Back to Journals » Drug Design, Development and Therapy » Volume 14
Perindopril in Breast Milk and Determination of Breastfed Infant Exposure: A Prospective Observational Study
Authors Leggett C, Lwin EMP , Ritchie U, Song Y , Gerber JP, Turner S, Hague WM , Stark M, Upton R , Garg S
Received 22 November 2019
Accepted for publication 10 February 2020
Published 2 March 2020 Volume 2020:14 Pages 961—967
DOI https://doi.org/10.2147/DDDT.S239704
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Yan Zhu
Catherine Leggett,1,* Ei Mon Phyo Lwin,2,* Usha Ritchie,1 Yunmei Song,2 Jacobus P Gerber,2 Sean Turner,1 William M Hague,3,4 Michael Stark,3,5 Richard Upton,2 Sanjay Garg2
1SA Pharmacy, Women’s and Children’s Hospital, North Adelaide, SA 5006, Australia; 2School of Pharmacy and Medical Sciences, Reid Building, City East Campus, University of South Australia, Adelaide, SA 5000, Australia; 3Robinson Research Institute, University of Adelaide, North Adelaide, SA 5006, Australia; 4Obstetric Medicine, Women’s and Children’s Hospital, North Adelaide, SA 5006, Australia; 5Neonatal Medicine, Women’s and Children’s Hospital, North Adelaide, SA 5006, Australia
*These authors contributed equally to this work
Correspondence: Sanjay Garg
Centre for Pharmaceutical Innovation and Development (CPID), School of Pharmacy and Medical Sciences, University of South Australia, Adelaide, SA 5000, Australia
Tel +61 8 830 21575
Fax +61 8 830 22389
Email [email protected]
Objective: This study aimed to quantify the amount of perindopril and its active metabolite perindoprilat present in breast milk and corresponding maternal and infant plasma concentrations.
Design: Prospective, longitudinal, observational.
Setting: Tertiary specialist paediatric and obstetric hospital in Adelaide, South Australia.
Population: Breastfeeding women actively treated with perindopril for hypertensive disorders postpartum.
Methods: Eight breast milk samples and a single plasma sample were collected from each participant over a 24 hrs period, and plasma samples were taken from eligible breastfed infants. Breast milk and plasma concentrations of perindopril and perindoprilat were analysed using a validated Liquid Chromatography tandem-Mass Spectrometry (LC-MS/MS) method.
Main Outcome Measures: Mean breast milk concentrations of perindopril and perindoprilat, Relative Infant Dose (RID) < 10%, and Theoretical Infant Dose (TID).
Results: Ten women and three infants participated in the study. The mean concentration of perindopril in breast milk for each participant ranged from 0.003 to 1.2 ng/mL and perindoprilat 0.2– 36 ng/mL. RID for perindopril was 0.0005– 0.2% and perindoprilat 0.03– 4.6%. TID for perindopril was 0.00045– 0.18 μg/kg/day and perindoprilat 0.032– 5.4 μg/kg/day. Infant plasma levels for perindopril ranged from 0.44 to 1.12 ng/mL and perindoprilat undetectable – 10.14 ng/mL. Maternal reports described normal infant growth and development.
Conclusion: Infant exposure to perindopril and perindoprilat through breast milk is low. However, some infants were found to have plasma perindoprilat concentrations consistent with pharmacodynamic effects. Perindopril may be used in mothers of healthy term infants, provided the infant is carefully monitored.
Keywords: perindopril, perindoprilat, LC-MS/MS, human plasma, human milk, clinical lactation, infant drug exposure
Introduction
Breast milk is the optimal source of nutrition for infants and the benefits of breastfeeding are well established for both mother and child.1,2 The World Health Organization (WHO) states that ‘breastfeeding is an unequalled way of providing ideal food for the healthy growth and development of infants’ and recommends exclusive breastfeeding for 6 months.3,4 Maternal medication use has been highlighted as a potential barrier to breastfeeding due to concern regarding infants’ “exposure” through human milk.5
Hypertension has been reported to occur in 10–15% of pregnancies and often persists into the postpartum period, requiring pharmacotherapy.6,7 Hypertension may be pre-existing or arise from pregnancy complications, such as pre-eclampsia. Hypertensive disorders during pregnancy and postpartum can lead to a persistently increased cardiovascular disease risk and the need for long-term antihypertensive therapy.8–10 Angiotensin-converting enzyme (ACE) inhibitors are commonly used in the management of hypertension and are suitable first-line agents outside of pregnancy.11 ACE inhibitors are favoured for the treatment of hypertension during the postpartum period as they have fewer adverse central nervous system effects (ie. sedation) and are therapeutically superior to commonly used agents during pregnancy, such as methyldopa and labetalol.
