Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 8
New approach to the understanding of keloid: psychoneuroimmune–endocrine aspects
Authors Hochman B, Isoldi F , Furtado F , Masako Ferreira L
Received 21 June 2014
Accepted for publication 21 September 2014
Published 10 February 2015 Volume 2015:8 Pages 67—73
DOI https://doi.org/10.2147/CCID.S49195
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Jeffrey Weinberg
Bernardo Hochman†, Felipe Contoli Isoldi, Fabianne Furtado, Lydia Masako Ferreira
Plastic Surgery Division, Federal University of São Paulo, São Paulo, Brazil
†Bernardo Hochman passed away on April 21, 2014
Abstract: The skin is a dynamic and complex organ that relies on the interrelation among different cell types, macromolecules, and signaling pathways. Further, the skin has interactions with its own appendages and other organs such as the sebaceous glands and hair follicles, the kidney, and adrenal glands; systems such as the central nervous system; and axes such as the hypothalamic–pituitary–adrenal axis. These continuous connections give the skin its versatility, and when an injury is caused, some triggers start a cascade of events designed to restore its integrity. Nowadays, it is known that this psychoneuroimmune–endocrine intercommunication modulates both the homeostatic condition and the healing process. In this sense, the skin conditions before a trauma, whether of endogenous (acne) or exogenous origin (injury or surgical incision), could regulate the process of tissue repair. Most skin diseases such as psoriasis and atopic dermatitis, among others, have in their pathophysiology a psychogenic component that triggers integrated actions in the nervous, immune, and endocrine systems. However, fibroproliferative disorders of wound healing, such as hypertrophic scar and keloid, are not yet included in this listing, despite showing correlation with stress, especially with the psychosocial character. This review, by understanding the "brain–skin connection", presents evidence that allows us to understand the keloid as a psychomediated disease.
Keywords: keloid, stress, psychological, psychoneuroimmunology, wound healing
Introduction
Keloid is considered a fibroproliferative disorder of skin wound healing, and its pathophysiological mechanisms are not fully known. As keloid only occurs in humans in which the psychological chronic stress component is already demonstrated, it is now also reported from the “psychomediation” perspective.
Skin scar diseases cannot be understood merely as esthetic complaints. They are associated with a variety of physiological reactions that can be modulated by many events, such as psychological ones. It begins with understanding about the interconnection between the skin and the central nervous system at an embryonic level. Their common origin is the ectoderm. Thus, depending on the reference, it can be said that the skin would be the external surface of the brain, or the brain would be the skin’s deepest layer.1
Some skin diseases such as psoriasis,2 atopic dermatitis,3 seborrheic dermatitis,4 herpes simplex,5 vitiligo,6,7 acne,8 alopecia,9 and urticaria10 are already included in the “psychophysiological disorders” category.11 Keloid and hypertrophic scars should also be included in this category because of the impact of psychoemotional stress in their clinical features. The trigger, the maintenance, and/or the worsening of the signs and symptoms of these skin scar diseases are closely related to a stressful event (internal/psychological and/or external/environmental)12 that activates neuroimmune–endocrine circuits.13
In this context, this review aims to open up new fronts for the comprehension of this pathological scar, looking for a better understanding of the patient as a whole in a psychoneuroimmune–endocrine way.14,15
Psychoneuroimmune–endocrine systems and the skin
The skin has several elements of the nervous,16 immune,17 and endocrine systems.18 Some examples are corticotropin-releasing hormone (CRH) production by keratinocytes, melanocytes, and pilosebaceous units;19 adrenocorticotropic hormone (ACTH) and α-melanocyte-stimulating hormone (α-MSH) by keratinocytes, melanocytes, pilosebaceous units, fibroblasts, and endothelial cells;20 and cortisol by keratinocytes and pilosebaceous units, among other hormones.21 Cytokines and growth factors such as interleukin 1 (IL-1) are also synthesized by keratinocytes, melanocytes, fibroblasts, and endothelial cells; IL-6 by the keratinocytes, fibroblasts, pilosebaceous units, and endothelial cells; tumor necrosis factor-α (TNF-α) by keratinocytes and melanocytes;22,23 and interferon-γ by keratinocytes and fibroblasts, among other elements of character, mostly proinflammatory. Therefore, the skin has a power plant of its own, with the capacity to produce locally all these factors related to other systems, speaking a common biochemical language and communicating via a complete bidirectional circuit involving shared ligands.24 In addition to this local and bidirectional communication among systems, there is also a central–peripheral communication (Figure 1).25
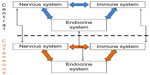  | Figure 1 Integration of central and cutaneous neuro immune–endocrine systems. |
In this review, regulatory events of the skin homeostasis will be studied under the prism of psychogenic stimuli.26 Besides the 5-hydroxytryptamine, acetylcholine, and inflammatory cytokines, signaling from areas of the limbic system (hippocampus, amygdale, and medial prefrontal cortex)27 is able to release CRH from the hypothalamus paraventricular nuclei (Figure 2A). Adding to its classical performance in the pituitary gland, releasing ACTH, which acts on the adrenal glands cortex, providing cortisol, the CHR also elicits sympathetic responses in the locus coeruleus of the brain stem, releasing norepinephrine by peripheral nerve endings, and norepinephrine and epinephrine by the adrenal glands medulla (Figure 2B). The activation of the sympathetic nervous system also enables the production of other substances, including the catecholamines.
