Back to Journals » Journal of Pain Research » Volume 12
Non-inferiority trial of a pedal controlled injection device: a step toward enhancing patient safety and operator independence during neural blockade
Authors Aziz SR, Smith DI , Mbaye RN , Gusman JT , Garza EI, Wang B , Feng C, Tran NH
Received 23 February 2018
Accepted for publication 21 August 2018
Published 7 February 2019 Volume 2019:12 Pages 571—577
DOI https://doi.org/10.2147/JPR.S166335
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor E Alfonso Romero-Sandoval
Syed Reefat Aziz,1 Daryl I Smith,2 Rose N Mbaye,1 Jacob T Gusman,1 Estefania I Garza,1 Bokai Wang,3 Changyong Feng,3 Nobuyuki-Hai Tran2
1Department of Biomedical Engineering, University of Rochester, School of Medicine and Dentistry, Rochester, NY, USA; 2Department of Anesthesiology, University of Rochester, School of Medicine and Dentistry, Rochester, NY, USA; 3Department of Biostatistics and Computational Biology, University of Rochester Medical Center, Rochester, NY, USA
Background and objectives: Limitations in manpower in health care facilities, both in civilian and military settings, can severely affect patient safety as well as overall outcomes. Regional anesthesia via neural blockade is an effective means of managing uncontrolled acute pain, which has been associated with cardiopulmonary, endocrine, immunologic, and hematologic derangement in addition to the development of potentially life-threatening coagulopathy. We have designed a remote-controlled injection device that may expedite the performance of regional nerve blocks in these situations.
Methods: This work examines how the device affects the ability of the operator to act independently with respect to various block component times by statistically comparing device-assisted blockade with usual or clinically relevant techniques. The classic or two-person technique was compared with the foot-controlled technique.
Results: The results validated the hypothesis that the novel mechanism of performing a nerve block is not inferior to the classic technique with regard to the specified endpoints within our experimental design.
Conclusion: This confirmation indicates that the use of this device may be feasible when the use of another technique could be cumbersome, or otherwise untenable.
Keywords: regional anesthesia, neuromuscular blockade, medical device, non-inferiority trial, biomedical engineering
Background and objectives
Limitations in manpower in health care facilities, both in civilian and combat settings, can severely affect patient safety as well as overall outcomes. One challenge in the acute setting is the treatment of pain caused by trauma or by surgical procedures. Regional anesthesia via neural blockade is an effective means of managing uncontrolled acute pain. Uncontrolled acute pain has been associated with cardiopulmonary, endocrine, immunologic, and hematologic derangement in addition to the development of potentially life-threatening coagulopathy. Training and junior attendings to perform these blocks in less than optimal environments (diminished staff availabilities, severe time, and ergonomic environments) is often a function of performing enough repetitions of a procedural technique to gain proficiency.1–4 We have designed a remote-controlled injection device that may expedite the performance of and improve operator independence of regional nerve blocks in these situations.5 The device involves a linear actuator to provide automated pushing and pulling of a syringe in response to input from a physician anesthesiologist. A foot pedal interface is utilized, where transversal foot motion – to the left or to the right – triggers fluid flow in the accompanying syringe pump in a forward direction (dispensing) or reverse direction (aspiration), respectively. The system is constructed mostly out of high-density polyethylene and ultra-high molecular weight polyethylene, which have low density, making the device relatively portable. This study was deemed exempt from formal Institutional Review Board/ Research Subjects Review Board approval because it retrospectively examined trainee times used in an instructional setting.
Methods
Our first objective was to evaluate the aforementioned foot-controlled (FC) injection device in order to determine instructor and trainee preferences and tendencies. Some of these tendencies may provide insight into operator variability when teaching block techniques to trainees. Certain operator preferences may be quantitated with the goal of arriving at a consensus of optimal, consistent teaching methods. This serves as an initial step in standardizing technical instruction and helps establish legitimacy in trainee assessment.
Our second objective was to compare the times necessary to perform regional blocks using a pedal-based technique vs standard techniques for injection of local anesthetic. This objective will examine the time it takes for completion of various timed components of the neural block in the ergonomic settings in which common nerve blocks are performed. Data will be recorded from the trial of each examinee in each blockade setting. The mean times for examinees in each discipline and at each training level will be compared.
