Back to Journals » International Journal of Nanomedicine » Volume 9 » Issue 1
Nanomedicines in the treatment of hepatitis C virus infection in Asian patients: optimizing use of peginterferon alfa
Received 17 December 2013
Accepted for publication 29 January 2014
Published 25 April 2014 Volume 2014:9(1) Pages 2051—2067
DOI https://doi.org/10.2147/IJN.S41822
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Chen-Hua Liu,1–3 Jia-Horng Kao1–3
1Department of Internal Medicine, National Taiwan University Hospital, Taipei, Taiwan; 2Hepatitis Research Center, National Taiwan University Hospital, Taipei, Taiwan; 3Graduate Institute of Clinical Medicine, National Taiwan University College of Medicine, Taipei, Taiwan
Abstract: Asia is endemic for hepatitis C virus (HCV) infection, which is the leading cause of cirrhosis, hepatic decompensation, hepatocellular carcinoma , and liver transplantation worldwide. HCV has six major genotypes and each HCV genotype has its specific geographic distribution. HCV genotypes 1, 2, 3, and 6 are common in Asia. The aim of HCV treatment is to eradicate the virus by effective therapeutic agents; viral clearance is durable after long-term post-treatment follow-up. In most Asian countries, peginterferon alfa (PEG-IFN α) in combination with ribavirin remains the standard of care, and the overall sustained viral response (SVR) rate in Asian HCV patients is higher than that in Western patients. The differences are most significant in patients with HCV genotype 1 (HCV-1) infection, which is attributed to the higher frequency of IFN-responsive or favorable interleukin-28B (IL-28B) genotype in Asian populations than in other ethnic populations. In addition, the introduction of response-guided therapy, where the optimized treatment duration is based on the early viral kinetics during the first 12 weeks of treatment, increases the SVR rate. Recently, telaprevir or boceprevir-based triple therapy was found to further improve the SVR rate in treated and untreated HCV-1 patients and has become the new standard of care in Western and some Asian countries. Many novel direct-acting antiviral agents, either in combination with PEG-IFN α plus ribavirin or used as IFN-free regimens are under active investigation. At the time of this writing, simeprevir and sofosbuvir have been approved in the US. Because the SVR rates in Asian HCV patients receiving PEG-IFN α plus ribavirin therapy are high, health care providers should judiciously determine the clinical usefulness of these novel agents on the basis of treatment duration, anticipated viral responses, patient tolerance, financial burdens, and drug accessibility.
Keywords: PEG-IFN α, HCV, SVR, RGT, IL28B
Introduction
Hepatitis C virus (HCV) infection, the leading cause of cirrhosis, hepatic decompensation, hepatocellular carcinoma (HCC), and liver transplantation, affects ~170 million people worldwide.1 It is estimated that approximately 20% of individuals who are infected with HCV will develop cirrhosis within 20–30 years, and that the risk of HCC development in these patients is 1%–4% per annum.2 The aim of HCV therapy is the eradication of HCV, and the current definition of successful treatment is sustained virologic response (SVR), referring to undetectable serum HCV RNA levels at 12 (SVR12) or 24 (SVR24) weeks after the cessation of antiviral therapy. The durability of viral response after achieving SVR has been confirmed by many observational studies, showing that over 98% of patients who achieve SVR can maintain non-viremic status after long-term post-treatment follow-up, regardless of HCV monoinfection, coinfection with hepatitis B virus (HBV) or human immunodeficiency virus (HIV), alanine aminotransferase (ALT) levels, or patient ancestry.3,4 Compared to the development of conventional interferon alpha (IFN α) for the treatment of chronic HCV infection, which results in only 6%–19% of SVR after 24–48 weeks of treatment, the use of peginterferon alfa (PEG-IFN α) in combination with ribavirin (RBV) has greatly improved the overall SVR rate to 42%–52% in HCV genotypes 1/4 patients and 76%–82% in HCV genotype 2/3 patients, respectively.5–7 In addition, the introduction of response-guided therapy (RGT), where the optimized treatment duration is on the basis of early viral kinetics during the first 12 weeks of treatment, has further increased the SVR rate to 70%–75%.8,9 Interestingly, the SVR rates of Asian patients with HCV genotype 1 (HCV-1) infection receiving PEG-IFN α plus RBV combination therapy are higher than those of Western HCV-1 patients. In contrast, the response rates between Asian and Western non-HCV-1 patients are comparable. The higher response rate in Asian HCV-1 patients than in Western patients was explained by the discovery of human interleukin-28B (IL-28B) genetic polymorphisms (rs12979860 or rs8099917) that strongly affect the SVR rates in HCV-1 patients receiving dual therapy of PEG-IFN α plus RBV.10,11 Recently, the development of telaprevir (TVR) or boceprevir (BOC)-based triple therapy with PEG-IFN α plus RBV further improved the SVR rate to 66%–76% in treated and untreated HCV-1 patients and has become the standard of care (SOC) for Western HCV-1 patients.12–15 Many novel direct-acting antiviral agents (DAAs), either in combination with PEG-IFN α plus RBV or used as IFN-free regimens are under active investigation. This review will focus on the recent advances in the epidemiology, natural history, and therapy of Asian patients with HCV infection, with special attention on the role of PEG-IFN α.
