Back to Journals » Journal of Inflammation Research » Volume 14
Overexpression of HOXB4 Promotes Protection of Bone Marrow Mesenchymal Stem Cells Against Lipopolysaccharide-Induced Acute Lung Injury Partially Through the Activation of Wnt/β-Catenin Signaling
Authors Lin S , Chen Q , Zhang L, Ge S, Luo Y, He W, Xu C, Zeng M
Received 9 May 2021
Accepted for publication 19 July 2021
Published 27 July 2021 Volume 2021:14 Pages 3637—3649
DOI https://doi.org/10.2147/JIR.S319416
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Monika Sharma
Shan Lin, 1, 2 Qingui Chen, 1, 2 Lishan Zhang, 1, 2 Shanhui Ge, 1, 2 Yuling Luo, 1, 2 Wanmei He, 1, 2 Caixia Xu, 3 Mian Zeng 1, 2
1Department of Medical Intensive Care Unit, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, 510080, People’s Republic of China; 2Institute of Pulmonary Diseases, Sun Yat-sen University, Guangzhou, Guangdong, People’s Republic of China; 3Research Center of Translational Medicine, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510080, People’s Republic of China
Correspondence: Mian Zeng
Department of Medical Intensive Care Unit, The First Affiliated Hospital, Sun Yat-sen University, No. 58 Zhongshan Road 2, Guangzhou, Guangdong, 510080, People’s Republic of China
Email [email protected]
Caixia Xu
Research Center of Translational Medicine, The First Affiliated Hospital, Sun Yat-sen University, No. 58 Zhongshan Road 2, Guangzhou, Guangdong, 510080, People’s Republic of China
Email [email protected]
Purpose: Pulmonary vascular endothelial cell (EC) injury is recognized as one of the pathological factors of acute lung injury/acute respiratory distress syndrome (ALI/ARDS). Bone marrow mesenchymal stem cell (BMSC)-based cytotherapy has attracted substantial attention over recent years as a promising therapeutic approach for ALI/ARDS; however, its use remains limited due to inconsistent efficacy. Currently, gene modification techniques are widely applied to MSCs. In the present study, we aimed to investigate the effect of BMSCs overexpressing Homeobox B4 (HOXB4) on lipopolysaccharide (LPS)-induced EC injury.
Methods: We used LPS to induce EC injury and established EC-BMSC coculture system using transwell chambers. The effect of BMSCs on ECs was explored by detecting EC proliferation, apoptosis, migration, tube formation, and permeability, and determining whether the Wnt/β-catenin pathway is involved in the regulatory mechanism using XAV-939, inhibitor of Wnt/ β-catenin.
Results: As compared to BMSCWT, BMSCHOXB4 coculture promoted EC proliferation, migration, and tube formation after LPS stimulation and attenuated LPS-induced EC apoptosis and vascular permeability. Mechanistically, BMSCHOXB4 coculture prevented LPS-induced EC injury by activating the Wnt/β-catenin pathway, which is partially reversible by XAV-939. When cocultured with BMSCHOXB4, pro-inflammatory factors were dramatically decreased and anti-inflammatory factors were greatly increased in the EC medium compared to those in the LPS group (P< 0.05). Additionally, when compared to BMSCWT coculture, the BMSCHOXB4 coculture showed an enhanced modulation of IL-6, TNF-α, and IL-10, but there was no statistically significant effect on IL-1β and IL-4.
Conclusion: Coculturing of BMSCHOXB4 prevented LPS-induced EC injury by reversing the inactivation of the Wnt/β-catenin signaling pathway. An in vivo study remains warranted to ascertain whether engraftment of BMSCHOXB4 can be an attractive strategy for the treatment of ALI/ARDS.
Keywords: acute lung injury, mesenchymal stem cells, endothelial cells, HOXB4
Corrigendum for this paper has been published
Background
Sepsis is a complex clinical syndrome with physiological, biological, and biochemical abnormalities.1 It develops from a dysregulated host response to infection.1 The accumulated understanding of the pathophysiological mechanisms of sepsis reveal that it involves host immune imbalance, inflammatory mediator outbreaks, coagulation abnormalities, and apoptosis. The lung is the most susceptible organ to suffer from sepsis and can develop acute respiratory distress syndrome (ARDS).2–5 Alveolar type II epithelial cell and capillary endothelial cell (EC) injury are the primary underlying causes in the development of acute lung injury (ALI)/ARDS.5 Previously, studies focused on alveolar type II epithelial cells, but increasing evidence suggests that alveolar capillary EC damage may play a more critical role in promoting the progression of ALI/ARDS.6,7 Alveolar capillary EC destruction that results from sepsis, trauma, and severe infections leads to impaired structural integrity and function. This subsequently causes the loss of permeability and consequent fluid accumulation in the interstitial and alveolar spaces of the lungs.7–9 Therefore, the contribution of alveolar capillary EC damage in the progression of ARDS cannot be underestimated.
