Back to Journals » OncoTargets and Therapy » Volume 11
Natural borneol is a novel chemosensitizer that enhances temozolomide-induced anticancer efficiency against human glioma by triggering mitochondrial dysfunction and reactive oxide species-mediated oxidative damage
Authors Liu WJ, Yin YB, Sun JY, Feng S, Ma JK, Fu XY, Hou YJ, Yang MF, Sun BL, Fan CD
Received 17 May 2018
Accepted for publication 17 July 2018
Published 4 September 2018 Volume 2018:11 Pages 5429—5439
DOI https://doi.org/10.2147/OTT.S174498
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Tohru Yamada
Wen-Jian Liu,1,* Yi-Bo Yin,1,* Jing-Yi Sun,2,* Sai Feng,3 Jin-Kui Ma,4 Xiao-Yan Fu,5 Ya-Jun Hou,5 Ming-Feng Yang,5 Bao-Liang Sun,1,5 Cun-Dong Fan5
1Department of Neurology, Affiliated Hospital of Taishan Medical University, Taian, Shandong, People’s Republic of China; 2Department of Orthopaedics, Wonju Severance Christian Hospital, Yonsei University Wonju College of Medicine, Wonju, Gangwon, Korea; 3Guangzhou New BenFu Technology Co., Ltd, Guangzhou, Guangdong, People’s Republic of China; 4Faculty of Bioresource Sciences, Akita Prefectural University, Shimoshinjo-Nakano, Akita-shi, Akita, Japan; 5Key Lab of Cerebral Microcirculation in Universities of Shandong, Taishan Medical University, Taian, Shandong, People’s Republic of China
*These authors contributed equally to this work
Background: Temozolomide (TMZ)-based chemotherapy represents an effective way for treating human glioma. However, its clinical application is limited because of its side effects and resistance to standard chemotherapy. Hence, the search for novel chemosensitizers to augment their anticancer efficiency has attracted much attention. Natural borneol (NB) has been identified as a potential chemosensitizer in treating human cancers. However, the synergistic effect and mechanism of NB and TMZ in human glioma have not been investigated yet.
Materials and methods: U251 human glioma cells were cultured, and the cytotoxicity and apoptosis of NB and/or TMZ were examined by MTT assay, flow cytometric analysis and Western blot. Nude mice tumor model was also employed to evaluate the in vivo anticancer effect and mechanism.
Results: The results showed that the combined treatment of NB and TMZ more effectively inhibited human glioma growth via triggering mitochondria-mediated apoptosis in vitro, accompanied by the caspase activation. Combined treatment of NB and TMZ also caused mitochondrial dysfunction through disturbing Bcl-2 family expression. Further investigation revealed that NB enhanced TMZ-induced DNA damage through inducing reactive oxide species (ROS) overproduction. Moreover, glioma tumor xenograft growth in vivo was more effectively inhibited by the combined treatment with NB and TMZ through triggering apoptosis and anti-angiogenesis.
Conclusion: Taken together, our findings validated that the strategy of using NB and TMZ could be a highly efficient way to achieve anticancer synergism.
Keywords: glioma, borneol, temozolomide, DNA damage, apoptosis
Corrigendum for this paper has been published.
Introduction
Glioma is the most common primary malignant tumor in the adult central nervous system.1 The 5-year survival rate is less than 5%.2–4 Because of its high recurrence rate, single surgery, chemotherapy or radiotherapy was not appropriate for the treatment of human glioma.5 Combined therapies have emerged as excellent strategies. Studies have shown that postoperative chemotherapy had achieved great outcome for human glioma.6 However, the existence of blood–brain barrier (BBB) confined the clinical application of chemotherapy.7–9 Hence, the search for novel chemosensitizers to promote the BBB permeability and improve the chemotherapy of human glioma is urgently needed.10
Natural borneol (NB), a simple bicyclic monoterpene, was isolated from the resin of Dryobalanops aromatica Gaertn F, which showed multiple pharmacological properties. It is reported that NB could improve the oral bioavailability of anti-tumor drugs by regulating the permeability of the BBB.10 Increased evidence have confirmed that NB can increase cell membrane mobility and drug solubility, decrease mucus stickiness and elasticity and inhibit drug efflux.11,12 Recently, studies showed that NB can act as a chemosensitizer to enhance anticancer drug-induced apoptosis in human cancer cells through triggering DNA damage and regulation of MAPKs and AKT pathways.13,14 However, the potential of NB as a chemosensitizer in human glioma has not been elucidated yet.
