand.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Guerrero MA, Ballard BR, Grau AM. Malignant phyllodes tumor of the breast: review of the literature and case report of stromal overgrowth. Surg Oncol. 2003;12(1):27–37.
2. Spitaleri G, Toesca A, Botteri E, et al. Breast phyllodes tumor: a review of literature and a single center retrospective series analysis. Crit Rev Oncol Hematol. 2013;88(2):427–436. doi:10.1016/j.critrevonc.2013.06.005
3. Zhang Y, Kleer CG. Phyllodes tumor of the breast: histopathologic features, differential diagnosis, and molecular/genetic updates. Arch Pathol Lab Med. 2016;140(7):665–671. doi:10.5858/arpa.2016-0042-RA
4. Mishra SP, Tiwary SK, Mishra M, Khanna AK. Phyllodes tumor of breast: a review article. ISRN Surg. 2013;2013:361469. doi:10.1155/2013/361469
5. Mangi AA, Smith BL, Gadd MA, Tanabe KK, Ott MJ, Souba WW. Surgical management of phyllodes tumors. Arch Surg. 1999;134(5):
6. Granic M, Opric D, Pupic G, et al. [Surgical methods for the treatment of breast phyllodes tumors–a report of 319 cases]. Acta Chir Iugosl. 2006;53(1):57–62.
7. Chaney AW, Pollack A, McNeese MD, et al. Primary treatment of cystosarcoma phyllodes of the breast. Cancer. 2000;89(7):1502–1511. doi:10.1002/1097-0142(20001001)89:7<1502::aid-cncr13>3.0.co;2-p
8. Guillot E, Couturaud B, Reyal F, et al. Management of phyllodes breast tumors. Breast J. 2011;17(2):129–137. doi:10.1111/j.1524-4741.2010.01045.x
9. Sabban F, Collinet P, Lucot JP, Boman F, Leroy JL, Vinatier D. [Phyllodes tumor of the breast: analysis of 8 patients]. J Gynecol Obstet Biol Reprod (Paris). 2005;34(3 Pt 1):252–256.
10. Reinfuss M, Mitus J, Duda K, Stelmach A, Rys J, Smolak K. The treatment and prognosis of patients with phyllodes tumor of the breast: an analysis of 170 cases. Cancer. 1996;77(5):910–916. doi:10.1002/(sici)1097-0142(19960301)77:5<910::aid-cncr16>3.0.co;2-6
11. Bernstein L, Deapen D, Ross RK. The descriptive epidemiology of malignant cystosarcoma phyllodes tumors of the breast. Cancer. 1993;71(10):3020–3024. doi:10.1002/1097-0142(19930515)71:10<3020::aid-cncr2820711022>3.0.co;2-g
12. Gatta G, Iaselli F, Parlato V, Di Grezia G, Grassi R, Rotondo A. Differential diagnosis between fibroadenoma, giant fibroadenoma and phyllodes tumour: sonographic features and core needle biopsy. Radiol Med. 2011;116(6):905–918. doi:10.1007/s11547-011-0672-y
13. Yilmaz E, Sal S, Lebe B. Differentiation of phyllodes tumors versus fibroadenomas. Acta Radiol. 2002;43(1):34–39.
14. Krishnamurthy S, Ashfaq R, Shin HJ, Sneige N. Distinction of phyllodes tumor from fibroadenoma: a reappraisal of an old problem. Cancer. 2000;90(6):342–349.
15. Barrio AV, Clark BD, Goldberg JI, et al. Clinicopathologic features and long-term outcomes of 293 phyllodes tumors of the breast. Ann Surg Oncol. 2007;14(10):2961–2970. doi:10.1245/s10434-007-9439-z
16. Choi N, Kim K, Shin KH, et al. The characteristics of local recurrence after breast-conserving surgery alone for malignant and borderline phyllodes tumors of the breast (KROG 16-08). Clin Breast Cancer. 2019. doi:10.1016/j.clbc.2019.04.003
17. Ouyang Q, Li S, Tan C, et al. Benign phyllodes tumor of the breast diagnosed after ultrasound-guided vacuum-assisted biopsy: surgical excision or wait-and-watch? Ann Surg Oncol. 2016;23(4):1129–1134. doi:10.1245/s10434-015-4990-5
18. Lu Y, Chen Y, Zhu L, et al. Local recurrence of benign, borderline, and malignant phyllodes tumors of the breast: a systematic review and meta-analysis. Ann Surg Oncol. 2019;26(5):1263–1275. doi:10.1245/s10434-018-07134-5
19. Carlson RW, Allred DC, Anderson BO, et al. Metastatic breast cancer, version 1.2012: featured updates to the NCCN guidelines. J Natl Compr Canc Netw. 2012;10(7):821–829.
20. Zeng S, Zhang X, Yang D, Wang X, Ren G. Effects of adjuvant radiotherapy on borderline and malignant phyllodes tumors: a systematic review and meta-analysis. Mol Clin Oncol. 2015;3(3):663–671. doi:10.3892/mco.2015.503
21. Gnerlich JL, Williams RT, Yao K, Jaskowiak N, Kulkarni SA. Utilization of radiotherapy for malignant phyllodes tumors: analysis of the National Cancer Data Base, 1998-2009. Ann Surg Oncol. 2014;21(4):1222–1230. doi:10.1245/s10434-013-3395-6
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