Mesenchymal stem cells (MSCs) are an important family of stem cells with great therapeutic potential.10 MSC-based cell therapy has been attractive as a potential treatment for ARDS due to multiple aspects, including the secretion of several paracrine factors, assistance in tissue repair, immunomodulation, and reduction in the severity of bacterial infections.11–13 To date, some clinical studies have confirmed the therapeutic effects of MSCs in ARDS, including that caused by coronavirus disease 2019 (COVID-19).14–19 In the pre-experiment, we found that pre-treatment of bone marrow MSCs (BMSCs) with Ghrelin (growth hormone-releasing peptide, an endogenous ligand for the growth hormone-releasing hormone receptor) markedly strengthened the therapeutic effect of BMSCs in an animal model of lipopolysaccharide (LPS)-associated ALI/ARDS. This was evidenced by a significant reduction in alveolar lumen exudate, we found a significant upregulation of Homeobox B4 (HOXB4) by transcriptome sequencing of Ghrelin-pretreated BMSCs supernatants. HOXB4 is a member of the homeobox (HOX) gene family with essential roles in the regulation of cell renewal and differentiation.20 However, there are only a few studies on the interaction between HOXB4 and BMSCs in ALI/ARDS. Moreover, it is unclear whether HOXB4 can strengthen the therapeutic effect of BMSCs in ALI/ARDS; the underlying molecular mechanism deserves to be investigated. Therefore, in the present study, we aimed to investigate the role of HOXB4 in the protection of BMSCs against ALI/ARDS.
Materials and Methods
Cell Culture and Preparation
The study was approved by the Ethical Review Committee of the First Affiliated Hospital of Sun Yat-sen University. The animal part of this study was performed in accordance with the guidelines of the Animal Care and Use Committee of the First Affiliated Hospital of Sun Yat-sen University and the China Laboratory Animal Welfare Guidelines [GB/T 35892-2018]. The method of isolation and culture of rat BMSCs was performed in accordance with our previous studies and the protocol of Grässel et al.21–23 When adherent cell cultures reached 80–90% confluence, BMSCs were recovered for subculture using 0.25% trypsin EDTA (Gibco, Carlsbad, CA, USA).
Recombinant lentiviral vectors overexpressing HOXB4 and empty vectors were constructed by Shanghai Genechem (China). All vectors carried the anti-purine bacteriocin gene and green fluorescent protein. We performed the corresponding pre-experiments before lentiviral transfection by inoculating 100 µL of complete medium (containing 5000 cells) in a 96-well plate and replacing the new complete medium after 24 h of incubation. Then, BMSCs were transfected with a multiplicity of infection (MOI) of 1, 10, 50, and 100, respectively, and transfection enhancement solution HitransG A (Shanghai Genechem, China) was added. The new medium was replaced after 16 h of incubation followed by subsequent changes according to cell growth. After 72 h, the fluorescence expression abundance was observed under a fluorescence microscope (Olympus, Japan), and the group with 80% infection efficiency with well-grown cells was selected for subsequent experiments. The final MOI per cell was used as 100, and the transfected cells were selected after 96 h using 2 µg/mL of puromycin.
EA.hy926 human umbilical vein ECs were purchased from Procell (Wuhan, China) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin (all from Gibco) under standard conditions of 5% CO2 at 37°C.
The co-culture regime was established in transwell chambers with a pore size of 0.4 μm (Corning, NY, USA). The upper chamber was incubated with wild-type (WT) BMSCs (1×105 cells/mL), vector BMSCs (1×105 cells/mL), and HOXB4 overexpressing BMSCs (1×105 cells/mL) respectively, and the lower chamber was incubated with ECs (1×105 cells/mL). On the basis of our preliminary experiments and previous study, we used LPS (O127:B8, Sigma, St. Louis, MO, USA) at a concentration of 150 μg/mL to act on ECs cocultured with different BMSCs for 24 h simultaneously.24 Overall, there were five groups: control, EC+LPS (150 μg/mL), BMSCWT+EC+LPS (150 μg/mL), BMSCVector+EC+LPS (150 μg/mL), and BMSCHOXB4+EC+LPS (150 μg/mL).
To verify the protective effect of BMSCHOXB4 on LPS-induced EC injury via the Wnt/β-catenin pathway, we also evaluated ECs pretreated for 12 h with XAV-939 (APExBIO, Houston, TX, USA), a specific inhibitor of the Wnt/β-catenin pathway. Together with the pre-experiments and previous study, XAV-939 acted on ECs at a concentration of 10 µM dissolved in dimethyl sulfoxide.23
5-Ethynyl-20-Deoxyuridine (EdU) Assay
The EdU kit (Ribobio, China) was used to detect cell proliferation. Briefly, after 2 h of incubation with EdU, the cells were fixed with 4% paraformaldehyde and permeabilized with 0.5% Triton X-100. Apollo® staining reaction solution was subsequently added followed by incubation for 30 min in the dark. Next, the cells were washed three times with phosphate-buffered saline (PBS) followed by incubation for 30 min with Hoechst 33342 reaction solution. The cells were then washed and kept moist with PBS, and they were observed using fluorescence microscopy. The procedure of the assay is detailed in the instructions provided with the kit, and the cell proliferation capacity is reflected by calculating the ratio of EdU-positive cells to the total number of DAPI-positive cells.
Wound Scratch Assay
ECs were inoculated at a density of 1×106 cells per well in 6-well culture plates and grown to confluence. Cell monolayers were scraped off using a 200 μL pipette tip, then washed twice with PBS, and cultured for 24 h with fresh serum-free medium and the different co-culture regimes described above. Photographs were taken after cell scraping and 24 h after co-culture to observe cell migration, and wound healing percentage was determined using the ImageJ software, ie (0 h scratch area – 24 h scratch area)/0 h scratch area × 100%.
Tube Formation Assay
The growth factor reduced Matrigel (Corning) was plated at 300 μL per well in 24-well plates for 30 min at 37°C. ECs (2×105) were cultured in 300 μL DMEM supplemented with 10% FBS (all from Gibco). Calcein AM fluorescent dye (5 µL) (KeyGen, China) was added 5 to 10 min prior to fluorescence microscopy visualization to enhance the visibility of tube and network formation in Matrigel. Tubular structures were examined under a fluorescence microscope 3 h after cell inoculation, and the extent of tubule formation was quantified by counting vascular crossings in three randomly selected fields of view in each well. The results were analyzed using the ImageJ software.
