Back to Journals » Journal of Inflammation Research » Volume 14
Nicotinamide Mononucleotide Alleviates Hyperosmolarity-Induced IL-17a Secretion and Macrophage Activation in Corneal Epithelial Cells/Macrophage Co-Culture System
Authors Meng YF, Pu Q , Dai SY, Ma Q, Li X, Zhu W
Received 18 November 2020
Accepted for publication 27 January 2021
Published 22 February 2021 Volume 2021:14 Pages 479—493
DOI https://doi.org/10.2147/JIR.S292764
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Yi-Fang Meng,1,2,* Qi Pu,1,* San-You Dai,3,* Qian Ma,1 Xinyu Li,1 Wei Zhu2
1Department of Ophthalmology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, People’s Republic of China; 2Department of Ophthalmology, Changshu No. 2 People’s Hospital, Changshu, People’s Republic of China; 3Department of Ophthalmology, Lixiang Eye Hospital of Soochow University, Suzhou, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xinyu Li
Department of Ophthalmology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, 1095 Jiefang Ave, Wuhan, People’s Republic of China
Email [email protected]
Wei Zhu
Department of Ophthalmology, Changshu No. 2 People’s Hospital, Changshu, People’s Republic of China
Tel/Fax +86-027-83663456
Email [email protected]
Background: Hyperosmosis stress (HS) was a key pathological factor in the development of dry eye disease (DED). Nicotinamide mononucleotide (NMN) demonstrated protective effects in the corneal damage, however, its role in the HS-induced DED remained unclear.
Methods: A NaCl based HS in-vitro model (500 mOsm) was generated and used in a co-culture system including corneal epithelial cells (CEC) and macrophage cell line RAW264.7. The effect of NMN on NAD+ metabolism and the expression of HS biomarker, tonicity-responsive element binding protein (TonEBP), was studied in the CEC. The cellular activity, including cell viability, apoptosis status and lactate dehydrogenase (LDH) release through trypan blue staining, flow cytometry and LDH assay, respectively. The mitochondrial membrane potential (MMP) assay would be conducted using the JC1 kit. The expression of IL-17a were detected using RT-PCR, ELISA and Western blot. After co-culture with the CEC in different group for 24 h, the phagocytosis ability and macrophage polarization were assessed in RAW264.7 cells co-cultured with CEC with or without HS or NMN treatment. Besides, the involvement of Notch pathway in the RAW264.7 would be analyzed. The potential involvement of Sirtuin 1 (SIRT1) and IL-17a in the crosstalk between CEC and macrophage was studied with SIRT1 inhibitor EX 527 and anti-IL-17a monoclonal antibody, respectively.
Results: NMN treatment increased NAD+ concentration and thus improved cell viability, reduced apoptotic rate and decreased the LDH release in HS-treated CEC. Besides, NMN alleviated HS-induced MMP, intracellular ROS and LDH release. Besides, it was confirmed NMN improve SIRT1 function and decreased the HS related IL-17a expression in CEC and then alleviated macrophage phagocytosis ability and M1 polarization based on a CEC-macrophage co-culture system. Moreover, NMN treatment of CEC in the CEC could moderate the subsequent macrophage activation through Notch pathway. SIRT1 activation and IL-17a inhibition was regarded as key progress in the function of NMN based on the application of EX 527 and anti-IL-17a antibody in the CEC-macrophage co-culture system.
Conclusion: The findings demonstrated that NMN could alleviated HS-induced DED status through regulating the CEC/macrophage interaction. Our data pointed to the role of SIRT1, IL-17a and Notch pathway in the function of NMN and then provided updated knowledge of potential NMN application in the management of DED.
Keywords: dry eye disease, hyperosmosis stress, nicotinamide mononucleotide, SIRT1, notch pathway, macrophage, corneal epithelial cells
Background
Dry eye disease (DED) was regarded as one of the most prevalent ocular disorders and would lead to significant ocular discomforts in the patients with DED. DED, which was a multi-factor disease in the ocular surface, was characterized by loss of tear film homeostasis and accompanying ocular surface symptoms.1,2 Its etiologies included unstable tear film, tear hyperosmolarity, inflammation and injury on the ocular surface, and neurosensory abnormalities.3 Among all the pathological progresses of DED, a key progress was the tear hyperosmolarity, which was both a clinical manifestation as well as an indicator of the subsequent ocular inflammation. Detecting the potential interventions of tear hyperosmolarity would help in both understanding of the pathological mechanism as well as advanced therapy of DED.
Even the etiology of DED was still not clearly understood by now, hyperosmolarity-induced inflammation has been concerned. Tear hyperosmolarity could lead to various pathological effects related with the development of DED, the ultimate damage appears to be caused inflammatory cytokines in the ocular surface. The epithelial layer was located in the outermost in the cornea tissue and it could work as the first cell barrier during both extrinsic damages as well as eye drops treatment. Through analyzing human corneal epithelial cells (CEC) exposed to hyperosmosis stress (HS) and control status, more than 150 unique phospholipids and sphingolipids were annotated and this lipid histochemical study highlighted the relationship between lipid changes in corneal cells exposed to HS and inflammatory responses.4 Besides, in a previous study, it was found that HS would lead to an excessive reactive oxygen species release and lead to decreased autophagy and decreased cell activity in DED.5 Protection of CEC in the pathological status would help in the management of DED and this research approach provided us various potential therapies for DED.6,7
Macrophage was a key immune cell recruited to the cornea during the incidence of ocular surface disorders8,9 and thus the interaction between CEC and macrophage would be an important progress in DED. As macrophage is a key source of inflammatory factors, the up-regulated inflammatory factors during pathological condition would injure the normal function of CEC.10 In advance, as CEC was the outset cell layer of cornea and the biological function of macrophage would be regulated after CEC treated by different pathological progresses. For instance, it was reported that Aspergillus fumigatus-stimulated CEC induces the pyroptosis of macrophages by secreting thymic stromal lymphopoietin.11 IL-17 is the most important effector in Th17 cells, and it is currently believed that its family members include six highly homologous cytokines, IL-17a ~ f, among which IL-17a was the main cytokine of IL-17 family. As reported in previous studies, IL-17a was involved in the pathological processes of ocular surface diseases and play an important role in DED incidence.12 Macrophages show great variability in different microenvironments, and were generally classified into classical activated type (also known as M1-type) and alternative activated type (also known as M2-type), however, the effects of CEC secreted IL-17a on macrophage remained unclear.
