Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Optimization of Nebulized Budesonide in the Treatment of Acute Exacerbation of Chronic Obstructive Pulmonary Disease
Authors Zhang R , Zhu J , Liu Y , Li Y , Liu W , Zhang M , Chen B , Zhu S
Received 25 October 2019
Accepted for publication 20 December 2019
Published 26 February 2020 Volume 2020:15 Pages 409—415
DOI https://doi.org/10.2147/COPD.S235125
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chunxue Bai
Rui Zhang,* Jiechen Zhu,* Yanan Liu,* Yuanqin Li, Wenjing Liu, Maowei Zhang, Bi Chen, Shuyang Zhu
Department of Respiratory Medicine, Affiliated Hospital of Xuzhou Medical College, Xuzhou, Jiangsu, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Shuyang Zhu
Department of Respiratory Medicine, Affiliated Hospital of Xuzhou Medical College, Xuzhou, West Huaihai Road #99, Xuzhou 221000, People’s Republic of China
Email [email protected]
Background: Clinical studies have suggested nebulized budesonide (NB) as an alternative to systemic corticosteroids for patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD). However, the optimal budesonide dose for AECOPD remains unclear.
Objectives: To compare the efficacy and safety of different doses of NB in the management of AECOPD.
Patients and Methods: A total of 321 AECOPD patients with moderate-to-severe exacerbation were randomly divided into three groups and treated with NB. The low dose group (L) was given 4 mg/day (n=95, 1 mg Q6h), while high-dose group 1 (H1, n=111, 2 mg Q6h) and high-dose group 2 (H2, n=115, 4 mg Q12h) were given 8 mg/day. Patients also received routine treatment including oxygen therapy, expectorant, nebulization bronchodilators, antibiotics, and fluid rehydration. The COPD assessment test (CAT), lung function, and artery blood gas were evaluated before and after 3 hrs and 5 days of treatment. In addition, hospital stay, frequency of acute exacerbations within 3 months of discharge, and adverse events during treatment were compared.
Results: H1 and H2 showed improved spirograms and CAT score faster than L. In H2, forced expiratory volume in 1 s (FEV1%) at 3 hrs and FEV1%, forced expiratory flow after 50% of the forced vital capacity has been exhaled (FEF50%), mean forced expiratory flow between 25% and 75% of forced vital capacity (FEF25– 75%) and CAT score at 5 days were significantly improved compared to L. FEV1% improved most in H2, moderately in H1, and least in L, with significant differences between groups at 5 days. No differences between groups were observed in adverse effects, hospital stay, and frequency of exacerbations within 3 months of discharge.
Conclusion: Compared to the conventional dose (4 mg/day), a high dose (8 mg/day) of NB improved pulmonary function and symptoms more effectively in the early treatment of AECOPD, especially when given as 4 mg twice daily.
Keywords: obstructive pulmonary disease, exacerbation, nebulized budesonide, dose, pulmonary function
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic and progressive disease. The 2016 Global Burden of Disease study estimated that worldwide, COPD caused 2.93 million deaths1 and affected 65 million people.2 COPD caused more deaths than any other chronic respiratory disease1 and was the third leading cause of death worldwide in 2016.3 The 2014 Global Burden of Lung Disease study showed that 56% (38.6 billion euros) of the European Union expenditure on respiratory diseases was spent on COPD.4 In the United States, medicaid incurred $2118/year in incremental costs due to COPD.5
Acute Exacerbation of COPD (AECOPD) is defined as an acute deterioration of respiratory symptoms requiring additional treatment.6 In COPD patients, exacerbations typically occur 0.5–3.5 times each year,7 and cause decreased quality of life, frequent hospitalizations, rapid lung function decline, and increased mortality, disability rate, and healthcare costs.8–11 In the United States, the median cost of inpatient care for AECOPD was estimated at $5844 per patient in 2010.12 The management of AECOPD must be improved.
