Back to Journals » Clinical Epidemiology » Volume 10
Parental nicotine replacement therapy and offspring bronchitis/bronchiolitis and asthma – a nationwide population-based cohort study
Authors Molero Y, Zetterqvist J , Lichtenstein P, Almqvist C, Ludvigsson JF
Received 17 April 2018
Accepted for publication 8 June 2018
Published 25 September 2018 Volume 2018:10 Pages 1339—1347
DOI https://doi.org/10.2147/CLEP.S171401
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Henrik Sørensen
Yasmina Molero,1–3 Johan Zetterqvist,1 Paul Lichtenstein,1 Catarina Almqvist,1,4 Jonas F Ludvigsson1,5–7
1Department of Medical Epidemiology and Biostatistics, Karolinska Institutet, Stockholm, Sweden; 2Centre for Psychiatry Research, Department of Clinical Neuroscience, Karolinska Institutet, & Stockholm Health Care Services, Stockholm County Council, Stockholm, Sweden; 3Department of Psychiatry, Warneford Hospital, University of Oxford, Oxford, UK; 4Paediatric Allergy and Pulmonology Unit at Astrid Lindgren Children’s Hospital, Karolinska University Hospital of Paediatrics, Stockholm, Sweden; 5Department of Paediatrics, Örebro University Hospital, Örebro, Sweden; 6Division of Epidemiology and Public Health, School of Medicine, University of Nottingham, Nottingham, UK; 7Department of Medicine, Columbia University College of Physicians and Surgeons, New York, NY, USA
Background: Prior evidence shows that environmental tobacco smoke is a risk factor for respiratory tract infections, wheeze, and asthma. Nicotine replacement therapy has been shown to increase smoking cessation. However, no prior studies have explored if parental use decreases the risk of bronchitis/bronchiolitis and asthma in the offspring.
Objective: To examine whether nicotine replacement therapy varenicline, given to parents, was associated with a reduction in bronchitis/bronchiolitis and/or asthma in their children.
Methods: This study is a population-based cohort study, linking data from nationwide registers, and using a within-individual design that minimizes selection effects and controls for time-invariant confounding factors. Participants included 37,420 parents with a collected prescription of varenicline with 72,392 offspring <18 years of age. Exposure was defined as collected prescriptions of varenicline among the parents. Primary outcomes were offspring hospital visits for bronchitis/bronchiolitis (ICD10: J20 or J21) and offspring hospital visits for asthma (ICD10: J45).
Results: Parental varenicline treatment was associated with a lower rate of visits for bronchitis/bronchiolitis in their children (incidence rate ratio [IRR]=0.67; 95% CI=0.50–0.91), but no association was found for asthma (IRR=1.08; 95% CI=0.97–1.19). The rate reduction of bronchitis/bronchiolitis was similar when we restricted data to children aged 0–3 years (IRR=0.71; 95% CI=0.52–0.97) and to maternal varenicline treatment (IRR=0.64; 95% CI=0.43–0.96). When restricting the outcomes to unplanned visits only (ie, excluding booked appointments, follow-ups, and referrals), no associations were found (IRR=0.72, 95% CI=0.51–1.02).
Conclusion: In this cohort study, nicotine replacement treatment in parents was associated with reduced hospital visits for bronchitis/bronchiolitis in their children.