Epidemiology
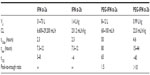
HCV infection remains a global health problem. It is estimated that approximately 3–4 million people are newly infected, and 170 million people (3% of the world’s population) are chronically infected with HCV. The majority of chronic HCV-infected carriers come from East Asia (about 60 million) and Southeast Asia (30–35 million). The prevalence of the antibody to HCV (anti-HCV) in Asia varies widely by geographical area (Table 1). In general, East Asia and Southwest Asia are estimated to have a high prevalence (>3.5%), whereas South and Southeast Asia are estimated to have a moderate prevalence (1.5%–3.5%).16 The distribution of HCV genotype/subtype also varies widely across different Asian regions. HCV genotype 1b and genotype 2a (HCV-1b/HCV-2a) predominate in East Asia; HCV genotype 3 (HCV-3) predominates in South Asia; HCV genotype 6 (HCV-6) predominates in Southeast Asia.17 Compared with HCV-1 patients in the US, where subtype 1a infection comprises more than 50% of the population, most Asian HCV-1 patients are infected with subtype 1b.6
  | Table 1 Prevalence rates of HCV infection and distribution of HCV genotypes in Asia |
Natural history
Since the identification of the HCV particle in 1989, the natural history of patients with HCV infection has been well documented in subsequent prospective and retrospective studies.2 In patients with acute HCV infection, about 65%–80% of these patients without receiving effective antiviral treatment will evolve into chronic HCV infection. Most patients with chronic HCV infection will lead to hepatitis and hepatic fibrosis, which may be accompanied by non-specific symptoms, such as fatigue, anorexia, and right upper quadrant discomfort.2,18 After 20–30 years of chronic infection, nearly 20% of these patients develop cirrhosis, which is followed by the development of hepatocellular carcinoma (HCC) (1.5% per annum), variceal bleeding (1.1% per annum), hepatic encephalopathy (0.4% per annum), and ascites (2.5% per annum).19 Many host and viral factors have been considered to adversely affect the progression of hepatic fibrosis (old age, HBV infection, HIV infection, and heavy alcohol consumption, etc), the development of HCC (high serum HCV RNA level and HCV genotype 1b), and hepatic or extrahepatic mortality (high serum HCV RNA level).20–25
With the use of IFN-based therapy to treat HCV infection, many studies have indicated that more than 98% of patients who achieve SVR have durable undetectable serum HCV RNA by long-term post-treatment follow-up.3,4 Patients with SVR have improved liver histology on necroinflammation and fibrosis, as well as decreased liver-related mortality, hepatic decompensation, and HCC.26,27 Furthermore, successful antiviral treatment may have beneficial clinical outcomes even for HCV-infected patients with decompensated cirrhosis.28 These lines of evidence highlight that SVR is the closest to a life-long virologic cure for HCV infection possible and leads to the improvement of clinical outcomes.
Pharmacology of peginterferon
Process of pegylation
The process of pegylation was first developed in the 1970s. Pegylation involves attaching an inert polyethylene glycol (PEG) molecule to a core protein. It is now a well-established method to modify the pharmacological properties of proteins.29 Pegylation involves the substitution of a PEG hydroxyl group with an electrophilic functional group that is covalently attached, via an amide or urethane bond, to a lysine (Lys) or histidine (His) residual or to the N-terminus of the proteins, and thus creates a larger molecule with an increased molecular weight and apparent Stokes radius. An alternative to attaching multiple PEG units is the use of a single branched PEG moiety. Compared with linear PEG conjugates, branched-chain PEG conjugates tend to have greater pH and thermal stability, as well as being resistant to proteolytic degradation.