In this study, U251 human glioma cells and nude mice-bearing glioma xenograft were employed to evaluate the combined effect and mechanism of NB and TMZ against human glioma cell growth in vitro and in vivo. The results indicated that NB had the potential to enhance TMZ-mediated anticancer efficiency against human glioma through triggering mitochondrial dysfunction and reactive oxide species (ROS)-induced DNA damage.
Materials and methods
Chemicals
NB was obtained from BenFu Technology Co., Ltd. (Guangzhou, China) DMEM, PBS, FBS, 2’.7’-dichlorofluorescin diacetate (DCFH-DA) probe and mitochondria-targeted MitoSOX probe (M36008) were all purchased from Thermo Fisher Scientific (Waltham, MA, USA). Temozolomide (TMZ), MTT and propidium iodide (PI) were obtained from Sigma-Aldrich Co. (St Louis, MO, USA). TUNEL-DAPI kit, MitoSOX probe and BCA assay kit were purchased from Beyotime Institute of Biotechnology (Shanghai, China). All antibodies used in this study were obtained from Cell Signaling Technology (Beverly, MA, USA). All solvents used were of high performance liquid chromatography (HPLC) grade.
Cell culture, drug treatment and determination of cell viability
U251 human glioma cells were acquired from American Type Culture Collection (ATCC, Manassas, VA, USA) and were cultured in DMEM with 10% FBS, penicillin (100 U/mL) and streptomycin (100 U/mL) at 37°C in a 5% CO2 incubator. Cell was seeded in 96-well plates and treated with 10–160 μM TMZ or 5–80 μg/mL of NB for 48 hours. For combined treatment, the cells were treated with TMZ (20 and 40 μM) and NB (40 and 80 μg/mL) for 48 hours. Cell viability of U251 cells was detected by the MTT assay. After treatment, 20 μL/well of MTT solution (5 mg/mL) was added to the cells and incubated for 5 hours at 37°C. The supernatants were aspirated and 150 μL of DMSO was added. Then, the absorbance was measured on a scanning multi-well spectrophotometer. Cell viability was calculated as a percentage of control (as 100%).
Flow cytometric analysis
Cell cycle distribution and apoptosis were assessed by flow cytometric analysis as previously used.15 The cells treated with NB and/or TMZ were harvested by centrifugation and washed with PBS, and then the cells were fixed with 70% alcohol and stained with PI solution for 2 hours. Labeled cells were washed with PBS and then analyzed by flow cytometry (FCM). The proportions of cells in G0/G1, S and G2/M phases were represented as DNA histograms. Apoptotic cells with hypodiploid DNA contents were measured by quantifying the sub-G1 peak. For each experiment 10,000 events per sample were recorded.
Detection of early and late apoptoses
Early and late apoptoses of U251 cells were detected by Annexin V–PI staining and TUNEL-DAPI staining, respectively. Cells cultured in a chamber slide were treated with 3.7% formaldehyde for 10 minutes and permeabilized in PBS with 0.1% Triton X-100. Then, the cells were administered with the Annexin V–PI staining and TUNEL-DAPI staining kits for measuring the early and late apoptoses, respectively. The experiments were conducted according to the manufacturer’s instructions. The images were obtained from a fluorescence microscope.
Evaluation of mitochondrial dysfunction
Mitochondrial function was evaluated by the mitochondrial membrane potential (Δψm) and mitochondrial morphology, which were examined by JC-1 and Mito-Tracker probes, respectively. Briefly, the cells seeded in a 6 cm plate were exposed to NB and/or TMZ. After treatment, the cells were washed and incubated with 10 μM JC-1 or Mito-Tracker for 15 minutes in the dark. Then, the cells were washed and imaged under an inverted fluorescence microscope.