Keywords: asthma, wheezing, nicotine replacement therapy, varenicline, environmental tobacco smoke, child environmental tobacco smoke exposure
Introduction
Obstructive bronchitis, sometimes called bronchiolitis, is common in childhood.1 It has been estimated that 20% of all newborns need medical attention during their first year of life because of the most dominant agent, namely respiratory syncytial virus.2 In infants, bronchitis can lead to poor appetite and apnea. Affected infants may also develop pneumonia and death may occur in exceptional circumstances.3 There is no curative treatment for viral bronchitis.1
Asthma is characterized by airway inflammation, bronchospasm, and airflow obstruction translating into recurrent cough, wheezing, and dyspnea.4 Whereas genetic factors are important in asthma,5 environmental factors explain a large share of its variability, especially wheezing in small children.6 Apart from respiratory infections and allergens, environmental tobacco smoking (ETS) exposure is an important cause of asthma,7 and smoking can trigger a sudden ventilatory impairment in asthma.8
About 40% of all children worldwide are exposed to ETS through second-hand smoke, with even higher numbers reported in some countries.9 In Sweden, approximately 11% of Swedish parents to infants aged 8 months smoke,10 although it is unknown how many of these children are exposed to ETS. ETS exposure is also affected by third-hand smoke, ie, residual exposure from tobacco smoke on clothing, surfaces, and dust. Third-hand smoke reacts with other compounds and forms secondary pollutants that are reemitted as a gas.11 The household is an important source of ETS exposure; a study of Italian children showed that two-thirds of children were exposed to ETS in their homes. Even children who lived with smokers who did not smoke at home showed increased urinary cotinine levels (ie, the main metabolite for nicotine) as compared to children living with non-smokers.12 ETS is a risk factor for respiratory tract infections, wheeze, and asthma,7 and seems to be particularly associated with bronchitis/bronchiolitis in small children.13 It has been calculated that ETS exposure in British children could explain 8% of wheezing in children aged <3 years, and 4% of asthma in 3- to 4-year-olds.14 Current and former smokers have lower forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) as evidence of obstructive lung disease.15 Furthermore, smoking may decrease the response to inhaled corticosteroids in patients with asthma.8
In a 2012, meta-analysis based on 18 trials, Rosen et al reported that parents exposed to interventions were more likely to quit smoking (23.1%) than parents not exposed to these interventions (18.4%).16 The authors noted, however, that although smoking cessation can be effective, many parents fail to quit despite motivational support.16
In recent years, smoking cessation treatment, such as nicotine replacement therapy (NRT), has been introduced to decrease smoking dependence. Varenicline is a partial nicotine receptor antagonist that aims to decrease both rewards and cravings of smoking. Two meta-analyses by Cahill et al found that varenicline is superior to both placebo and other therapies regarding smoking cessation.17,18 Varenicline more than doubled the chances of smoking abstinence at 6 months compared to placebo, and was also associated with increased smoking abstinence when compared to bupropion (risk ratio [RR]=1.39; 95% CI=1.25–1.54) and NRT (RR=1.25; 96% CI=1.14–1.37).18
In this study, we used a within-individual design that more fully adjusts for time-invariant confounding factors associated with respiratory problems (eg, individual vulnerability and historic factors). We hypothesized that parental use of varenicline, used here as a proxy for smoking cessation, would lead to fewer hospital visits for bronchiolitis/bronchitis and/or asthma in their offspring.
Materials and methods
Study design
In this cohort study, information about the participants was collected from Swedish population-based registers with national coverage. Register linkage was performed from November 22, 2006 (ie, the introduction of varenicline in Sweden) to December 31, 2013, using the unique personal identity number assigned to all Swedish citizens/residents. All data were pseudonymized, and none of the researchers had access to the key used to recode the data.
Participants
Using the Swedish Prescribed Drug Register, we identified all individuals who had collected a prescription of varenicline (Chantix or Champix; Pfizer, New York, NY, USA; ATC code N07BA03) in Sweden. Parental data were then linked to data on offspring health care use through the Multigeneration Register. From this linkage, we identified children who were aged <18 years at the start of parental varenicline treatment.
Nicotine replacement therapy
The Swedish Prescribed Drug Register includes information on all collected drugs since July 2005.19 We used this register to identify parents with a collected prescription of varenicline. Varenicline therapy is recommended as a 12-week treatment. Each varenicline prescription was thus equaled to a 12-week treatment period. Because varenicline is often divided into several prescriptions for the same 12-week treatment, we regarded collected prescriptions within 12 weeks of the first collected prescription to be part of the same treatment period. If a prescription collection occurred more than 12 weeks after a previous collection, we regarded this as a new treatment period. An individual could thus be exposed to several varenicline treatment periods.
Bronchitis/bronchiolitis and asthma
The Swedish Patient Register started in 1964, is nationwide, and includes data on inpatient and specialized outpatient care. The positive predictive value of most diagnoses, including asthma, is about 85%–95%.20,21 Bronchitis/bronchiolitis was defined as an inpatient/outpatient hospital or specialized outpatient clinic visit coded with ICD10: J20 or J21. Asthma was defined as an inpatient/outpatient hospital or specialized outpatient clinic visit coded with ICD10: J45. First, we examined all types of visits, both planned (ie, booked appointments, follow-ups, and referrals) and unplanned (ie, urgent or emergency visits, either to hospital emergency departments or to specialized outpatient clinics). Second, we restricted the outcomes to unplanned visits. The reason for this stratification was that planned visits are more likely associated with long-term disease activity, while unplanned visits reflect immediate disease severity.