Perindopril is an ACE inhibitor, exhibiting high lipophilicity and local inhibition of the renin-angiotensin-aldosterone system in tissues such as the heart, kidneys, adrenal glands and blood vessels.11 It is marketed as two separate salt formulations (erbumine and arginine), which are considered bioequivalent. The efficacy, safety and tolerability of perindopril are well established in adult patients for the treatment of hypertension and heart failure.12 Perindopril has been shown to have a longer duration of action, providing 24 hour blood pressure control with a single daily dose.13 Notably, perindopril may have advantages for clinical practice due to once daily dosing and potential for improved adherence with therapy in breastfeeding mothers.
Few studies have investigated the use of ACE inhibitors in women who are breastfeeding. This study aimed to quantify the amount of perindopril and its active metabolite perindoprilat present in breast milk and the corresponding maternal and infant plasma concentrations in order to inform clinical practice.
Method
Design and Setting
This prospective, longitudinal observational study was conducted at the Women’s and Children’s Hospital Adelaide, a tertiary specialist paediatric and obstetric hospital in South Australia. Recruitment took place over an 18-month period from January 2016 to June 2017. Eligible participants required a diagnosis of a hypertensive condition post-partum. Approval was granted by the Women’s and Children’s Health Network (WCHN) Human Research Ethics Committee and the University of South Australia Research Ethics Committee, and our study was conducted in accordance with the Declaration of Helsinki.
Study Participants
Breastfeeding women actively treated with perindopril were referred to the investigation team by WCHN clinicians. Women were eligible for inclusion in the study if they were (1) ≥18 years of age and able to provide informed consent, (2) on a stable dose of perindopril arginine or perindopril erbumine (at steady state), (3) breastfeeding (defined as either breastfeeding or expressing) and (4) willing to provide breast milk and plasma samples. Infants eligible for inclusion were (1) ≥4 weeks corrected age, (2) receiving exclusively breastmilk and (3) not in a critical care setting.
Information on maternal age, weight, postpartum status, and factors potentially affecting pharmacokinetics (including smoking, alcohol intake, diet, ethnicity, medical conditions and concomitant medications) were recorded from interview with the mother. Infant age, sex, weight (birth and current) and known medical conditions (maternal participant reported) were also documented.
Measurement
Breast milk and plasma concentrations of perindopril and perindoprilat were analysed using our previously validated Liquid Chromatography tandem-Mass Spectrometry (LC-MS/MS) method.14
Data Collection
Eight breast milk samples (up to 10 mL each) were collected over a 24 hour period (i.e. at least one dosing interval). Samples were collected from the same breast by electric pump or manual expression. Participants attended the WCHN to provide the first breast milk sample, one paired maternal blood sample (up to 10 mL, taken within 30 mins of the breast milk sample) and, if the woman consented, one infant blood sample (up to 0.5 mL). Subsequent breast milk samples were collected in the participant’s home, using pre-labelled containers. These samples were stored in the participant’s domestic refrigerator and returned to the WCHN within 24 hrs. The samples were coded and stored at −80°C until analysis.
Written informed consent was obtained from participating women and on behalf of their infants for the collection and analysis of breast milk and plasma samples.
Data Analysis
Pharmacokinetic (PK) parameters of perindopril and perindoprilat in maternal breast milk were calculated in accordance with the FDA guidance document for undertaking clinical lactation studies,15 including the area under the milk concentration–time curve from time zero to time of last measurable concentration (AUClast), the dose-normalized area under the milk concentration–time curve over 24 hrs (AUC_D; AUC0–24), the dose-related peak concentration (Cmax_D), the time to peak plasma concentration (Tmax), the average steady-state milk drug concentration during multiple-dose administration (Cavg) and the terminal half-life (t½). The dose-normalized AUC (AUC_D) was calculated by dividing the AUClast by the dose. Non-compartmental PK analysis with PhoenixTM Winnonlin® 7.0 software was used to calculate the pharmacokinetic parameters.