The skin is able to express CRH as its receptors (CRH-R). The CRH-R1α isoform is the predominant CRH receptor in the skin, and it is expressed in all major cell populations of epidermis, dermis, and subcutaneous tissue. By contrast, CRH-R2 is expressed predominantly in hair follicles, sebaceous and eccrine glands, and muscle and blood vessels.28 In addition to CRH, the skin also expresses urocortin I and urocortin II messenger ribonucleic acid.28 CRH-R1 binds to urocortin I but not to urocortin II, while CRH-R2 binds to urocortin II but not to urocortin I,29,30 leading to the belief that the skin has a depth of responsiveness and interaction to the environment that is little understood. Finally, skin produces the precursor protein proopiomelanocortin protein (POMC) and POMC-derived peptides that give rise to ACTH and other polypeptide products.19,31 Keloid fibroblasts express POMC.32
Ito et al33 have shown that human hair follicles can synthesize cortisol, and its synthesis is regulated by endogenous feedback controls. Thus, the skin apparently has a peripheral equivalent to the hypothalamic–pituitary–adrenal (HPA) axis that is fully functional. The peripheral skin HPA axis may coordinate or fine-tune peripheral stress responses with the central HPA axis. In addition to expressing components of the HPA axis, the skin also produces a number of other neuroendocrine signals, including prolactin,34–36 melatonin,37 and catecholamines.38
Besides the HPA axis, the skin is highly innervated with sensory nerves that produce neurotrophins and neuropeptides. Sensory nerves derive from the dorsal root ganglion in the skin, and C-fibers form the cutaneous sensory nervous system. Psychological stress leads to increased concentrations of cutaneous nerve growth factor (NGF).39 NGF has a number of biological activities, including 1) axon sprouting of peptidergic and sympathetic neurons; 2) promoting cross-talk between neural cells, glia, and immune cells; and 3) facilitating monocyte/macrophage migration through vascular endothelium.40 NGF upregulates substance P (SP) nerve fibers in the dermis of stressed mice. Calcitonin gene-related peptide (CGRP), a potent vasodilator, is also upregulated in response to NGF.39 SP and CGRP have different distributions within the skin, with SP nerve fibers detected in the dermis and subcutis, and CGRP nerve fibers are in the epidermis around the distal hair follicle and the arrector pili muscle.41
From this evidence, there is no way to understand skin diseases and their symptoms decoupled from psychological aspects. Clinical and experimental data support the assertion that the minds can start, stop, and influence biological events in the skin.42
Psychoneuroimmune–endocrine system and keloid
At the moment of the loss of skin integrity
Considering the three super-systems, the nervous system is the one that responds more rapidly to noxious stimuli in the skin, due to its electrical nature. After a skin wound (endogenous origin such as acne, or exogenous such as a surgical incision or a mosquito bite), the loss of cutaneous integrity generates a “short circuit” in the skin battery, changing the outer skin pole to positive; this is responsible for generating an electrical current at the site of injury and initiating eletrotaxia. This current remains present until the skin regains its integrity by wound healing, restoring its natural negative polarization. This current of injury helps eletrotaxia (primarily neutrophils and monocytes) initiate tissue repair (for better understanding of these definitions, see Hochman et al43).
At the same time as the nerve endings directly influence the bioelectrical condition, they also lead afferences to the dorsal root ganglion, reporting the cutaneous status. Some of these stimuli reach the cerebral cortex. The efferent response is the neuropeptides and neurotransmitters release. The neuroelectric and neuropeptidergic components of the healing are inseparable parts of the neurogenic inflammation. The neurogenic inflammation level dictates the intensity of the subsequent phases of the healing.