Systemic corticosteroids are recommended in the treatment of AECODP by almost all international guidelines.6,13,14 However, because COPD patients are often elderly and relatively immobilized, high doses and frequent use of systemic corticosteroids15,16 may cause increased complications, such as osteoporosis, hyperglycemia, anxiety, depression, and the risk of infection.17–20 Recently, clinical studies and Meta-analysis have shown that nebulized budesonide (NB) is as effective as systemic corticosteroids in controlling nonacidotic AECOPD, as evaluated by clinical symptoms, lung function, and blood gas analysis. Furthermore, NB treatment reduces side effects17–22 and medicine costs23 compared to systemic corticosteroids. GOLD guidelines suggest NB as a suitable alternative to systemic corticosteroids in the treatment of exacerbations in some patients.6
In patients with acute asthma, FEV1 improved significantly after NB alone for 3 hrs,24 and different frequency and doses of budesonide have shown differences in improving pulmonary function and symptoms.25–27 To some extent, the pathogenesis of airway inflammation in AECOPD and acute asthma is similar, and the treatment of AECOPD with NB and bronchodilators is promising. Because few studies have systematically investigated the frequency and dose of NB in AECOPD management, optimal treatment parameters are unknown.
In this study, we aimed to study the efficacy and safety of NB administered at different frequencies and doses for managing AECOPD. We evaluated CAT score, pulmonary function, blood gas analysis, hospital stay, frequency of exacerbations within 3 months after discharge, and adverse effects during treatment.
Materials and Methods
Patients
We prospectively enrolled AECOPD patients hospitalized in the Department of Respiratory Medicine at the Affiliated Hospital of Xuzhou Medical University from October 2017 to May 2019. Patients were randomly divided into three groups: a low dose group (group L, 1 mg Q6h) received 4 mg/day, while high-dose group 1 (group H1, 2 mg Q6h) and high-dose group 2 (group H2, 4 mg Q12h) received 8 mg/day. Patients provided written informed consent, the study was conducted in accordance with the Declaration of Helsinki, and the study protocol was approved by the ethics committee of the center (ID: XYFY2017-KL148).
Inclusion criteria were as follows: age more than 50 years; smoking history of at least 20 pack-years; meet GOLD diagnostic criteria for COPD6 (show major COPD symptoms [chronic cough, expectoration, and/or dyspnea] and a history of exposure to risk factors, exhibit persistent airflow limitation, spirometry shows the presence of a post-bronchodilator FEV1/FVC < 0.70), and diagnosed with AECOPD6 (have an acute worsening of respiratory symptoms including dyspnea, chronic cough, or sputum production that results in additional therapy in COPD patients).
Patients were excluded from the study if they: had a personal history of asthma, allergic rhinitis, or atopy; had cancer, serious heart, liver, kidney, gastrointestinal diseases, or hepatorenal endocrine disease; were at risk of acute respiratory failure requiring mechanical ventilation or admission to the intensive care unit (ICU); had deterioration caused by specific reasons such as pneumonia, pneumothorax, or congestive heart failure; or had been exposed to systemic corticosteroids in the 30 days before admission.
Treatment During Hospitalization
Patients with AECOPD were randomly assigned to receive 4 mg NB per day (group L, 1 mg Q6h) or 8 mg NB per day (group H1, 2 mg Q6h; group H2, 4 mg Q12h). Patients also received routine treatments including oxygen therapy to ensure SaO2 > 90%, expectorant, nebulization bronchodilator (Compound Ipratropium Bromide Solution for Inhalation, 1 mg Q6h), antibiotics and fluid rehydration. No other steroids were used during the treatment period.
NB (1.0 mg/2 mL, Pulmicort respules®, AstraZeneca, London, England) and compound ipratropium bromide (0.5 mg ipratropium bromide and 3 mg salbutamol sulfate, Combivent®, Boehringer Ingelheim, Ingelheim, Germany) were driven by oxygen power with an oxygen flow rate of 4 to 6 L/min.