Demographic measures
Information on parental civil status, education, employment, and type of occupation in 2006 was extracted from the Longitudinal Integration Database for Health Insurance and Labor Market Studies, maintained by Statistics Sweden. Parental completed education was coded as basic education (ie, primary and lower secondary school), upper secondary education, and tertiary education (ie, university or college). Type of parental occupation was coded as a labor job (ie, occupation with on-the-job training), intermediate job (ie, an occupation that requires upper secondary school, occupation-specific training, or both), technical job or skilled trade (ie, an occupation that requires a college education or apprenticeship training), and professional job (ie, management occupation or an occupation that requires a university education).
Asthma medications
Information on offspring asthma medication was collected from the Swedish Prescribed Drug Register and included inhaled ß2-agonists (ATC code R03AC), inhaled glucocorticoids (ATC codes R03BA and R03AK), and leukotriene receptor antagonists (ATC code R03DC).20 Data on medications were used for descriptive purposes only.
Ethics
This study (2013/5:8) was approved by the Ethics Review Board in Stockholm, Sweden (12 September 2013). Informed consent was waived by the Review Board because of the register-based design.
Statistical analyses
We compared the rate of offspring bronchitis/bronchiolitis and asthma diagnoses during varenicline treatment periods to the rate of these outcomes during all other non-treatment periods by using a within-individual design with conditional Poisson regression to calculate incidence rate ratios (IRRs). To adjust for the age of the child, which is a time-varying factor associated with parental smoking cessation, as well as with bronchitis/bronchiolitis and asthma, we added the child’s age (measured in months) to the model as a covariate. To avoid potential time-varying confounding by season, we used calendar month as a categorical covariate. We also used quadratic and cubic functions of child age as covariates to allow for nonlinear effects of age. In sensitivity analyses, we also coded age as a categorical time-varying covariate, with one category for each whole month since birth. Finally, since some children in the study were siblings, the standard errors were estimated using a cluster-robust sandwich estimator. This design has previously been used in pharmacoepidemiological studies, including studies of varenicline.22 Information on the design is provided elsewhere.23 For statistical analysis, we used SAS version 9.4 and STATA version 14.1.
Sensitivity analyses
We defined two types of sensitivity analyses a priori: 1) Because the influence of exposure to maternal smoking may differ from the influence of paternal smoking,24 we performed subanalyses to examine associations separately for mothers and fathers. Moreover, considering that visits for airway disease are more common in smaller children, we carried out sub-analyses stratifying the cohort to children aged 0–3 and 4–17 years. 2) Although the aim of varenicline treatment is to reduce smoking during medication, its effects on smoking behavior may be long term. It may also take time to achieve a smoke-free environment for the child. In a separate analysis, we therefore compared the first 6 and 12 months after the start of parental varenicline treatment to all other non-treatment periods. Hospital visits for bronchitis/bronchiolitis and asthma may be associated with subsequent hospital visits. Also, such visits may influence parents to initiate varenicline treatment in order to reduce the child’s bronchitis/bronchiolitis or asthma. To address this, we carried out a separate analysis where we excluded children who had been treated for bronchitis/bronchiolitis and asthma, respectively, up to 6 months before the start of each varenicline treatment period.
Results
Background data
We identified 37,420 parents with a collected prescription of varenicline during follow-up (Table 1). Of those, 21,984 were mothers (58.7%) and 15,436 were fathers. Over 75% of parents were aged 30–49 years at the start of varenicline treatment. Other demographic characteristics are given in Table 1.
In all, 72,392 children had experienced an interval of parental varenicline treatment before they were 18 years old. Of those, approximately 40% (n=30,128) were <10 years at the start of parental varenicline treatment, and 13.6% (n=9,867) were aged 0–2 years. During follow-up, 2.4% (n=1,755) of the children had a hospital or specialized outpatient clinic visit for bronchitis/bronchiolitis, and 7.7% (n=5,581) had a visit for asthma. Approximately 21% (n=15,073) had collected a prescription for asthma medications (Table 1).