Akaishi et al44 proposed a neurogenic inflammation hypothesis for keloid formation. Other authors have also followed this perspective.45–47 However, all of them have analyzed just the peripheral and local nature of the lesion as a result of stressful stimuli (skin stretch and mechanotransduction). Considering the existence of central–peripheral ways of communication between the central and the cutaneous nervous system, it is possible to extrapolate the opposite route, based on the same grounds, in which central nervous stimuli would trigger a similar response in the skin. The neurogenic inflammation level of the healing process is directly related to the amount and the functionality of cutaneous nerve fibers. Kadanoff48 was the pioneer in demonstrating the relationship of innervation with keloid. Hochman et al49 showed higher density and depth of nerve fibers in the keloid than the skin. A higher density of nerve fibers was also demonstrated in hypertrophic scars when compared to normal scars.50–52 In turn, the lower density of nerve fibers in the keloid epidermis could be related to chronic stimulation of conductive nerve endings of itching, similar to the fibers of the nociceptive stimuli, and it would be an autoregulatory mechanism for modulation of intensity and persistence of sensory inputs.53 In this context, botulinum toxin has recently come to prominence as a form of treatment.54–57 For the future, capsaicin, the functional denervator, may also appear as an alternative.58
Before the loss of skin integrity
The basal skin condition is a very important factor in healing disorders. The cutaneous inflammatory status is influenced, among other factors, by ultraviolet radiation,12,59 feeding,60–62 obesity and metabolic syndrome, and stress of psychological origin.63 This could explain the bizarre temporal character of keloid, in that an individual can develop it in different periods of their life.64 Black skin absorbs more ultraviolet radiation than the other skin color types. This is a disadvantage because the more radiation absorbed, the more skin inflammation, increasing this important risk factor which corresponds to a higher prevalence of keloids in this population.
The immune response and the immune system can be modulated by the interactions between essential fatty acids, eicosanoids, and free radicals coming from the diet. For the essential fatty acids, if there are enhanced arachidonic acid levels plus deficiency of precursors (linoleic acid, g-linolenic acid, and dihomo-g-linolenic acid) and some inflammatory competitors (dihomo-g-linolenic acid and eicosapentaenoic acid), it will result in an inevitable overproduction of proinflammatory metabolites, which enhances a pathologic inflammation, both systemic and local, in the skin. This scenario is seen in patients with keloid.65
Psychological stress can amplify both the bioelectric and neuropeptidergic components of the neurogenic phase of the healing inflammation. Sweat and sebaceous secretion as a result of emotional factors66,67 increase the electrical conductivity of the deeper layers and the impedance of the stratum corneum, respectively, increasing the current of injury and the eletrotaxia at the moment of injury. On the other hand, the stress chronically stimulates the neuroimmune–endocrine circuit and the sensory system.68–70 The binding of neurotransmitters, neuropeptides, and neurohormones (acetylcholine, CGRP, substance P, CRH) in the mast cells receptors makes them degranulate. From the degranulation, several proinflammatory mediators such as IL-1, IL-6, growth factors (NGF, vascular endothelial growth factor [VEGF], transforming growth factor-β [TGF-β]), TNF-α, and histamine are released and refeed the cycle, creating a chronic inflammatory condition.12 The intimate relationship and interaction between mast cells and myofibroblasts support the important role of mast cells and their mediators in the pathogenesis of the keloid.71,72 TGF-β,73–76 VEGF,74 histamine,77 TNF-α,78 and IL-679–81 have already been implicated in the pathogenesis and symptomatology of the keloid.82
In summary, stressed patients exacerbate the neuroimmune–endocrine system, and that causes basal inflammatory metabolic activity on the individual skin, which enhances the neurogenic inflammation phase during the healing process. In other words, people bring on the inflammation themselves when their “nerves are on edge”.43
Furtado et al83 were the first to show a direct correlation between stress and keloid. Patients with fibroproliferative scars who were candidates for surgical resection and postoperative radiotherapy underwent evaluation of psychological stress on the day before the surgery. The parameters evaluated were pain and itching, quality of life, perceived stress, depression and anxiety – through questionnaires and scales – salivary cortisol, and minimum and maximum galvanic skin response (GSR) at rest and under stress (when filling in questionnaires). The patients were evaluated postoperatively at 3, 6, 9, and 12 months. Each time the patients returned to the medical service, two specialists classified the lesions in nonrelapsed and relapsed patients. The relapsed group presented higher values in GSR during the stress situation. The chance of relapse increased 34% for each increase of 1,000 arbitrary units, at maximum GSR in stress. Thus, psychological stress increases the chance of relapse in postoperative keloid.