Clinical Efficacy
CAT score, lung function, and blood gas analysis (PaO2 and PaCO2) were evaluated before treatment, 3 hrs after nebulizing, and after 5 days of treatment. Lung function was evaluated by FVC%, FEV1%, FEV1/FVC%, FEF50%, FEF25–75%, and residual volume (RV)/total lung capacity (TLC). Arterial blood gas analysis was performed while breathing room air at rest.
Length of hospital stay and frequency of acute exacerbations within 3 months after discharge were also compared. Exacerbation after discharge was defined as an unscheduled visit to any medical unit due to increase in cough, purulent sputum, or dyspnea. Patient status after discharge was assessed by monthly phone calls for 3 months.
Adverse Events
Adverse events included hoarseness, hyperglycemia, and sleep disruption. In addition, pulmonary function, COPD deterioration, ICU admission, early discharge, and patient withdrawal for any reason were monitored and recorded.
Statistical Analysis
Clinical data were analyzed using SPSS 22.0 software. All quantitative data were expressed as mean ± SD. ANOVA and paired t-tests were used to compare changes in continuous variables between and within groups, respectively. All categorical data were expressed as rate and percentage, with Chi-square tests used to compare groups. A two-sided p-value < 0.05 was considered statistically significant.
Results
A total of 378 patients were enrolled and randomly assigned to the three groups. During the study, 57 patients were excluded due to uncompleted pulmonary function tests (n=24), rapid deterioration or admission to ICU (n=10), early discharge (n=9), or refusing to cooperate with treatment (n=14). Dropout rates were similar among the three groups (17.2% L, 15.7% H1, 15.4% H2). Groups L, H1, and H2 had 95, 111, and 115 patients complete treatment, respectively.
General Characteristics
The three groups of patients did not have significant differences in general characteristics, including age, gender, current smoking index, time of exacerbation, eosinophil count, glucose, complications, concomitant treatment before randomization, CAT score, PaO2, PaCO2, and lung function (p>0.05 for all parameters) (Table 1).
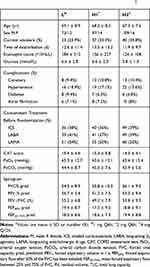 |
Table 1 General Characteristics of Patients at Admission‡ |
Short-Term Clinical Efficacy
Compared to before nebulization, PaO2 was significantly improved in all three groups after 5 days (p<0.001 for all parameters). There were no significant differences between groups at 3 hrs or 5 days (p=0.529 and p=0.748, respectively) (Table 2).
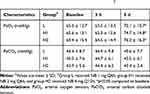 |
Table 2 Artery Blood Gas After Hospitalization‡ |
At 5 days, CAT scores had improved in the L, H1, and H2 groups by 5.3 ± 2.1, 5.9 ± 2.3, and 6.4 ± 2.6, respectively, with significant improvements in all groups compared to baseline (p=0.021, p=0.038 and p=0.009, respectively). The H2 group showed significantly greater changes compared to group L (p=0.048) (Table 3).
 |
Table 3 CAT Scores After Hospitalization‡ |
Although initial spirometric parameters were similar between groups, absolute values after 3 hrs and 5 days were higher in H2 than L and H1. In groups L, H1, and H2, FEV1% improved 0.7 ± 0.8%, 1.1 ± 0.7%, and 1.4 ± 0.5% at 3 hrs, and 3.3 ± 4.7%, 4.3 ± 4.6%, and 5.6 ± 5.4% at 5 days, respectively. The H2 group showed significant improvement in FEV1% at 3 hrs (p=0.033), and there were significant differences between the three groups at 5 days (p<0.05 for all parameters). In addition, FEF50% and FEF25–75% for H2 at 5 days was significantly improved compared to L (p=0.022 and p=0.041, respectively) (Table 4).
 |
Table 4 Lung Function After Hospitalization‡ |
The hospital stay among three groups was similar, with groups L, H1, and H2 averaging 6.5 ± 2.4 days, 6.2 ± 1.7 days, and 6.5 ± 4.7 days, respectively (p=0.895).
