Back to Journals » Local and Regional Anesthesia » Volume 8
Out-of-plane ultrasound-guided paravertebral blocks improve analgesic outcomes in patients undergoing video-assisted thoracoscopic surgery
Authors Amlong C, Guy M, Schroeder K, Donnelly M
Received 18 April 2015
Accepted for publication 11 August 2015
Published 15 December 2015 Volume 2015:8 Pages 123—128
DOI https://doi.org/10.2147/LRA.S86853
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Stefan Wirz
Corey Amlong, Moltu Guy, Kristopher M Schroeder, Melanie J Donnelly
Department of Anesthesiology, University of Wisconsin Hospital and Clinics, Madison, WI, USA
Purpose: Paravertebral blocks (PVBs) are a method of limiting postoperative pain for patients undergoing video-assisted thoracoscopic surgery (VATS). We began providing ultrasound-guided PVBs for patients undergoing VATS in the spring of 2011, using an out-of-plane approach. The aim of this study was to evaluate this practice change.
Methods: Following institutional review board approval, we reviewed the charts of 114 patients undergoing VATS by one surgeon at our institution between January 2011 and July 2012. Of the 78 eligible patients, 49 patients received a PVB prior to surgery. We evaluated opioids administered in the perioperative period, pain scores, and side effects from pain medications.
Results: Patients who received a preoperative PVB required fewer narcotics intraoperatively and during their hospital stay (P=0.001 and 0.011, respectively). Pain scores on initial assessment and in recovery were lower in patients who received a PVB (P=0.005), as were dynamic and resting pain scores at 24 hours after surgery (P=0.003 and P<0.001, respectively). Patients receiving a PVB had fewer episodes of treated nausea both in the postanesthesia care unit (P=0.004) and for the first 24 hours after surgery (P=0.001). These patients also spent less time in recovery (P=0.025) than the patients who did not receive a block.
Conclusion: The current study suggests improved outcomes in patients who underwent VATS with a preoperative PVB. All variables showed a trend toward improved results in patients who obtained a preoperative PVB.
Keywords: nerve blocks, video-assisted thoracoscopic surgery, anesthesia, pain
Introduction
Video-assisted thoracoscopic surgery (VATS) has increased in popularity worldwide. The benefits of VATS include decreased postoperative pain, improved cosmetic surgical wounds, decreased morbidity, and shorter hospital stays when compared to open thoracic procedures.1–3
Pain associated with thoracic surgery is multifactorial and involves muscular pain, neuropathic pain, pleural irritation, and referred pain. While patients undergoing VATS have been shown to have less acute pain compared to thoracotomy,2,4,5 the development of chronic pain is similar.6–10 Furthermore, the degree of acute post-thoracotomy pain has been shown to predict the presence and significance of chronic pain related to these procedures,6 increasing the importance of acute postoperative pain control. Paravertebral blocks (PVBs) are a validated method of limiting postoperative pain for patients undergoing VATS and other thoracic procedures.11–17 Our institution began providing ultrasound-guided PVBs for patients undergoing VATS in the spring of 2011 with positive feedback from both patients and surgeons. Unlike many published descriptions of ultrasound-guided PVB, in which the operator approaches the PVB space in-plane to the ultrasound beam with the probe held in a transverse mediolateral fashion, we utilize an “out-of-plane” approach with the ultrasound probe in a parasagittal plane combined with intermittent low-volume hydrodissection to guide needle advancement. The aim of this study was to retrospectively evaluate the analgesic efficacy of the preoperative PVBs provided for patients undergoing VATS.