Measurement of ROS generation and superoxide anion
The intracellular ROS and superoxide anion were measured by DCFH-DA and MitoSOX probes in live cells, respectively. Briefly, after treatment with NB and/or TMZ, the cells were incubated with 10 μM DCFH-DA or 0.5 μM MitoSOX. After reaction, the cells were washed and observed by an inverted fluorescence microscope for the detection of ROS (green fluorescence) and superoxide anion (red fluorescence). The images shown here were obtained from three independent experiments.
Western blot analysis
Total protein was prepared from the cells treated with NB and/or TMZ and quantified by the BCA kit. SDS-PAGE was performed in 10% tricine gel, and 40 μg/lane total protein was loaded. After electrophoresis, proteins were transferred from the gel onto a nitrocellulose membrane at 110 V for 1 hour. Then, the membrane was blocked with 5% nonfat milk for 1 hour, and the membranes were hatched with primary antibodies at 1:1,000 dilution in 5% nonfat milk over night at 4°C. Finally, the membrane was treated with horseradish peroxidase-coupled secondary antibody at 1:2,000 for 1 hour at room temperature. Protein was imaged under an Image-System (Bio-Rad Laboratories Inc., Hercules, CA, USA). β-Actin was used as the equal loading.
In vivo study
U251 cell suspension (109 cells) was inoculated in the armpit of 6-week-old Balb/c nude mice. After 1-week growth, the tumor-bearing mice were randomly divided into four groups (10 mice/group). The control group was given the corresponding solvent, and the other groups were given 20 mg/kg/day NB and/or 10 mg/kg/day TMZ every day for 2 weeks. At the end of the experiment, tumor was separated, and the tumor volume and weight were measured, using the formula: volume=l×w2/2, with l being the maximal length and w being the width. Sections of tumors were used for Western blotting and immunohistochemical (IHC) assay. All animal experiments were approved by the Animal Experimentation Ethics Committee of Shandong in China.
Statistical analysis
All data and images were done from three independent experiments at least, and all data were indicated as mean±standard error. Statistical analysis was performed by SPSS statistical package (SPSS 13.0 for Windows; SPSS Inc., Chicago, IL, USA). The data of two groups were analyzed by Student’s t-test, and the data of three or more groups were analyzed by one-way analysis of variance multiple comparisons to assess significant differences, and differences with P<0.05 or P<0.01 were considered statistically significant. Bars with different letters indicate statistical difference at the P<0.05 level.
Ethical statement
This study was performed in Key Lab of Cerebral Microcirculation in Universities of Shandong, Taishan Medical University. All the in vitro and in vivo experiments were performed in accordance with the relevant guidelines and regulations of Taishan Medical University. The in vivo experiments were approved by the Taishan Medical University Ethics Committee. All surgeries were performed under 10% chloral hydrate, and all efforts were made to minimize suffering.
Results
NB enhances TMZ-induced cytotoxicity in human glioma cells
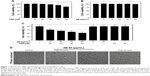
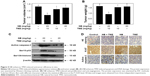
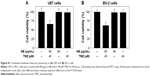
In order to optimize the dosages for combined treatment, the cytotoxicity of NB or TMZ alone toward human glioma cells was screened by the MTT assay. As shown in Figure 1A and B, treatment of U251 cells with TMZ (0–40 μM) for 48 hours caused no significant cytotoxicity. Only treatment with 80 and 160 μM TMZ significantly inhibited U251 cell growth. U251 cells exposed to NB (0–80 μM) for 48 hours showed no cytotoxicity. However, combined treatment of NB and TMZ significantly inhibited U251 cell growth (Figure 1C). For instance, the treatment of cells with 80 μM NB or 40 μM TMZ alone showed no significant changes in the U251 cell viability. However, combined treatment of NB (80 μM) and TMZ (40 μM) significantly inhibited the cell viability to 52.8%. Similar cytotoxicity effect of NB and/or TMZ was also detected in U87 human glioma cells and BV-2 mice microglial cells (Figure S1). Combined treatment-induced growth inhibitory effect was further confirmed by the morphological changes in U251 cells. As shown in Figure 1D, the treatment of U251 cells with NB or TMZ alone caused no significant change in cell morphology. However, combined treatment of NB and TMZ dramatically resulted in the cell morphological changes, such as the cell shrinkage, decrease in the cell number and loss of cell-to-cell contact. Taken together, these results indicated that NB had the potential to enhance TMZ-induced cytotoxicity in human glioma cells.