NAD+, as a coenzyme that exists in human body and is closely related to thousands of enzymes involved in cellular life activities,13 has the potential of delaying senescence and prolonging life with its direct precursor substance, nicotinamide mononucleotide (NMN). As aging is a risk factor of DED based on the TFOS DEWS II,14 the application of NMN, which is a potential anti-aging drug might help in the management of DED. In two most recent experiments, it was found that NMN application could promote epithelial homeostasis by regulating the biosynthesis of NAD+.15 Besides, local supplementation of NMN can effectively prevent corneal endothelial cell apoptosis from UVB exposure by reactivating AKT signaling.16 To detect the effects of NMN on the CEC-macrophage interaction in the development of DED, we conduct an in-vitro study and the aims of this study included (1) detection of the protective effects of NMN on HS-induced CEC and (2) clarification of the effects of IL-17a on biological functions of macrophage.
Methods
Cell Culture and Treatment
Mouse primary CEC and macrophage cell line RAW264.7, were purchased from Meisen Cell Biotechnology Co. Ltd (Hangzhou, China) and cultured in the culture was conducted in Complete Medium for Mouse Corneal Epithelial Cells (Meisen, China), which was Keratinocyte serum-free medium (KSFM) based special growth medium without Ca2+. Cells between passages 3 and 5 would be used in the following experiments. The RAW264.7 cell was cultured in high-glucose DMEM culture medium supplemented with 10% of fetal bovine serum (FBS) and 100 U/mL of penicillin. Both cells would be maintained in the incubator in 37°C in a 5% CO2 atmosphere. The culture media was changed the next day, and the cell growth status was observed daily.
To generate an in-vitro HS model, 40 mM and 90 mM NaCl was added in the culture media to generate a hyperosmotic medium in 400 and 500 mOsm, respectively. To exclude the effect of Na+/Cl− in the HS in-vitro model, 80 mM and 180 mM mannitol added in the culture media to induce a control HS in-vitro model. NMN in a purity of over 99.5% was purchased from Hygieia Biotechnology Co., Ltd (Shenzhen, China) and NMN would be dissolved in PBS for the management of in-vitro systems.
A non-contact co-culture system was generated in this experiment with a six-well transwell plate with 0.4 μm pores. This co-culture system is divided into upper and lower layers and there is a layer of microporous membrane between the upper and lower layers. The CEC/macrophage co-culture would be developed by seeding the macrophage onto the inner chamber and CEC onto the outer chamber. The CEC and macrophage in different groups were pre-treated with HS/NMN/Sirtuin 1 (SIRT1) or IL-17a inhibitor simultaneously and then fresh media was replaced to generate a co-culture system. The macrophage in the co-culture system would be observed and then collected for advanced experiments. Cells between the upper and lower layers cannot pass and only soluble molecules can penetrate and thus the cells on both layers can interact with each other.
Cell Viability
In this current study, the trypan blue staining method was adopted to detect cell viability. The cultured CEC in different groups were digested with 0.25% trypsin and single cell suspension was prepared. The diluted cells suspension in 1×106 cells/mL was mixed with 0.4% trypan blue solution at a rate of 9:1. Microscopically, dead cells would be stained light blue, while alive cells remained clear without any staining. The amount of alive and dead cells was counted using a counting board in three minutes and the cell viability was calculated according to the following formula: cell viability (%) = (amount of living cells/amount of total cells) ×100%.
NAD+ Quantification
Intracellular NAD+ levels were quantified with a NAD+ assay kit (Sigma-Aldrich, USA). The kit was based on an enzymatic cycle reaction in which the NADH formed reduces the methyl MAz reagent. The reduced product color intensity measured at OD565 nm could be proportional to the NAD+/NADH concentration in each sample cells. Three independent measurements of each group would be conducted and the mean values were used in the final analyses.
Apoptosis Detection
The cells in each group would be rinsed and collected by centrifugation for Annexin V-FITC/PI staining (Beyotime, China). Each pellet was resuspended in 500 μL of binding buffer and then a total of 5 μL of FITC in combination of 5 μL of PI were added to each well. The detected cells were incubated with the mixed staining solution for 15 min at 37 °C in dark. The apoptotic ratios were then determined by flow cytometry (Beckman, USA). AnnexinV-FITC positive cells were demonstrated on the X axis while Y axis indicated PI positive cells. The living and apoptotic status would be defined by quarter gate gating tool. Q2 (AnnexinV-FITC positive and PI positive cells) demonstrated late apoptotic status and Q3 (AnnexinV-FITC positive and PI negative cells) indicated early apoptotic status. The number of combinations of Q2 and Q3 would be used in the analyses of apoptosis status in this study.