The increase of drug size after pegylation prolongs the drug-absorption time and the half-life, which enables once weekly dosing. Several mechanisms are proposed to account for the increase in half-life, including interference with the interaction between the carbohydrate chains and the specific receptors, masking specific amino acid motifs for the corresponding cellular receptors, diminished proteolysis and antigenicity, and limiting the glomerular filtration. Furthermore, pegylation has been suggested to reduce the immunogenicity and antigenicity for the core protein, which may decrease the possibility of a hypersensitivity reaction.30 Despite the favorable pharmacologic properties, pegylation has been shown to reduce the activity of the original protein in an in vitro system, suggesting that pegylation may perturb the interaction between the original protein and the target receptor (Table 2).31
  | Table 2 Advantages and disadvantages of pegylation |
Chemical structure of peginterferon α
Peginterferon α-2a
Peginterferon α-2a (Pegasys, Hoffmann-La Roche, Basel, Switzerland) is formed by a covalent bond between a 40 kDa branched PEG moiety to IFN α-2a (Figure 1). The process of pegylation during drug manufacturing results in a mixture of four major positional isomers at Lys31, Lys121, Lys131, and Lys134 and four minor positional isomers at Lys49, Lys70, Lys83, and Lys112. The antiviral activity of PEG-IFN α-2a is about 1% of IFN α-2a. The major positional isomers at Lys31 and Lys134 have greater antiviral activity than the other positional isomers and possess about 2% of the antiviral activity of IFN α-2a.32
  | Figure 1 Chemical structure of peginterferon α-2a and α-2b.Abbreviations: PEG-IFN, peginterferon; IFN, interferon; Lys, lysine; His, histidine; Cys, cysteine; Ser, serine. |
Peginterferon α-2b
Peginterferon α-2b (Peg-Intron; Schering-Plough Group, Berlin, Germany) is formed by a covalent bond between a 12 kDa linear PEG moiety to IFN α-2b. The process of pegylation during drug manufacturing results in a mixture of mono-pegylated positional isomers.31 The primary site of conjugation (>50% of all positional isomers) is located on the His34 amino acid residue. The sites of conjugation in the remaining positional isomers are distributed among the various Lys, His, cysteine, and serine residues. Compared with IFN α-2b, the antiviral activity of peginterferon α-2b is about 28% by weight of IFN α-2b core protein. However, the positional isomer at His34 has greater antiviral activity and possesses about 37% of the antiviral activity of IFN α-2b.
Pharmacokinetics of peginterferon α
PEG-IFN α-2a is absorbed at a sustained rate and has a prolonged absorption half-life (t1/2abs, 50 hours) (Table 3 and Figure 2).29,34 PEG-IFN α-2a at a single dose of 180 μg in healthy volunteers produces a mean time to maximum plasma drug concentration (tmax) of 72–96 hours. After multiple doses of PEG-IFN α-2a, the tmax value was about 45 hours. The mean elimination half-life (t1/2β) is 65 hours. At the steady state, which is usually attained after 5–8 weeks of drug therapy, the serum peak-to-trough ratio of peginterferon α-2a is about 1.5, implying that the drug concentration can be sustained during the 1-week dosing interval. Furthermore, the serum PEG-IFN α-2a will be undetectable about 4–6 weeks after drug discontinuation.
  | Figure 2 Mean peginterferon α-2a (A) or α-2b (B) concentration–time profiles.Notes: Week 1 (solid line) and week 4 (dashed line) were in non-cirrhotic patients with chronic hepatitis C treated with peginterferon α-2a 180 μg per week (adapted from peginterferon α-2a product monograph)34 or peginterferon α-2b 1.0 μg per kilogram of body weight per week. Data from Glue et al33 and package inserts.34,35 |
Compared with PEG-IFN α-2a, the t/12abs of PEG-IFN α-2b is much shorter (4.6 hours) and is about twice the t1/2abs of conventional IFN α-2a or α-2b (2.3 hours) (Table 3 and Figure 2).29,33,35 Because of this rapid drug absorption, the tmax after a single dose or multiple doses of PEG-IFN α-2b is about 15–44 hours (mean 20 hours). The t1/2β of PEG-IFN α-2b (40 hours) is shorter than that of PEG-IFN α-2a. Although PEG-IFN α-2b can be used once per week, the serum peak-to-trough ratio is >10.