In the first stage of the study, a focus group of 32 anesthesiology clinicians of varying practice and training levels was constructed. This helped to determine ergonomic and operational preferences as well as establish customer validation for the electromechanical design of the device. Focus group participants completed a questionnaire created to examine trends in their procedural tendencies and preferences. Questions included trainee preferences regarding the direction of the fluid being dispensed and aspirated (left/right) and orientation of the pedal system (vertical/horizontal). Additionally, participants were able to rate (1, lowest through 5, highest) how well the device performs various design requirements, such as speed of dispensing/aspiration as well as ergonomic comfort with respect to orientation and device placement. Although a formal validation was not performed for the 1–5 scoring scheme, this was acceptable since the purpose was to qualitatively identify trends in operator preferences. Each study participant was randomly assigned a number according to the Latin square randomization array indicated in Table 1. As such, all data obtained and analyzed from study participants were de-identified or anonymized, therefore written informed consent was not required.
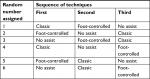  | Table 1 The setup of the Latin square randomization array Note: A random number generator (www.random.org) was used to assign a number from 1 to 6 to determine each participant’s testing sequence. |
The second stage of this work was to compare the device with more conventional or usual techniques, that is, the two-operator or the single-operator technique using manual injection. The criteria to evaluate the device vs the usual techniques are the times to complete individual components of the nerve block. We examined how the device affects the ability of the operator to act independently with respect to various block component times. This pilot trial involved 21 trainees at various levels in the anesthesiology residency program at the University of Rochester (Rochester, NY, USA).
The outcomes examined were total time to perform the nerve block (tt), time to visualize (ttv) the block needle in the block phantom (gel); time of the needle to target (ttt) embedded in the block phantom and time to injection (tti). The pre-trial analysis serves to validate the experimental format and the ability of our phantom models to approximate the clinical settings encountered in usual regional neural blockade practice. We examined the outcomes described previously in each of the ergonomic settings and compared the classical approach (two operators), the single-operator approach (needle released in order to control the injection syringe); and the foot-controller-assisted approach. A system-level design of the device is visually depicted in Figure 1.
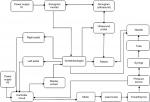  | Figure 1 A schematic of the foot-controlled injection system with integration of ultrasound guidance and operator/patient interfaces. |
Latin squares randomization of the sequence of blocks performed is used in order to account for intra-exam learning and acquired facility from technique to technique by the individual operator in three different orientations of neural blockade: gravity, neutral, and anti-gravity based. The block orientations were chosen as they related to the ultrasound probe and needle orientations used for neural blockade procedures at our site. The gravity-based technique mimics a block performed upon a patient in the supine position as in the performance of a femoral nerve block. The neutral-based technique describes the orientation of the ultrasound probe and block needle that would be employed to perform a thoracic paravertebral block in a patient in the sitting position. The anti-gravity-based technique mimics the ultrasound probe and block needle orientation that would be used to perform a sciatic nerve block at the popliteal fossa with the patient in the supine position. The Latin square array (Table 1) was used to control the nuisance variability of operators’ “learning” or practicing ultrasound-guided neural blockade as they performed blocks moving from technique to technique. This learning or practice would have specifically consisted of decreasing time to ultrasound-guided needle and target localization, as well as a decrease in time to maneuver the needle to the target.
We hypothesized that the FC method is not inferior to the classic method (CL). The one-sample non-inferiority test was used to test our hypothesis. For all tests, the significance level was set at 0.05. The analysis was implemented with SAS 9.2 (SAS Institute Inc., Cary, NC, USA). The null hypothesis is that the mean time in the FC setting is greater than the mean time for the CL (margin 0.01), and the alternative hypothesis is that the mean time for performance of the nerve block in the FC method is less than that of the CL method (margin 0.01).
Results
In addressing the first objective of our work, we were able to determine the preferences of individuals within the focus group. These individuals had relevant experience in technical procedures, such as neuromuscular blockades. Tables 2 and 3 and Figures 2 and 3 visually depict the trainee preferences and rated device outcomes obtained in the focus group. Results indicate which direction of the FC system proved to be more intuitive for users. For attending physicians, dispensing to the right seemed more intuitive than the left. Half of all respondents believed the dispensing speed (1.5 cc/s) was too fast (Table 3). A 5-mL syringe was used in this focus group instead of the 30-mL syringe that is usually used at our institution, which could have accounted for the perception of greater injection speed. Clinical base year (interns) seemed to prefer vertical foot pedal motion, while residents and attendings strongly favored the horizontal motion.
