Back to Journals » Transplant Research and Risk Management » Volume 11
Circulating nucleic acids as biomarkers for allograft injury after solid organ transplantation: current state-of-the-art
Authors Pattar SK, Greenway SC
Received 5 February 2019
Accepted for publication 23 April 2019
Published 27 June 2019 Volume 2019:11 Pages 17—27
DOI https://doi.org/10.2147/TRRM.S204233
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Qing Yi
Sabrina K Pattar,1 Steven C Greenway1–3
1Department of Biochemistry and Molecular Biology, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada; 2Department of Pediatrics and Alberta Children’s Hospital Research Institute, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada; 3Department of Cardiac Sciences and Libin Cardiovascular Institute of Alberta, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada
Abstract: Immune-mediated injury of a transplanted organ can lead to allograft dysfunction and even patient death. Acute cellular rejection typically occurs within the first months post-transplantation but patients are at life-time risk, particularly if there is medication non-compliance or reduction of immunosuppression due to complications. Therefore, safe and accurate monitoring of the donated organ for signs of rejection is essential for long-term survival of the transplanted organ and recipient. The current gold standard for rejection surveillance is through tissue biopsy and histology, which is costly, invasive, and subjective. Thus, efforts to develop non-invasive methods for the detection of rejection post-transplantation are a priority in the field. The first FDA-approved non-invasive assay, AlloMap, was developed in 2006 and monitored the peripheral expression of 11 genes associated with immune system activation. More recently, there has been a shift towards interrogating the status of the transplanted organ directly. Fragments of genomic DNA are released into the blood during cellular apoptosis and levels of cell-free DNA (cfDNA) have been shown to be elevated in the presence of organ injury, including after transplantation. Since the genomic characteristics of DNA are maintained in cfDNA (eg, sequence variants), this circulating molecule represents a promising organ-specific biomarker for allograft injury. DNA sequence variants have been used to distinguish donor and recipient cfDNA with or without a priori donor genotyping in a variety of solid organs post-transplant. Current research has established the groundwork and future multi-center trials will determine if this novel molecular diagnostic tool represents a viable alternative to tissue biopsy. Other nucleic acid molecules released from the transplanted organ (eg, microRNAs) are presently less well developed in comparison to cfDNA but may also represent potential novel biomarkers. This review summarizes current literature and evaluates the promises and pitfalls of circulating nucleic acids as biomarkers for allograft injury post-transplant.
Keywords: cell-free DNA, biomarkers, transplantation, rejection, microRNAs, gene expression
Introduction
Immune-mediated injury of a transplanted organ is a serious problem, as rejection can lead to allograft dysfunction and, in severe cases, patient death. Acute cellular rejection (ACR) most often occurs within the first 6 months post-transplant.1 This form of rejection involves the accumulation of mononuclear cells, specifically CD4+ and CD8+ T-cells, in the interstitial space of the allograft as a result of antigens on the donated organ being identified as foreign to the recipient.1 These T-cells ultimately initiate an immune cascade that leads to the apoptosis of the targeted cells.1 As these cells die, the nucleic acids within become fragmented, resulting in approximately 120–160 base pair (bp) pieces of double-stranded cell-free DNA (cfDNA) that are released into the blood and ultimately excreted in the urine.2
Circulating nucleic acids, including cfDNA, have been leveraged as diagnostic tools to replace invasive biopsies in other areas of medicine. For example, the detection of fetal cfDNA in maternal blood has been used to detect genetic abnormalities in the fetus, thereby replacing highly invasive amniocentesis, and has seen rapid commercialization and clinical uptake.3,4 Another common use of cfDNA has been in oncology, since the cells that comprise the tumor are known to release fragments of genomic DNA into the circulation.5,6 As such, “liquid biopsies” can be employed to isolate cfDNA (derived from both normal and tumor tissue) from serum or plasma, and then interrogated using massively-parallel sequencing to identify the presence of the cancer-causing mutation(s) and enable diagnosis or monitor response to chemotherapy.5,7,8 Whereas the clinical scenarios of transplantation, pregnancy, and cancer involve the presence of two different populations of cfDNA derived from distinct sources, the recognition that epigenetic changes (ie DNA methylation) also remain intact in fragments of cfDNA has expanded the use of cfDNA to become a biomarker capable of identifying tissue-specific injury.2 Since cfDNA can be obtained from a simple blood draw, its concentration within the serum or plasma accurately measured and its nucleotide sequence or modifications determined, cfDNA is an excellent candidate for a non-invasive biomarker for multiple human diseases including rejection after solid organ transplantation.