Materials and methods
Following approval of an exemption application from the University of Wisconsin Health Sciences Institutional Review Board, we used a billing database to identify consecutive adult patients undergoing VATS by a single thoracic surgeon at our institution between January 2011 and July 2012. A retrospective review of these patients’ electronic medical records was performed. Patients who underwent additional procedures at the time of the VATS, required postoperative care and continued mechanical ventilation in an intensive care unit, had chronic pain as defined as a baseline preoperative pain score of 5 or greater, were under the age of 18, or who presented with injuries sustained as part of trauma were excluded from our analyses. Additionally, two patients were excluded secondary to postoperative death attributed to long-standing hepatic disease and postoperative stroke attributed to the patient’s history of cardiovascular disease. Neither patient who died was in the block group. Of the 78 eligible patients, 49 patients received a PVB prior to their planned surgical procedure. All patients on the operating room schedule during the day were considered for a nerve block. Reasons for exclusion of patients would include hemodynamic instability, patient intubated and sedated already, coagulopathy, extreme obesity, prior back surgery, and a patient with active infection. The block occurred in a dedicated holding room and was performed by either a resident or a fellow, and a staff anesthesiologist assigned to the block team. Blocks performed by residents or fellows were done under the direct supervision of a staff anesthesiologist on the regional anesthesia team. Blocks were performed with minimal sedation using intravenous midazolam (1–2 mg) and fentanyl (50–100 μg). After assisting the patient into the sitting position, ultrasound guidance in the sagittal plane was used to identify transverse process, costotransverse ligament, and pleura on the side of the surgery at levels appropriate based on the surgeon’s proposed port and chest tube sites. Once ultrasound visualization was adequate, the patient’s skin was infiltrated with 2% lidocaine (1–3 mL), and a 22-gauge (50 mm) or 21-gauge (100 mm) UniPlex NanoLine regional anesthesia needle (Pajunk® Medizin Technologie, Geisingen, Germany) was inserted out-of-plane relative to the ultrasound probe. Hydrodissection with sterile saline was used as the needle was advanced until anterior pleural deflection was visualized on ultrasound. Bupivacaine (0.5%) with 2.5 μg/mL epinephrine was injected in divided doses after negative aspiration. In total, 10–20 mL of bupivacaine was injected at each level, with most patients receiving 10 mL or 15 mL per level. Total block volume from all levels was 25–30 mL in most cases, although a single patient received a total of 40 mL. All patients received blocks at either two or three levels ranging from T3–4 to T6–7 on the side ipsilateral to the proposed procedure. Using patients’ electronic medical records, the amount of opioids administered intraoperatively, in the postanesthesia care unit (PACU), for the first 24 hours after discharge from the PACU and during the hospital stay were recorded. To facilitate comparisons, we converted all administered opioids to intravenous morphine equivalents using standard conversion factors as follows: morphine – 10, hydromorphone – 1.5, fentanyl – 0.1, and meperidine – 75.18 The use of additional analgesics, including nonopioid intravenous and oral analgesics, and intraoperative incision infiltration by the surgeon were also recorded.
Once the patient was admitted to the floor, the patient reported numerical pain scores ranging from 0 (least pain) to 10 (most pain) that were recorded by nursing staff. Patient’s pain was frequently reevaluated by nursing staff throughout the patient’s hospital stay. The following patient-reported numerical pain scores were recorded or calculated using medical records: patients’ initial pain score on arriving to the PACU, highest pain score while in the PACU, mean pain score while in the PACU, pain score on initial assessment once the patient reached the floor, resting and dynamic pain scores at 24 hours after discharge from the PACU, and mean resting and dynamic pain scores for the first 24 hours following discharge from the PACU. Mean pain scores were computed using all-nurse recorded patient-reported numerical pain scores during the respective time frame. The amount of time spent in the PACU for each patient was also recorded. PACU time was defined as the amount of time the patient was physically present in the PACU before meeting nursing discharge criteria. All patients undergoing VATS procedures, with or without a preoperative PVB, received pain medications ordered by the surgery and anesthesia team in the perioperative period. Intraoperative opioids were administered at the discretion of the in-room anesthesia staff based on clinical signs of inadequate analgesia such as increased heart rate and blood pressure during the procedure. Opioids in the PACU were administered at the patient’s request or based on the patient’s verbal pain score at the discretion of the PACU nurses. Once admitted to the floor, pain medications were administered at the discretion of the surgical service and floor nurses. The standard pain medication regimen included a hydromorphone patient controlled analgesic pump with a nurse bolus available. Patients were ultimately advanced to oral opioids, usually in the form of oxycodone. Most patients were placed on scheduled oral acetaminophen unless contraindicated, and most received scheduled doses of either intravenous or oral nonsteroidal anti-inflammatory drug (NSAID) medication. Lastly, the presence or absence of postoperative nausea or vomiting was recorded. Patients who received a rescue antiemetic in either the PACU or within the first 24 hours postoperatively were considered to have postoperative nausea. Our primary interest was whether PVBs led to improved pain scores and reduced opioid consumption. Intergroup comparisons (PVB vs no PVB) were performed using the Student’s t-test, Fisher’s exact test, and Mann–Whitney U test for continuous, binary, and ordinal variables, respectively. An independent statistician performed statistical analysis of data. A P-value of <0.05 was considered significant.