  | Table 2 Responses regarding speed of withdrawal from syringe, comfort in horizontal foot motion, and placement of the foot pedal, ranked 1 (worst) through 5 (best) |
  | Table 3 Clinician preferences (%) |
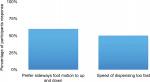  | Figure 2 Operator preferences. Note: Summary graphs of specific operator preferences with respect to pedal activation and assessment of injection speed. |
  | Figure 3 Operator ratings (mean) of device performance: injectate dispensing and withdrawal; comfort of directional foot movement for pump activation; ergonomic orientation of the activating pedal. |
In addressing the second objective of our work, we validated the hypothesis that the novel mechanism of performing a nerve block via the FC device is not clinically worse with regard to the specified endpoints within our experimental setup. Our results are summarized in Table 4, which consists of comparisons among the gravity-, neutral-, and anti-gravity-based techniques. The time intervals examined are ttv, ttt, and tti. It should be noted that both the needle and the target must be visible on the ultrasound monitor, both before and during the entire time the injection takes place; the time the needle is removed from the block site (no- or needle out); and the tt.
Discussion
Statistical analysis was performed, which compared techniques. The no-assistance (na) (single operator without the benefit of the FC injection device) technique was compared with the classic and the FC techniques. The na or single-operator model vs the classic (two-person) model (CL) showed statistical significance in ttv (P≤0.05) and near statistical significance (P≤0.051) between the same categories with respect to time of needle to the target, and total block time. This comparison is not depicted in Table 4.
The classic technique was also compared with the FC technique and Table 4 depicts the results of the non-inferiority trial on the device. This validates the hypothesis that the mechanism of performing a nerve block via the FC device is not clinically worse with regard to the specified endpoints within our experimental setup. This confirmation indicates that the use of this device may be feasible when the use of another technique (such as the classical procedure with two operators, or a single unassisted operator) could be cumbersome, or otherwise untenable.
Teaching trainees to perform ultrasound-guided nerve blocks typically lacks true objective consistency regarding the “best” method because of operator, both instructor and trainee, variability. While this may be unavoidable to some degree, there are certain operator preferences that offer a deeper understanding of optimal tendencies and strengthen the interest of identifying which techniques may warrant emphasis as well as providing some information on inter-instructor cautions and the development of safeguards to improve overall patient safety. The responses from the focus group begin to indicate the preferences of individuals with relevant technical expertise that can pave the path to the standardization of instruction for this procedure.
The development of the foot-actuated device is the result of the need to efficiently and safely perform neural blockade in multiple settings and when staffing conditions may be suboptimal. The initial question asked in the second stage of the study was as follows: can the device save time in the performance of a given block? Considerations for set-up time and access to patients were initially the predominant concerns but these gave way to the realization that with subsequent iterations of the device, these issues would be easily managed. We focused our attention on the need to evaluate the usefulness of the device in a variety of clinical, ergonomic settings. The initial criteria were, as examined in this paper, the relative times to complete various essential components of the procedure. Our prototype allowed for testing on gel block phantoms under the conditions described. The establishment of non-inferiority of the device when compared with the usual techniques employed to complete these procedures is the first step in validating its usefulness and indicates to us that pursuing further refinements are worthwhile. Areas of concern in which the device does not appear to assist in the completion of neural blockade involve those factors that are currently a function of the individual operator’s skill set. These factors cannot be modified or improved by this device in its current iteration. The specific factors measured in the study were ttv, ttt, and tti. There was no significant difference in these times either with or without the use of the device.
It should be noted that with each of the factors the specific ability of the individual operator to coordinate movement of his or her hands in a synchronized fashion and maintain the appropriate coordination throughout the entire procedure, including agent injection, was the critical component. The complete needle visualization was confirmed throughout the procedure by both the operator and the instructor/trainee. Whenever visualization was lost, appropriate time was taken to re-acquire the ultrasound image before the procedure progressed. The timer ran throughout the image re-acquisition period.
Conclusion
This device may be feasible when the use of another technique (such as the classical procedure with two operators, or a single unassisted operator) proves cumbersome, unsafe, or otherwise untenable. The focus group respondents indicated various considerations for ergonomic developments, such as a safety stop-gap mechanism as well as the need for a liquid-crystal display of pressure and volume to quantify neural compliance. Possible future developments also include a more stringent flow rate monitoring mechanism in order to regulate speed of injection. Further analyses and trials with the incorporation of these design enhancements are warranted.