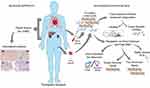
Frequent and accurate monitoring of allograft health is essential for long-term survival of the transplant recipient. For heart transplantation (HT), the current gold standard for diagnosing rejection is the endomyocardial biopsy (EMB).9 However, this procedure is expensive and has significant limitations (Table 1), many of which are common to all organ or tissue biopsies.10,11 In addition, the invasive nature of the biopsy may lead to potential complications, such as puncture of the adjacent carotid artery during catheter insertion, cardiac perforation with tamponade, pneumothorax, damage to the tricuspid valve, air embolism, atrial arrhythmias, and prolonged bleeding.11–13 Alternative, non-invasive diagnostic methods would permit easier, safer, and possibly even more frequent monitoring of the transplanted organ, which might allow for earlier detection of rejection and the implementation of appropriate treatment. Unfortunately, current non-invasive methods, including echocardiography and cardiac magnetic resonance imaging, lack adequate specificity and sensitivity.14–17 Blood-based biomarkers currently represent the “holy grail” for a minimally-invasive approach for the detection of rejection and, recently, circulating nucleic acids isolated from the plasma or urine have shown promise and have even been implemented into clinical practice (Figure 1).18–21
 |
Table 1 Advantages and disadvantages of diagnostic methods for the detection of rejection after solid organ transplantation |
Transcriptomics-based molecular diagnostics
AlloMap
Substantial effort has been made to develop non-invasive assays that could replace or reduce the use of EMB, especially for patients who are asymptomatic or who are >1 year post-transplant and in whom the likelihood of finding significant rejection is very low. In these patients, the probability of biopsy-related complications, although low, is more likely than the detection of subclinical allograft rejection requiring treatment. Perhaps logically, initial efforts focused on monitoring the recipient’s immune response and gene-expression profiling of peripheral blood mononuclear cells (PBMCs) was used to detect the presence of rejection.22 More specifically, an assay that utilized 11 informative genes was developed to discriminate between the absence of rejection (International Society of Heart and Lung Transplantation (ISHLT) grade 0R) and moderate-to-severe rejection (ISHLT grade ≥2R) non-invasively.22 By combining information from the literature regarding pathways involved in immune activation, recruitment, and mobilization during rejection, bioinformatics, and publicly-available cDNA libraries of stimulated and resting leukocytes, a total of 7,370 genes were represented on a custom microarray. Analysis of this microarray was conducted using patient PBMC samples, whereby 252 candidate genes were identified and subjected to real-time PCR (RT-PCR) using samples from patients with and without a confirmed episode of rejection. Overall, four individual genes (ARHU, PDCD1, ITGA4, and SEMA7A) and three aggregates of gene expression (ITGAM, FLT3, and IL1R2; G6B and PF4; WDR40A and MIR) provided a total of 11 gene classifiers that could be used to effectively distinguish between rejection and quiescent samples. The majority of these genes were associated with T-cell production, activation, and mobilization, which is consistent with their role in driving allograft rejection. Lastly, through the use of a linear discriminant equation, a score between 0 and 40 was assigned to each sample based on gene expression, where a score ≥20 indicated the presence of rejection. This work resulted in the AlloMap test, the first FDA-approved, non-invasive method for the detection of rejection following cardiac transplantation that showed sufficient correlation with results obtained from an EMB. Follow-up clinical studies have been conducted to systematically confirm the effectiveness of this assay in comparison to the EMB.
The Invasive Monitoring Attenuation through the Gene Expression (IMAGE) trial conducted by Stanford University gathered 602 heart transplant recipients from 13 US centers between January 2005 and October 2009.23 It should be noted that participants had received a heart transplant between 6 months and 5 years prior to the study and were randomized to either undergo routine EMB or be monitored using the AlloMap assay in a 1:1 ratio.23 On average, the participants were 54 years of age, and the majority of the study group were caucasian males.23 The IMAGE trial defined the primary outcome as the first occurrence of rejection with hemodynamic compromise and/or graft dysfunction due to other causes, death, or retransplantation. Secondary outcomes included death from any cause, the number of biopsies performed, and biopsy-related complications.23 In addition to this, information regarding the participants’ quality-of-life and satisfaction with their assigned monitoring method was also collected. It was found that patients in the gene-profiling group were more satisfied with the non-invasive protocol than those in the biopsy group, and the level of satisfaction increased in the second year, while the scores in the biopsy group remained lower and consistent over the same 2-year period.23 Notably, a total of 34 episodes of rejection were detected in the gene-profiling group out of 1,190 serum samples obtained and analyzed throughout the trial.23 In comparison, 47 episodes of rejection were observed out of the 1,249 EMBs performed in the biopsy group over the course of the study.23 In the gene-profiling group, six of the 34 rejection events were treated as a result of a gene-expression score >30, which warranted EMB confirmation. In the biopsy group, 22 treated episodes of rejection were asymptomatic and were detected on routine biopsy alone.23 Overall, the use of the AlloMap test, in conjunction with clinical observation and echocardiograms, safely reduced the number of biopsies performed without increasing the risk of serious cardiovascular events.23 However, more recently there has been a shift from monitoring the recipient’s immune response to approaches that directly interrogate the health of the donated organ, including the use of organ-derived circulating nucleic acids.
Circulating messenger RNAs
Much like the strategy employed for the development of AlloMap to non-invasively detect rejection in heart transplant recipients, changes in the expression of specific genes have been identified to detect primary graft dysfunction (PGD) in lung transplant recipients.24 A multicenter prospective cohort study, conducted between 2008 and 2010, across three US transplant centers (the University of Pennsylvania, Columbia University, and the University of Wisconsin) enrolled 106 lung transplant recipients between the ages of 16–70.24 PGD was defined as grade 3 rejection within the first 72 hours post-transplant, and the expression of 100 genes typically associated with an immune response were assessed in each patient.24 Blood samples were drawn from all 106 participants at 2 hours, 1 week, 6 weeks, 3 months, 6 months, and 1 year post-transplant. Of these 106 lung-transplant recipients, 24 patients met the criteria for PGD.24
Notably, within the patients who experienced PGD, 18 genes were found to be differentially expressed over the course of the first year, with the majority of these genes being involved in both innate and adaptive immunity.24 Furthermore, 11 genes, including nod-like receptor family, pyrin domain-containing 3 (NLRP3), were overexpressed in patients with PGD, compared to those without, within 2 hours after transplantation.24 After the first week post-transplant, patients with PGD displayed nine overexpressed genes, which included toll-like receptor-4 and -2 (TLR4 and TLR2), as well as nod-like receptor family, caspase recruitment domain-containing 4 and 3 (NLRC4 and NLRP3).24 Overall, these levels of expression indicated that genes encoding innate immune and inflammasome-related proteins were highly represented during this early period of rejection. In contrast to this, 11 genes were underexpressed in patients with PGD, compared to those without, as early as 2 hours after reperfusion, most of which were associated with T-cell regulation.24 Of note, FOXP3, a master regulator for regulatory T-cell development and function, as well as Fas ligand (FASLG), an apoptosis trigger essential for adaptive immune regulation, were underexpressed within 2 hours of reperfusion and remained differentially expressed at 7 days post-transplant.24 Thus, overexpression of genes associated with innate immune and inflammasome activation, as well as suppression of T-cell regulatory responses, may act as a strong indicator for PGD within the first week post-lung transplant and would, therefore, necessitate further measures to ensure survival of the allograft and recipient.