Results
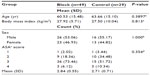
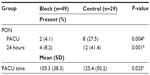
The charts of 114 patients who underwent unilateral VATS procedures were reviewed. Patients were excluded from our analysis for the following reasons: baseline pain score of >5 either from pain related to reason for surgery or from an unrelated chronic issue (n=17), remained intubated postoperatively (n=12), presented as a multiple injury trauma (n=4), deceased on postoperative day 1 (n=1), stroke on postoperative day 0 (n=1), and had a prolonged narcotic infusion lasting into PACU as part of a total-intravenous anesthetic (n=1). Of the 78 eligible patients, 49 received an ultrasound-guided PVB, while the remaining 29 did not. Chart review revealed that among the patients receiving a PVB, four received a block at three levels, while the remaining 45 patients received a block at two levels. Two three-level blocks were performed at levels T4, 5, and 6, one at T1, 3, and 5, and one at T2, 3, and 5. Twenty-six patients received a two-level block at levels T4 and 6, nine at T3 and 5, five at T5 and 7, four at T3 and 6, and a single patient received a block at T1 and 5. The two groups (PVB vs no PVB) were comparable in terms of patient age, sex (%), body mean index, and American Society of Anesthesiologists score (Table 1). Our analysis showed that all patients received as-needed intravenous opioids, frequently in the form of patient-controlled analgesia. All but six patients in the PVB group and five patients in the control group additionally received scheduled acetaminophen once admitted to the floor. Results in Table 2 show that mean intraoperative opioid consumption in morphine equivalents was significantly lower for patients who received preoperative PVB compared to those who did not (P=0.001). Mean opioid consumption in the PACU and for the first 24 hours after surgery appeared to be less in the PVB group compared to controls; however, this small difference failed to achieve statistical significance (P=0.246 and 0.467, respectively). Mean opioid consumption for the duration of hospital stay was significantly lower in patients who received PVB (P=0.011). Patient-reported numerical pain scores immediately upon arrival to the PACU were significantly lower in the PVB group (P=0.005; Table 3). Peak pain scores in the PACU (P=0.097) and mean pain scores in the PACU (P=0.061) were lower in the PVB group but also failed to reach statistical significance. Pain scores on initial assessment once patients reached the floor were also significantly lower in patients receiving a PVB (P=0.031). Both dynamic and resting pain scores at the 24-hour time point after surgery were significantly lower in the PVB group (P=0.003 and P<0.001, respectively); however, mean dynamic and rest pain scores over this 24-hour period, while lower in the PVB group, failed to reach statistical significance (P=0.089 and P=0.078, respectively). Patients who received preoperative PVB had a lower incidence of nausea requiring treatment both in the PACU (4% vs 28%, P=0.004) and for the first 24 hours after PACU discharge (8% vs 43%, P=0.001) compared to the control group (Table 4). Lastly, patients who received PVB spent significantly less time in the PACU before meeting discharge criteria compared to those who did not receive PVB (105 vs 125 minutes; P=0.025). We found that 40 of 49 (82%) patients in the block group received scheduled postoperative acetaminophen, while 25 of 29 (86%) patients who did not receive a block received scheduled acetaminophen. Thirteen of 49 patients (27%) in the block group received at least one dose of scheduled NSAID medication in the form of intravenous or oral ibuprofen, diclofenac, or ketorolac. Six of 29 patients (21%) who did not receive a block received NSAID medication. Lastly, 24 of 49 patients (49%) in the block group received intraoperative wound infiltration with 0.25% bupivacaine by the surgeon, while 24 of 29 patients (83%) in the control group received intraoperative local anesthetic wound infiltration.