Main results
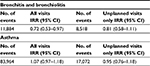
Comparing 12 weeks of parental varenicline treatment to all other non-treatment periods (Table 2), results showed that parental varenicline treatment was associated with a lower rate of all hospital or specialized outpatient clinic visits for bronchitis/bronchiolitis (IRR=0.67; 95% CI=0.50–0.91), but no association was found for asthma (IRR=1.08; 95% CI=0.97–1.19). Restricting the outcome to unplanned visits only (ie, excluding booked appointments, referrals, and follow-ups), results for bronchitis/bronchiolitis were of the same magnitude (IRR=0.72; 95% CI=0.51–1.02).
Sensitivity analyses
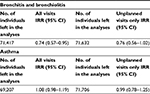
The influence of maternal smoking may differ from that of paternal smoking. In a second analysis, we therefore examined the mothers and fathers separately (Table 3). Our analysis showed reductions in visits for bronchitis/bronchiolitis during periods of maternal varenicline treatment (IRR=0.64; 95% CI=0.43–0.96). Similar results were noted during periods of paternal varenicline treatment (IRR=0.70; 95% CI=0.48–1.03). No associations were found when examining visits for asthma or when restricting bronchitis/bronchiolitis or asthma to unplanned visits only (Table 3).
Because hospital visits for bronchitis/bronchiolitis and asthma are more often seen in younger children, we carried out a subanalysis in which we stratified for the child’s age at the start of parental varenicline treatment (0–3 and 4–17 years). The analysis demonstrated that parental varenicline treatment was associated with a reduced rate of all visits for bronchitis/bronchiolitis in children aged 0–3 years (IRR=0.71; 95% CI=0.52–0.97). Varenicline treatment was associated with a similar IRR in older children (IRR=0.64; 95% CI=0.36–1.11). When restricting outcomes to unplanned visits only, reductions of bronchitis/bronchiolitis were only shown for children aged 4–17 years (IRR=0.36; 95% CI=0.13–0.96). No associations were found for asthma visits (Table 3).
Although the aim of varenicline treatment is to reduce smoking during treatment, its effect on smoking behavior may be long term. It may also take time to achieve a smoke-free environment for the child. Accordingly, a third analysis was done to compare the rate of bronchitis/bronchiolitis and asthma diagnoses received during 6 or 12 months after the start of varenicline treatment. These extended periods were then compared with the rate of bronchitis/bronchiolitis and asthma during all other non-treatment periods. No associations were found (Table S1). Furthermore, age was recorded as a categorical time-varying covariate. Results in these analyses were similar to the main results (Table S2). Lastly, we excluded children who had been treated for bronchitis/bronchiolitis and asthma, respectively, to account for the effect of previous hospitalizations on subsequent hospitalizations. After this exclusion, results remained similar to the overall results (Table S3).
Discussion
In this nationwide population-based study, we found that varenicline treatment, used here as a proxy for smoking cessation, was associated with a reduction in the rate of hospital or specialized outpatient clinic visits for bronchitis/bronchiolitis in the offspring. No association was found for visits due to asthma.
Earlier literature
Although research has shown that parents are susceptible to interventions for smoking cessation, most parents (over 75%) do not quit.16 Two Cochrane analyses have recently examined interventions aiming to reduce the exposure of children to ETS. Interventions on parental education and counseling programs to reduce children’s ETS did not demonstrate their effectiveness, whereas motivational interviewing or intensive counseling that is provided in clinical settings seems to be more effective.25 Another important factor is that maternal smoking in pregnancy appears to increase the risk of wheeze and asthma in children who are not exposed to maternal smoking after birth.7
Varenicline is an effective oral smoking cessation therapy.17,18 A pooled analysis of 19 randomized controlled trials showed that smoking abstinence rates for varenicline were 49% at weeks 9–12 (ie, towards the end of a varenicline treatment period) and 22% at week 52.26 We are only aware of studies exploring the benefits of varenicline in relation to smoking habits and health benefits.18,27 However, there are no studies systematically exploring pharmaceutical interventions in parent smokers to decrease the risk of bronchitis/bronchiolitis and asthma in their offspring. While drug safety concerns (including cardiovascular side effects and suicidality) initially hindered the use of varenicline, recent data suggest that the medication can be used safely.22,28,29 Furthermore, varenicline treatment in parent smokers may be an efficient way to reduce the risk of bronchitis/bronchiolitis in children.