The examination of changes in gene expression, based on levels of mRNAs in plasma and urine, has also been implemented for the non-invasive detection of acute rejection in patients who have received a kidney transplant. Of note, T-cell immunoglobulin and mucin domain 3 (TIM-3), which has been found to play a role in both alloimmune and autoimmune responses, ultimately dictates tolerance induction, and, therefore, graft survival, in kidney transplantation.25 In addition to TIM-3, kidney injury molecule-1 (KIM-1) has been found to be highly expressed in epithelial cells of an injured proximal tubule, which makes it an excellent candidate for a kidney-specific biomarker.25 One such study investigated both urinary and blood TIM-3 mRNA expressions, urinary KIM-1 mRNA expression, as well as urinary and serum KIM-1 proteins in 85 renal allograft recipients diagnosed with ACR (n=24), chronic allograft dysfunction (CAD) (n=19), and those with stable transplants (n=42).25 Overall, it was found that patients that experienced either ACR or CAD patients had significantly greater urinary and blood TIM-3 mRNA expressions, urinary KIM-1 mRNA expression, and urinary and serum KIM-1 protein levels, in comparison to stable kidney transplant recipients and healthy controls.25 Interestingly, patients with elevated levels of both TIM-3 and KIM-1 mRNA, as well as corresponding protein levels, in the serum and urine tended to also display a lower glomerular filtration rate (GFR).25 Furthermore, while TIM-3 mRNA levels declined in the plasma and urine of patients who experienced ACR, following anti-rejection therapy, these levels have been found to remain consistently high in CAD patients.25,26 Therefore, the level of expression of TIM-3 can be used to non-invasively identify patients who may have surpassed ACR and are now experiencing CAD. In addition to this, persistently elevated levels of KIM-1 has been associated with graft loss within 5 years of transplantation, which indicates the predictive value of this potential non-invasive biomarker.25
Circulating organ-specific microRNAs
MicroRNAs (miRNAs) are a class of small non-coding RNAs which play an integral role in the regulation of gene expression and the modulation of many cellular processes through post-transcriptional suppression.27 Changes in the tissue expression of miRNAs have been linked to pathological conditions in humans including malignant,28 infectious,29 autoimmune,30,31 renal,32 and cardiovascular diseases.33 Furthermore, miRNAs may be passively released from dying cells encapsulated in apoptotic bodies that effectively protect the miRNA from immediate degradation in the circulation.19,34 Therefore, the potential use of these endogenous circulating miRNAs, which are highly stable in blood and resistant to temperature changes, as diagnostic markers for the detection of rejection following transplantation has gained a great deal of attention.19,34 Circulating miRNAs as a non-invasive biomarker for the detection of immune-mediated rejection following transplantation were first evaluated using animal models,35–39 but more recent testing using human samples has helped further our understanding of the kinetics and role of circulating human organ-specific miRNAs during acute rejection events.
MicroRNAs have been found to also be detectable in urine and can, therefore, serve as potential biomarkers of acute rejection in humans after kidney transplantation.40–44 In one study, urinary miRNAs of stable transplant patients and transplant patients with acute rejection were profiled, which revealed a strong dysregulation of miR‐210 expression in the urine of patients presenting with acute rejection.40 More specifically, miR‐210 expression was downregulated in patients with acute rejection in comparison to controls.40 Interestingly, low miR‐210 levels were significantly associated with a decline in GFR shortly after transplantation compared to levels observed in stable patients with and without urinary tract infections.40 Thus, miR-210 has been considered a strong potential non-invasive blood biomarker for the detection of acute rejection and kidney failure following renal transplantation. In addition to the downregulation of miR-210, an upregulation of miR-150, miR-155, miR-663, and miR-638 has also been observed in the serum of patients during an acute rejection event.44
In terms of liver transplantation, although several differentially-expressed liver-specific miRNAs associated with rejection have been identified,45 it has been noted that human hepatocytes express large amounts of miR-122.46–48 This miRNA plays a pivotal role in the regulation of cholesterol metabolism, in conjunction with a more widely expressed miRNA, miR-33, as well as in iron homeostasis, alongside the broadly expressed miR-210.46,47,49,50 Due to its generally high expression, miR-122 can be detected in serum and tends to be present in high concentrations in patients with viral, alcoholic, or chemical-related hepatotoxicity.46,48 In one study, serum samples were obtained from 12 healthy controls and 43 liver transplant recipients, 13 of which displayed histologically-proven ACR.46 Liver-specific miRNAs in the serum of liver transplant recipients experiencing an episode of acute rejection were analyzed and levels of miR-122 were found to be significantly elevated during rejection.46 On average, a 9-fold increase was observed at the time of a rejection episode when compared to the levels of miR-122 observed 6 months later following the resolution of ACR.46 In addition to this, miR-122 showed higher sensitivity compared to other biomarkers of hepatic injury, as the rise in levels preceded that of the aminotransferases.46
The use of circulating miRNAs as potential non-invasive biomarkers for immune-mediated injury has also been demonstrated for cardiac transplantation.20,50–54 One study compared 30 patients with acute biopsy-proven rejection to 30 matched control patients without rejection.20 Overall, four cardiac miRNAs; miR-10a, miR-31, miR-92a, and miR-155, were found to be differentially expressed in the serum of patients experiencing ACR and peripheral blood levels strongly correlated with levels observed within cardiac tissue.20 Interestingly, miR-10a, miR-155, miR-31, and miR-92a were also found to play a role in initiating a myocardial immune response which could account for their differential expression during an ACR event.20 Therefore, these miRNAs, which are both cardiac-specific and associated with an immune response, have the potential for being robust non-invasive biomarkers for those experiencing ACR. However, further prospective studies will have to be conducted to better understand the kinetics of these miRNAs and establish threshold levels associated with rejection.