ROS Activity Assay
For the detection of intracellular ROS content, a commercial kit was purchased from Beyotime Biotechnology (China). After separating the cells into the flow tubes, the cells were centrifugally washed in PBS for three times. Diluting THE DCFH-DA probe at 1:1000, adding 1mL diluted probe into each tube and incubating it in the incubator for 20 min according to the manufacturer’s instructions. Intracellular ROS concentrations would be detected using flow cytometry (Beckman, USA).
Mitochondrial Membrane Potential (MMP) Assay
MMP was measured with a Mitochondrial Membrane Potential Assay Kit (Beyotime, China). Briefly, after culturing in the conditioned medium, cells were rinsed and washed with PBS 3 times, and incubated with 50 nM JC-1 at 37°C for 20 minutes. JC-1 is an ideal fluorescent probe widely used to detect mitochondrial membrane potential ΔΨm. When the mitochondrial membrane potential is high, JC-1 aggregates in the mitochondrial matrix to form a polymer, which can produce red fluorescence; when the mitochondrial membrane potential is low, JC-1 cannot gather in the mitochondrial matrix, when JC-1 is a monomer, it can produce green fluorescence. After the incubation, the cells were washed with PBS and analyzed by flow cytometry when the excitation wavelength was 488 nm and the emission wavelength was 575 nm.
Lactate Dehydrogenase (LDH) Release Assay
LDH release was analyzed using the Pierce™ LDH Cytotoxicity Assay Kit (Thermo Scientific, USA). The target cells were adjusted to 2×105/mL and added into the 96-well round bottom cell culture plate. A total of 100 μL were added to each well and then cultured for 4 ~ 6 hours in an incubator at 37°C with 5% CO2. After adding the lactate dehydrogenase related regents, the plates were placed at room temperature for 20 minutes. The optical density (OD value) of each hole was measured on the enzyme linked detector with a detection wavelength of 492 nm and a reference wavelength of 650 nm.
Phagocytosis Assay
RAW264.7 cells in different groups were used in the phagocytosis assay. After incubating with Nile red fluorescent microspheres beads (Invitrogen, USA) in a concentration of 5×105 for 2 h, the cells were then washed with PBS to quench the fluorescence of extracellular beads and then fixed with 4% for 15 min. Fixedcells would be stained with DAPI and then observed under fluorescence microscope.
Real-Time PCR (RT-PCR) Assay
Total RNA from the cultured cells were extracted with TRIzol (Invitrogen, USA). The cDNA was synthesized using an Omniscript RT kit (Qiagen, Germany). The mRNA expressions of relevant genes were measured by RT-PCR assay with a QuantiTect SYBR Green PCR kit (Qiagen, Germany) using an ABI PRISM 7000 system (Applied Biosystems, USA). The primers used in this experiment were provided in Table 1. To normalize the expressions of all the genes of interest, we adopted β‐actin as an endogenous control and calculated the relative expression using the 2−ΔΔCt formula.
 |
Table 1 The Primers for RT-PCR in This Experiment |
ELISA
Measurement of IL-17a in the supernatant was conducted by ELISA method. Soluble IL-17a in each group was detected with a commercial ELISA kit (BD, USA) according to manufacturer’s instruction. The absorbance rate at 450 nm was transformed to the IL-17a concentration through a standard curve on a 96 well plate spectrophotometer.
Western Blot Analysis
The Western blot was performed to detect the protein level of TonEBP, SIRT1, IL-17a, intracellular domain of Notch 1 (NICD1) and Jagged1. Briefly, the harvested cells in different groups were lysed using RAPI regent (Beyotime, China) with 1% protease inhibitor cocktail (Sigma-Aldrich, USA). A total of 25–30 μg protein extraction per sample from both CEC and RW264.7 cells were separated with SDS-PAGE and the proteins were electroblotted onto a nitrocellulose membrane. Then the membranes were blocked with 10% (w/v) fat-free milk in TBST (TBS with 0.1% Tween-20) for 1 h at room temperature. The blots were incubated overnight at 4 °C with primary antibodies. The following antibodies were used in this experiment: anti-TonEBP (1:1000, Abcam, UK), anti-SIRT1 (1:1000, CST, USA), anti-IL-17a (1:1000, Abcam, UK), anti-NICD1 (1:1000, Abcam, UK), anti-Jagged1 (1:500, Abcam, UK) and anti-β-actin (1:1000, Santa Cruz, USA). After washing the membranes with TBST, they were further incubated with secondary antibodies for 1 h at room temperature. The transfers were visualized using an ECL kit according to the manufacturer’s instructions.
Statistical Analysis
Statistical analyses were performed with GraphPad software package 8.0 (CA, USA) in this study. The continuous variables would be presented as the mean ± standard deviation (SD) in independent experiments. Unpaired t-test and one-way ANOVA method would be used in the detection of difference in two and more than three groups, respectively. Post-hoc analyses were conducted using Student-Newman-Keuls (SNK) method for multiple comparison during the one-way ANOVA analyses. A P value of less than 0.05 would be considered statistically significant.