The volume of distribution (Vd) is affected by the size of the PEG-protein conjugate. The Vd of PEG-IFN α-2a is 8–12 L, which is 4–6-fold less than that of conventional IFN α-2a (31–73 L), suggesting that the drug is mainly distributed in the intravascular compartment. However, the Vd of PEG-IFN α-2b is 0.99 L/kg, which is slightly less than that of conventional IFN α-2b (1.4 L/kg). PEG-IFN α-2a may be given as a single fixed dose, whereas PEG-IFN α-2b should be administered by weight-based dosing.
The clearance (CL) of PEG-IFN α-2a and PEG-IFN α-2b are about one-hundredth and one-tenth that of conventional IFN α, respectively. Both types of PEG are metabolized by the liver and cleared by the kidneys. No dose adjustments are needed for PEG-IFN α-2a and PEG-IFN α-2b in patients with creatinine clearance above 20 mL/minute and 50 mL/minute, respectively. In patients with end-stage renal disease, PEG-IFN α-2a at a dose of 135 μg per week produces a similar pharmacokinetic profile to 180 μg per week in patients with normal renal function.36
Pharmacodynamics of peginterferon α
The activity of 2′5′-oligoadenylate synthetase (OAS), the key effector protein stimulated in response to IFN, is involved in the inhibition of viral function and replication. Serum OAS levels increase rapidly after the administration of conventional IFN α-2a or PEG-IFN α-2a, and the peak OAS levels occur at 24 and 48 hours after the administration. Although the OAS levels decline within 24 hours after IFN α-2a administration, they remain near the peak level for up to 168 hours after PEG-IFN α-2a administration, which is consistent with the pharmacokinetic profiles as described above.37 The pharmacodynamics assessment of PEG-IFN α-2b shows dose-related increases of serum neopterin, which is synthesized by macrophages in response to IFN stimulation and serves as a marker for cellular immune system activation, and non-dose-related increases of serum OAS levels.33
Treatment of hepatitis C virus infection
Overview
The goal of treating HCV infection is sustained virologic response (SVR), defined as undetectable serum HCV RNA levels at 12 weeks (SVR12) or 24 weeks (SVR24) after the cessation of therapy.38,39 The durability of viral response is supported by many observational studies, showing that over 98% of patients who achieve SVR can maintain non-viremic status after long-term post-treatment follow-up, regardless of HCV monoinfection, HBV or HIV coinfection, ALT levels, or patient ancestry.3,4 Furthermore, patients with SVR are shown to have decreased liver and non-liver related morbidity and mortality.27,28,40–42 SVR is thus the short-term surrogate marker to predict long-term viral suppression and improved clinical outcome.
The evolution of treatment for chronic HCV infection has greatly improved over the past 20 years (Figure 3). Ever since early 1990 when conventional IFN α monotherapy was used to treat chronic HCV infection with a SVR rate of about 6%–19%, PEG-IFN α in combination with RBV for 24–48 weeks has achieved an overall SVR rate of about 54%–63% across various HCV genotypes.5–7 The introduction of TVR and BOC, the first-generation DAA in May 2011, in combination with PEG-IFN α and RBV has further improved the SVR rates to 68%–75%.12–15 The newer generation of DAA, simeprevir or sofosbuvir, in combination with PEG-IFN α and RBV has an overall SVR rate of >80%, and the regimen has recently been approved by the US Food and Drug Administration (FDA) to treat HCV-1 patients in December 2013.43,44
Although PEG-IFN α-2a or PEG-IFN α-2b in combination with RBV is effective in treating patients with chronic HCV infection, controversies have been raised with regard to the antiviral responses between the two formulas of PEG-IFN α. Two recent meta-analyses which directly compared the safety and efficacy of PEG-IFN α-2a and PEG-IFN α-2b indicated that compared with patients who received PEG-IFN α-2b plus RBV, those who received PEG-IFN α-2a plus RBV tended to have greater SVR rates. Furthermore, the safety profiles were comparable between the two formulas of PEG-IFN α.45,46 In Asian patients with chronic HCV infection, Miyase et al also demonstrated that the SVR rate was statistically greater in patients with PEG-IFN α-2a plus RBV therapy than those with PEG-IFN α-2b plus RBV therapy.47 The more favorable pharmacokinetic profile of PEG-IFN α-2a than that of PEG-IFN α-2b may account for the superior antiviral effects of PEG-IFN α-2a when compared to PEG-IFN α-2b.