Long-Term Clinical Efficacy
During the 3 months after discharge, 59.3%, 61.6%, and 56.3% of patients in the L, H1, and H2 groups, respectively, used inhaled corticosteroids (ICS). At the same time, 62.6%, 65.0%, and 62.1% of patients used long-acting β2 agonists and 69.9%, 68.2%, and 67.5% of patients used long-acting anticholinergic drugs, respectively. No differences were seen between groups (p>0.05 for all parameters). Exacerbation frequency was 0.17 ± 0.07 times in the L group, 0.11 ± 0.09 times in the H1 group, and 0.14 ± 0.12 times in the H2 group, with no significant differences between groups (p=0.513).
Adverse Events
Only 15.7%, 15.3%, and 16.5% of patients in groups L, H1, and H2 experienced adverse events, and there were no significant differences between groups in the rate of adverse events. In group L, six patients had hoarseness, four patients had hyperglycemia (three with diabetes), and five patients had insomnia. In group H1, ten patients had hoarseness, three patients had hyperglycemia (three with diabetes), and four patients had insomnia. In group H2, twelve patients had hoarseness, five patients had hyperglycemia (four with diabetes), and two patients had insomnia (Table 5).
 |
Table 5 Adverse Reactions in the Three Groups‡ |
Discussion
In this study, we investigated the efficacy and safety of different doses of NB for managing AECOPD. The results demonstrated that 8 mg/day of NB effectively improves short-term clinical outcomes for AECOPD patients compared to 4 mg/day of NB. Additionally, among patients treated with 8 mg/day, NB administered 4 mg Q12h improved FEV1% more effectively than when administered 2 mg Q6h within 3 hrs, suggesting that a high dose of budesonide rapidly improved pulmonary function and symptoms.
Our study showed that NB administered 8 mg/day more effectively improved pulmonary function and symptoms than when administered 4 mg/day. Relatively little information is available on clinical effects of different doses of budesonide in patients with AECOPD. Inconsistent with our data, Akgun20 showed no differences in FEV1 or PaO2 improvement when comparing NB at 8 mg/day and 4 mg/day for 24 hrs, 48 hrs, and before hospital discharge. The inconsistent results may be due to the patient population and the follow-up time for evaluation. Akgun et al noted significant baseline differences in FEV1 between groups, suggesting that the degree of airway obstruction was different in each group, which may bias the results to some extent. Moreover, Akgun et al studied a relatively small number of patients and did not study small airway function improvement after NB. Further research is needed to evaluate a larger sample of patients in multicenter clinical studies.
Few studies have investigated the efficacy of nebulizing the same dose of budesonide at different frequencies in patients with AECOPD. Our study found that, compared to 2 mg Q6h, NB administered 4 mg Q12h significantly improved FEV1%, FEF50%, FEF25–75%, and CAT score at 5 days. Some studies28,29 have shown that glucocorticoids affect both genetic and non-genetic pathways through cytoplasmic and cell membrane receptors, but its action process is initiated by a membrane-bound hormone receptor. A high hormone concentration is often required due to the small quantity and low binding affinity of cell membrane receptors, and higher doses hormone cause stronger effects. In vitro experiments have confirmed that budesonide atomization takes effect a few minutes after nebulization, and improves airway and lung function in patients with chronic airway inflammation within a few hours.27 Only 3 hrs after 4 mg NB was administered with bronchodilators, FEV1% significantly improved, reflecting the rapid effects of corticosteroids. This intergroup difference is attributed to different corticosteroid doses because each group was treated with the same bronchodilator frequency and dose. Additionally, substantial evidence indicates that the combination of ICS and β2 agonists has a synergistic effect. First, ICS improve β2-adrenoceptor signaling by increasing β2-adrenoceptor density30 and reducing functional desensitization of the receptor.31 Second, ICS inhibit inflammatory gene expression, which is enhanced by β2 agonists.32 Furthermore, ICS rapidly enhance the effects of bronchodilators, especially in combination with β2 agonists,33 which could be used as a rescue therapy in asthma and AECOPD.