Apoptosis Assays
TdT-mediated dUTP Nick-End Labeling (TUNEL) (Beyotime, China) and Annexin V-fluorescein isothiocyanate (AV-FITC)/propidium iodide (PI) (BD Biosciences, San Jose, CA, USA) were used to evaluate cell apoptosis. Among these assays, the TUNEL positive cells to total cells ratio was calculated to assess the level of apoptosis, and the Annexin V-FITC/PI assay was performed to analyze the percentage of early apoptotic as well as late apoptotic cells. The above assays were performed by referring to the manufacturers’ instructions and our previous studies.22,24
Endothelial Cell Permeability Assay
Transwell permeability assay was used to study the permeability of ECs.25 We seeded 2×105 ECs in 300 μL medium in 6.5 mm transwell chambers (Corning) and cultured the cells until the formation of a confluent monolayer. Then, the ECs were co-cultured in a 24-well plate inoculated with BMSCWT, BMSCVector, and BMSCHOXB4 and simultaneously administered with LPS. After 24 h, the chambers inoculated with ECs were transferred to a 24-well plate, the top medium of the chambers was removed and refilled with medium containing streptavidin-horseradish peroxidase. After 5 min of incubation, 20 μL of medium was collected from the lower chamber and moved to a new 96-well plate. Next, 50 µL of TMB substrate was added to each well of the 96-well plate and allowed to react for 5–20 min at room temperature, followed by the addition of 25 µL of stop solution (Sigma) to each well. Absorbance was detected at 450 nm using an enzyme linked immunosorbent assay (ELISA) reader.
Western Blotting
The proteins were extracted from ECs using RIPA lysis buffer (Beyotime, China) containing protease inhibitors. The protein extracts were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were then blocked with quick-blocking buffer (Beyotime, China) for 15 min at room temperature and incubated overnight at 4°C with the corresponding primary antibody concentration of 1 to 1000 (anti-β-catenin, anti-VE-cadherin, anti-Bcl-2, anti-BAX and anti-GAPDH) (Cell Signaling Technology, Beverly, MA, USA). After incubation with the secondary antibody (Cell Signaling Technology, Beverly, MA, USA) for 1 h, the bands were visualized by chemiluminescence using the Image Quant LAS 4000 system.
ELISA
The concentration of inflammatory factors in the culture medium, including the pro-inflammatory factors interleukin (IL)-1, IL-6, and TNF-α as well as the anti-inflammatory factors IL-4 and IL-10, was determined using ELISA kits (NeoBioscience, China) according to the manufacturer’s instructions.
Statistical Analysis
All variables were expressed as mean ± standard deviation (SD). One-way ANOVA and post hoc tests were used for comparison between multiple groups. All statistical analyses were performed using R (http://www.R-project.org, version 3.6.1) and GraphPad Prism 8. P values of less than 0.05 were considered to be statistically significant.
Results
Validation of BMSCHOXB4
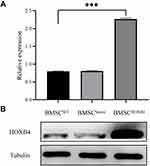
The efficiency of lentivirus-transfected rat BMSCs overexpressing HOXB4 was analyzed by Western blotting. As shown in Figure 1A and B, the expression of HOXB4 was significantly increased in BMSCHOXB4 group compared to that in BMSCWT group; there was no difference in HOXB4 expression between the BMSCWT and BMSCVector groups.
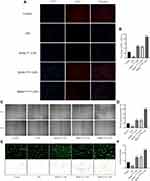
BMSCHOXB4 Coculture Promotes the Proliferation, Migration, and Tube Formation Ability of ECs
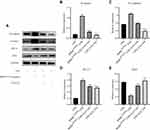
The EdU proliferation assay results showed that the proliferation capacity of ECs was remarkably reduced after LPS stimulation compared to that in the control group (P <0.01). After co-culturing with different groups of BMSCs, EC proliferation capacity was differentially increased, and BMSCHOXB4 coculture dramatically promoted EC proliferation capacity after LPS-induced injury compared to BMSCWT and BMSCVector coculture groups (P <0.01) (Figure 2A and B). The scratch assay results revealed that the migration ability of ECs decreased after LPS stimulation and increased after co-culturing with different groups of BMSCs. Compared to BMSCWT and BMSCVector coculture groups, the migration ability of ECs was significantly increased in the BMSCHOXB4 coculture group (P <0.01) (Figure 2C and D). Finally, to assess the role of BMSCHOXB4 in angiogenesis in vitro, EC tube-formation assay was performed. As seen in Figure 2E, there was almost no tube formation in the LPS group. However, coculture with BMSCHOXB4 significantly promoted the EC tube formation ability compared to that in BMSCWT and BMSCVector coculture groups (P <0.01) (Figure 2F).