Final considerations
Keloid management is still considered a challenge for physicians and researchers. This is a healing benign neoplasm exclusive to humans whose therapeutic modalities are unsatisfactory and present a high rate of recurrence.84,85 Several hypotheses regarding its physiopathogenesis have been postulated.44,86–89 Currently, a genetic cause has received the greater prominence, with a large number of publications about it.90–92
However, it must be understood that genetic and environmental influences are interconnected in a complex way. Currently, a widely accepted model is the stress–diathesis model, according to which stressful situations (the precipitating factor) will affect people in different ways due to inherited vulnerabilities (a predisposing factor). Moreover, according to this model, a genetic predisposition to disease development (“diathesis”) may remain latent until stress events, which can be represented by psychosocial factors interacting with biological characteristics of a person, make the disease unfold.42
Environmental factors are certainly critical in defining phenotypes during early development, and they continue to influence phenotypes throughout the life cycle of an organism. Nearly every aspect of development and behavior is affected by both personal experience, that is gained through the environment, and the genetic makeup. Environmental factors can affect and alter gene expression, while genes can define how people respond to different environments. Indeed, it is actually superficial to debate whether nature or nurture is more important. In truth, the relationship between genetic determinants and the environment is so completely entwined that you cannot look at an individual and judge which contribution is more valuable. Together, the continual interplay of both genes and ever-changing environmental factors determines who people are.
Keloid and hypertrophic scars are different phenotypic expressions of the same phenomenon and can exemplify the complexity of the disorder.92 It is certain that the treatment of these healing disorders cannot focus on one single aspect. It is necessary to understand the wide spectrum of communication between the three super-systems. The release of hormones, neural transmitters, substances, and immune cells creates an inflammatory environment that stimulates the fibrogenesis (the expression of proinflammatory genes are inserted in this context).89,93–95 On the other hand, personality and coping styles reflect individual differences in appraisal and response to stressors that may influence neuroimmune–endocrine functions.
Thus, skin disorders should be interpreted in a holistic way; in other words, a good practice evaluates the individual as a whole, under a psychoneuroimmune–endocrine functional perspective. Since most, or perhaps all, cases of cutaneous somatization have been long neglected, they now become the target of research in which the psychogenic part has gained prominence, because there is no dermatosis in which the psychological factor is not involved.43 The trigger to wound healing depends on neurogenic factors (electrical and neurosecretory) which, in turn, are premodulated by psychological aspects and adjusted by immune and endocrine factors. Any change in these circuits may interfere in the normal formation of a scar, resulting in wound healing deficit (atrophic scars and ulcerations) until hyperproliferative healing (hypertrophic scars and keloid).
Based on these principles, the treatment of patients with keloid should be conducted by a multidisciplinary team. The joint and integrated approach of physicians, psychologists, nurses, nutritionists, and physical therapists knowledgeable of its psychoneuroimmune–endocrine etiology may be able to provide relief to those who suffer from a deformative, stigmatizing disease that is still without a cure.
Acknowledgment
Tribute is paid to the great master Professor Bernardo Hochman. This is a posthumous tribute to him, he devoted much of his life to studying, understanding, teaching, and disseminating what the keloid really is. We will remember forever his words “If the eyes are the mirror of the soul, the skin is the mirror of the mind.”
Disclosure
The authors report no conflicts of interest in this work.