To the best of our knowledge, few studies have evaluated the long-term efficacy of different doses of NB in AECOPD patients. In our study, the three treatment groups had similar exacerbation frequencies during the 3 months after discharge. Intravenous corticosteroids reduced the risk of acute exacerbation;34 however, it is unclear whether increased doses of budesonide reduce the risk of acute exacerbation. Gunen et al35 compared exacerbation frequency within 1 month of discharge in patients treated with placebo, NB 8 mg/day (1.5 mg Q6h), and intravenous prednisolone 40 mg/day. On the corticosteroid arms, these rates became almost half the rate in placebo arm, however these were not statistically significant. Ding et al21 compared acute aggravation times with NB 6 mg/day (2 mg Q8h) and intravenous methylprednisolone 40 mg/day during the 12 months after discharge and found no significant differences between the two groups. However, this study did not account for different treatments after discharge between the two groups, limiting the interpretation of these results.
Administering NB 4 mg Q12h was more effective than 1 mg Q6h or 2 mg Q6h, but there were no significant differences in adverse effects and hospital stay among the three groups. Budesonide is a long-acting glucocorticoid, with a high binding affinity for the glucocorticoid receptor, small particle size, long pulmonary residence time, and lipid conjugation, which enhance its efficacy. Meanwhile, its low oropharyngeal exposure, in situ activation in the lung, negligible oral bioavailability, high protein binding, and rapid systemic clearance enhance its safety.28 Taken together, these pharmacokinetic and pharmacodynamic characteristics give budesonide a high topical anti-inflammatory activity with a low level of systemic activity.15,25,35 The incidence of adverse reactions in our study was similar to that observed in previous studies, indicating the safety of frequent high-dose NB. Moreover, giving the drug twice daily instead of four times daily could reduce medical costs and labor while increasing patient compliance.
This study was conducted in a single center with a small sample size and short follow-up time. Future multicenter studies with larger sample sizes and longer follow-up times are needed to better understand the use of high-dose budesonide in AECOPD as well as to understand its mechanism of action.
Conclusions
In conclusion, in patients with AECOPD, high-dose NB (4 mg Q12h) rapidly improved FEV1%, small airway function, and exacerbation symptoms. Furthermore, high-dose NB treatment increased patient compliance, reduced manpower and material resources, and reduced medical resource consumption.
Acknowledgments
This study was supported in part by grants from the National Natural Science Foundation of China (Grant No. 81600044), the Six Talent Peaks Project in Jiangsu Province, China (Grant No. WSN-081), and the Xuzhou City Bureau of Science and Technology Project (Grant No. KC KC18058). Written informed consent was obtained from patients for publication of this manuscript and any accompanying images.
Disclosure
The authors have no conflicts of interest to declare.
References
1. Naghavi M, Abajobir AA, Abbafati C, et al. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1151–1210. doi:10.1016/S0140-6736(17)32-152-9
2. World Health Organization. Chronic respiratory diseases: chronic obstructive pulmonary disease. Available from: https://www.who.int/respiratory/copd/burden/en/.
3. World Health Organization. The top 10 causes of death. Available from: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death.
4. Ferkol T, Schraufnagel D. The global burden of respiratory disease. Ann Am Thorac Soc. 2014;11(3):404–406. doi:10.1513/AnnalsATS.201311-405PS
5. D’Souza AO, Shah M, Dhamane AD, Dalal AA. Clinical and economic burden of COPD in a medicaid population. COPD. 2014;11(2):212–220. doi:10.3109/15412555.2013.836168
6. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (2019 report). Available from: https://goldcopd.org/gold-reports/.