NB enhances TMZ-induced apoptosis in human glioma cells
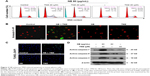
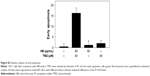
To verify the cell death mechanism, U251 cells after treatment with NB and/or TMZ were analyzed by FCM. As shown in Figure 2A, the treatment of U251 cells with NB or TMZ alone caused no significant apoptosis and no effects on cell cycle distribution. However, enhanced cell apoptosis was dramatically detected after combined treatment of NB and TMZ. For instance, the combined treatment of NB (40 and 80 μM) and TMZ (20 μM) triggered U251 cell apoptosis to 28.1% and 49.2%, respectively. Annexin V–PI and TUNEL-DAPI staining further confirmed combined treatment-induced early and late apoptoses, respectively (Figure 2B and C). The statistical analysis of early apoptosis further confirmed this conclusion (Figure S2). Moreover, the enhanced cell apoptosis was further confirmed in the protein level. As shown in Figure 2D, enhanced activation of caspase-3, -7 and -9 was detected after combined treatment of NB and TMZ. The activation of caspase-7 and caspase-9 indicated the contribution of mitochondria-mediated apoptosis. Taken together, these results demonstrated that NB had the potential to enhance TMZ-induced apoptosis in human glioma cells.
NB enhances TMZ-induced mitochondria dysfunction by regulating Bcl-2 family
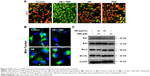
Mitochondrion integrates the internal and external apoptotic signals and plays a significant role in deciding cells’ fate. The depletion of mitochondrial membrane potential (Δψm) will lead to the release of multiple apoptosis factors from the mitochondria into the cytoplasm. Hence, both the Δψm and the mitochondrial morphological change were evaluated using JC-1 and Mito-Tracker probes, respectively. As shown in Figure 3A, U251 cells exposed to treatment with NB or TMZ alone showed no significant change in the Δψm and the mitochondrial morphology. However, the combined treatment of NB and TMZ significantly caused the loss of Δψm, as reflected by the fluorescence shift from red to green. The statistical analysis of Δψm further confirmed this conclusion (Figure S3). Moreover, combined treatment also resulted in the mitochondrial fragmentation. As shown in Figure 3B, the mitochondria in healthy cells and in cells treated with NB or TMZ alone all showed filamentous network with extensive interconnection throughout the cytoplasm. However, combined treatment markedly caused the mitochondrial fragmentation from protonema to punctiform. Bcl-2 family proteins have been depicted as essential regulators of the mitochondrial apoptosis pathway. Therefore, the Bcl-2 family was also identified. As shown in Figure 3C, exposure of cells to NB or TMZ alone caused no obvious effects on the Bcl-2 family expression. Combined treatment of NB and TMZ effectively increased the expression of Bax and Bad, but decreased the expression of Bcl-2 and Bcl-XL. Taken together, these abovementioned results revealed that NB had the potential to enhance TMZ-induced mitochondrial dysfunction through affecting Bcl-2 family balance.