Results
NMN Moderated NAD+ Metabolism and HS Biomarker Expression in CEC
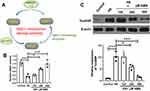
As described in a previous published review, the salvage pathway occupied 85% of all the NAD+ and thus regarded as a key metabolism pathway.13 NAD+ would be transformed to nicotinamide (NAM) through NAD+ consuming enzymes, and then became nicotinamide dinucleotide (NMN) after being catalyzed by nicotinamide phosphoribosyl transferase (NAMPT). NMN would then be catalyzed by nicotinamide mononucleotide adenylyl transferase 1–3 (NMNAT1-3). The detailed salvage pathway was presented in Figure 1A and NAMPT was the rate-limiting enzyme in the salvage pathway of NAD+ mentalism. Comparing with the control group, a significant decreased NAD+ content was detected (P=0.001) in the HS group and improved NAD+ concentration was detected in the 250 and 500 μM NMN treatment groups comparing with the HS group for 24h (P=0.017 and P<0.001, respectively, Figure 1B). To validate the in-vitro model, higher hyperosmotic biomarker, TonEBP, was up-regulated in both NaCl and mannitol based 400 and 500 mOsm hyperosmotic culture medium (P<0.05, Supplementary Figure 1). In the NaCl based hyperosmotic culture medium in a 500 mOsm, 250 and 500 μM NMN significantly moderated the expressions of TonEBP (P=0.031 and P=0.003, respectively, Figure 1C). In the following experiments, a dose of 500 μM NMN would be used in the in-vitro experiments.
NMN Improved Cell Viability and Apoptotic Status of HS-Treated CEC
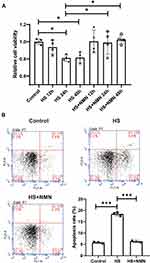
As HS was one of the most common pathological progress of DED, the potential protective effects of NMN on the cellular function of CEC. As showed in Figure 2A, a significant decreased cell viability could be detected in 24h and 48h after HS treatment (P<0.05). When a 500 μM NMN was added in the HS group, there was no significant difference in all the time points comparing with the control group. When comparing the HS group and NMN-treated HS cell groups, improved cell viability was detected in 24 h and 48 h (P<0.05). Considering that there was no significant difference in the cell viability between 24h and 48h in HS group and there was an improved cell viability since NMN treatment for 12 h, a time point of 24 h was used in the following experiment. Considering that CEC apoptosis played a key role in the progression of DED, the biological function of NMN on cellular apoptosis was detected. Through flow cytometry, it was found that cellular total apoptosis was induced by HS and these cellular biological progresses were alleviated by NMN treatment (P<0.001, Figure 2B).
NMN Alleviated HS-Induced MMP, Intracellular ROS and LDH Release Increase in CEC
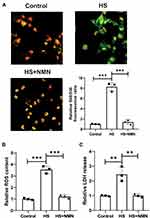
In this study, it was found that HS treatment significantly increased MMP. NMN treatment significantly improved the MMP status (P<0.001, Figure 3A) and it suggest that NMN demonstrated a significant protective effect in the MMP maintenance. Considering that mitochondria was a key source of intracellular ROS, the results of intracellular ROS was consistent with the MMP status. As showed in Figure 3B, NMN alleviated intracellular ROS induced by HS in cultured CEC. When the LDH release was considered, it was found that LDH release was significantly after HS treatment and advanced study demonstrated that NMN treatment would normalize the LDH release in the HS-treated CEC (P<0.001, Figure 3C).
HS-Induced IL-17a Expression Was Moderated by NMN Treatment Through SIRT1
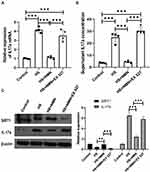
As CEC played a key role in the inflammation of ocular surface, the effect of HS on the expression of an important cytokine, IL-17a and its potential regulation mechanism was detected. Besides, the effect of NMN treatment on IL-17a expression was detected in this current experiments. As showed in Figure 4A–C, the expression of IL-17a in mRNA level, secreted IL-17a in supernatant of cell culture media and CEC samples were increased in the HS group (P<0.001) and 500 μM NMN treatment significantly moderated the IL-17a expression (P<0.001). In the advanced experiment, the effect of NMN administration on IL-17a demonstrated that NMN was a negative regulator of IL-17a secretion. As SIRT1 selective inhibitor (EX 527, 100 nM) treatment significantly neutralize the protective effect of NMN, it was found that NMN regulated the expression of IL-17a through SIRT1.
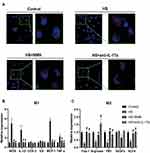
NMN Regulated Macrophage Phagocytosis and Polarization in the CEC/Macrophage Co-Culture System
To detect the effect of secreted IL-17a from CEC on the function of macrophage, we conducted an in-vitro study based on CEC-RAW264.7 co-culture system. The CEC and RWA264.7 cells in different groups were cultured independently for 24h and would be maintained in the co-culture system for 12h before advanced experiments. To detect the effect of NMN regulated CEC on macrophage phagocytosis and polarization, the RAW264.7 cells would be used in the advanced experiments. Before advanced experiments, the control IgG (1μg/mL, Sigma, USA) and anti-IL-17a (1μg/mL, R&D System, USA) would be applied in the RAW264.7 in the only HS and anti-IL-17a group, respectively. As showed in Figure 5A, increased phagocytosis ability of macrophage when co-cultured with the HS-treated CEC (P<0.05). While application of NMN in the CEC and anti-IL-17a would help to relieve the increased phagocytosis status of macrophage. Macrophage polarization was detected in each group and M1/M2 related biomarkers were detected. Higher concentrations of M1 related biomarkers, including iNOS, IL-1β, MCP-1 and TNF-α, were detected in macrophage co-cultured with the HS-treated CEC (P<0.05, Figure 5B). When the M2 related biomarkers were considered, lower Fizz-1, Arginase, YM1 and KLF4 concentrations were detected in the macrophages co-cultured with CEC in HS status (P<0.05, Figure 5C). The different expressed M1 and M2 related biomarkers would be moderated by NMN treatment in CEC and anti-IL-17a in the macrophage.