7. Cai BQ, Cai SX, Chen RC, et al. Expert consensus on acute exacerbation of chronic obstructive pulmonary disease in the People’s Republic of China. Int J Chron Obstruct Pulmon Dis. 2014;9:381–395. doi:10.2147/COPD.S58454
8. Rosenberg SR, Kalhan R. An integrated approach to the medical treatment of chronic obstructive pulmonary disease. Med Clin N Am. 2012;96(4):811–826. doi:10.1016/j.mcn-a.2012.05.002
9. Ramsey SD, Hobbs FD. Chronic obstructive pulmonary disease, risk factors, and outcome trials: comparisons with cardiovascular disease. Proc Am Thorac Soc. 2006;3(7):635–640. doi:10.1513/pats.200603-094SS
10. Mullerova H, Shukla A, Hawkins A, Quint J. Risk factors for acute exacerbations of COPD in a primary care population: a retrospective observational cohort study. BMJ Open. 2014;4(12):e006171. doi:10.1136/bmjopen-2014-006171
11. Soler-Cataluna JJ, Martinez-Garcia MA, Roman SP, Salcedo E, Navarro M, Ochando R. Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax. 2005;60(11):925–931. doi:10.1136/thx.2005.040527
12. Perera PN, Armstrong EP, Sherrill DL, Skrepnek GH. Acute exacerbations of COPD in the United States: inpatient burden and predictors of costs and mortality. COPD. 2012;9(2):131–141. doi:10.3109/15412555.2011.650239
13. Wedzicha JA, Miravitlles M, Hurst JR, et al. Management of COPD exacerbations: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J. 2017;49(3):1600791. doi:10.1183/13993003.00791-2016
14. NICE Guideline Updates Team (UK). Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2018 report). Available from: https://www.nice.org.uk/.
15. Maltais F, Ostinelli J, Bourbeau J, et al. Comparison of nebulized budesonide and oral prednisolone with placebo in the treatment of acute exacerbations of chronic obstructive pulmonary disease: a randomized controlled trial. Am J Respir Crit Care Med. 2002;165(5):698–703. doi:10.1164/ajrccm.165.5.2109093
16. Donaldson GC, Seemungal TAR, Patel IS, et al. Airway and systemic inflammation and decline in lung function in patients With COPD. Chest. 2005;128(4):1995–2004. doi:10.1378/chest.128.4.1995
17. Decramer M, Lacquet LM, Fagard R, Rogiers P. Corticosteroids contribute to muscle weakness in chronic airflow obstruction. Am J Respir Crit Care Med. 1994;150(1):11–16. doi:10.1164/ajrccm.150.1.8025735
18. McEvoy CE, Ensrud KE, Bender E, et al. Association between corticosteroid use and vertebral fractures in older men with chronic obstructive pulmonary disease. Am J Resp Crit Care. 1998;157(3):704–709. doi:10.1164/ajrccm.157.3.9703080
19. Henzen C, Suter A, Lerch E, Urbinelli R, Schorno XH, Briner VA. Suppression and recovery of adrenal response after short-term, high-dose glucocorticoid treatment. Lancet. 2000;355(9203):542–545. doi:10.1016/S0140-6736(99)06290-X
20. Akgun M. Two different dosages of nebulized steroid versus parenteral steroid in the management of COPD exacerbations: a randomized control trial. Med Sci Monitor. 2014;20:513–520. doi:10.12659/MSM.890210
21. Ding Z, Li X, Lu Y, et al. A randomized, controlled multicentric study of inhaled budesonide and intravenous methylprednisolone in the treatment on acute exacerbation of chronic obstructive pulmonary disease. Respir Med. 2016;121:39–47. doi:10.1016/j.rmed.2016.1-0.013