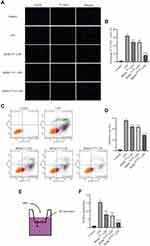
BMSCHOXB4 Coculture Attenuates the Apoptosis and Vascular Permeability of ECs
TUNEL and AV-FITC/PI assays were used to assess the role of BMSCHOXB4 in LPS-induced EC apoptosis. EC apoptosis was significantly increased upon LPS stimulation, as evidenced by an increase in TUNEL-positive cells; BMSC coculture demonstrated varying degrees of anti-EC apoptosis; as compared to BMSCWT and BMSCVector coculture groups, BMSCHOXB4 remarkably attenuated EC apoptosis (P <0.01) (Figure 3A and B). Similarly, the AV-FITC/PI assay results showed a remarkable reduction in the apoptosis rate in BMSCHOXB4 coculture group (apoptosis rate: 28.62%) compared to that in BMSCWT (apoptosis rate: 44.31%) and BMSCVector (apoptosis rate: 43.10%) coculture groups (P <0.01) (Figure 3C and D). Vascular permeability is one of the indicators referring to the barrier function of ECs. Hence, we measured the vascular permeability by endothelial cell leakage assay (Figure 3E), and found that the permeability of ECs was significantly increased after LPS treatment and greatly decreased after coculture with BMSCs, where the vascular permeability of BMSCHOXB4 coculture group was markedly lower than that of BMSCWT and BMSCVector coculture groups (P <0.01) (Figure 3F).
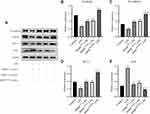
BMSCHOXB4 Coculture Protects Against LPS-Induced EC Injury by Activating the Wnt/β-Catenin Pathway
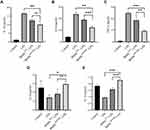
To investigate the molecular mechanism underlying the protective effect of HOXB4 on ECs, we assessed the expression of β-catenin, VE-cadherin, BAX, and BCL-2 using Western blotting (Figure 4A). Compared to LPS and BMSCWT coculture group, the expression of β-catenin and VE-cadherin in ECs was significantly increased in the BMSCHOXB4 coculture group; the expression of anti-apoptotic protein BCL-2 was considerably increased, while the expression of the apoptotic protein BAX was remarkably decreased (Figure 4B–E). Notably, a reduced expression level of β-catenin was observed in LPS group compared to that in the control group (Figure 4B), implying that LPS-induced EC injury is closely associated with the inhibition of Wnt/β-catenin pathway. Collectively, these findings prompted us to investigate whether BMSCHOXB4 coculture regulates the Wnt/β-catenin pathway in LPS-induced EC injury (Figure 5A). Interestingly, our study showed that LPS-induced inhibition of the Wnt/β-catenin pathway was blunted by BMSCHOXB4 coculture, resulting in substantially higher β-catenin levels than those in the LPS group (P <0.01) (Figure 5B). Conversely, the protective effect was reverted by XAV-939, a specific inhibitor of the Wnt/β-catenin pathway, accompanied by an upregulation of BAX, along with a downregulation of BCL-2 and VE-cadherin (Figure 5C–E).
Measurement of Inflammatory Factors
The levels of inflammatory factors in the cell culture medium were measured by ELISA (Figure 6). After LPS administration, the pro-inflammatory factors (IL-1β, IL-6, and TNF-α) were elevated, and after coculturing with BMSCs, the levels of IL-1β, IL-6, and TNF-α were differentially decreased (Figure 6A–C). When compared to BMSCWT, the levels of IL-6 (P <0.001) and TNF-α (P <0.01) were dramatically lower in the BMSCHOXB4 coculture group, while the levels of IL-1β did not differ in BMSCHOXB4 and BMSCWT coculture groups (P >0.05). Similarly, the anti-inflammatory factors IL-4 and IL-10 were significantly reduced after LPS stimulation (Figure 6D and E), and BMSCHOXB4 coculture significantly increased IL-10 levels compared to BMSCWT coculture group (P <0.001), but IL-4 levels were not statistically different in BMSCHOXB4 and BMSCWT coculture groups (P >0.05).
Discussion
Throughout the last two decades, significant advances have been made in elucidating the molecular mechanisms involved in ALI/ARDS. Nonetheless, the pathways that regulate vascular stability have been relatively poorly characterized. In this study, we determined the protective effects of BMSCHOXB4 coculture on LPS-induced ECs via the Wnt/β-catenin pathway, including antiapoptosis, promoting proliferation, boosting tube formation, as well as maintaining permeability and immunomodulatory effects.