Milk to plasma ratio, theoretical infant dose (TID) and relative infant dose (RID) of perindopril and perindoprilat were calculated (Equations 1–3).16,17
(1) (2) (3)
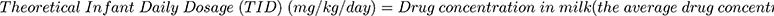
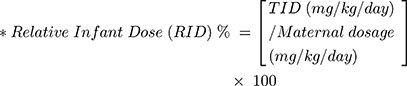
*Begg (1999), Bennett (1988), Hagg (2000) and Kristensen (1999) state that 150 mL/kg/day is the standardised milk consumption for a 2-month-old infant.15
Breast milk concentrations of perindopril and perindoprilat are presented as mean (range) for each participant. Mean concentrations were determined by averaging the concentration of perindopril or perindoprilat concentrations across all samples collected for each participant. The range included the lowest and highest concentration limits within the samples collected for each participant.
Results
Participant Characteristics
Ten women participated in the study, including three mother-infant pairs. Participant characteristics are described in Table 1. All women were actively breastfeeding at the time of the study. All women were non-smokers and were not using other medications known to interact with perindopril(at) or assay methods. The three infants studied were all born at term.
 |
Table 1 Maternal Participant Data |
A total of 79 breast milk samples were collected from the cohort of participants (mean 7.9 samples per participant). Breast milk samples were collected between 8 and 119 days post-partum (median 36.5 days).
Breast Milk and Plasma Concentrations
Perindopril
The mean concentration of perindopril in breast milk samples collected over the 24 hour study period per participant varied from 0.0035 to 1.35 ng/mL, with Cmax (range 0.03–4.1 ng/mL) observed at 2–4 hrs post last maternal dose taken. Perindopril was undetectable in breast milk in four participants at 6–12 hrs after dosing. Participant 6 had only one detectable breast milk concentration of 0.03 ng/mL at 3.4 hrs. Paired mother-infant perindopril plasma concentrations were 2.17 and 0.91 ng/mL at 4 hrs (Participant 2), 2.43 and 1.12 ng/mL at 4.3 hrs (Participant 4) and 18.13 and 0.44 ng/mL at 3.2 hrs (Participant 10).
Perindoprilat
The mean concentration of perindoprilat in breast milk samples collected over the 24 hour study period per participant varied from 0.41 to 36.83 ng/mL. The breast milk concentration for each participant remained relatively constant, resulting in a Tmax of 3–7 hrs, although in three participants, perindoprilat was undetectable in breast milk at a number of time points. Paired mother-infant perindoprilat plasma concentrations were 44.58 and 5.23 ng/mL at 4 hrs (Participant 2), 28.44 and 10.14 ng/mL at 4.3 hrs (Participant 4) and 12.48 ng/mL and <LOD at 3.15 hrs (Participant 10).
Pharmacokinetics
Estimated pharmacokinetic parameters for perindopril and perindoprilat for each participant are shown in Tables 2 and 3.
 |
Table 2 Estimated PK Parameters of Perindopril in Breast Milk |
 |
Table 3 Estimated PK Parameters of Perindoprilat in Breast Milk |
Milk-to-plasma (M/P) ratios for perindopril and perindoprilat calculated at specific individual time points ranged from 0.04–0.7 and 0.2–5.2, respectively. M/P ratios remained less than one for nine participants. The higher perindoprilat M/P for participant 9 (5.2) was observed 29 hrs following the last dose of perindopril.
The mean breast milk concentrations for each individual were used to calculate the theoretical infant dose (TID) and relative infant dose (RID). The TID for perindopril was 0.00045–0.18 µg/kg/day and perindoprilat 0.032–5.4 µg/kg/day. The RID for perindopril was 0.0005–0.2% and perindoprilat 0.03–4.6%.
Infant Outcomes
All parental reports for each breastfed infant indicated normal growth and development.