References
Juhan D. Job’s Body. Skin as Surface of the Brain. Barrytown, NY: Station Hill Press, Inc.; 2013:35–41. | |
Hall JM, Cruser D, Podawiltz A, Mummert DI, Jones H, Mummert ME. Psychological stress and the cutaneous immune response: roles of the HPA axis and the sympathetic nervous system in atopic dermatitis and psoriasis. Dermatol Res Pract. 2012;2012:403908. | |
Suárez AL, Feramisco JD, Koo J, Steinhoff M. Psychoneuroimmunology of psychological stress and atopic dermatitis: pathophysiologic and therapeutic updates. Acta Derm Venereol. 2012;92(1):7–15. | |
Misery L, Touboul S, Vinçot C, et al. Stress and seborrheic dermatitis. Ann Dermatol Venereol. 2007;134(11):833–837. | |
Chida Y, Mao X. Does psychosocial stress predict symptomatic herpes simplex virus recurrence? A meta-analytic investigation on prospective studies. Brain Behav Immun. 2009;23(7):917–925. | |
Manolache L, Benea V. Stress in patients with alopecia areata and vitiligo. J Eur Acad Dermatol Venereol. 2007;21(7):921–928. | |
Yu R, Huang Y, Zhang X, Zhou Y. Potential role of neurogenic inflammatory factors in the pathogenesis of vitiligo. J Cutan Med Surg. 2012;16(4):230–244. | |
Yosipovitch G, Tang M, Dawn AG, et al. Study of psychological stress, sebum production and acnevulgaris in adolescents. Acta Derm Venereol. 2007;87(2):135–139. | |
Hadshiew IM, Foitzik K, Arck PC, Paus R. Burden of hair loss: stress and the underestimated psychosocial impact of telogen effluvium and androgenetic alopecia. J Invest Dermatol. 2004;123:455–457. | |
Arck P, Paus R. From the brain-skin connection: the neuroendocrine-immune misalliance of stress and itch. Neuroimmunomodulation. 2006;13(5–6):347–356. | |
Lugović-Mihić L, Ljubešić L, Mihić J, Vuković-Cvetković V, Troskot N, Šitum M. Psychoneuroimmunologic aspects of skin diseases. Acta Clin Croat. 2013;52:337–345. | |
Arck PC, Slominski A, Theoharides TC, Peters EM, Paus R. Neuroimmunology of stress: skin takes center stage. J Invest Dermatol. 2006;126(8):1697–1704. | |
Paus R, Theoharides TC, Arck PC. Neuroimmunoendocrine circuitry of the ‘brain-skin connection’. Trends Immunol. 2006;27(1):32–39. | |
Ader R, Cohen N, Felten D. Psychoneuroimmunology: interactions between the nervous system and the immune system. Lancet. 1995; 345(8942):99–103. | |
Furtado F, Hochman B, Ferrara SF, et al. What factors affect the quality of life of patients with keloids? Rev Assoc Med Bras. 2009;55(6):700–704. | |
Roosterman D, Goerge T, Schneider SW, Bunnett NW, Steinhoff M. Neuronal control of skin function: the skin as a neuroimmunoendocrine organ. Physiol Rev. 2006;86(4):1309–1379. | |
Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today. 1993;14(2):75–78. | |
Zouboulis CC. Human skin: an independent peripheral endocrine organ. Horm Res. 2000;54(5–6):230–242. | |
Kono M, Nagata H, Umemura S, Kawana S, Osamura RY. In situ expression of corticotrophin-releasing hormone (CRH) and proopiomelanocortin (POMC) genes in human skin. FASEB J. 2001;15(12):2297–2299. | |
Wakamatsu K, Graham A, Cook D, Thody AJ. Characterisation of ACTH peptides in human skin and their activation of the melanocortin-1 receptor. Pigment Cell Res. 1997;10(5):288–297. | |
Slominski A, Zbytek B, Nikolakis G, et al. Steroidogenesis in the skin: implications for local immune functions. J Steroid Biochem Mol Biol. 2013;137:107–123. | |
Luger TA. Epidermal cytokines. Acta Derm Venereol Suppl (Stockh). 1989;151:61–76. | |
Ansel J, Perry P, Brown J, et al. Cytokine modulation of keratinocyte cytokines. J Invest Dermatol. 1990;94(Suppl 6):101S–107S. | |
Blalock JE. The immune system as the sixth sense. J Inter Med. 2005;257(2):126–138. | |
Ferreira LM, Gragnani A, Furtado F, Hochman B. Control of the skin scarring response. An Acad Bras Cienc. 2009;81(3):623–629. | |