Circulating long non-coding RNAs
In contrast to miRNAs, long non-coding RNAs (lncRNAs) are characterized as being greater than 200 nucleotides in length.55 There is an emerging understanding of the role of these nucleic acids in not only regulating the genome and proteome, but also the potential utility as a biomarker for acute organ injury, and by extension could predict organ survival.55,56 These lncRNAs have been found to be detectable in plasma samples taken from patients who have experienced acute kidney injury.56 However, there has been a shift to detect these nucleic acids in urine, as it is easily accessible and can detect any changes occurring within the kidneys that may be attributed to ACR.57 One such study employed 62 patients with biopsy-proven ACR and 31 stable, control, patients with no evidence of rejection.56,57 Overall, ACR was detected in 20 urine samples at 6 weeks, 28 urine samples at 3 months, and 14 urine samples at 6 months after kidney transplantation.56,57 This indicated that lncRNAs are in fact stable in urine, and can, therefore, be feasibly isolated, detected, and function as potential biomarkers for ACR in transplanted kidneys. Interestingly, three intergenic lncRNAs, LNC-MYH13-3:1 (L321), RP11–395P13.3–001 (L327), and RP11-354P17.15–001 (L328), were found to be differentially expressed when comparing patients with ACR, and those with no evidence of rejection. L327 and L328 were both found to be upregulated in patients with ACR, when compared with stable kidney transplant recipients. It should be noted that, after successful antirejection therapy, the levels of L328 normalized in patients with ACR. However, the upregulation of L328 was also associated with a greater decline in GFR 1 year after transplantation.57 Therefore, L328 may be an excellent candidate for a non-invasive biomarker of acute cell mediated rejection in kidneys, and has predicative value for determining the likelihood of long-term graft function.
Recipient- vs donor-derived cell-free DNA
Donor-recipient sex-mismatch
Since the presence of cfDNA in the plasma is due to the natural process of apoptosis, all individuals have some detectable levels of cfDNA in their blood.57,58 For healthy individuals, the majority of the cfDNA detected in the plasma is derived from hematopoietic cells.57,58 The use of circulating nucleic acids, such as cfDNA, as a biomarker for rejection has several advantages, as it is derived directly from the cells of the donated organ that have been injured by the cytotoxic immune response. In addition to this, cfDNA can be easily obtained through a simple and minimally-invasive blood draw or urine sample. Furthermore, cfDNA maintains all of the genetic features of genomic DNA, thereby allowing for differentiation between genetic material released from the donated organ and cfDNA derived from the recipient’s cells undergoing routine apoptosis.58 In cases where the organ donor is male and the recipient is female, this sex-mismatch can be leveraged in order to identify and quantify donor-derived cfDNA (dd-cfDNA).21 As such, injury to the donated organ as a result of rejection is expected to lead to an increase in the copy number of cfDNA derived from the donor’s Y chromosome.21 This principle was demonstrated in urine samples taken from sex-mismatched female renal transplant recipients, whereby patients who had experienced rejection had elevated levels of dd-cfDNA in their urine, specifically containing regions found within the Y chromosome, as compared to patients without rejection.21 Although this approach allows for confident assessment of rejection in the allograft, sex-mismatch between the donor and recipient is a relatively uncommon event and, therefore, not broadly applicable in transplantation. Methodology that relies upon identifying differences between the donor and recipient cfDNA based on single nucleotide polymorphisms (SNPs) in cfDNA would be more universally applicable.
Genome transplant dynamics – “two genomes” model
Given that an organ transplant is also a genome transplant, the concept of genome transplant dynamics (GTD) relies on informative genetic differences between the donor and recipient whereby, at a particular locus, the recipient ideally is homozygous for a single base (eg, AA) and the donor is homozygous for a different base (eg, GG).58,59 Given the genetic heterogeneity between individuals, this potentially amounts to tens of thousands of potentially useful loci across the genome that could be used to discriminate dd-cfDNA from recipient cfDNA. Thus, high-throughput sequencing of cfDNA allows for the identification of a unique donor “genetic fingerprint”. This donor-specific signature can be used to calculate the quantity of dd-cfDNA present within the recipient circulation.58,59 This concept was first illustrated using previously-collected blood samples and banked splenocytes from the donor to determine the genotype for each donor-recipient pairing.59 After sequencing the purified cfDNA, the percentage of donor-specific molecules was determined based on the total number of informative bases. In samples taken at the time of a biopsy-proven rejection event, the percentage of donor-specific SNPs was increased, while the rate of errors due to sequencing and incorrect genotyping information remained unchanged.59 In addition to this, it was also noted that the percentage of dd-cfDNA within the recipient’s circulation in the absence of rejection was relatively negligible (<1%).59 However, this percentage increased to >3–4% during, or even immediately preceding, biopsy-proven rejection, which represented a significant increase in the amount of dd-cfDNA within the recipient’s blood.59
Since the establishment of the GTD assay, prospective studies have been conducted to validate this approach. Both pediatric and adult heart and lung transplant patients were recruited, and the genotypes of the donors and recipients were determined using whole-genome sequencing (WGS).60 Over the course of the study, serial plasma samples were collected and, upon sequencing the isolated cfDNA, based on the previously-determined SNP differences between the donor-recipient pairs, an average of 53,423 informative SNP markers were available per pairing.60 Overall, the predictive capacity of this assay was superior to that of the AlloMap test, and often showed early detection of acute rejection.60 However, despite the accuracy of the GTD assay, important limitations are the requirement for a priori knowledge of the donor and recipient’s genotypes and the requirement for WGS. While the recipient’s genetic information can be easily obtained, this is not always true for the donor and WGS for each donor-recipient pair is costly, labor intensive, and time-consuming. Thus, alternative strategies have been developed to address these weaknesses.