Over the recent years, the therapeutic effects of BMSCs have been observed in a wide variety of diseases, such as sepsis, myocardial infarction, and trauma, all of which have led to advances in cellular therapies that have attracted an interest in the therapeutic use of BMSCs.26–28 Originally, the therapeutic effects of BMSCs were primarily focused on direct restorative effects on impaired cells and tissues.29,30 With the progress of cellular technology, it was identified that the beneficial effects of BMSCs do not require direct cellular contact. BMSCs are predominantly engaged in the repair of cellular damage through the paracrine mechanism, in which contents contain, but not limited to, exosomes.31–33 In our study, the non-direct contact beneficial effects of BMSCs were also observed through the transwell coculture regime. Notably, we first overexpressed HOXB4 in BMSCs and observed a potentially greater protective effect compared to that in wild-type BMSCs, this finding may aid in strengthening and optimizing the therapeutic effect of BMSCs. Otherwise, the most recent research on HOXB4 is related to embryonic stem cells and hematopoietic stem cells, whereas the studies on HOXB4 and BMSCs were rarely available.34–36 Our study revealed that HOXB4 positively modulated the Wnt/β-catenin pathway, which is a canonical signaling pathway with pivotal functions in cell growth and differentiation.37 In particular, β-catenin is a polyfunctional protein that interacts with VE-cadherin at the cell junctions and contributes to the construction of intercellular barriers. The free-form β-catenin is accessible to the nucleus for the modulation of gene expression, and abnormal expression or activation of this free-form β-catenin causally triggers tumors.38–40 Among the previous studies, the biological functions of β-catenin in cellular and animal models of LPS-induced injury have yielded controversial results. Some studies found that β-catenin expression was decreased following LPS exposure and then increased by interventions that exerted a range of beneficial effects, indicating that β-catenin has a positive regulatory effect on cell viability;41–43 similarly, in animal experiments, substantial evidence suggested that activation of the Wnt pathway protects against LPS-induced ALI, as evidenced by Cai et al who found that activation of the Wnt/β-catenin pathway increased preservation of MSCs in the lungs, promoted differentiation of MSCs to AT II cells, and reduced lung inflammation, as well as inhibited lung fibrosis, compared to that in wild-type MSCs.44 Likewise, Zhang et al and Villar et al found a beneficial therapeutic effect of Wnt/β-catenin activation on ALI.45,46 Alternatively, other studies held opposite opinions, as they found that β-catenin expression is increased after LPS treatment and the therapeutic effects are mediated via interventions that reduce β-catenin expression, suggesting that β-catenin is a comparatively detrimental agent for cell survival.47–49 For example, Cheng et al found that activation of β-catenin promotes LPS-induced ALI by driving the Th17 response in mice, whereas LPS-induced ALI and lung inflammation were somewhat ameliorated by the use of the Wnt/β-catenin pathway inhibitor, dickkopf1.50 Sun et al also revealed that the inhibition of aberrantly activated Wnt/β-catenin signaling in rats would promote epithelial differentiation of MSCs, thereby repairing lung injury and reducing lung fibrosis.51 Villar et al similarly found that Wnt/β-catenin was abnormally activated in patients with early sepsis-related ARDS and was associated with lung inflammation and pro-fibrosis.52 In our study, we found that β-catenin expression decreased after LPS treatment and increased after coculturing with BMSCs, which in turn exhibited a therapeutic effect on the cells. In fact, the contradictory findings in the studies on β-catenin are mostly dependent on the cell type and the experimental approach. Furthermore, there is some interaction between the Wnt/β-catenin pathway and other signaling pathways, which allows for more accurate modulation of cellular responses in response to injury stimuli.53,54 As for example, in the case of inflammatory responses, the balancing of Wnt/β-catenin activity and inflammatory pathways plays a fundamental role in the maintenance of intracellular homeostasis in the face of LPS provocation, where active Wnt/β-catenin signaling inhibits pro-inflammatory responses and protects cells from devastating effects of inflammation; insufficient inflammatory responses may lead to downregulation of Wnt/β-catenin signaling, which enhances the immune response.55
In terms of clinical trials, from the first MSC clinical trials on ARDS to the COVID-19 pandemic, it appears from the published data that MSCs-based cell therapy for ARDS has an exciting and alluring prospect.16,19 The properties of MSCs, whether direct repair or paracrine effects, are beneficial effects that permeate the pathogenesis of ALI/ARDS. The therapeutic effect of MSCs is highly variable among individuals, and the species source, the mode of input, and the dose of MSCs can lead to erratic efficacy.56 The dose of MSCs is one of the factors affecting the efficacy, and the optimal dose depends on different diseases and severity as well as the route of input, which needs to be explored by clinical studies and dose escalation trials. In addition, the inherent properties of MSCs are equally fundamental to their therapeutic effects, and currently it is suggested that optimizing the therapeutic effects of MSCs by gene modification techniques has good prospects for clinical applications.57 In our study, we revealed for the first time that HOXB4 gene-modified MSCs could enhance protection against LPS-induced EC injury, which may contribute to the optimization of MSC-based therapies. However, animal-level studies are warranted to further confirm the therapeutic effects of HOXB4 gene-modified MSCs.
The existence of limitations in our study should not be overlooked. First, we could not determine whether the therapeutic effect was exosomally exerted, but only further revealed the non-contact therapeutic effect of BMSCs by using the transwell system. Nevertheless, we believe that HOXB4 plays a predominant role in this study mainly by way of extracellular vesicles carrying HOXB4 since we used transwell chambers with a pore size of 0.4 μm, where exosomes are 30 to 100 nm in diameter and microvesicles are 100 nm to 1 μm.58 Additionally, we did not perform RNA sequencing of exosomes in the BMSCHOXB4 group as well as in the BMSCWT group, so we were unable to ascertain whether there are any remaining potential paracrine substances that play a role in the BMSCHOXB4 group other than HOXB4. In a recent study, a significant reduction in the therapeutic effect of dental pulp stem cells on ECs was found after the use of the exosome secretion inhibitor, GW4869; however, the effect was still higher than that in the control group, even though the authors did not give a statistical analysis of the relevant data on this aspect. This could suggest that exosomes play a key role in the treatment, but not entirely.59 Second, we found that the therapeutic effect of the BMSCHOXB4 was better than that of BMSCWT only at the cellular stage; the effects in animals are yet to be elucidated. Third, as is well known, ALI/ARDS is not solely an incident of gram-negative bacterial infection, but a complex condition featured by gram-negative and gram-positive bacterial infections with systemic issues concerning the respiratory system, urinary system, gastrointestinal tract, and central nervous system.1 Consequently, the cellular model used in our study cannot thoroughly address the potential mechanisms of endothelial barrier dysfunction and cellular damage associated with ALI/ARDS. Nevertheless, at least, we were able to obtain a better insight into the effect exerted by gram-negative infections. Fourth, we could not ascertain the interaction between HOXB4 and Wnt/β-catenin pathway; whether it is a direct or indirect action is something that needs to be explored in future studies. However, previous research showed that HOXB4 directly acts on the Wnt/β-catenin pathway and exerts its anti-tumor effects by reducing Wnt/β-catenin activity.60
Conclusion
In summary, our study findings showed that LPS-induced EC injury is attributed to a reduction in β-catenin, whereas co-culturing with BMSCHOXB4 apparently reverses the inactivation of the Wnt/β-catenin signaling pathway. This suggests that BMSCs can protect ECs from LPS-induced cell injury by altering the Wnt/β-catenin signaling pathway. Ultimately, further in vivo studies are warranted to establish whether engraftment of BMSCHOXB4 can be an attractive strategy for the treatment of ALI/ARDS.