Slominski AT, Zmijewski MA, Zbytek B, Tobin DJ, Theoharides TC, Rivier J. Key role of CRF in the skin stress response system. Endocr Rev. 2013;34(6):827–884. | |
Jankord R, Herman JP. Limbic regulation of hypothalamo-pituitary-adrenocortical function during acute and chronic stress. Ann N Y Acad Sci. 2008;1148:64–73. | |
Slominski A, Pisarchik A, Tobin DJ, Mazurkiewicz JE, Wortsman J. Differential expression of a cutaneous corticotropin-releasing hormone system. Endocrinology. 2004;145(2):941–950. | |
Hsu SY, Hsueh AJ. Human stresscopin and stresscopin-related peptide are selective ligands for the type 2 corticotropin-releasing hormone receptor. Nat Med. 2001;7(5):605–611. | |
Grammatopoulos DK, Chrousos GP. Functional characteristics of CRH receptors and potential clinical applications of CRH-receptor antagonists. Trends Endocrinol Metab. 2002;13(10):436–444. | |
Slominski A, Szczesniewski A, Wortsman J. Liquid chromatography-mass spectrometry detection of corticotropin-releasing hormone and proopiomelanocortin-derived peptides in human skin. J Clin Endocrinol Metab. 2000;85(10):3582–3588. | |
Teofoli P, Motoki K, Lotti TM, Uitto J, Mauviel A. Propiomelanocortin (POMC) gene expression by normal skin and keloid fibroblasts in culture: modulation by cytokines. Exp Dermatol. 1997;6(3):111–115. | |
Ito N, Ito T, Kromminga A, et al. Human hair follicles display a functional equivalent of the hypothalamic-pituitary-adrenal axis and synthesize cortisol. FASEB J. 2005;19(10):1332–1334. | |
Foitzik K, Krause K, Nixon AJ, et al. Prolactin and its receptor are expressed in murine hair follicle epithelium, show hair cycle-dependent expression, and induce catagen. Am J Pathol. 2003;162(5):1611–1621. | |
Langan EA, Foitzik-Lau K, Goffin V, Ramot Y, Paus R. Prolactin: an emerging force along the cutaneous-endocrine axis. Trends Endocrinol Metab. 2010;21(9):569–577. | |
Ramot Y, Bíró T, Tiede S, et al. Prolactin – a novel neuroendocrine regulator of human keratin expression in situ. FASEB J. 2010;24(6):1768–1779. | |
Slominski A, Tobin DJ, Zmijewski MA, Wortsman J, Paus R. Melatonin in the skin: synthesis, metabolism and functions. Trends Endocrinol Metab. 2008;19(1):17–24. | |
Weihe E, Schütz B, Hartschuh W, Anlauf M, Schäfer MK, Eiden LE. Coexpression of cholinergic and noradrenergic phenotypes in human and nonhuman autonomic nervous system. J Comp Neurol. 2005;492(3):370–379. | |
Joachim RA, Kuhlmei A, Dinh QT, et al. Neuronal plasticity of the “brain-skin connection”: stress-triggered up-regulation of neuropeptides in dorsal root ganglia and skin via nerve growth factor-dependent pathways. J Mol Med (Berl). 2007;85(12):1369–1378. | |
Levi-Montalcini R, Skaper SD, Dal Toso R, Petrelli L, Leon A. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci. 1996;19(11):514–520. | |
Peters EM, Botchkarev VA, Botchkareva NV, Tobin DJ, Paus R. Hair-cycle-associated remodeling of the peptidergic innervations of murine skin, and hair growth modulation byneuropeptides. J Invest Dermatol. 2001;116(2):236–245. | |
Urpe M, Buggiani G, Lotti T. Stress and psychoneuroimmunologic factors in dermatology. Dermatol Clin. 2005;23(4):609–617. | |
Hochman B, Furtado F, Isoldi FC, Nishioka MA, Ferreira LM. Psychological stress and skin wound healing: new highlights. In: Cavalcanti L, Azevedo S, editors. Psychology of Stress. New York, NY: Nova Science Publishers, Inc.; 2013:1–48. Available from: https://www.novapublishers.com/catalog/product_info.php?products_id=42015&osCsid=a79df5592f9331df1e8f8cf43e6796c1. Accessed September 24, 2014. | |
Akaishi S, Ogawa R, Hyakusoku H. Keloid and hypertrophic scar: neurogenic inflammation hypotheses. Med Hypotheses. 2008;71(1):32–38. | |
Yagmur C, Akaishi S, Ogawa R, Guneren E. Mechanical receptor-related mechanisms in scar management: a review and hypothesis. Plast Reconstr Surg. 2010;126(2):426–434. | |
Ogawa R. Mechanobiology of scarring. Wound Repair Regen. 2011; 19 Suppl 1:s2–s9. | |