“Single genome” model
An alternative “single-genome” method utilizes all genotyped SNPs as opposed to relying only on those homozygous SNPs that differ between the recipient and donor.61 Therefore, a priori knowledge of the donor’s specific genotype is not required in order to determine the proportion of dd-cfDNA within the circulation of the recipient.61 Due to the fact that heart and lung transplants do not typically occur between closely related individuals (unlike kidney and liver), the developed algorithm for heart and lung transplants assumes that the donor genotype is randomly selected from a human population.60 Based on this, the probability of a specific donor allele is assumed to be its frequency in the population, as determined from databases such as the 1,000 Genomes Project.60 The performance of this model was assessed by directly comparing the estimated levels of dd-cfDNA to levels determined using both the donor and recipient’s genotypes, and it was found that the two algorithms were highly comparable for lung transplantation.61 However, for heart transplant recipients, the estimated levels of dd-cfDNA were highly correlated, but not as strongly as was seen in the lung cohort.61 This may be due to lower levels of dd-cfDNA in the heart cohort, which made the estimation more difficult. It should be noted that the predictive value of this assay was greatly reduced when the abundance of dd-cfDNA fell below 0.2% or exceeded 25%, which prevented the application of this assay for the detection of rejection in liver and bone marrow transplants.61,62 Thus, further improvements to these algorithms are likely required to increase the sensitivity and specificity for the detection of dd-cfDNA within the circulation of transplant recipients, especially for patients who have recently received a transplant when the levels of circulating cfDNA are known to be elevated.
The requirement of WGS for both the donor and recipient can not only be an unrealistic requirement for time-critical organ transplants from deceased donors, but it can quickly become excessively costly to perform such analysis for every donor-recipient pair. As such, an alternative approach that is rapid, does not require WGS or a priori knowledge of the donor and recipient’s genotypes, and only requires plasma collected from the transplant recipient has been developed.63 This method was used to determine the level of dd-cfDNA within the plasma of both pediatric and adult heart transplant recipients as well as a case of pediatric human liver cell transplant.63 Instead of WGS, a commercially-available panel of 124 highly polymorphic SNPs was utilized rather than interrogating the entire genome. This significantly reduced the total amount of sequencing required and, therefore, substantially reduced the sequencing and analysis time. Furthermore, in order to model the mixture of dd-cfDNA and recipient cfDNA, a novel algorithm based on the assumption that the donor and recipient would display biallelic loci was designed.63 Using this assay, dd-cfDNA was detectable after the infusion of donor hepatocytes up to 24 weeks post-transplantation, and levels of dd-cfDNA were detectable in plasma obtained from adult and pediatric heart transplant recipients.63 Notably, due to the algorithm employed, this assay is limited to circumstances where the dd-cfDNA fraction is <14%.63 Therefore, this assay would likely be best suited for the detection of rejection in stable heart transplant recipients who are >1 month post-transplant or those patients who are not lung or liver transplant recipients where the proportion of dd-cfDNA can be substantially higher.62 Also, since this assay relies upon differences in minor allele frequency between individuals, this assay would likely not be robustly accurate in the situation of closely related donor–recipient pairs, such as occurs in living-related kidney donation. Finally, this assay remains to be validated for the accurate detection of ACR.
Quantification of dd-cfDNA using digital droplet PCR
The idea of utilizing polymorphic (minor allele frequency >0.4) SNPs for the targeted quantification of dd-cfDNA has been taken one step further by applying the technology of digital droplet PCR (ddPCR). The use of ddPCR allows for the high-throughput processing of samples with small volumes and low concentrations (ie, cfDNA isolated from plasma), thereby effectively reducing the cost and required materials and time while maintaining sensitivity and specificity.64–68 As applied to transplantation, 41 highly polymorphic and informative SNPs were interrogated for the detection of dd-cfDNA in the plasma of 10 liver, nine kidney, and eight heart transplant recipients.64 Stable kidney and heart transplant recipients showed dd-cfDNA levels of 2–3%, while stable liver transplant recipients had dd-cfDNA levels around 7%.64 In the case of liver transplant recipients, it was found that failure for dd-cfDNA to fall below 15% by 10 days post-transplant or a significant rise in dd-cfDNA levels were indicative of rejection.64 Overall, this cost-effective and rapid (same-day results) test has shown promise for the accurate quantification of dd-cfDNA levels. However, it should be noted that this protocol required pre-amplification of the isolated cfDNA, which may impact the reproducibility of these findings in future clinical trials. Thus, testing of this assay in larger clinical trials may support its introduction into routine post-transplant care, and reduce the need for invasive biopsies to monitor for the development of allograft rejection.
Donor-derived cell-free DNA kinetics during quiescence and rejection
Levels of cfDNA can vary for several reasons apart from rejection, such as surgery, trauma, infection, or even exhaustive exercise.2,67 Therefore, understanding the kinetics of the release of dd-cfDNA into the recipient’s plasma is imperative for determining the likelihood of organ survival, as different solid organ transplants result in distinguishing levels of dd-cfDNA. At 1 day post-heart transplant, the level of dd-cfDNA has been observed to reach 3.8±2.3%.61 However, the level of dd-cfDNA rapidly declined within the first week and remained low in the absence of rejection, thereby displaying single-decay kinetics.61 Patients who have received a kidney transplant displayed very similar kinetics for the level of dd-cfDNA.68 In contrast to this, on the first postoperative day for bilateral lung transplants, the level of dd-cfDNA was observed to be 26±14%.62 In addition to this, the decline in dd-cfDNA displayed two-step decay kinetics, whereby the level dd-cfDNA declined rapidly within the first week but then slowed in decline and generally remained elevated throughout the post-transplant course, in comparison to levels observed in stable heart and kidney transplant recipients.61,62,68 During episodes of acute rejection in lung transplant patients, the level of dd-cfDNA spiked to 14–15% from the baseline levels of 1–3% just prior to the event.62 Similar to this, when an episode of acute rejection was experienced by a heart transplant recipient, the plasma dd-cfDNA levels jumped to 4–5%, from the baseline level of ~0.06% observed during quiescence.59
The characteristic levels of dd-cfDNA observed following each of these solid organ transplants can be attributed to the variation in tissue mass and the corresponding cell turnover rate. For example, when comparing bilateral and single-lung transplants, the rate of turnover has been observed to be, on average, 107 and 58 cells per second, respectively.61 Conversely, more than 14 days post-heart transplant, where no rejection event had been experienced, an average turnover rate of eight cells per second tended to be observed.61,69 The vast differences in cell mass and cell turnover alone contribute to the discrepancies in dd-cfDNA levels observed, even in stable solid organ transplant recipients. Thus, an understanding of the expected levels of dd-cfDNA over time associated with a given solid organ transplant not only allows for the identification of any perturbations, which may be a sign of acute rejection, but also to determine the best form of rejection monitoring. As mentioned above, current non-invasive assays require dd-cfDNA levels to not exceed 14%, which tends to be the case for lung transplant recipients for at least the first 10 days post-transplant.61,63 Therefore, designing assays that account for these elevated levels would be a point of interest for the future of non-invasive rejection monitoring.