Abbreviations
BMSC, bone marrow mesenchymal stem cell; HOXB4, homeobox B4; EC, endothelial cell; IL, interleukin; TNF-α, tumor necrosis factor-α; LPS, lipopolysaccharide; ALI/ARDS, acute lung injury/acute respiratory distress syndrome; COVID-19, coronavirus disease 2019; EdU, 5-ethynyl-20-deoxyuridine; TUNEL, TdT mediated dUTP Nick-End Labeling; AV-FITC/PI, Annexin V-fluorescein isothiocyanate/propidium iodide; MOI, multiplicity of infection; DMEM, Dulbecco’s modified Eagle’s medium; FBS, fetal bovine serum; WT, wild-type; PVDF, polyvinylidene fluoride; SDS-PAGE, sodium dodecyl sulfate polyacrylamide gel electrophoresis; PBS, phosphate-buffered saline; ELISA, enzyme linked immunosorbent assay.
Acknowledgments
Thanks to the researchers at the Research Center of Translational Medicine for their generous assistance with the experiments.
Funding
This work was supported by the National Natural Science Foundation of China [Grant number: 81670066]; the Critical Care Research Funding of the Aesculap Academy (2017); and the Guangdong Basic and Applied Basic Research Foundation, China [Grant number: 2019A1515011198]; the Guangdong Science and Technology Program [Grant number: 2019A030317003].
Disclosure
The authors report no conflicts of interest.
References
1. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA. 2016;315(8):801–810. doi:10.1001/jama.2016.0287
2. van der Poll T, Opal SM. Host-pathogen interactions in sepsis. Lancet Infect Dis. 2008;8(1):32–43. doi:10.1016/S1473-3099(07)70265-7
3. Levi M, van der Poll T. Inflammation and coagulation. Crit Care Med. 2010;38(2 Suppl):S26–S34. doi:10.1097/CCM.0b013e3181c98d21
4. Hotchkiss RS, Nicholson DW. Apoptosis and caspases regulate death and inflammation in sepsis. Nat Rev Immunol. 2006;6(11):813–822. doi:10.1038/nri1943
5. Thompson BT, Chambers RC, Liu KD. Acute respiratory distress syndrome. N Engl J Med. 2017;377(6):562–572. doi:10.1056/NEJMra1608077
6. Millar FR, Summers C, Griffiths MJ, Toshner MR, Proudfoot AG. The pulmonary endothelium in acute respiratory distress syndrome: insights and therapeutic opportunities. Thorax. 2016;71(5):462–473. doi:10.1136/thoraxjnl-2015-207461
7. Huppert LA, Matthay MA, Ware LB. Pathogenesis of acute respiratory distress syndrome. Semin Respir Crit Care Med. 2019;40(1):31–39. doi:10.1055/s-0039-1683996
8. Huppert LA, Matthay MA. Alveolar fluid clearance in pathologically relevant conditions: in vitro and in vivo models of acute respiratory distress syndrome. Front Immunol. 2017;7(8):371.
9. Fan E, Brodie D, Slutsky AS. Acute respiratory distress syndrome: advances in diagnosis and treatment. JAMA. 2018;319(7):698–710. doi:10.1001/jama.2017.21907
10. Bianco P, Robey PG, Simmons PJ. Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell. 2008;2(4):313–319. doi:10.1016/j.stem.2008.03.002
11. Gupta N, Su X, Popov B, Lee JW, Serikov V, Matthay MA. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179(3):1855–1863. doi:10.4049/jimmunol.179.3.1855
12. Gupta N, Krasnodembskaya A, Kapetanaki M, et al. Mesenchymal stem cells enhance survival and bacterial clearance in murine Escherichia coli pneumonia. Thorax. 2012;67(6):533–539. doi:10.1136/thoraxjnl-2011-201176
13. Lee JW, Krasnodembskaya A, McKenna DH, Song Y, Abbott J, Matthay MA. Therapeutic effects of human mesenchymal stem cells in ex vivo human lungs injured with live bacteria. Am J Respir Crit Care Med. 2013;187(7):751–760. doi:10.1164/rccm.201206-0990OC
14. Wilson JG, Liu KD, Zhuo H, et al. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015;3(1):24–32. doi:10.1016/S2213-2600(14)70291-7
15. Liu KD, Wilson JG, Zhuo H, et al. Design and implementation of the START (STem cells for ARDS treatment) trial, a phase 1/2 trial of human mesenchymal stem/stromal cells for the treatment of moderate-severe acute respiratory distress syndrome. Ann Intensive Care. 2014;4:22. doi:10.1186/s13613-014-0022-z
16. Zheng G, Huang L, Tong H, et al. Treatment of acute respiratory distress syndrome with allogeneic adipose-derived mesenchymal stem cells: a randomized, placebo-controlled pilot study. Respir Res. 2014;15(1):39. doi:10.1186/1465-9921-15-39
17. Lopes-Pacheco M, Robba C, Rocco PRM, Pelosi P. Current understanding of the therapeutic benefits of mesenchymal stem cells in acute respiratory distress syndrome. Cell Biol Toxicol. 2020;36(1):83–102. doi:10.1007/s10565-019-09493-5
18. Sengupta V, Sengupta S, Lazo A, Woods P, Nolan A, Bremer N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020;29(12):747–754. doi:10.1089/scd.2020.0080
19. Leng Z, Zhu R, Hou W, et al. Transplantation of ACE2- mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis. 2020;11(2):216–228. doi:10.14336/AD.2020.0228
20. Antonchuk J, Sauvageau G, Humphries RK. HOXB4-induced expansion of adult hematopoietic stem cells ex vivo. Cell. 2002;109(1):39–45. doi:10.1016/S0092-8674(02)00697-9
21. Grässel S, Stöckl S, Jenei-Lanzl Z. Isolation, culture, and osteogenic/chondrogenic differentiation of bone marrow-derived mesenchymal stem cells. Methods Mol Biol. 2012;879:203–267.