Combination therapy of peginterferon α and ribavirin in Asian patients with chronic HCV infection
HCV genotype 1 infection
In Western patients with HCV-1 infection, the overall SVR rates were far from satisfactory, ranging from 39%–52% by 48 weeks of PEG-IFN α-2a plus weight-based RBV (daily 1,000 mg for patients with body weight <75 kg; daily 1,200 mg for patients with body weight ≥75 kg).5–7,48 However, the SVR rates of Asian HCV-1 patients receiving 48 weeks of PEG-IFN α-2a plus weight-based RBV were higher than those of Western patients.49–55 Two studies enrolling East Asian HCV-1 patients in Taiwan indicated that combination therapy with PEG-IFN α-2a plus RBV for 48 and 24 weeks had SVR rates of 76%–79% and 56%–59%, respectively.53,54 With regard to PEG-IFN α-2b plus weight-based RBV for Asian HCV-1 patients, the SVR rates of 48 weeks of treatment were 44%–51%, which was similar to the response rates for Western HCV-1 patients.47,56,57 One study in the US which enrolled HCV patients from different ethnic groups (Asian, White, Hispanic, and African-American) confirmed that Asian HCV-1 patients had the highest response rates to IFN-based therapy (Table 4).58
HCV genotype 2/3 infection
In contrast to HCV-1 patients, where racial differences may affect the SVR rates to combination therapy, the overall SVR rates in HCV-2/3 patients who received PEG-IFN α-2a or PEG-IFN α-2b plus flat-dose RBV (daily 800 mg) for 24 weeks were similar in different ethnic groups (Western versus [vs] Asian patients: 70%–82% vs 71%–95%).5–7,48,49,51,52,55,59–62 In East Asian HCV-2 patients treated with PEG-IFN α-2a plus weight-based RBV, the SVR rates were comparable for those receiving 24 or 16 weeks of treatment (95% vs 94%, respectively).59
HCV genotype 4 infection
Three studies are available with regard to the efficacy of PEG-IFN α-2a plus RBV for 48 weeks in Southwest Asian HCV-4 patients.63–65 The overall SVR rate was 50%–68%, which was higher than that for HCV-4 patients receiving PEG-IFN α-2b plus RBV (44%–45%).66,67
HCV genotype 6 infection
Several studies have indicated that the SVR rates in HCV-6 patients receiving 48 weeks of PEG-IFN α-2a or PEG-IFN α-2b plus weight-based RBV were superior to those in HCV-1 patients receiving the same treatment regimen. The SVR rates were 70%–86% and 60%–82% for HCV-6 patients with 48 and 24 weeks of treatment, respectively.68–73 Two randomized studies for Southeast Asian HCV-6 patients indicated that 24 weeks of treatment can achieve comparable response rates to 48 weeks of treatment.72,73
Response-guided therapy for Asian patients with chronic HCV infection
In early 2000, the HCV viral kinetic models found for PEG-IFN α-based therapy showed two phases of early viral decline: the first phase has a rapid viral decline within the first 48 hours, indicating an effect of the direct inhibition of viral replication; the second phase has a slower viral decline after 48 hours, indicating the elimination of the infected virus in hepatocytes.74 Patients with good viral declines at both phases are rapid responders; those with good first phase but with poor second phase declines are slow responders; those with poor or no viral declines at both phases are null responders. Numerous studies in the following 5–10 years have confirmed that the measurements of serum HCV viral load during the first 12 weeks of treatment may help determine the optimized treatment duration in HCV-infected patients receiving PEG-IFN α-based therapy. The notion of response-guided therapy (RGT) for HCV infection is based on the dogma that the more rapidly the viral load declines, the shorter the duration of treatment can be.