When analyzing mothers and fathers separately, bronchitis/bronchiolitis was associated with a decreased rate in both genders (IRR ratio 0.64 with maternal exposure as opposed to 0.70 with paternal exposure). We found similar associations with varenicline use and offspring bronchitis/bronchiolitis both before and after the age of 3 years, even if bronchitis/bronchiolitis is less frequent in older children1 (which explains why the 95% CIs were wider for the older age group).
In a sensitivity analysis (Tables S1–S3), we compared the first 6 and 12 months after the start of parental varenicline treatment to nontreatment periods. This analysis yielded somewhat higher IRRs that failed to attain statistical significance. The lack of significance may be due to lower smoking abstinence rates over time.26 Obviously, a weakness of the study is our lack of data on actual smoking. There is a risk that the IRR estimates are biased because of carryover effects. For instance, parental medication with varenicline could influence the diagnosis rate of bronchitis/bronchiolitis and asthma on a later occasion. Similarly, previous hospitalizations may influence subsequent hospitalizations. We therefore carried out sensitivity analyses where we excluded children with hospital visits for bronchitis/bronchiolitis or asthma up to 6 months before the start of parental varenicline treatment. Results remained similar to the overall results, suggesting that the effects associated with varenicline treatment were not attributed to carryover effects from the previous treatment.
Strengths and limitations
This study was based on more than 72,000 children exposed to parental varenicline treatment. We used a within-individual design that minimized confounding by time-invariant factors (eg, genes, smoking during pregnancy, and invariant exposure to other smokers in the household), as IRRs for bronchitis and asthma were compared within the same stratum (ie, child) during medication and non-medication periods. Still, we acknowledge that age may introduce bias, especially because the pattern of wheezing/asthma is highly age dependent.30 To adjust for the effect of age on bronchitis/bronchiolitis and asthma, we introduced age as a time-varying covariate in all analyses.
In Sweden, varenicline is sold only on prescription. The Swedish Prescribed Drug Register has a very high coverage of prescribed drugs, with patient identity data missing for less than 0.3% of all prescriptions.19 Of note, this study was limited to children of varenicline users. Thus, if children to such users differ systematically from other ETS-exposed children, this may limit the external validity of our study.
We obtained data on bronchitis/bronchiolitis and asthma from the Swedish Patient Register, independently of our exposure (varenicline). This register is highly specific but may have lower sensitivity for mild respiratory disease, since children with mild disease were not likely to have been treated in a hospital or at a specialized outpatient clinic. Hence, because our data are primarily valid for severe disease, we cannot extrapolate our findings to minor respiratory disease. Furthermore, we carried out two different analyses: one where we used diagnoses received during all types of visits, and one where we used only diagnoses received during unplanned visits (ie, excluding booked appointments, referrals, and follow-ups). Unplanned visits reflect immediate disease severity, as opposed to planned visits, that are associated with long-term disease activity. Revisits and referrals are often planned for those children with the most severe respiratory disease, so a reduction in planned revisits is likely associated with the child having less respiratory symptoms over an extended period.
We have no data on adherence to varenicline or the proportion of parents who actually stopped smoking while on varenicline. This problem is comparable with non-adherence in clinical trials. In this sense, our within-individual estimate is parallel to the intention-to-treat analysis used in randomized controlled trials (eg, in a study where an antihypertensive medication is linked to a reduced risk of myocardial infarction but where there are no data on the proportion of patients taking the drug or their mean reduction of blood pressure). Furthermore, we did not have data on smoke exposure from other individuals in the same household. Although our model adjusts for exposure to constant factors, this would likely underestimate associations between varenicline treatment and outcomes. Despite this, we could demonstrate that children had fewer hospital visits for bronchitis/bronchiolitis during periods when parents were prescribed varenicline, and we believe this risk reduction is mediated through a reduction in smoking. The rate reduction associated with unplanned visits for bronchitis/bronchiolitis was similar (0.72) to that of any visit (0.67). In contrast, we found no associations with asthma, suggesting that ETS may have less effect on asthma exacerbations than on bronchitis/bronchiolitis.13 In a meta-analysis of 60 papers, Jones et al found that ETS had its most striking effect on the risk of bronchiolitis (exposure to household smoking led to a 2.5 increased risk of bronchiolitis in the offspring).13 However, we also acknowledge that we may have underestimated the effect of varenicline on asthma, if children with asthma see general practitioners rather than hospital-based pediatricians at exacerbation. It is also possible that with lower exposure to ETS, some children could reduce their medications for asthma. Being able to reduce asthma medication is positive for the child but may have concealed a beneficial effect of varenicline for that outcome.