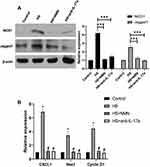
Notch Pathway Involved in the Effect of IL-17a/NMN on CEC/Macrophage Co-Culture System
As Notch pathway had been reported to play a key role in the function of IL-17a on the macrophage, its role in the function of NMN on the CEC/macrophage co-culture system. In this experiment, the expressions of both the key protein as well the Notch pathway related genes were analyzed. As showed in Figure 6A, increased protein level of NICD and Jagged1 were detected in the CEC in HS co-cultured with RAW274.6 group. However, the activation of Notch pathway would be suppressed when NMN added in the CEC and anti-IL-17a added in the RAW264.7. When the Notch pathway related genes were considered, higher expressions of CXCL1, Hes1 as well as Cyclin D1 were detected in the HS-treated CEC/macrophage co-culture system (P<0.05, Figure 6B). Moderated Notch pathway related genes expressions could be detected when NMN treatment in CEC and anti-IL-17a on RAW274.6 cells.
Discussion
DED was one of the most prevalent ocular disease in which tear hyperosmolarity was one of the key pathological progresses during the development of DED. Both CEC and macrophage played important effects in the hyperosmolarity-induced ocular surface, however, the interaction between CEC and macrophage was still eagerly required to be investigated. NMN had been reported to be related with the incidence of a variety of chronic diseases17 and recently reported to play a protective effect in ocular diseases.16 In the present study, it was confirmed HS treatment would lead to an increased expression of IL-17a in CEC and thus induce the activation of macrophage based on a CEC/macrophage co-culture system. Moreover, NMN could significantly alleviate HS-induced IL-17a secretion and moderate the subsequent macrophage activation through Notch pathway. As NMN was a direct activator of SIRT1 and selective inhibition of SIRT1 alleviated the protective effects of NMN in HS-treated CEC. Therefore, these findings provided us important clues in the understanding of the effects of NMN on DED development as well as the important pathological progress of CEC/macrophage interaction.
As DED was a multifactor disease and there are several relatively independent pathological progresses involving in the development of DED, among them HS has been regarded as one of the decisive factors.18 Increased tear hyperosmolarity was a clinical sign of incidence of DED and it could trigger the subsequent ocular inflammation. In previous studies, aberrantly hyperosmosis status would increase the apoptosis and ROS activation of CEC and thus cause ocular surface damage in the development of DED.19,20 Detecting the protective regents for CEC, such as application of high-affinity liposomal astaxanthin21 and Polygonum cuspidatum (PCE) aqueous extract,22 demonstrated potential innovative therapies for DED. However, the effects of NMN, which was regarded as a powerful pre-drug for cellular senescence, in the progression of DED remained unclear. In a recent study, it was reported that aging was related with corneal and ocular surface homeostasis in mice,23 thus it provided us powerful tools for potential application of anti-aging drug in the treatment of DED. In a most recent study, it was found that hyperglycemia decreased the biosynthesis of NAD+ and impaired epithelial wound healing in DM mice.24 Supplementation of NAD+ and its precursors promotes corneal wound healing and nerve regeneration in patients with diabetes mellitus. In this current study, NMN application in the HS-induced dry eye in-vitro model demonstrated significant protective effects, including improvement in cell viability and alleviated apoptotic status. This was the first study focusing the effect of NMN in HS-induced DED and the preliminary conclusion demonstrated that NMN could be regarded as a hopeful drug for DED management.
Abnormal increased inflammatory factors, including 1β, IL-6, IL-8, IL-10, interferon-γ, IFN-γ, and tumor necrosis factor-α, however, evidence was less strong for IL-2 and IL-17A.25 While it was found that Th17 was a key immune cell that demonstrated main role in chronic ocular surface disorders.26 The IL-17 cytokine family was named CTLA8 when it was discovered in 1993.27 It is secreted by T helper cell 17, Th17. It was subsequently discovered that it can induce synovial cells and mesenchyme. The cell secretes IL-6 and IL-8 and is renamed IL-17. Among the 6 members of the IL17 family, IL-17a is the symbol of the family and the most widely studied factor.28 It can protect the host against extracellular pathogens, but it can also promote the inflammatory pathology of autoimmune diseases, similar to IL-17c. Previous experiments confirmed that the IL-17 family can activate anti-cytokines and chemokines in the MAPK, NF-κB and C/EBPS pathways. As reported in a series of clinical studies, higher IL-17a in tear were detected in patients with contact lens discomfort and more severe dry eye related symptoms.12,29 Besides, it was demonstrated that increased IL-17a level was detected in the DED cases with diabetes comparing with the DED cases without diabetes and it highlighted the effect of IL-17a caused by the systemic inflammation on the DED incidence.12 In this current experiment, we provided evidences demonstrating that increased hyperosmolarity would induce up-regulation of IL-17a in the CEC and then lead to the activation of macrophage. These results highlight the important role of IL-17a in the incidence of DED and confirmed the function if IL-17a on macrophage. It should be noticed that IFN-γ, which was both up-regulated in DED as well as a key regulator of macrophage,30 might produce potential key role in the incidence of DED. In the following experiments, IFN-γ was once be considered as the target gene in the detection of the molecular mechanism DED development. Besides, Notch pathway had been reported to be an important pathway involved in the biological activity of IL-17a in macrophage. To date, several attempts have been conducted to analyze the effect of IL-17a on the Notch pathway and it was reported that Notch signaling pathway was tightly associated with IL-17a in airway hyperresponsiveness and skin inflammation.31,32 Activated action of Notch pathway and increased expression of its target genes provided us powerful and conclusive evidence on the biological activity of IL-17a through Notch. Advanced experiments should be conducted to detect the potential protective effects of inhibitors of IL-17a and Notch pathway on DED incidence.