The viral responses during the 12 weeks of treatment are defined and the clinical implications of early viral responses are described in the next sections “Rapid virologic response”, “Week 8 virologic response”, and “Early virologic response”.8,38
Rapid virologic response
Rapid virologic response (RVR), which is the key determinant for SVR, is defined as undetectable serum HCV RNA level at week 4 of treatment. Patients who achieved RVR had high SVR rate (88%–100%) by PEG-IFN α-2a plus RBV therapy across various HCV genotypes.75 Baseline factors predictive of RVR included HCV genotype, age, viral load, ALT levels, and hepatic fibrosis. The RVR rates in patients with HCV genotypes 1, 2, 3, and 4 receiving PEG-IFN α-2a plus RBV were 16%, 71%, 60%, and 38% in the large international trials.6,7,76 Furthermore, RVR outweighed other baseline factors to predict SVR. Therefore, patients with HCV-2/3 infection can have greater SVR rates than those with HCV-1/4 infection.
Although the standard duration of PEG-IFN α plus RBV therapy was 48 weeks for HCV-1 patients, two meta-analysis studies indicated that patients with low baseline viral load (HCV viral load <400,000 IU/mL) and with RVR had comparable SVR rates by 24 or 48 weeks of PEG-IFN α plus RBV therapy.9,77 In East Asian HCV-1 patients who achieved RVR, two independent studies indicated that the SVR rate for 24 weeks of PEG-IFN α-2a plus RBV therapy is similar to 48 weeks of combination therapy in HCV-1 patients with baseline viral load <400,000 IU/mL (94%–96% vs 100%, respectively).53,54 In contrast, patients who failed to achieve RVR or patients who achieved RVR but had high viral load (>400,000–800,000 IU/mL) should receive 48 weeks of therapy to secure the SVR rates.53,54
In HCV-2/3 patients who achieved RVR after PEG-IFN α plus RBV therapy, a meta-analysis study indicated that treatment for 24 weeks achieved greater SVR rates than treatment for 12–16 weeks.9 However, the overall SVR rates were similar in these patients if they received PEG-IFN α plus weight-based RBV therapy. Compared with HCV-3 patients, the beneficial effects of 24 weeks over 12–16 weeks of treatment were limited in HCV-2 patients.9 In East Asian HCV-2 patients who achieved RVR, Yu et al showed that the SVR rates were 98%–100% if they received PEG-IFN α-2a plus weight-based RBV therapy for 16–24 weeks.59 In addition, Sato et al showed that the SVR rates were 94% and 93% if East Asian HCV-2 patients achieved super-rapid virologic response (defined as undetectable HCV RNA at week 2 of treatment) and RVR by PEG-IFN α-2b plus weight-based RBV therapy for 12 and 24 weeks, respectively.61
In HCV-4 patients who achieved RVR after PEG-IFN α plus RBV therapy, the international expert panel from the European Association for the Study of Liver Diseases (EASL) recommends that patients can receive 24 weeks of PEG-IFN α plus weight-based RBV therapy only if they do not have poor response predictors (high baseline viral load >800,000 IU/mL, advanced hepatic fibrosis, and high basal insulin resistance).78 In contrast, patients who achieve RVR but have poor predictors of response should receive 48 weeks of treatment.
In Southeast Asian HCV-6 patients, PEG-IFN α-2a plus weight-based RBV for 24 weeks achieved a comparable SVR rate to 48-week treatment if they achieved RVR (75% vs 85%, respectively).73
Week 8 virologic response
Week 8 virologic response (Wk-8R) is defined as undetectable serum HCV RNA level at week 8 of treatment in the absence of RVR. Mangia et al conducted individualized treatment for HCV-1 patients who received 24, 48, or 72 weeks of PEG-IFN α-2a or PEG-IFN α-2b plus weight-based RBV if the serum HCV RNA level first became undetectable at weeks 4, 8, or 12 weeks of treatment, taking the standard 48 weeks of combination as the control group.79 The SVR rates were similar in patients who achieved Wk-8R and those with a standard duration of treatment (72% vs 70%, respectively). In contrast, for patients who failed to achieve Wk-8R but who achieved first undetectable HCV RNA level at week 12 of treatment, treatment for 72 weeks showed a superior response to treatment for 48 weeks (63% vs 38%, respectively). Liu et al conducted a randomized trial to compare the efficacy of 48 and 72 weeks of PEG-IFN α-2a plus weight-based RBV in East Asian HCV-1 patients. In patients who achieved Wk-8R, the SVR rates were similar for those receiving treatment for 72 and 48 weeks (84% vs 85%, respectively); patients who failed to achieve Wk-8R, extending the treatment duration of therapy to 72 weeks, had greater SVR rates than at 48 weeks of treatment (50% vs 26%, respectively).80