The comparison of varenicline treatment to all other non-treatment periods (both before and after varenicline treatment) could lead to an underestimation of effects, as many individuals remain smoke-free after the end of treatment.17 However, comparing varenicline treatment to only pretreatment periods would make adjustment for age problematic, since higher age is likely to be negatively associated with the outcome. Therefore, a protective effect of varenicline treatment would be difficult to separate from the effect of higher age in such a design. On the other hand, not adjusting for age in this design would instead lead to an overestimation of the effect, if the effect is assumed to be a proxy for smoking cessation.
Finally, we did not have information on smoking exposure among the children, ie, if the parents smoked indoors or outdoors. However, it has been suggested that outdoor smoking too is associated with ETS, and that outdoor smoke can drift into adjacent indoor areas and affect air quality.31 Smoking rates are lower in Sweden (14% as opposed to 23% in the rest of the European Union).32 Should our results prove to be valid outside Sweden, the public health benefits of varenicline-treated parents may be greater than in our study.
Conclusion
In conclusion, this study suggests that NRT (ie, varenicline) in parents may be associated with a reduction in morbidity from bronchitis/bronchiolitis in their children. If our findings replicate in other designs and settings, this could indicate that treating parents with varenicline may be a helpful addition to the limited arsenal of preventive measures against bronchitis/bronchiolitis in children.
Data sharing
Other researchers can be granted individual access to our data through the Swedish National Board of Health and Welfare.
Acknowledgment
We acknowledge financial support from the Swedish Research Council through the Swedish Initiative for Research on Microdata in the Social and Medical Sciences (SIMSAM) framework grant no. 340-2013-5867.
Ethics approval
This project (2013/5:8) was approved by the Ethics Review Board in Stockholm, Sweden, on September 12, 2013.
Author contributions
YM, JFL, and JZ conceived and designed the study with input from the other authors. YM and JFL wrote the first draft of the paper. JFL supervised the project. PL and CA obtained funding for data collection and register-based linkages. YM and JZ analyzed the data. All authors interpreted the data and contributed to the writing of the paper. All authors revised and approved the final version. YM had full access to all the data and takes responsibility for the integrity of the data and the accuracy of the data analyses.
Disclosure
The principal author affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained. The authors report no conflicts of interest in this work.
References
Meissner HC. Viral bronchiolitis in children. N Engl J Med. 2016;374(1):62–72. | ||
Hall CB, Weinberg GA, Iwane MK, et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med. 2009;360(6):588–598. | ||
Stein RT, Bont LJ, Zar H, et al. Respiratory syncytial virus hospitalization and mortality: Systematic review and meta-analysis. Pediatr Pulmonol. 2017;52(4):556–569. | ||
Asher I, Pearce N. Global burden of asthma among children. Int J Tuberc Lung Dis. 2014;18(11):1269–1278. | ||
Ullemar V, Magnusson PK, Lundholm C, et al. Heritability and confirmation of genetic association studies for childhood asthma in twins. Allergy. 2016;71(2):230–238. | ||
von Mutius E, Martinez FD, Fritzsch C, Nicolai T, Roell G, Thiemann HH. Prevalence of asthma and atopy in two areas of West and East Germany. Am J Respir Crit Care Med. 1994;149(2 Pt 1):358–364. | ||
Neuman Å, Hohmann C, Orsini N, et al. Maternal smoking in pregnancy and asthma in preschool children: a pooled analysis of eight birth cohorts. Am J Respir Crit Care Med. 2012;186(10):1037–1043. | ||
Tomlinson JE, Mcmahon AD, Chaudhuri R, Thompson JM, Wood SF, Thomson NC. Efficacy of low and high dose inhaled corticosteroid in smokers versus non-smokers with mild asthma. Thorax. 2005;60(4):282–287. | ||