NAD+ was the substrate of longevity III protein lysine deacetylase Sirtuins. SITR1 existed in the cytoplasm. Sirtuins played an important role in cell resistance, energy metabolism, apoptosis and aging, so they are called longevity proteins. SIRT1 had been regarded as a potential protective target for ocular damages.33,34 It had confirmed that NMN after systemic or local application would always be transformed to NAD+ and then demonstrated important regulation effects for kinds of diseases.35,36 SIRT1 was one of the most confirmed NAD+ consuming enzyme, thus higher SIRT1 would be induced by NMN application in various disorders and biological progresses, including hepatocellular carcinoma,37 adaptive thermogenesis38 and osteogenesis.39 In this current experiment, it was found that NMN-induced expression of SIRT1 could lead to a decreased level of IL-17a and this result might explain the biological mechanism of NMN application on DED, at least in part. This finding was consistent with previous result based on animal experiments of allograft rejection, T cell-driven retinal disease and germinal matrix hemorrhage.40–42 The mounting evidences on the relationship between SIRT1 and IL-17a in various biological progresses demonstrated that they could be a potential regulation mechanism in other disorders. There was one limitation should be discussed in this experiment. Only SIRT1 and IL-17a inhibitors, EX-527 and IL-17a monoclonal antibody, were used in the mechanism investigation. Even these two regents were target selective, the off-target effects of these two inhibitors should be addressed in the understanding of pathological effects of SIRT1/IL-17a pathway in the CEC-macrophage crosstalk in the development of DED. The vector regents or control Fc antibody would provide better knowledge in the validation of mechanism of action for SIRT1/IL-17a pathway. Besides, advanced studies based on SIRT1 or IL-17a knock-out mice would help in the detailed molecular mechanism detection in the following experiments.
In this current study, we provided the knowledge of NMN application on the expression of SIRT1 and IL-17a and then described the potential molecular mechanism in the CEC-macrophage co-culture system. To describe the regulation effects of downstream functional proteins, the Notch pathway was described and it improved our understanding of the DED. Besides, the interaction between CEC and macrophage was described in this current study and thus provide us deeper understanding in the biological progress of DED. The central role of CEC was confirmed and the co-culture system could be used in the following DED analyses. However, one limitation should be mentioned in this study was that only in-vitro data was provided in this study. Considering the complex regulation relationship of different genes and cell types were studied in this study, it might be relatively hard to re-conduct this experiment in animal models. The macrophage cell line, RAW264.7, was not primary cultured cells and it would be partly from the really macrophage in the mice. In the advanced, primary cultured macrophage would be used in the experiment. Besides, considering the off-target effects could not be ignored in the inhibitor use in the experiments, more animal experiments based on SIRT1−/- or IL-17a−/- mice would be conducted. The apoptosis related proteins, including Bax/Bcl2 and caspase family, would help our understanding in the effects of NMN on HS-related CEC apoptosis. Based on the fact that there were limitations on the cellular experiments, we thought that Tunnel assay in animal experiments would help to improve our knowledge. These experiments would be conducted in the animal experiments. In the future, advanced application of activators and inhibitors of SIRT1 and Notch pathway would be used and thus provide us better understanding in the specific drugs for the treatment of DED.
Conclusions
In summary, NMN could induce up-regulation of SRIT1 and down-regulation of IL-17a in the CEC. Application of NMN would alleviate cellular damage of CEC caused by hyperosmolarity. Besides, IL-17a secreted from CEC would activate macrophage and lead to M1 polarization thus demonstrated harmful effects. Our collective data pointed to the molecular mechanisms and then provided the potential benefits of NMN in the management of DED.
Abbreviations
NMN, nicotinamide mononucleotide; DED, dry eye disease; HS, hyperosmosis stress; CEC, corneal epithelial cells; LDH, lactate dehydrogenase; MMP, mitochondrial membrane potential; IL-17a, interleukin 17a; iNOS, inducible nitric oxide; COX-2, cyclooxygenase 2; Fizz-1, found in inflammatory zone 1; TNF-α, tumor necrosis factor-α; Arg, Arginase-1; TGF-β1, transforming growth factor-β1; IL1β, interleukin 1β; IL6, interleukin 6; MCP-1, monocyte chemoattractant protein 1; YM1, chitinase3-like3; VEGFa, vascular endothelial growth factor a; CXCL1, chemokine ligand 1; NICD, intracellular domain of Notch.
Data Sharing Statement
Data was available through request to corresponding author (Li XY: [email protected]; Zhu W: [email protected]).
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This work was supported in whole or in part by National Nature Science Foundation Project for Young Scientists of China (Grant No. 81700804), the Foundation for Young Medical Talents of Jiangsu Province (Grant No. QNRC2016211) and Scientific Research Project of Jiangsu Health Commission (Z2019044).
Disclosure
The authors declare no competing interests in this work.