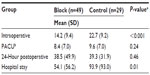  | Table 2 Opioid requirements in intravenous morphine equivalents (mg) |
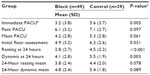  | Table 3 Patient-reported numerical pain scores |
Discussion
The findings of this study support the assertion that preoperative PVBs performed with an out-of-plane needle insertion technique improve analgesic outcomes in the immediate postoperative period and improve the quality of the anesthetic. Specifically, our patients had lower reported pain scores, lower perioperative opioid requirements, fewer incidences of treated postoperative nausea or vomiting, and shorter stays in the PACU. All analyzed outcomes showed a trend toward improved results in patients who obtained preoperative PVB compared to those who did not. Statistically, this fact is in itself significant and provides an objective correlation to the subjective positive feedback we have received from both surgeons and patients. The analgesic efficacy of PVBs in patients undergoing both video-assisted thoracoscopic procedures and open thorcatomies has been documented.11–14,16,17 Furthermore, the use of paravertebral catheters for postoperative pain control for thoracic procedures has also been investigated.12 Classically, PVBs have been placed using landmark techniques with or without nerve stimulation, often by walking the needle off of transverse process and advancing an additional 1 cm or until the “pop” of the transverse costal ligament is felt.19 More recently, ultrasound guidance has grown in popularity with several different techniques being used for block placement, most commonly placing the ultrasound probe in the transverse plane with injection of local anesthetic using an in-plane technique.20,21 Other ultrasound-guided techniques with out-of-plane needle placement rely on contact with the transverse process or loss of resistance to saline injection once the transverse costal ligament is passed to ensure block placement in the paravertebral space.22 The results of the present study demonstrate that using an out-of-plane ultrasound-guided method of single-injection PVB placement, we were able to improve our anesthetic via improved pain scores, less postoperative nausea, and less recovery time in the PACU.
One limitation of the current study is a lack of documented dermatomal sensory blockade after block placement. It is not our practice to check sensory levels following placement of these blocks, and therefore, the rate of epidural spread and block failure is unknown. In addition, the retrospective nature of this study and the bias that was introduced due to the unblinded nature of this study introduces bias. Providers may have expected less pain in the block patients, and therefore, gave less medication. Despite these issues, these data do provide further evidence of the analgesic efficacy of PVB in patients undergoing VATS procedures and introduce the possibility that a PVB catheter would add value by potentially prolonging our PACU results into postoperative day 1. In this chart review, we cannot determine how long the single-injection blocks might have lasted. The literature supports a duration of single-shot PVB ranging from 6 hours postoperatively to 12 hours or even longer. We know that the block lasted long enough to get the patients through the PACU, so perhaps by placing catheters, we can extend the analgesia in a meaningful way for patients. If we get improved analgesia on the floor, then perhaps we begin to impact the ability of patients to ambulate and avoid respiratory complications. Further prospective studies are warranted to determine the ideal analgesic regimen for VATS patients. The out-of-plane approach utilized at our institution may put patients at elevated risk of misplaced injection of local anesthetic, and this needs to be carefully considered. Though no adverse events from the PVBs were noted in our chart review, we clearly did not have enough patients to draw any conclusions regarding block safety.
Conclusion
In summary, the current study presents data suggesting that ultrasound-guided paravertebral blockade using an out-of-plane needle approach may offer a number of clinical benefits to patients undergoing VATS procedures. Further investigation is required to determine ideal analgesic regimen.
Acknowledgment
The authors would like to thank Christopher Warren for his assistance with statistics for this paper.
Disclosure
The authors report no conflicts of interest in this work.