Discussion
Interpretation and Main Findings
To our knowledge, this is the first study reporting on the excretion of perindopril and its active metabolite perindoprilat into breast milk. There has been considerable discussion regarding the safety of medications during breastfeeding and the optimal method for evaluating infant exposure.18 Specialised lactation references refer to parameters such as M/P, RID, TID, infant plasma concentrations and adverse effects in order to determine potential safety.16,19-22 Our study assists clinical decision-making in mothers diagnosed with a hypertensive disorder in the post-partum period, where treatment with perindopril is being considered, by providing an assessment of infant exposure via breast milk using the parameters described above.
Breast milk concentrations of perindopril and perindoprilat varied among the 10 maternal participants in our study. Overall, the low or undetectable concentrations of perindopril in breast milk can be attributed in part to relatively low exposure and accumulation of the parent compound in plasma due to its short plasma half-life (0.8–1 hrs) and extensive hepatic metabolism.23 The relatively higher breast milk concentrations of perindoprilat were anticipated, due to pharmacokinetic parameters favouring transfer: low protein binding (10–20%), low molecular weight (368.5 free acid form), and long half-life (30–120 hrs).16,23 Our findings are consistent with published literature for other ACE inhibitors (captopril, enalapril, quinapril), which have reported low or undetectable concentrations in breast milk.24–28 In contrast to these studies, breast milk concentrations of the active metabolite were detectable in several of our participants.23
Strengths and Limitations
Milk to plasma (M/P) ratios less than one are generally indicative of a low extent of transfer into breast milk.16 We reported M/P ratios of less than one for perindopril and perindoprilat in all but one participant. The higher M/P ratio for perindoprilat in this participant could be explained by the prolonged 29 hour interval between sampling and previous dose administration, due to an unintentional dose omission. However, M/P ratios in isolation are considered to be of limited use as they do not provide an indication of the absolute quantity of drug delivered to the breastfed infant, which may be negligible and clinically insignificant.16
The RID for perindopril and perindoprilat was 0.0005–0.2% and 0.03–4.6%, respectively, with values of less than 10% generally regarded as compatible with breastfeeding.16 Our results are consistent with those reported for other ACE inhibitors: enalapril 0.16%/enalaprilat 0.08%,19 captopril 0.014%19 and quinapril 1.6%.19 It is important to consider the relative toxicity when assessing breastfeeding suitability, as some drugs are inappropriate despite a low RID (e.g. cancer chemotherapy). It is noteworthy that the TID of perindopril (0.00045 to 0.18 µg/kg/day) is considerably lower than an adult dose of approximately 0.14 mg/kg/day, based on a 70 kg person taking 10 mg of perindopril arginine. A slightly higher TID was calculated for perindoprilat (0.032–5.4 µg/kg/day); however, the low oral bioavailability in adults (25%), indicates the potential for limited absorption by the breastfed infant.23
Although TID and RID are useful to estimate breastfed infant exposure, infant plasma concentrations are arguably the most direct method. Plasma concentrations of perindoprilat were detected in two of the three infants (5.28 and 10.14 ng/mL). However, these were single plasma samples taken at a time approximately in the range of maternal peak plasma concentration. Peak plasma concentrations of perindoprilat in adults are known to increase linearly with dose. In adult pharmacokinetic studies involving perindopril erbumine, peak perindoprilat concentrations were 19.6 pmol/L (6.67 ng/mL), 44.5 pmol/L (15.15 ng/mL) and 107 pmol/L (36.42 ng/mL) on daily doses of 2 mg, 4 mg and 8 mg, respectively.23 Of interest, the mother of the breastfed infant with undetectable perindoprilat in plasma (participant 10) was taking the general adult maximum dose of 10 mg daily, which was double the dose taken by another participant with detectable infant concentrations (participant 4), possibly indicating the potential for interpatient pharmacokinetic or genetic variability. Perindoprilat concentrations of 2–3 pmol/L (0.68–1.02 ng/mL) are required to achieve an antihypertensive effect in adults.23 Perindopril is currently not used to manage hypertension in infants and therefore the plasma concentration required for therapeutic or toxic effects has not been established. While the absence of maternally reported acute adverse effects in the infants, accompanied by normal growth and development assessments is reassuring, further characterisation in a larger sample size of infants of mothers receiving perindopril while breastfeeding is required.