References
1. Tsubota K, Pflugfelder SC, Liu Z, et al. Defining dry eye from a clinical perspective. Int J Mol Sci. 2020;21(23):9271. doi:10.3390/ijms21239271
2. Kojima T, Dogru M, Kawashima M, Nakamura S, Tsubota K. Advances in the diagnosis and treatment of dry eye. Prog Retin Eye Res. 2020;78:100842. doi:10.1016/j.preteyeres.2020.100842
3. Talens-Estarelles C, Garcia-Marques JV, Cervino A, Garcia-Lazaro S. Use of digital displays and ocular surface alterations: a review. Ocul Surf. 2020. doi:10.1016/j.jtos.2020.10.001
4. Magny R, Regazzetti A, Kessal K, et al. Lipid annotation by combination of UHPLC-HRMS (MS), molecular networking, and retention time prediction: application to a lipidomic study of in vitro models of dry eye disease. Metabolites. 2020;10(6):225. doi:10.3390/metabo10060225
5. Wang B, Peng L, Ouyang H, et al. Induction of DDIT4 impairs autophagy through oxidative stress in dry eye. Invest Ophthalmol Vis Sci. 2019;60(8):2836–2847. doi:10.1167/iovs.19-27072
6. Hong SC, Ha JH, Lee JK, Jung SH, Kim JC. In vivo anti-inflammation potential of aster koraiensis extract for dry eye syndrome by the protection of ocular surface. Nutrients. 2020;12(11):3245. doi:10.3390/nu12113245
7. Huang HY, Wang MC, Chen ZY, et al. Gelatin-epigallocatechin gallate nanoparticles with hyaluronic acid decoration as eye drops can treat rabbit dry-eye syndrome effectively via inflammatory relief. Int J Nanomedicine. 2018;13:7251–7273. doi:10.2147/IJN.S173198
8. Yang FM, Fan D, Yang XQ, et al. The artemisinin analog SM934 alleviates dry eye disease in rodent models by regulating TLR4/NF-kappaB/NLRP3 signaling. Acta Pharmacol Sin. 2020. doi:10.1038/s41401-020-0484-5
9. Soiberman U, Kambhampati SP, Wu T, et al. Subconjunctival injectable dendrimer-dexamethasone gel for the treatment of corneal inflammation. Biomaterials. 2017;125:38–53. doi:10.1016/j.biomaterials.2017.02.016
10. Zhou Y, Lin J, Peng X, et al. The role of netrin-1 in the mouse cornea during aspergillus fumigatus infection. Int Immunopharmacol. 2019;71:372–381. doi:10.1016/j.intimp.2019.03.047
11. Ji Q, Wang L, Liu J, et al. Aspergillus fumigatus-stimulated human corneal epithelial cells induce pyroptosis of THP-1 macrophages by secreting TSLP. Inflammation. 2020. doi:10.1007/s10753-020-01367-x
12. Liu R, Ma B, Gao Y, Ma B, Liu Y, Qi H. Tear inflammatory cytokines analysis and clinical correlations in diabetes and nondiabetes with dry eye. Am J Ophthalmol. 2019;200:10–15. doi:10.1016/j.ajo.2018.12.001
13. Xie N, Zhang L, Gao W, et al. NAD(+) metabolism: pathophysiologic mechanisms and therapeutic potential. Signal Transduct Target Ther. 2020;5(1):227.
14. Wang MTM, Muntz A, Lim J, Kim JS, Lacerda L, Arora A. Ageing and the natural history of dry eye disease: a prospective registry-based cross-sectional study. Ocul Surf. 2020;18(4):736–741. doi:10.1016/j.jtos.2020.07.003
15. Li Y, Ma X, Li J, et al. Corneal denervation causes epithelial apoptosis through inhibiting NAD+ biosynthesis. Invest Ophthalmol Vis Sci. 2019;60(10):3538–3546. doi:10.1167/iovs.19-26909
16. Zhao C, Li W, Duan H, et al. NAD(+) precursors protect corneal endothelial cells from UVB-induced apoptosis. Am J Physiol Cell Physiol. 2020;318(4):C796–C805. doi:10.1152/ajpcell.00445.2019
17. Cambronne XA, Kraus WL. Location, location, location: compartmentalization of NAD(+) synthesis and functions in mammalian cells. Trends Biochem Sci. 2020;45(10):858–873. doi:10.1016/j.tibs.2020.05.010
18. Guzman M, Miglio M, Keitelman I, et al. Transient tear hyperosmolarity disrupts the neuroimmune homeostasis of the ocular surface and facilitates dry eye onset. Immunology. 2020;161(2):148–161. doi:10.1111/imm.13243
19. Rokohl AC, Trester M, Guo Y, et al. Dry anophthalmic socket syndrome - standardized clinical evaluation of symptoms and signs. Ocul Surf. 2020;18(3):453–459. doi:10.1016/j.jtos.2020.05.001
20. Downie LE, Ng SM, Lindsley KB, Akpek EK. Omega-3 and omega-6 polyunsaturated fatty acids for dry eye disease. Cochrane Database Syst Rev. 2019;12:CD011016. doi:10.1002/14651858.CD011016.pub2
21. Shimokawa T, Yoshida M, Fukuta T, Tanaka T, Inagi T, Kogure K. Efficacy of high-affinity liposomal astaxanthin on up-regulation of age-related markers induced by oxidative stress in human corneal epithelial cells. J Clin Biochem Nutr. 2019;64(1):27–35. doi:10.3164/jcbn.18-27