Conclusions
Costly and invasive tissue biopsies for the detection of allograft rejection are not only associated with significant potential complications, but also lack sensitivity due to the restricted sampling area and subjective grading assessment. As such, the use of non-invasive assays that accurately assess the health of the transplanted organ as a whole could allow for safer and more frequent monitoring while improving the ability of clinicians to detect and treat significant rejection. In combination with novel assays to monitor total levels of immunosuppression (for example, based on viral diversity),71,72 personalized immunosuppression and monitoring could be realized to reduce morbidity, mortality, and improve long-term outcomes for organ transplantation. Overall, while the interrogation of the recipient’s immune response has been able to reduce the number of biopsies performed for a transplant recipient, efforts to directly examine the health of the transplanted organ appear promising. Techniques that circumvent the need for WGS and the donor genotype are likely the easiest to implement clinically, but their robust validity remains to be proven. The field of non-invasive diagnostics has expanded greatly in recent years, and research efforts may soon make the tissue biopsy redundant and obsolete.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Cornell LD, Smith RN, Colvin RB. Kidney transplantation: mechanisms of rejection and acceptance. Annu Rev Pathol Mech Dis. 2008;3:189–220. doi:10.1146/annurev.pathmechdis.3.121806.151508
2. Lehmann-Werman R, Neiman D, Zemmour H, et al. Identification of tissue-specific cell death using methylation patterns of circulating DNA. Proc Natl Acad Sci. 2016;113:E1826–E1834. doi:10.1073/pnas.1519286113
3. Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350:485–487.
4. Fan HC, Gu W, Wang J, Blumenfeld YJ, El-Sayed YY, Quake SR. Non-invasive prenatal measurement of the fetal genome. Nature. 2012;487:320–324. doi:10.1038/nature11251
5. Diaz LA, Bardelli A, Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol. 2014;32:579–586. doi:10.1200/JCO.2013.54.6911
6. Schwarzenbach H, Hoon DSB, Pantel K. Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer. 2011;11:426–437. doi:10.1038/nrc3066
7. Han X, Wang J, Sun Y. Circulating tumor DNA as biomarkers for cancer detection. Genomics Proteomics Bioinformatics. 2017;15:59–72. doi:10.1016/j.gpb.2016.10.006
8. Salvi S, Gurioli G, De Giorgi U, et al. Cell-free DNA as a diagnostic marker for cancer: current insights. Onco Targets Ther. 2016;9:6549–6559. doi:10.2147/OTT
9. Sakakibara S, Konno S. Endomyocardial biopsy. Jpn Heart J. 1962;3:537–543.
10. Khan T, Selvakumar D, Trivedi S, et al. The value of endomyocardial biopsy in diagnosis and guiding therapy. Pathology. 2017;49:750–756. doi:10.1016/j.pathol.2017.08.004
11. McMinn JF, Lang NN, McPhadden A, Payne JR, Petrie MC, Gardner RS. Biomarkers of acute rejection following cardiac transplantation. Biomark Med. 2014;8:815–832. doi:10.2217/bmm.14.56
12. Baraldi-Junkins C, Levin HR, Kasper EK, et al. Complications of endomyocardial biopsy in heart transplant patients. J Heart Lung Transplant. 1993;12:63–67.
13. Mason JW. Techniques for right and left ventricular endomyocardial biopsy. Am J Cardiol. 1978;41:887–892. doi:10.1016/0002-9149(78)90729-4
14. Kindel SJ, Hsu HH, Hussain T, et al. Multimodality noninvasive imaging in the monitoring of pediatric heart transplantation. J Am Soc Echocardiogr. 2017;30:859–870. doi:10.1016/j.echo.2016.10.014
15. Lunze FI, Colan SD, Gauvreau K, et al. Tissue Doppler imaging for rejection surveillance in pediatric heart transplant recipients. J Hear Lung Transplant. 2013;32:1027–1033. doi:10.1016/j.healun.2013.06.016
16. Greenway SC, Dallaire F, Kantor PF, et al. Magnetic resonance imaging of the transplanted pediatric heart as a potential predictor of rejection. World J Transplant. 2016;6:751–758. doi:10.5500/wjt.v6.i4.751
17. Butler CR, Savu A, Bakal JA, et al. Correlation of cardiovascular magnetic resonance imaging findings and endomyocardial biopsy results in patients undergoing screening for heart transplant rejection. J Hear Lung Transplant. 2015;34:643–650. doi:10.1016/j.healun.2014.12.020
18. Strimbu K, Tavel JA. What are biomarkers?. Curr Opin HIV AIDS. 2010;5:463–466. doi:10.1097/COH.0b013e32833ed177