NB enhances TMZ-induced DNA damage through ROS accumulation
ROS could enter nucleus to cause DNA damage by which many anticancer agents induce cancer cell apoptosis.16 Therefore, the oxidative status in U251 cells treated with NB and/or TMZ was examined. As shown in Figure 4A, treatment with NB or TMZ alone only caused slight ROS generation. However, the combined treatment of NB and TMZ significantly caused ROS accumulation, as indicated by the enhanced green fluorescence. The statistical analysis of ROS generation further confirmed combined treatment-induced ROS accumulation (Figure S4). Furthermore, the mitochondrial superoxide overproduction was also detected by MitoSOX, a specific mitochondria-targeted probe, and the results suggested that enhanced production of superoxide was vividly observed in U251 cells after combined treatment with NB and TMZ (Figure 4B). ROS generation caused DNA damage, which is effective to combat human cancers. Hence, combined treatment-induced DNA damage was investigated. As shown in Figure 4C, no significant DNA damage was observed after treatment with NB or TMZ alone. As expected, combined treatment of NB and TMZ significantly caused DNA damage, as demonstrated by the enhanced phosphorylation level of histone (Ser139), p53 (Ser15), ATR (Ser428) and ATM (Ser1981), all of which were DNA damage markers.17 ROS inhibition by glutathione (GSH) effectively inhibited combined treatment-induced cell death (Figure S5), which further confirmed the significant role of ROS in combined treatment-induced cell growth inhibition against human glioma cells. Taken together, these results suggested that NB had the potential to enhance TMZ-induced DNA damage through ROS overproduction.
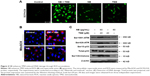
NB enhances TMZ-induced anticancer efficiency in vivo
To explore the combined treatment-induced anticancer effect in vivo, the immunodeficient nude mice were employed. The results showed that treatment with NB or TMZ alone only slightly inhibited the glioma growth, as indicated by the decreased tumor volume and tumor weight. However, combined treatment of NB and TMZ significantly inhibited tumor volume and tumor weight compared to that in treatment with NB or TMZ alone (Figure 5A and B). Additionally, combined treatment-induced anticancer mechanism was similar to that of the in vitro mechanism. As shown in Figure 5C, after combined treatment of NB and TMZ, enhanced cell apoptosis and DNA damage were significantly observed, as indicated by the increased expression of active caspase-3 and the phosphorylation level of histone (Ser139) and p53 (Ser15) (Figure 5C). The staining of Ki-67 and active caspase-3 in tumor tissue further confirmed combined treatment-induced anti-proliferation and apoptosis, respectively (Figure 5D). Moreover, combined treatment also abolished the tumor angiogenesis. As shown in Figure 5D, HE staining revealed that combined treatment significantly decreased the number of erythrocyte-containing blood vessels. The staining of CD-31, a marker of vascular endothelial cells, further confirmed the combined treatment-induced anti-angiogenesis. Taken together, these results suggested that NB had the potential to enhance TMZ-induced anticancer efficiency against human glioma in vivo.
Discussion
Single agent-based chemotherapy was not appropriate for the treatment of human glioma. Recently, combined treatment of chemosensitizer and chemotherapeutic drugs has been applied in clinic to improve the anti-tumor effect. Novel chemosensitizers can improve the efficiency of existing anticancer drugs and overcome their resistance and side effects, and hence has been accepted as a new strategy in the field of chemistry and pharmacology.18 Induction of tumor cell apoptosis and/or cell cycle arrest all contributed to the cytotoxic mechanisms for anti-cancer effect of medicines to kill cancer cells.19 Apoptosis plays an important role in the development, homeostasis and prevention of cancer. Mitochondria-mediated (intrinsic) pathway and death receptor-mediated (extrinsic) pathway can be activated in response to apoptotic stimuli by induction of a series of caspase cascades.20,21 In this study, combined treatment of NB and TMZ significantly induced U251 cell apoptosis accompanied by caspase activation, indicating that NB had the potential to enhance TMZ-induced apoptosis against human glioma.