22. Park B, Lee IS, Hyun SW, Jo K, Lee TG, Kim JS. The protective effect of polygonum cuspidatum (PCE) aqueous extract in a dry eye model. Nutrients. 2018;10(10):1550. doi:10.3390/nu10101550
23. De Silva MEH, Hill LJ, Downie LE, Chinnery HR. The effects of aging on corneal and ocular surface homeostasis in mice. Invest Ophthalmol Vis Sci. 2019;60(7):2705–2715. doi:10.1167/iovs.19-26631
24. Li Y, Li J, Zhao C, et al. Hyperglycemia-reduced NAD(+) biosynthesis impairs corneal epithelial wound healing in diabetic mice. Metabolism. 2021;114:154402. doi:10.1016/j.metabol.2020.154402
25. Roda M, Corazza I, Bacchi Reggiani ML, Pellegrini M, Taroni L, Giannaccare G. Dry eye disease and tear cytokine levels-a meta-analysis. Int J Mol Sci. 2020;21(9):3111. doi:10.3390/ijms21093111
26. Fan NW, Dohlman TH, Foulsham W, McSoley M, Singh RB, Chen Y. The role of Th17 immunity in chronic ocular surface disorders. Ocul Surf. 2020. doi:10.1016/j.jtos.2020.05.009
27. Girondel C, Levesque K, Langlois MJ, et al. Loss of interleukin-17 receptor D promotes chronic inflammation-associated tumorigenesis. Oncogene. 2020;40(2):452–464. doi:10.1038/s41388-020-01540-4
28. Trivedi S, Labuz D, Anderson CP, et al. Mucosal-associated invariant T (MAIT) cells mediate protective host responses in sepsis. Elife. 2020;9. doi:10.7554/eLife.55615
29. Gad A, Vingrys AJ, Wong CY, Jackson DC, Downie LE. Tear film inflammatory cytokine upregulation in contact lens discomfort. Ocul Surf. 2019;17(1):89–97. doi:10.1016/j.jtos.2018.10.004
30. Murray PJ, Wynn TA. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 2011;11(11):723–737. doi:10.1038/nri3073
31. Zeng Z, Wang L, Ma W, et al. Inhibiting the notch signaling pathway suppresses Th17-associated airway hyperresponsiveness in obese asthmatic mice. Lab Invest. 2019;99(12):1784–1794. doi:10.1038/s41374-019-0294-x
32. Wang Y, Li X, Xing X, Xue H, Qi R, Ji H. Notch-hes1 signaling regulates IL-17A(+) gammadelta (+)T cell expression and IL-17A secretion of mouse psoriasis-like skin inflammation. Mediators Inflamm. 2020;2020:8297134. doi:10.1155/2020/8297134
33. Li Y, Li J, Zhao C, et al. Hyperglycemia-reduced NAD(+) biosynthesis impairs corneal epithelial wound healing in diabetic mice. Metabolism. 2020;154402.
34. Li J, Qi X, Wang X, Li W, Li Y, Zhou Q. PTEN inhibition facilitates diabetic corneal epithelial regeneration by reactivating Akt signaling pathway. Transl Vis Sci Technol. 2020;9(3):5. doi:10.1167/tvst.9.3.5
35. Okur MN, Fang EF, Fivenson EM, Tiwari V, Croteau DL, Bohr VA. Cockayne syndrome proteins CSA and CSB maintain mitochondrial homeostasis through NAD(+) signaling. Aging Cell. 2020;19(12):e13268. doi:10.1111/acel.13268
36. Xie W, Zhu T, Zhou P, et al. Notoginseng leaf triterpenes ameliorates OGD/R-induced neuronal injury via SIRT1/2/3-Foxo3a-MnSOD/PGC-1alpha signaling pathways mediated by the NAMPT-NAD pathway. Oxid Med Cell Longev. 2020;2020:7308386. doi:10.1155/2020/7308386
37. Garten A, Grohmann T, Kluckova K, Lavery GG, Kiess W, Penke M. Sorafenib-induced apoptosis in hepatocellular carcinoma is reversed by SIRT1. Int J Mol Sci. 2019;20(16):4048. doi:10.3390/ijms20164048
38. Yamaguchi S, Franczyk MP, Chondronikola M, et al. Adipose tissue NAD(+) biosynthesis is required for regulating adaptive thermogenesis and whole-body energy homeostasis in mice. Proc Natl Acad Sci U S A. 2019;116(47):23822–23828. doi:10.1073/pnas.1909917116
39. Song J, Li J, Yang F, et al. Nicotinamide mononucleotide promotes osteogenesis and reduces adipogenesis by regulating mesenchymal stromal cells via the SIRT1 pathway in aged bone marrow. Cell Death Dis. 2019;10(5):336. doi:10.1038/s41419-019-1569-2
40. Ye Q, Zhang M, Wang Y, Fu S, Han S, Wang L. Sirtinol regulates the balance of Th17/Treg to prevent allograft rejection. Cell Biosci. 2017;7(1):55. doi:10.1186/s13578-017-0182-2
41. Gardner PJ, Joshi L, Lee RW, Dick AD, Adamson P, Calder VL. SIRT1 activation protects against autoimmune T cell-driven retinal disease in mice via inhibition of IL-2/Stat5 signaling. J Autoimmun. 2013;42:117–129. doi:10.1016/j.jaut.2013.01.011
42. Liu SP, Huang L, Flores J, et al. Secukinumab attenuates reactive astrogliosis via IL-17RA/(C/EBPbeta)/SIRT1 pathway in a rat model of germinal matrix hemorrhage. CNS Neurosci Ther. 2019;25(10):1151–1161. doi:10.1111/cns.13144
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.