19. Huo Q, Zhou M, Cooper DK, et al. Circulating miRNA or circulating DNA-Potential biomarkers for organ transplant rejection. Xenotransplantation. 2018:e12444. doi:10.1111/xen.12444
20. Duong Van Huyen J-P, Tible M, Gay A, et al. MicroRNAs as non-invasive biomarkers of heart transplant rejection. Eur Heart J. 2014;35:3194–3202. doi:10.1093/eurheartj/ehu152
21. Sigdel TK, Vitalone MJ, Tran TQ, et al. A rapid noninvasive assay for the detection of renal transplant injury. Transplant J. 2013;96:97–101. doi:10.1097/TP.0b013e318295ee5a
22. Deng MC, Eisen HJ, Mehra MR, et al. Noninvasive discrimination of rejection in cardiac allograft recipients using gene expression profiling. Am J Transplant. 2006;6:150–160. doi:10.1111/ajt.2006.6.issue-1
23. Pham MX, Teuteberg JJ, Kfoury AG, et al. Gene-expression profiling for rejection surveillance after cardiac transplantation. N Engl J Med. 2010;362:1890–1900. doi:10.1056/NEJMoa0912965
24. Diamond JM, Cantu E, Porteous MK, et al. Peripheral blood gene expression changes associated with primary graft dysfunction after lung transplantation. Am J Transplant. 2017;17:1770–1777. doi:10.1111/ajt.2017.17.issue-7
25. Shahbaz SK, Pourrezagholi F, Barabadi M, et al. High expression of TIM-3 and KIM-1 in blood and urine of renal allograft rejection patients. Transpl Immunol. 2017;43–44:11–20. doi:10.1016/j.trim.2017.07.002
26. Shahbaz SK, Barabadi M, Ahmadpour P, et al. Sequential monitoring of TIM-3 mRNA expression in blood and urine samples of renal transplant recipients. Transpl Immunol. 2018. doi:10.1016/j.trim.2018.10.007
27. Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–355. doi:10.1038/nature02871
28. Chen X, Ba Y, Ma L, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18:997–1006. doi:10.1038/cr.2008.282
29. Vasilescu C, Rossi S, Shimizu M, et al. MicroRNA fingerprints identify miR-150 as a plasma prognostic marker in patients with sepsis. PLoS One. 2009;4:e7405. doi:10.1371/journal.pone.0007405
30. Hezova R, Slaby O, Faltejskova P, et al. microRNA-342, microRNA-191 and microRNA-510 are differentially expressed in T regulatory cells of type 1 diabetic patients. Cell Immunol. 2010;260:70–74. doi:10.1016/j.cellimm.2009.10.012
31. Tang Y, Luo X, Cui H, et al. MicroRNA-146a contributes to abnormal activation of the type I interferon pathway in human lupus by targeting the key signaling proteins. Arthritis Rheum. 2009;60:1065–1075. doi:10.1002/art.v60:4
32. Wang G, Kwan BC-H, Lai F-M-M, et al. Intrarenal expression of miRNAs in patients with hypertensive nephrosclerosis. Am J Hypertens. 2010;23:78–84. doi:10.1038/ajh.2009.208
33. Nossent AY, Hansen JL, Doggen C, Quax PH, Sheikh SP, Rosendaal FR. SNPs in MicroRNA binding sites in 3′-UTRs of RAAS genes influence arterial blood pressure and risk of myocardial infarction. Am J Hypertens. 2011;24:999–1006. doi:10.1038/ajh.2011.92
34. Gilad S, Meiri E, Yogev Y, et al. Serum MicroRNAs are promising novel biomarkers. PLoS One. 2008;3:e3148. doi:10.1371/journal.pone.0003148
35. Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T. Identification of tissue-specific MicroRNAs from mouse. Curr Biol. 2002;12:735–739. doi:10.1016/S0960-9822(02)00809-6
36. Morita M, Chen J, Fujino M, et al. Identification of microRNAs involved in acute rejection and spontaneous tolerance in murine hepatic allografts. Sci Rep. 2015;4:6649. doi:10.1038/srep06649
37. Oda H, Ikeguchi R, Yurie H, et al. Plasma microRNAs are potential biomarkers of acute rejection after hindlimb transplantation in rats. Transplant Direct. 2016;2:e108. doi:10.1097/TXD.0000000000000620
38. Wei L, Wang M, Qu X, et al. Differential expression of MicroRNAs during allograft rejection. Am J Transplant. 2012;12:1113–1123. doi:10.1111/j.1600-6143.2011.03958.x
39. Van Aelst LNL, Summer G, Li S, et al. RNA profiling in human and murine transplanted hearts: identification and validation of therapeutic targets for acute cardiac and renal allograft rejection. Am J Transplant. 2016;16:99–110. doi:10.1111/ajt.13688
40. Lorenzen JM, Volkmann I, Fiedler J, et al. Urinary miR-210 as a mediator of acute T-cell mediated rejection in renal allograft recipients. Am J Transplant. 2011;11:2221–2227. doi:10.1111/ajt.2011.11.issue-10
41. Zununi Vahed S, Zonouzi AP, Ghanbarian H, Ghojazadeh M, Samadi N, Ardalan M. Upregulated expression of circulating MicroRNAs in kidney transplant recipients with interstitial fibrosis and tubular atrophy. Iran J Kidney Dis. 2017;11:309–318.