There are several limitations to our study. Firstly, the sample size was limited to 10 maternal and 3 infant participants, which may not accurately reflect variations in population pharmacokinetics and other parameters. However, this is a relatively large sample (total of 79 breast milk samples) in the context of comparable breastfeeding studies involving ACE inhibitors. Recruitment of infants was challenging; many were ineligible due to prematurity (which is associated with maternal hypertensive conditions), they were not exclusively breastfed or because consent for plasma samples was declined due to maternal reluctance over the perceived invasive nature of sampling. Secondly, due to logistical practicalities in the timing of sample collection, maternal and infant plasma samples were mostly, but unintentionally, taken at peak maternal dose concentration (2 to 4 hrs after last dose). It would have been preferable to have taken multiple plasma samples paired with breast milk samples over the dosing interval. Thirdly, breast milk samples were collected in the home environment and were reliant on participant recording of collection times. We attempted to control for this through the provision of detailed instructions and data collection labels were provided to reduce variability and improve reliability. Fourthly, a single maternal plasma sample was taken for each participant over the study period; therefore, M/P ratios were calculated from a single time point and would be subject to greater variation. Finally, we did not undertake an objective measurement of adverse effects on the breastfed infants. This was a considered decision as it is difficult to obtain clinically meaningful data for non-invasively measured infant blood pressure and heart rate at a single time point in such a small sample size. In addition, assessment of renal function and blood glucose concentrations was deemed to be too invasive and unlikely to yield relevant data, based on extrapolation from other ACE inhibitor breastfeeding studies.
Conclusion
We believe our study is the first to report on the excretion of perindopril and its active metabolite into breast milk, and supports therapeutic decision-making for both women and clinicians. Overall, infant exposure to perindopril and perindoprilat through breast milk appears to be relatively low. However, while our study found undetectable plasma concentrations in one infant, the remaining two infants had single point perindoprilat plasma samples, taken at the time of maternal dose peak concentration, whose concentrations were equivalent to therapeutic peak plasma concentrations in adults and exceeded the plasma concentrations required to exert an antihypertensive effect in adults. Further studies, with a larger number of infant plasma samples are sought to validate our findings. The decision to treat a breastfeeding woman with perindopril should be made with careful consideration of the benefit of maternal treatment and assessment of potential risks to the infant. Given the known maternal and infant benefits of breastfeeding, we believe that perindopril could be used safely in mothers of healthy term infants, with careful monitoring of the infant for sedation, growth, poor feeding, weight gain and developmental milestones, if there are concerns over maternal non-adherence to treatment and therefore significant advantages for once daily dosing.
Details of Ethics Approval
The ethics application of Evaluation of Medicines in Breast Milk and Clinical Effects in the Breastfed Infant (HREC/14/WCHN/115) was granted full ethics approval on 12th Nov, 2014. The ethics application meets the requirements of the National Statement on Ethical Conduct in Human Research and the study was conducted in accordance with the Declaration of Helsinki.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Gartner LM, Morton J, Lawrence RA, et al. Breastfeeding and the use of human milk. Pediatrics. 2005;115(2):496–506.
2. Hoddinott P, Tappin D, Wright C, Breast feeding. BMJ. 2008;336(7649):881–887. doi:10.1136/bmj.39521.566296.BE
3. Geneva SW. The Optimal Duration of Exclusive Breastfeeding. A Systematic Review. Geneva: WHO; 2001. Available from: https://www.who.int/nutrition/publications/infantfeeding/WHO_NHD_01.09/en/.
4. NHMRC.Infant Feeding Guidelines: Information for Healthcare Workers. NHMRC; 2012. Available from: https://nhmrc.gov.au/about-us/publications/infant-feeding-guidelines-information-health-workers.
5. Pearlstein TB, Zlotnick C, Battle CL, et al. Patient choice of treatment for postpartum depression: a pilot study. Arch Womens Ment Health. 2006;9(6):303–308.
6. Program NH. Report of the national high blood pressure education program working group on high blood pressure in pregnancy. Am J Obstet Gynecol. 2000;183(1):s1–s22. doi:10.1067/mob.2000.107928
7. Nelson-Piercy C. Handbook of Obstetric Medicine. CRC press; 2015 Apr, 8.
8. Dunietz GL, Strutz KL, Holzman C, et al. Moderately elevated blood pressure during pregnancy and odds of hypertension later in life: the POUCHmoms longitudinal study. BJOG. 2017;124(10):1606–1613.