Effective delivery of regional anesthesia, especially in the acute setting is of great value. Early post-injury mobility, decrease in the levels of stress hormone, reduction in the incidence of cardiovascular as well as pulmonary complications, improvement in gut motility as a result of reduced pain response as well as a decrease in opioid-related ileus, and a decrease in thromboembolic events have all been related to effective post-acute insult analgesia. These insults may be elective as in the case of scheduled surgical procedures, or emergent as in the case of trauma; however, the common denominator remains the need to provide effective, reliable analgesia.
To this end, we are in an ongoing endeavor to satisfy two important goals. First, to provide better preparation for our trainees; and, second, to develop adjuncts that can assist in the safe and efficient performance of these techniques. We begin to address the latter goal by analyzing the components of a sound technique to perform an actual regional anesthetic. The majority of modern nerve blocks used to provide regional anesthesia are performed with ultrasound guidance. Invariably this technique requires the use of two operators; the principal operator who controls the ultrasound probe and the needle; and the “assistant” who controls the syringe containing the blocking agent (local anesthetic, steroid adjunct, alpha-2 agonist adjunct, etc). In academic settings, the syringe controller is usually the instructor who, in addition to injecting the agent, is providing technical, didactic, and theoretical guidance to the less experienced primary operator. In the early stages of training, this is an optimal arrangement in that the trainee and instructor are closely tied to performing the procedure. However, as the trainee progresses to a more senior level, the goal is to create ever greater autonomy. In this setting, the block is performed under general supervision and the individual controlling the syringe is a junior clinician receiving direct orders from the more senior trainee.
The limitations of this arrangement are obvious, especially in a high-volume setting where a second or third blocking procedure can be performed with an attending working directly with a junior trainee. In the private practice setting, this need is compounded by the occurrence of decreased staff availability to provide assistance to a primary operator. When this occurs, the operator is left with two options. First, the operator may let go off the blocking needle in order to control the syringe to perform the injection. Or, second, the operator may release the ultrasound probe to accomplish the same task. Releasing the probe results in changing an ultrasound- guided technique into a blind technique; and releasing the needle to perform the injection relinquishes control over of the component most capable of causing significant damage, especially if an unexpected event (such as patient movement) occurs. In either event, we believe a suboptimal setting is created. In order to correct this condition, we have designed a remote-controlled injection device that may expedite the performance of regional nerve blocks in these situations.
This work describes, in part, the development of the device and its initial validation study.
Acknowledgments
Interim data from this work were presented at the 2016 American Society of Anesthesiologists Annual Meeting in Chicago, IL, USA; October 22–26, 2016,5 and at the 42nd Annual Regional Anesthesiology & Acute Pain Medicine Meeting; San Francisco, CA, USA; April 6–8, 2017.6
Disclosure
The authors report no conflicts of interest in this work.
References
Neal JM, Brull R, Chan VW, et al. The ASRA evidence-based medicine assessment of ultrasound-guided regional anesthesia and pain medicine: executive summary. Reg Anesth Pain Med. 2010;35(Suppl 2): S1–S9. | ||
Delvi MB. Training in ultrasound guided blocks. Saudi J Anaesth. 2011;5(2):119–120. | ||
Sites BD, Neal JM, Chan V. Ultrasound in regional anesthesia: where should the “focus” be set? Reg Anesth Pain Med. 2009;34(6):531–533. | ||
Sites BD, Spence BC, Gallagher JD, Wiley CW, Bertrand ML, Blike GT. Characterizing novice behavior associated with learning ultrasound-guided peripheral regional anesthesia. Reg Anesth Pain Med. 2007;32(2):107–115. | ||
Aziz S, Garza EI, Gusman JT, Mbaye RN, Smith D. Comparing instructor and trainee preferences in using a foot controlled injection device: an initial step in standardizing technical instruction. Available from: http://www.asaabstracts.com/strands/asaabstracts/abstract.htm?year=2016&index=12&absnum=5292. Accessed November 20, 2018. | ||
Aziz S, Tran T, Smith D. Evaluation of a Foot Controlled Injection Device: The First Step in Enhancing Patient Safety and Operator Independence during Neural Blockage. Available from: https://epostersonline.com/ASRASpring17/node/982. Accessed November 20, 2018. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