Ogawa R, Hsu CK. Mechanobiological dysregulation of the epidermis and dermis in skin disorders and in degeneration. J Cell Mol Med. 2013;17(7):817–822. | |
Kadanoff D. Neurotization and innervation of the human scar-keloids. Z Haut Geschlechtskr. 1969;44(21):925–930. | |
Hochman B, Nahas FX, Sobral CS, et al. Nerve fibres: a possible role in keloid pathogenesis. Br J Dermatol. 2008;158(3):651–652. | |
Parkhouse N, Crowe R, McGrouther DA, Burnstock G. Painful hypertrophic scarring and neuropeptides. Lancet. 1992;340(8832):1410. | |
Crowe R, Parkhouse N, McGrouther D, Burnstock G. Neuropeptide-containing nerves in painful hypertrophic human scar tissue. Br J Dermatol. 1994;130(4):444–452. | |
Zhang LQ, Laato M. Innervation of normal and hypertrophic human scars and experimental wounds in the rat. Ann Chir Gynaecol. 2001(215);90:29–32. | |
Tey HL, Maddison B, Wang H, et al. Cutaneous innervation and itch in keloids. Acta Derm Venereol. 2012;92(5):529–531. | |
Zhibo X, Miaobo Z. Intralesional botulinum toxin type A injection as a new treatment measure for keloids. Plast Reconstr Surg. 2009;124(5):275e–277e. | |
Uyesugi B, Lippincott B, Dave S. Treatment of a painful keloid with botulinum toxin type A. Am J Phys Med Rehabil. 2010;89(2):153–155. | |
Robinson AJ, Khadim MF, Khan K. Keloid scars and treatment with botulinum toxin type A: the Belfast experience. J Plast Reconstr Aesthet Surg. 2013;66(3):439–440. | |
Wilson AM. Eradication of keloids: surgical excision followed by a single injection of intralesional 5-fluorouracil andbotulinum toxin. Can J Plast Surg. 2013;21(2):87–91. | |
Kim LR, Whelpdale K, Zurowski M, Pomeranz B. Sympathetic denervation impairs epidermal healing in cutaneous wounds. Wound Repair Regen. 1998;6(3):194–201. | |
Scholzen TE, Brzoska T, Kalden DH, et al. Effect of ultraviolet light on the release of neuropeptides and neuroendocrine hormones in the skin: mediators of photodermatitis and cutaneous inflammation. J Investig Dermatol Symp Proc. 1999;4(1):55–60. | |
Louw L. The keloid phenomenon: progress toward a solution. Clin Anat. 2007;20(1):3–14. | |
Meeran SM, Singh T, Nagy TR, Katiyar SK. High-fat diet exacerbates inflammation and cell survival signals in the skin of ultraviolet B-irradiated C57BL/6 mice. Toxicol Appl Pharmacol. 2009;241(3):303–310. | |
Ferreira AC, Hochman B, Furtado F, Bonatti S, Ferreira LM. Keloids: a new challenge for nutrition. Nutr Rev. 2010;68(7):409–417. | |
Dhabhar FS. Psychological stress and immunoprotection versus immunopathology in the skin. Clin Dermatol. 2013;31(1):18–30. | |
Hochman B, Vilas Bôas FC, Mariano M, Ferreiras LM. Keloid heterograft in the hamster (Mesocricetus auratus) cheek pouch, Brazil. Acta Cir Bras. 2005;20(3):200–212. | |
Nirodi CS, Devalaraja R, Nanney LB, et al. Chemokine and chemokine receptor expression in keloid and normal fibroblasts. Wound Repair Regen. 2000;8(5):371–382. | |
Wilke K, Martin A, Terstegen L, Biel SS. A short history of sweat gland biology. Int J Cosmetic Sci. 2007;29(3):169–179. | |
Zouboulis CC. The sebaceous gland. Hautarzt. 2010;61(6):467–468. | |
Luger TA, Lotti T. Neuropeptides: role in inflammatory skin diseases. J Eur Acad Dermatol Venereol. 1998;10(3):207–211. | |
Harvima IT, Nilsson G, Naukkarinen A. Role of mast cells and sensory nerves in skin inflammation. G Ital Dermatol Venereol. 2010;145(2):195–204. | |
Harvima IT, Nilsson G. Stress, the neuroendocrine system and mast cells: current understanding of their role in psoriasis. Expert Rev Clin Immunol. 2012;8(3):235–241. | |
Lee YS, Vijayasingam S. Mast cells and myofibroblasts in keloid: a light microscopic, immunohistochemical and ultrastructural study. Ann Acad Med Singapore. 1995;24(6):902–905. | |
Shaker SA, Ayuob NN, Hajrah NH. Cell talk: a phenomenon observed in the keloid scar by immunohistochemical study. Appl Immunohistochem Mol Morphol. 2011;19(2):153–159. | |