22. Pleasants RA, Wang T, Xu X, et al. Nebulized corticosteroids in the treatment of COPD exacerbations: systematic review, meta-analysis, and clinical perspective. Respir Care. 2018;63(10):1302–1310. doi:10.4187/respcare.06384
23. Zhang J, Yao W, You X, Liu T, Liu Y. Comparative analysis of medical expenditure with nebulized budesonide versus systemic corticosteroids in hospitalized patients with acute exacerbations of chronic obstructive pulmonary disease in China. Int J Chron Obstruct Pulmon Dis. 2019;14:1195–1207. doi:10.2147/COPD.S182015
24. Engel T, Dirksen A, Heinig JH, Nielsen NH, Weeke B, Johansson SA. Single-dose inhaled budesonide in subjects with chronic asthma. Allergy. 1991;46(7):547–553. doi:10.1111/j.1398-9995.1991.tb00619.x
25. Morice AH, Morris D, Lawson-Matthew P. A comparison of nebulized budesonide with oral prednisolone in the treatment of exacerbations of obstructive pulmonary disease. Clin Pharmacol Ther. 1996;60(6):675–678. doi:10.1016/S0009-9236(96)90216-7
26. Rodrigo G, Rodrigo C. Inhaled flunisolide for acute severe asthma. Am J Respir Crit Care Med. 1998;157(3 Pt 1):698–703. doi:10.1164/ajrccm.157.3.9704022
27. Rodrigo GJ. Rapid effects of inhaled corticosteroids in acute asthma. Chest. 2006;130(5):1301–1311. doi:10.1378/chest.130.5.1301
28. Derendorf H, Nave R, Drollmann A, Cerasoli F, Wurst W. Relevance of pharmacokinetics and pharmacodynamics of inhaled corticosteroids to asthma. Eur Respir J. 2006;28(5):1042–1050. doi:10.1183/09031936.00074905
29. Kumar SD, Briver JL, Danta I, Wanner A. Transient effect of inhaled fluticasone on airway mucosal blood flow in subjects with and without asthma. Am J Respir Crit Care Med. 2000;161(3):918–921. doi:10.1164/ajrccm.161.3.9904106
30. Giembycz MA, Kaur M, Leigh R, Newton R. A Holy Grail of asthma management: toward understanding how long-acting beta(2)-adrenoceptor agonists enhance the clinical efficacy of inhaled corticosteroids. Br J Pharmacol. 2008;153(6):1090–1104. doi:10.1038/sj.bjp.0707-627
31. Chong LK, Drury DE, Dummer JF, Ghahramani P, Schleimer RP, Peachell PT. Protection by dexamethasone of the functional desensitization to beta 2-adrenoceptor-mediated responses in human lung mast cells. Br J Pharmacol. 1997;121(4):717–722. doi:10.1038/sj.bj-p.0701185
32. Cave AC, Hurst MM. The use of long acting beta(2)-agonists, alone or in combination with inhaled corticosteroids, in chronic obstructive pulmonary disease (COPD): a risk-benefit analysis. Pharmacol Ther. 2011;130(2):114–143. doi:10.1016/j.pharmthera.2010.12.008
33. Panettieri RA, Schaafsma D, Amrani Y, Koziol-White C, Ostrom R, Tliba O. Non-genomic effects of glucocorticoids: an updated view. Trends Pharmacol Sci. 2019;40(1):38–49. doi:10.1016/j.tips.2018.11.002
34. Madsen H, Brixen K, Hallas J. Screening, prevention and treatment of osteoporosis in patients with chronic obstructive pulmonary disease - a population-based database study. Clin Respir J. 2010;4(1):22–29. doi:10.1111/j.1752-699X.2009.00138.x
35. Gunen H, Hacievliyagil SS, Yetkin O, Gulbas G, Mutlu LC, In E. The role of nebulised budesonide in the treatment of exacerbations of COPD. Eur Respir J. 2007;29(4):660–667. doi:10.1183/09031936.00073506
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