22. Li B, Zhang H, Zeng M, et al. Bone marrow mesenchymal stem cells protect alveolar macrophages from lipopolysaccharide-induced apoptosis partially by inhibiting the Wnt/β-catenin pathway. Cell Biol Int. 2015;39(2):192–200. doi:10.1002/cbin.10359
23. Zeng M, Chen Q, Ge S, et al. Overexpression of FoxM1 promotes differentiation of bone marrow mesenchymal stem cells into alveolar type II cells through activating Wnt/β-catenin signalling. Biochem Biophys Res Commun. 2020;528(2):311–317. doi:10.1016/j.bbrc.2020.05.042
24. Huang C, Zheng H, He W, et al. Ghrelin ameliorates the human alveolar epithelial A549 cell apoptosis induced by lipopolysaccharide. Biochem Biophys Res Commun. 2016;474(1):83–90. doi:10.1016/j.bbrc.2016.04.074
25. Chen HR, Chuang YC, Lin YS, et al. Dengue virus nonstructural protein 1 induces vascular leakage through macrophage migration inhibitory factor and autophagy. PLoS Negl Trop Dis. 2016;10(7):e0004828. doi:10.1371/journal.pntd.0004828
26. Mei SH, Haitsma JJ, Dos Santos CC, et al. Mesenchymal stem cells reduce inflammation while enhancing bacterial clearance and improving survival in sepsis. Am J Respir Crit Care Med. 2010;182(8):1047–1057. doi:10.1164/rccm.201001-0010OC
27. Timmers L, Lim SK, Hoefer IE, et al. Human mesenchymal stem cell-conditioned medium improves cardiac function following myocardial infarction. Stem Cell Res. 2011;6(3):206–214. doi:10.1016/j.scr.2011.01.001
28. Tanrıverdi AK, Polat O, Elçin AE, et al. Mesenchymal stem cell transplantation in polytrauma: evaluation of bone and liver healing response in an experimental rat model. Eur J Trauma Emerg Surg. 2020;46(1):53–64. doi:10.1007/s00068-019-01101-9
29. Wong AP, Dutly AE, Sacher A, et al. Targeted cell replacement with bone marrow cells for airway epithelial regeneration. Am J Physiol Lung Cell Mol Physiol. 2007;293(3):L740–L752. doi:10.1152/ajplung.00050.2007
30. Liechty KW, MacKenzie TC, Shaaban AF, et al. Human mesenchymal stem cells engraft and demonstrate site-specific differentiation after in utero transplantation in sheep. Nat Med. 2000;6(11):1282–1286. doi:10.1038/81395
31. Morrison TJ, Jackson MV, Cunningham EK, et al. Mesenchymal stromal cells modulate macrophages in clinically relevant lung injury models by extracellular vesicle mitochondrial transfer. Am J Respir Crit Care Med. 2017;196(10):1275–1286. doi:10.1164/rccm.201701-0170OC
32. Phinney DG, Di Giuseppe M, Njah J, et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat Commun. 2015;6:8472. doi:10.1038/ncomms9472
33. Zhu YG, Feng XM, Abbott J, et al. Human mesenchymal stem cell microvesicles for treatment of Escherichia coli endotoxin-induced acute lung injury in mice. Stem Cells. 2014;32(1):116–125. doi:10.1002/stem.1504
34. Fan R, Bonde S, Gao P, et al. Dynamic HoxB4-regulatory network during embryonic stem cell differentiation to hematopoietic cells. Blood. 2012;119(19):e139–e147. doi:10.1182/blood-2011-12-396754
35. Wang L, Menendez P, Shojaei F, et al. Generation of hematopoietic repopulating cells from human embryonic stem cells independent of ectopic HOXB4 expression. J Exp Med. 2005;201(10):1603–1614. doi:10.1084/jem.20041888
36. Lee GS, Kim BS, Sheih JH, Moore M. Forced expression of HoxB4 enhances hematopoietic differentiation by human embryonic stem cells. Mol Cells. 2008;25(4):487–493.
37. MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17(1):9–26. doi:10.1016/j.devcel.2009.06.016
38. Liebner S, Corada M, Bangsow T, et al. Wnt/beta-catenin signaling controls development of the blood-brain barrier. J Cell Biol. 2008;183(3):409–417. doi:10.1083/jcb.200806024
39. Fodde R, Brabletz T. Wnt/beta-catenin signaling in cancer stemness and malignant behavior. Curr Opin Cell Biol. 2007;19(2):150–158. doi:10.1016/j.ceb.2007.02.007
40. Maretto S, Cordenonsi M, Dupont S, et al. Mapping Wnt/beta-catenin signaling during mouse development and in colorectal tumors. Proc Natl Acad Sci USA. 2003;100(6):3299–3304. doi:10.1073/pnas.0434590100
41. Zhang W, Zhang Y, Guo X, et al. Sirt1 protects endothelial cells against LPS-induced barrier dysfunction. Oxid Med Cell Longev. 2017;2017:4082102. doi:10.1155/2017/4082102
42. Li B, Zeng M, He W, et al. Ghrelin protects alveolar macrophages against lipopolysaccharide-induced apoptosis through growth hormone secretagogue receptor 1a-dependent c-Jun N-terminal kinase and Wnt/-catenin signaling and suppresses lung inflammation. Endocrinology. 2015;156(1):203–217. doi:10.1210/en.2014-1539
43. Li Y, Shi J, Yang J, et al. A Wnt/β-catenin negative feedback loop represses TLR-triggered inflammatory responses in alveolar epithelial cells. Mol Immunol. 2014;59(2):128–135. doi:10.1016/j.molimm.2014.02.002
44. Cai SX, Liu AR, Chen S, et al. Activation of Wnt/β-catenin signalling promotes mesenchymal stem cells to repair injured alveolar epithelium induced by lipopolysaccharide in mice. Stem Cell Res Ther. 2015;6(1):65. doi:10.1186/s13287-015-0060-y
45. Zhang J, Shao Y, He D, Zhang L, Xu G, Shen J. Evidence that bone marrow-derived mesenchymal stem cells reduce epithelial permeability following phosgene-induced acute lung injury via activation of wnt3a protein-induced canonical wnt/β-catenin signaling. Inhal Toxicol. 2016;28(12):572–579. doi:10.1080/08958378.2016.1228720
46. Villar J, Cabrera NE, Casula M, et al. WNT/β-catenin signaling is modulated by mechanical ventilation in an experimental model of acute lung injury. Intensive Care Med. 2011;37(7):1201–1209. doi:10.1007/s00134-011-2234-0
47. Monick MM, Carter AB, Robeff PK, Flaherty DM, Peterson MW, Hunninghake GW. Lipopolysaccharide activates Akt in human alveolar macrophages resulting in nuclear accumulation and transcriptional activity of beta-catenin. J Immunol. 2001;166(7):4713–4720. doi:10.4049/jimmunol.166.7.4713
48. Lee H, Bae S, Choi BW, Yoon Y. WNT/β-catenin pathway is modulated in asthma patients and LPS-stimulated RAW264.7 macrophage cell line. Immunopharmacol Immunotoxicol. 2012;34(1):56–65. doi:10.3109/08923973.2011.574704
49. Gong K, Zhou F, Huang H, Gong Y, Zhang L. Suppression of GSK3β by ERK mediates lipopolysaccharide induced cell migration in macrophage through β-catenin signaling. Protein Cell. 2012;3(10):762–768. doi:10.1007/s13238-012-2058-x
50. Cheng L, Zhao Y, Qi D, Li W, Wang D. Wnt/β-catenin pathway promotes acute lung injury induced by LPS through driving the Th17 response in mice. Biochem Biophys Res Commun. 2018;495(2):1890–1895. doi:10.1016/j.bbrc.2017.12.058
51. Sun Z, Gong X, Zhu H, et al. Inhibition of Wnt/β-catenin signaling promotes engraftment of mesenchymal stem cells to repair lung injury. J Cell Physiol. 2014;229(2):213–224. doi:10.1002/jcp.24436
52. Villar J, Cabrera-Benítez NE, Ramos-Nuez A, et al. Early activation of pro-fibrotic WNT5A in sepsis-induced acute lung injury. Crit Care. 2014;18(5):568. doi:10.1186/s13054-014-0568-z
53. Ma B, Hottiger MO. Crosstalk between Wnt/β-catenin and NF-κB signaling pathway during inflammation. Front Immunol. 2016;7:378. doi:10.3389/fimmu.2016.00378
54. McNeill H, Woodgett JR. When pathways collide: collaboration and connivance among signalling proteins in development. Nat Rev Mol Cell Biol. 2010;11(6):404–413. doi:10.1038/nrm2902
55. Blumenthal A, Ehlers S, Lauber J, et al. The Wingless homolog WNT5A and its receptor Frizzled-5 regulate inflammatory responses of human mononuclear cells induced by microbial stimulation. Blood. 2006;108(3):965–973. doi:10.1182/blood-2005-12-5046
56. Mastri M, Lin H, Lee T. Enhancing the efficacy of mesenchymal stem cell therapy. World J Stem Cells. 2014;6(2):82–93. doi:10.4252/wjsc.v6.i2.82
57. Kumar S, Chanda D, Ponnazhagan S. Therapeutic potential of genetically modified mesenchymal stem cells. Gene Ther. 2008;15(10):711–715. doi:10.1038/gt.2008.35
58. Mathivanan S, Ji H, Simpson RJ. Exosomes: extracellular organelles important in intercellular communication. J Proteomics. 2010;73(10):1907–1920. doi:10.1016/j.jprot.2010.06.006
59. Zhou H, Li X, Yin Y, et al. The proangiogenic effects of extracellular vesicles secreted by dental pulp stem cells derived from periodontally compromised teeth. Stem Cell Res Ther. 2020;11(1):110. doi:10.1186/s13287-020-01614-w
60. Lei D, Yang WT, Zheng PS. HOXB4 inhibits the proliferation and tumorigenesis of cervical cancer cells by downregulating the activity of Wnt/β-catenin signaling pathway. Cell Death Dis. 2021;12(1):105. doi:10.1038/s41419-021-03411-6
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.