Early virologic response
Early virologic response (EVR) is defined as ≥2-log reduction of serum HCV RNA from baseline to week 12 of treatment. In patients who achieve EVR, the viral responses are further classified into complete early virologic response (cEVR), which is defined as undetectable serum HCV RNA at week 12 of treatment in the absence of RVR, and partial early virologic response (pEVR), which is defined as detectable viremia at week 12 but ≥2-log reduction of serum HCV RNA from baseline to week 12 of treatment in the absence of RVR.8,38,81
The absence of EVR is a robust negative predictor of non-response to IFN-based therapy. Data from a large-scale international trial in HCV patients with genotypes 1–4 infection receiving PEG-IFN α-2a plus RBV therapy showed that as low a percentage as 3% of patients who failed to achieve EVR achieved SVR. Because of the low chance of viral response, non-EVR patients should discontinue treatment beyond week 12.6 None of the East Asian HCV-1 patients achieved SVR by 24 or 48 weeks of PEG-IFN α-2a plus RBV therapy if they did not have EVR.54 The utility of EVR is relatively less helpful in HCV-2/3 patients because few patients fail to clear the virus at week 12 of treatment.
Two meta-analyses showed that extending treatment duration from 48 weeks to 72 weeks could improve the SVR rates in HCV-1 patients who have slow responses to PEG-IFN α plus RBV therapy.9,82 The beneficial effect of extending treatment duration is maintained, regardless of the use of weight-based or flat-dose RBV. Among HCV-1 patients with cEVR, there were no differences in terms of SVR rate for patients receiving 48 or 72 weeks of therapy.9 Liu et al further examined Wk-8R in East Asian HCV-1 patients who achieved cEVR, and found that if these patients achieved Wk-8R, extending treatment duration to 72 weeks did not offer any beneficial effect on the SVR rate over 48 weeks of therapy (84% vs 85%, respectively). However, treatment for 72 weeks had a greater SVR rate than treatment for 48 weeks if these cEVR patients failed to achieve Wk-8R (50% vs 26%, respectively).80 For patients with pEVR, the SVR rates for 72 and 48 weeks of treatment were 26% and 17%, respectively.80
Until now, limited data are available with regard to the optimized treatment duration in HCV-2/3 patients with slow viral responses to PEG-IFN α plus RBV therapy. Cheinquer et al randomly assigned Western HCV-2/3 patients who failed to achieve RVR but who achieved cEVR or pEVR to receive PEG-IFN α-2a plus RBV therapy for 48 or 72 weeks.83 The SVR rates of 72 and 48 weeks of treatment were similar by intention-to-treat (ITT) and per-protocol (PP) analyses (61% vs 52%; 63% vs 52%, respectively). However, the SVR rate was improved if patients completed the extended duration of therapy (73% vs 54%, respectively, P=0.02). In Asian HCV-2/3 patients who failed to achieve RVR, Sato et al showed that PEG-IFN α-2b plus RBV therapy for 48 weeks had higher SVR rates than treatment for 24 weeks (77% vs 51%, respectively).61
The RGT for HCV-4 patients with slow viral responses was similar to that for HCV-1 patients.78 Limited data are available for Asian HCV-4 slow responders.
One study for Southeast Asian HCV-6 patients with cEVR or pEVR showed that these patients had poor responses to 24 or 48 weeks of PEG-IFN α-2a plus RBV therapy (0% vs 8%, respectively).73
Impact of interleukin-28B genotype on viral responses in Asian HCV patients receiving peginterferon α and RBV therapy
The racial factors affecting the treatment responses in HCV-1 patients remained elusive until four independent genome-wide association studies from America, Europe, and Japan reported that the single nucleotide polymorphisms at the loci of rs12979860 and rs8099917 near the IL-28 gene strongly predicted the SVR rates in HCV-1 patients receiving PEG-IFN α plus RBV therapy in 2009–2010.84–87 Patients with a favorable IL-28B genotype (rs12979860 CC or rs8099917 TT) had significantly higher SVR rates than those with unfavorable IL-28B genotypes (rs12979860 CT/TT or rs8099917 GT/GG) by PEG-IFN alfa plus RBV therapy. In the 5 years after the publication of these landmark articles, hundreds of studies have further examined the role of IL-28B genotyping in the treatment responses across various HCV genotypes.