Messadi DV, Le A, Berg S, Huang G, Zhuang W, Bertolami CN. Effect of TGF-beta 1 on PDGF receptors expression in human scar fibroblasts. Front Biosci. 1998;3:a16–a22. | |
Fujiwara M, Muragaki Y, Ooshima A. Upregulation of transforming growth factor-beta1 and vascular endothelial growth factor in cultured keloid fibroblasts: relevance to angiogenic activity. Arch Dermatol Res. 2005;297(4):161–169. | |
Campaner AB, Ferreira LM, Gragnani A, Bruder JM, Cusick JL, Morgan JR. Upregulation of TGF-beta1 expression may be necessary but is not sufficient for excessive scarring. J Invest Dermatol. 2006; 126(5):1168–1176. | |
Abdou AG, Maraee AH, Al-Bara AM, Diab WM. Immunohistochemical expression of TGF-β1 in keloids and hypertrophic scars. Am J Dermatopathol. 2011;33(1):84–91. | |
Kikuchi K, Kadono T, Takehara K. Effects of various growth factors and histamine on cultured keloid fibroblasts. Dermatology. 1995; 190(1):4–8. | |
McCauley RL, Chopra V, Li YY, Herndon DN, Robson MC. Altered cytokine production in black patients with keloids. J Clin Immunol. 1992;12(4):300–308. | |
Ghazizadeh M, Tosa M, Shimizu H, Hyakusoku H, Kawanami O. Functional implications of the IL-6 signaling pathway in keloid pathogenesis. J Invest Dermatol. 2007;127(1):98–105. | |
Ghazizadeh M. Essential role of IL-6 signaling pathway in keloid pathogenesis. J Nippon Med Sch. 2007;74(1):11–22. | |
Uitto J. IL-6 signaling pathway in keloids: a target for pharmacologic intervention? J Invest Dermatol. 2007;127(1):6–8. | |
Eishi K, Bae SJ, Ogawa F, Hamasaki Y, Shimizu K, Katayama I. Silicone gel sheets relieve pain and pruritus with clinical improvement of keloid: possible target of mast cells. J Dermatolog Treat. 2003;14(4):248–252. | |
Furtado F, Hochman B, Farber PL, Muller MC, Hayashi LF, Ferreira LM. Psychological stress as a risk factor for postoperative keloid recurrence. J Psychosom Res. 2012;72(4):282–287. | |
Durani P, Bayat A. Levels of evidence for the treatment of keloid disease. J Plast Reconstr Aesthet Surg. 2008;61(1):4–17. | |
Gauglitz GG. Management of keloids and hypertrophic scars: current and emerging options. Clin Cosmet Investig Dermatol. 2013;6:103–114. | |
Alonso PE, Rioja LF, Pera C. Keloids: a viral hypothesis. Med Hypotheses. 2008;70:156–166. | |
de Mesquita CJ. About strawberry, crab claws, and the Sir James Black’s invention. Hypothesis: can we battle keloids with propranolol? Med Hypotheses. 2010;74:353–359. | |
Huang C, Akaishi S, Hyakusoku H, Ogawa R. Are keloid and hypertrophic scar different forms of the same disorder? A fibroproliferative skin disorder hypothesis based on keloid findings. Int Wound J. Epub November 22, 2012. | |
Dong X, Mao S, Wen H. Upregulation of proinflammatory genes in skin lesions may be the cause of keloid formation [review]. Biomed Rep. 2013;1(6):833–836. | |
Shih B, Bayat A. Genetics of keloid scarring. Arch Dermatol Res. 2010;302(5):319–339. | |
Halim AS, Emami A, Salahshourifar I, Kannan TP. Keloid scarring: understanding the genetic basis, advances, and prospects. Arch Plast Surg. 2012;39(3):184–189. | |
Burd A, Huang L. Hypertrophic response and keloid diathesis: two very different forms of scar. Plast Reconstr Surg. 2005;116(7):150e–157e. | |
Nirodi CS, Devalaraja R, Nanney LB, et al. Chemokine and chemokine receptor expression in keloid and normal fibroblasts. Wound Repair Regen. 2000;8(5):371–382. | |
Zhang Q, Yamaza T, Kelly AP, et al. Tumor-like stem cells derived from human keloid are governed by the inflammatory niche driven by IL-17/IL-6 axis. PLoS One. 2009;4(11):e7798. | |
Qu M, Song N, Chai G, Wu X, Liu W. Pathological niche environment transforms dermal stem cells to keloid stem cells: a hypothesis of keloid formation and development. Med Hypotheses. 2013;81(5):807–812. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.