Mitochondria as the center for apoptotic cascades converges apoptosis signals from extrinsic and intrinsic apoptotic pathways, which play key role in regulating mitochondria-mediated apoptosis.20 Depletion of mitochondrial membrane potential will result in the opening of the mitochondrial permeability transition pore (MPTP), and the release of cytochrome c, apoptosis-inducing factors (AIFs), SMAC/Diablo and endonuclease G, which eventually leads to the caspase activation and apoptotic cascades.22 In this study, combined treatment of NB and TMZ significantly caused the mitochondrial dysfunction, as convinced by the loss of mitochondrial membrane potential and mitochondrial fragmentation, indicating that NB had the potential to enhance TMZ-induced mitochondrial dysfunction. Bcl-2 family protein, including pro-apoptotic members (such as Bax, Bid, Bad and Bim) and anti-apoptotic members (such as Bcl-2, Bcl-XL, Mcl-1 and Bcl-w), plays an important role in triggering mitochondrial apoptosis.23 Bcl-2 and Bcl-XL can bind the outer membrane of mitochondria and block efflux of cytochrome c.24 Overexpression of Bcl-2 and Bcl-XL in many cancer cells is one of the main causes of the resistance toward chemotherapeutic agents and radiotherapy.25 Hence, the balance of pro-apoptotic and anti-apoptotic members decides the stability and the integrity of the mitochondrial membrane.26 In this study, combined treatment of NB and TMZ significantly caused the imbalance of Bcl-2 family expression, which eventually resulted in mitochondria-mediated apoptosis in human glioma cells.
ROS, such as superoxide anion, hydrogen peroxide and hydroxyl radical, have been determined as potential regulators of apoptosis by modulating the extrinsic and intrinsic pathways of apoptosis.27 ROS may assault cell membrane lipids, proteins and DNA, cause oxidative damage and lead to cell apoptosis. Oxidative stress can irritate metabolic activity and cause mitochondrial dysfunction. Excessive production of ROS will lead to the accumulation of oxidized products of DNA, such as DNA strand breaks (DSB).28 p53 as a tumor suppressor can be activated in response to DNA damage and plays a key role in regulating cell cycle arrest and/or apoptosis.29 Activation of p53 can regulate the p21 expression that inhibits cyclin-dependent kinase activity.30 In this study, combined treatment of NB and TMZ significantly caused ROS accumulation, which eventually leads to the DNA damage, as convinced by the activation of DNA damage makers, such as ATM, ATR, p53 and histone. These results come to a conclusion that NB has the potential to enhance TMZ-induced DNA damage through ROS overproduction.
Conclusion
Our findings demonstrated that NB has the potential to enhance TMZ-mediated anticancer efficiency against human glioma through triggering mitochondrial dysfunction and ROS-induced DNA damage, which validated that the strategy of using NB and TMZ could be an effective way for therapy of human glioma.
Acknowledgment
This study was supported by the National Natural Science Foundation of China No 81501106 to C-DF and No 81471212 to B-LS.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Ostrom QT, Gittleman H, Farah P, et al. CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2006–2010. Neuro Oncol. 2013;15 Suppl 2:ii1–ii56. | ||
Carlsson SK, Brothers SP, Wahlestedt C. Emerging treatment strategies for glioblastoma multiforme. EMBO Mol Med. 2014;6(11):1359–1370. | ||
Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–132. | ||
Chen J, Mckay RM, Parada LF. Malignant glioma: lessons from genomics, mouse models, and stem cells. Cell. 2012;149(1):36–47. | ||
Maule F, Bresolin S, Rampazzo E, et al. Annexin 2A sustains glioblastoma cell dissemination and proliferation. Oncotarget. 2016;7(34):54632–54649. | ||
Hart MG, Garside R, Rogers G, Stein K, Grant R. Temozolomide for High Grade Glioma. Cochrane Database Syst Rev. 2013;(4):CD007415. | ||
Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. Structure and function of the blood-brain barrier. Neurobiol Dis. 2010;37(1):13–25. | ||
Trost A, Lange S, Schroedl F, et al. Brain and Retinal Pericytes: Origin, Function and Role. Front Cell Neurosci. 2016;10:20. | ||
Attwell D, Mishra A, Hall CN, O’Farrell FM, Dalkara T. What is a pericyte? J Cereb Blood Flow Metab. 2016;36(2):451–455. | ||
Berghiche H, Houamria M, Smagghe G, Soltani N. Activity of kk-42 in combinated treatment with RH-0345 or 20-hydroxyecdysone on morphometric measurements and free ecdysteroid in eggs of mealworms. Commun Agric Appl Biol Sci. 2005;70(4):837–841. | ||
Zhou Y, Li W, Chen L, Ma S, Ping L, Yang Z. Enhancement of intestinal absorption of akebia saponin D by borneol and probenecid in situ and in vitro. Environ Toxicol Pharmacol. 2010;29(3):229–234. | ||
Shen Q, Li X, Li W, Zhao X. Enhanced intestinal absorption of daidzein by borneol/menthol eutectic mixture and microemulsion. AAPS Pharm Sci Tech. 2011;12(4):1044–1049. | ||
Su J, Lai H, Chen J, et al. Natural borneol, a monoterpenoid compound, potentiates selenocystine-induced apoptosis in human hepatocellular carcinoma cells by enhancement of cellular uptake and activation of ROS-mediated DNA damage. PLoS One. 2013;8(5):e63502. | ||
Chen J, Li L, Su J, Li B, Chen T, Wong YS. Synergistic apoptosis-inducing effects on A375 human melanoma cells of natural borneol and curcumin. PLoS One. 2014;9(6):e101277. | ||
Chen T, Zheng W, Wong YS, Yang F. Mitochondria-mediated apoptosis in human breast carcinoma MCF-7 cells induced by a novel selenadiazole derivative. Biomed Pharmacother. 2008;62(2):77–84. | ||
Mizutani H. Mechanism of DNA damage and apoptosis induced by anticancer drugs through generation of reactive oxygen species. Yakugaku Zasshi. 2007;127(11):1837–1842. | ||
Sancar A, Lindsey-Boltz LA, Unsal-Kaçmaz K, Linn S. Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem. 2004;73:39–85. | ||
Kruh GD, Goldstein LJ. Doxorubicin and multidrug resistance. Curr Opin Oncol. 1993;5(6):1029–1034. | ||
Chen J, Li L, Su J, Chen T. Natural borneol enhances bisdemethoxycurcumin-induced cell cycle arrest in the G2/M phase through up-regulation of intracellular ROS in HepG2 cells. Food Funct. 2015;6(3):740–748. | ||
Kaufmann SH, Earnshaw WC. Induction of apoptosis by cancer chemotherapy. Exp Cell Res. 2000;256(1):42–49. | ||
Riedl SJ, Shi Y. Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol. 2004;5(11):897–907. | ||
van Gurp M, Festjens N, van Loo G, Saelens X, Vandenabeele P. Mitochondrial intermembrane proteins in cell death. Biochem Biophys Res Commun. 2003;304(3):487–497. | ||
Krantic S, Mechawar N, Reix S, Quirion R. Apoptosis-inducing factor: a matter of neuron life and death. Prog Neurobiol. 2007;81(3):179–196. | ||
Hikita H, Takehara T, Shimizu S, et al. Mcl-1 and Bcl-xL cooperatively maintain integrity of hepatocytes in developing and adult murine liver. Hepatology. 2009;50(4):1217–1226. | ||
Edlich F, Banerjee S, Suzuki M, et al. Bcl-x(L) retrotranslocates Bax from the mitochondria into the cytosol. Cell. 2011;145(1):104–116. | ||
Martinou JC, Youle RJ. Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell. 2011;21(1):92–101. | ||
Pelicano H, Carney D, Huang P. ROS stress in cancer cells and therapeutic implications. Drug Resist Updat. 2004;7(2):97–110. | ||
Circu ML, Ty A. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radical Bio Med. 2010;48:749–762. | ||
Cui Q, Yu JH, Wu JN, et al. P53-mediated cell cycle arrest and apoptosis through a caspase-3-independent, but caspase-9-dependent pathway in oridonin-treated MCF-7 human breast cancer cells. Acta Pharmacol Sin. 2007;28(7):1057–1066. | ||
Boulaire J, Fotedar A, Fotedar R. The functions of the cdk-cyclin kinase inhibitor p21WAF1. Pathol Biol. 2000;48(3):190–202. |
Supplementary materials
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.