Oberg M, Jaakkola MS, Woodward A, Peruga A, Prüss-Ustün A. Worldwide burden of disease from exposure to second-hand smoke: a retrospective analysis of data from 192 countries. Lancet. 2011;377(9760):139–146. | ||
The National Board of Health and Welfare. Statistics on smoking habits among parents of infants; 2016. Available from: http://www.socialstyrelsen.se/Lists/Artikelkatalog/Attachments/20314/2016-9-4.pdf. Accessed 9 September, 2017. | ||
Drehmer JE, Walters BH, Nabi-Burza E, Winickoff JP. Guidance for the clinical management of thirdhand smoke exposure in the child health care setting. J Clin Outcomes Manag. 2017;24(12):551–559. | ||
Protano C, Andreoli R, Manini P, Vitali M. How home-smoking habits affect children: a cross-sectional study using urinary cotinine measurement in Italy. Int J Public Health. 2012;57(6):885–892. | ||
Jones LL, Hashim A, Mckeever T, Cook DG, Britton J, Leonardi-Bee J. Parental and household smoking and the increased risk of bronchitis, bronchiolitis and other lower respiratory infections in infancy: systematic review and meta-analysis. Respir Res. 2011;12:5. | ||
Ferrence R. Passive smoking and children. BMJ. 2010;340(Mar 24): c1680. | ||
Woodruff PG, Barr RG, Bleecker E, et al. Clinical significance of symptoms in smokers with preserved pulmonary function. N Engl J Med. 2016;374(19):1811–1821. | ||
Rosen LJ, Noach MB, Winickoff JP, Hovell MF. Parental smoking cessation to protect young children: a systematic review and meta-analysis. Pediatrics. 2012;129(1):141–152. | ||
Cahill K, Stevens S, Perera R, Lancaster T. Pharmacological interventions for smoking cessation: an overview and network meta-analysis. Cochrane Database Syst Rev. 2013;5(5):Cd009329. | ||
Cahill K, Lindson-Hawley N, Thomas KH, Fanshawe TR, Lancaster T. Nicotine receptor partial agonists for smoking cessation. Cochrane Database Syst Rev. 2016;5(5):Cd006103. | ||
Wettermark B, Hammar N, Fored CM, et al. The new Swedish Prescribed Drug Register – opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16(7):726–735. | ||
Örtqvist AK, Lundholm C, Wettermark B, Ludvigsson JF, Ye W, Almqvist C. Validation of asthma and eczema in population-based Swedish drug and patient registers. Pharmacoepidemiol Drug Saf. 2013;22(8):850–860. | ||
Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450. | ||
Molero Y, Lichtenstein P, Zetterqvist J, Gumpert CH, Fazel S. Varenicline and risk of psychiatric conditions, suicidal behaviour, criminal offending, and transport accidents and offences: population based cohort study. BMJ. 2015;350:h2388. | ||
Allison PD. Fixed Effects Regression Models. Vol. 160. Thousand Oaks, CA: Sage publications; 2009. | ||
Jurado D, Muñoz C, Luna JD, Muñoz-Hoyos A. Is maternal smoking more determinant than paternal smoking on the respiratory symptoms of young children? Respir Med. 2005;99(9):1138–1144. | ||
Baxi R, Sharma M, Roseby R, et al. Family and carer smoking control programmes for reducing children’s exposure to environmental tobacco smoke. Cochrane Database Syst Rev. 2014;3(3):Cd001746. | ||
Agboola SA, Coleman T, Mcneill A, Leonardi-Bee J. Abstinence and relapse among smokers who use varenicline in a quit attempt-a pooled analysis of randomized controlled trials. Addiction. 2015;110(7):1182–1193. | ||
van Eerd EAM, van der Meer RM, van Schayck OCP, Kotz D, Cochrane Airways Group, Group CA. Smoking cessation for people with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2016;272(19):Cd010744. | ||
Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. The Lancet. 2016;387(10037):2507–2520. | ||
Kotz D, Viechtbauer W, Simpson C, van Schayck OC, West R, Sheikh A. Cardiovascular and neuropsychiatric risks of varenicline: a retrospective cohort study. Lancet Respir Med. 2015;3(10):761–768. | ||
Castro-Rodríguez JA, Holberg CJ, Wright AL, Martinez FD. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med. 2000;162(4 Pt 1):1403–1406. | ||
Sureda X, Fernández E, López MJ, Nebot M. Secondhand tobacco smoke exposure in open and semi-open settings: a systematic review. Environ Health Perspect. 2013;121(7):766–773. | ||
OECD. Health at a Glance: Europe 2012. Paris: OECD Publishing; 2012. |
Supplementary materials
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.