References
Grogan EL, Jones DR. VATS lobectomy is better than open thoracotomy: what is the evidence for short-term outcomes? Thorac Surg Clin. 2008;18:249–258. | |
Landreneau RJ, Hazelrigg SR, Mack MJ, et al. Postoperative pain related morbidity: video assisted thoracoscopic surgery vs. thoracotomy. Ann Thorac Surg. 1993;56:1285–1289. | |
Cheng D, Downey RJ, Kernstine K, et al. Video-assisted thoracic surgery in lung cancer resection: a meta-analysis and systematic review of controlled trials. Innovations. 2007;2(6):261–292. | |
Nomori H, Horio H, Naruke T, Suemasu K. What is the advantage of a thoracoscopic lobectomy over a limited thoracotomy procedure for lung cancer surgery? Ann Thorac Surg. 2001;72:879–884. | |
Nagahiro I, Andou A, Aoe M, Sano Y, Date H, Shimizu N. Pulmonary function, postoperative pain, and serum cytokine level after lobectomy: a comparison of VATS and conventional procedure. Ann Thorac Surg. 2001;72:362–365. | |
Katz J, Jackson M, Kavanagh BP, Sandler A. Acute pain after thoracic surgery predicts longterm post-thoracotomy pain. Clin J Pain. 1996;12:50–55. | |
Oey IF, Morgan MDL, Waller DA. Postoperative pain detracts from early health status improvement seen after video-assisted thoracoscopic lung volume reduction surgery. Eur J Cardiothorac Surg. 2003;24:588–593. | |
Hutter J, Miller K, Moritz E. Chronic sequelae after thoracoscopic procedures for benign diseases. Eur J Cardiothorac Surg. 2000;17:687–690. | |
Passlick B, Born C, Mandelkow H, Sienel W, Thetter O. Long-term complaints after minimal invasive thoracic surgery operations and thoracotomy. Chirurg. 2001;72:934–939. | |
Steegers MAH, Snik DM, Verhagen AF, van der Drift M, Wilder-Smith O. Only half of the chronic pain after thoracic surgery shows a neuropathic component. J Pain. 2008;9:955–961. | |
Hill SE, Keller RA, Stafford-Smith M, et al. Efficacy of single dose, multi-level paravertebral nerve blockade for analgesia after thoracoscopic procedures. Anesthesiology. 2006;104:1047–1053. | |
Fibla JJ, Molins L, Mier JM, Sierra A, Carranza D, Vidal G. The efficacy of paravertebral block using a catheter technique for postoperative analgesia in thoracoscopic surgery: a randomized trial. Eur J Cardiothorac Surg. 2011;40(4):907–911. | |
Komatsu T, Sowa T, Takahashi K, Fujinaga T. Paravertebral block as a promising analgesic modality for managing post-thoracotomy pain. Ann Thorac Cardiovasc Surg. 2014;20(2):113–116. | |
Kaya FN, Turker G, Mogol EB, Bayraktar S. Thoracic paravertebral block for video-assisted thoracoscopic surgery: single injection versus multiple injections. J Cardiothorac Vasc Anesth. 2012;26(1):90–94. | |
Agarwal A, Batra RK, Chhabra A, Subramaniam R, Misra MC. The evaluation of efficacy and safety of paravertebral block for perioperative analgesia in patients undergoing laparoscopic cholecystectomy. Saudi J Anaesth. 2012;6(4):344–349. | |
Vogt A, Stieger DS, Theurillat C, Curatolo M. Single-injection thoracic paravertebral nerve block for postoperative pain after thoracoscopic surgery. Br J Anaesth. 2005;95:816–821. | |
Richardson J, Sabanathan S, Jones J, Shah RD, Cheema S, Mearns AJ. A prospective, randomized comparison of preoperative and continuous balanced epidural or paravertebral bupivacaine on post-thoracotomy pain, pulmonary function and stress responses. Br J Anaesth. 1999;83(3):387–392. | |
Shaheen PE, Walsh D, Lasheen W, Davis M, Lagman R. Opioid equianalgesic tables: are they all equally dangerous? J Pain Symptom Manage. 2009;38(3):409–417. | |
Eason MJ, Wyatt TR. Paravertebral thoracic block – a reappraisal. Anaesthesia. 1979;34:638–642. | |
Conroy M. How I do it: ultrasound-guided thoracic paravertebral blockade. ASRA News. 2012:14–16. | |
Shibata Y, Nishiwaki K. Ultrasound-guided intercostal approach to thoracic paravertebral block. Anesth Analg. 2009;109(3):996–997. | |
Hara K, Sakura S, Nomura T, Saito Y. Ultrasound guided thoracic paravertebral block in breast surgery. Anaesthesia. 2009;64:223–225. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.