42. Bijkerk R, Florijn BW, Khairoun M, et al. Acute rejection after kidney transplantation associates with circulating MicroRNAs and vascular injury. Transplant Direct. 2017;3:e174. doi:10.1097/TXD.0000000000000699
43. Shapiro MD, Bagley J, Latz J, et al. MicroRNA expression data reveals a signature of kidney damage following ischemia reperfusion injury. PLoS One. 2011;6:e23011. doi:10.1371/journal.pone.0023011
44. Wilflingseder J, Regele H, Perco P, et al. miRNA profiling discriminates types of rejection and injury in human renal allografts. Transplantation. 2013;95:835–841. doi:10.1097/TP.0b013e31828ee12e
45. Shaked A, Chang B-L, Barnes MR, et al. An ectopically expressed serum miRNA signature is prognostic, diagnostic, and biologically related to liver allograft rejection. Hepatology. 2017;65:269–280. doi:10.1002/hep.28786
46. Farid WRR, Pan Q, van der Meer AJP, et al. Hepatocyte-derived microRNAs as serum biomarkers of hepatic injury and rejection after liver transplantation. Liver Transplant. 2012;18:290–297. doi:10.1002/lt.v18.3
47. Verhoeven CJ, Farid WRR, de Ruiter PE, et al. MicroRNA profiles in graft preservation solution are predictive of ischemic-type biliary lesions after liver transplantation. J Hepatol. 2013;59:1231–1238. doi:10.1016/j.jhep.2013.07.034
48. Zhang Y, Jia Y, Zheng R, et al. Plasma MicroRNA-122 as a biomarker for viral-, alcohol-, and chemical-related hepatic diseases. Clin Chem. 2010;56:1830–1838. doi:10.1373/clinchem.2010.147850
49. Rayner, K. J., et al. MiR-33 contributes to the regulation of cholesterol homeostasis. Science. 2010;328:1570–1573. doi: 10.1126/science.1189862
50. Qiao A, Khechaduri A, Kannan Mutharasan R, Wu R, Nagpal V, Ardehali H. MicroRNA-210 decreases heme levels by targeting ferrochelatase in cardiomyocytes. J Am Heart Assoc. 2013;2:e000121. doi:10.1161/JAHA.113.000121
51. Sukma Dewi I, Hollander Z, Lam KK, et al. Association of Serum MiR-142-3p and MiR-101-3p Levels with Acute Cellular Rejection after Heart Transplantation. PLoS One. 2017;12:e0170842. doi:10.1371/journal.pone.0170842
52. Di Francesco A, Fedrigo M, Santovito D, et al. MicroRNAs signature in heart transplant enhances diagnosis of different types of acute rejection. J Hear Lung Transplant. 2018;37:S39–S40. doi:10.1016/j.healun.2018.01.078
53. Bhatia AK, Phan JH, Mahle WT, Shehata B, Jo H. Identification of candidate MicroRNA as pathological markers of pediatric heart transplant rejection. J Hear Lung Transplant. 2015;34:S162. doi:10.1016/j.healun.2015.01.439
54. Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136:629–641. doi:10.1016/j.cell.2009.02.006
55. Lorenzen JM, et al. Circulating long noncoding RNA TapSAKI is a predictor of mortality in critically ill patients with acute kidney injury. Clin Chem. 2015;61:191–201. doi:10.1373/clinchem.2014.230359
56. Lorenzen JM, et al. Long noncoding RNAs in urine are detectable and may enable early detection of acute T cell-mediated rejection of renal allografts. Clin Chem. 2015;61:1505–1514. doi:10.1373/clinchem.2015.243600
57. Yamani MH, Taylor D. O. In Current Clinical Medicine. Elsevier; 2010:180–186. doi:10.1016/B978-1-4160-6643-9.00027-8
58. Snyder TM, Khush KK, Valantine HA, Quake SR. Universal noninvasive detection of solid organ transplant rejection. Source Proc Natl Acad Sci U S A. 2011;108:6229–6234. doi:10.1073/pnas.1013924108
59. De Vlaminck I, Valantine HA, Snyder TM, et al. Circulating cell-free DNA enables noninvasive diagnosis of heart transplant rejection. Sci Transl Med. 2014;6:241ra77. doi:10.1126/scitranslmed.3007803
60. Sharon, E., et al. Quantification of transplant-derived circulating cell-free DNA in absence of a donor genotype. PLoS Comput Biol. 2017;13:e1005629. doi:10.1371/journal.pcbi.1005629
61. De Vlaminck I, Martin L, Kertesz M, et al. Noninvasive monitoring of infection and rejection after lung transplantation. Proc Natl Acad Sci U S A. 2015;112:13336–13341. doi:10.1073/pnas.1517494112
62. Schütz E, et al. Graft-derived cell-free DNA, a noninvasive early rejection and graft damage marker in liver transplantation: A prospective, observational, multicenter cohort study. PLoS Med. 2017;14:e1002286. doi:10.1371/journal.pmed.1002286
63. Gordon PMK, Khan A, Sajid U, et al. An algorithm measuring donor cell-free DNA in plasma of cellular and solid organ transplant recipients that does not require donor or recipient genotyping. Front Cardiovasc Med. 2016;3:33. doi:10.3389/fcvm.2016.00033
64. Beck J, Bierau S, Balzer S, et al. Digital droplet PCR for rapid quantification of donor DNA in the circulation of transplant recipients as a potential universal biomarker of graft injury. Clin Chem. 2013;59:1732–1741. doi:10.1373/clinchem.2013.210328
65. Boer K, Baan CC, van Donk N, et al. Donor-derived cell-free DNA as minimally invasive tool to diagnose acute rejection after kidney transplantation. Transplantation. 2018;102:S689. doi:10.1097/01.tp.0000543639.38823.f8
66. Zou J, Duffy B, Steward N, Hachem R, Mohanakumar T. Detection of donor cell-free dna using digital PCR in lung transplant recipients with graft rejection and infection. Hum Immunol. 2015;76:68. doi:10.1016/j.humimm.2015.07.096
67. Atamaniuk J, Vidotto C, Tschan H, et al. Increased concentrations of cell-free plasma DNA after exhaustive exercise. Clin Chem. 2004;50:1668–1670. doi:10.1373/clinchem.2004.034553
68. Gielis EM, Beirnaert C, Dendooven A, et al. Plasma donor-derived cell-free DNA kinetics after kidney transplantation using a single tube multiplex PCR assay. PLoS One. 2018;13:e0208207. doi:10.1371/journal.pone.0208207
69. Bergmann O, Bhardwaj RD, Bernard S, et al. Evidence for cardiomyocyte renewal in humans. Science. 2009;324:98–102. doi:10.1126/science.1164680
70. Lorio MA, Rosa R, Suarez JF, et al. Influence of immune activation on the risk of allograft rejection in human immunodeficiency virus-infected kidney transplant recipients. Transpl Immunol. 2016;38:40–43. doi:10.1016/j.trim.2016.06.001
71. Simonetta F., Pradier A, Masouridi-Levrat S, et al. Torque teno virus load and acute rejection after orthotopic liver transplantation. Transplantation. 2017;101:e219–e221. doi:10.1097/TP.0000000000001588
72. Mendez-Eirin E, Barge-Caballero E, Paniagua-Martin M, et al. Impact of cytomegalovirus infection on long-term heart transplant outcome. J Hear Lung Transplant. 2017;36:S89. doi:10.1016/j.healun.2017.01.225
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.