9. Veerbeek JH, Hermes W, Breimer AY, et al. Cardiovascular disease risk factors after early-onset preeclampsia, late-onset preeclampsia, and pregnancy-induced hypertension. Hypertension. 2015;65(3):600–606.
10. Engeland A, Bjørge T, Klungsøyr K, Skjærven R, Skurtveit S, Furu K, Preeclampsia in pregnancy and later use of antihypertensive drugs. Eur J Epidemiol. 2015;30(6):501–508. doi:10.1007/s10654-015-0018-5
11. DiNicolantonio JJ, Lavie CJ, O’Keefe JH, Not all angiotensin-converting enzyme inhibitors are equal: focus on ramipril and perindopril. Postgrad Med. 2013;125(4):154–168. doi:10.3810/pgm.2013.07.2687
12. Ferrari R, Pasanisi G, Notarstefano P, Campo G, Gardini E, Ceconi C. Specific properties and effect of perindopril in controlling the renin–angiotensin system. Am J Hypertens. 2005;18(S5):142S–54S.
13. Perindopril. Micromedex Solutions. IBM Watson. Available from: https://www.micromedexsolutions.com/home/dispatch/ssl/true.
14. Lwin EM, Gerber C, Song Y, et al. A new LC-MS/MS bioanalytical method for perindopril and perindoprilat in human plasma and milk. Anal Bioanal Chem. 2017;409(26):6141–6148.
15. US Department of Health and Human Services (US DHHS), Food and Drug Administration (FDA), Centre for Drug Evaluation of Research (CDER), Centre for Biological Evaluation and research (CBER). Guidance for Industry; Clinical Lactation Studies – Study Design, Data Analysis, and Recommendations for Labelling. Maryland, USA: Food and Drug Administration; 2005.
16. Hale TW, Rowe HE. Medications & Mothers’ Milk. Springer. 2019.
17. Anderson PO, What do all the numbers mean? Breastfeed Med. 2016;11(6):277–279. doi:10.1089/bfm.2016.0076
18. Ilett KF, Kristensen JH, Wojnar-Horton RE, Begg EJ, Drug distribution in human milk. Aust Prescr. 1997;20(2):35–40. doi:10.18773/austprescr.1997.032
19. Lactmed. Available from: https://toxnet.nlm.nih.gov/newtoxnet/lactmed.htm.
20. Schaefer C, Peters PW, Miller RK, editors. Drugs During Pregnancy and Lactation: Treatment Options and Risk Assessment. Academic Press; 2014 Sep, 17.
21. Briggs GG, Freeman RK, Yaffe SJ. Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk.
22. e-lactancia. Available from: http://www.e-lactancia.org.
23. Sennesael J, Ali A, Sweny P, et al. The pharmacokinetics of perindopril and its effects on serum angiotensin converting enzyme activity in hypertensive patients with chronic renal failure. Br J Clin Pharmacol. 1992;33:93–99. doi:10.1111/bcp.1992.33.issue-1
24. Redman CW, Kelly JG, Cooper WD, The excretion of enalapril and enalaprilat in human breast milk. Eur J Clin Pharmacol. 1990;38(1):99. doi:10.1007/BF00314815
25. Begg EJ, Robson RA, Gardiner SJ, et al. Quinapril and its metabolite quinaprilat in human milk. Br J Clin Pharmacol. 2001;51(5):478–481.
26. Devlin RG, Fleiss PM, Captopril in human blood and breast milk. J Clin Pharmacol. 1981;21(2–3):110–113. doi:10.1002/jcph.1981.21.issue-2-3
27. Beardmore KS, Morris JM, Gallery ED, Excretion of antihypertensive medication into human breast milk: a systematic review. Hypertens Pregnancy. 2002;21(1):85–95. doi:10.1081/PRG-120002912
28. Shannon ME, Malecha SE, Cha AJ, Angiotensin converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs) and lactation: an update. J Hum Lact. 2000;16(2):152–155. doi:10.1177/089033440001600213
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