Recent meta-analysis studies indicated that IL-28B genotypes strongly affect the SVR rates in HCV-1/4 patients, but only marginally affect the SVR rates in HCV-2/3 patients receiving PEG-IFN α plus RBV therapy.88–90 Among HCV-1 patients, IL-28B genotypes are highly associated with SVR rates, regardless of ethnicity or HIV coinfection.88,89 The allele frequencies of IL-28B (rs12979860 C/T or rs8099917 T/G) vary in different ethnicity. The Asian population have the greatest frequency of the favorable IL-28B allele (rs12979860 C/T or rs809917 T/G =0.92/0.08), followed by the European–American population (rs8099917 T/G =0.83/0.17), the Hispanic population (rs12979860 C/T =0.56/0.44; rs8099917 T/G =0.69/0.31), and the African population (rs8099917 C/T =0.40/0.60; rs8099917 T/G =0.93/0.07).91,92 Furthermore, IL-28B genotypes are associated with the RVR rates in HCV-infected patients, especially in those with HCV-1 infection.78,80,91,93–97 Because the favorable IL-28 genotype highly predicts RVR and SVR in HCV-1 patients, Asian HCV-1 patients, who carry the highest frequency of the favorable IL-28B genotype, have greater SVR rates than HCV-1 patients of other ethnicities.
Based on the important role of IL-28B genotyping in the treatment responses to PEG-IFN α plus RBV therapy, the interplay of early viral kinetics and IL-28B genotypes on SVR rates was further examined. Thompson et al evaluated the effect of RVR and IL-28B genotypes from a large-scale international trial in HCV-1 patients receiving 48 weeks of PEG-IFN α plus RBV therapy. In patients who achieved RVR, the IL-28B genotypes did not predict the SVR rates; in patients who failed to achieve RVR, the SVR rates were greater in those with a favorable IL-28B genotype than those with unfavorable IL-28B genotypes.93
In East Asian HCV-1 patients who received 24 weeks of PEG-IFN α-2a plus RBV therapy, the IL-28B genotypes played a minor role to affect the overall SVR rates if they achieved RVR.80,94,95 In line with the report by Thompson et al, IL-28B genotypes strongly affected the SVR rates in East Asian HCV-1 patients if they failed to achieve RVR.96,98 However, the role of IL-28B genotypes was limited in East Asian HCV-1 patients who failed to achieve RVR if Wk-8R was taken into consideration.80 In Asian HCV-1 patients who relapsed to 24 weeks of PEG-IFN α-2a plus RBV therapy, patients with a favorable IL-28B genotype achieved greater SVR rates than those with unfavorable genotypes by 48 weeks of retreatment (67% vs 14%, respectively).99 With regard to the determination of the early stopping rule at week 4 of treatment in Asian HCV-1 non-responders, Yu et al and Huang et al evaluated patients receiving PEG-IFN α-2a plus RBV therapy for 48 weeks. In patients with HCV RNA >10,000 IU/mL or <3 log HCV viral decline at week 4 of treatment, patients with unfavorable IL-28B genotypes had low SVR rates (0%–18%) and should stop further treatment beyond week 4.100,101
In the era of RGT, the role of IL-28B genotypes in HCV-2/3 patients treated with PEG-IFN α plus RBV therapy has been evaluated in three independent studies from Europe and Asia.97,102,103 All HCV-2/3 patients who achieved RVR had similar SVR rates, regardless of IL-28B genotypes or the duration of therapy. However, there were controversies with regard to the role of IL-28B genotypes on the SVR rates in HCV-2/3 patients who failed to achieve RVR. Furthermore, IL-28B genotypes played no role to predict SVR in East Asian HCV-2 relapsers or non-responders who received 48 weeks of retreatment with PEG-IFN α-2a plus RBV.104
In line with HCV-1 patients, the role of IL-28B genotypes in HCV-4 patients who received PEG-IFN α plus RBV by RGT was limited, and may not select patients with the abbreviated duration of therapy.105 The role of IL-28B genotypes in the treatment of Asian HCV-4 patients remains largely unclear.
In Southeast Asian HCV-6 patients, a recent report evaluated the role of IL-28B in 60 patients treated with PEG-IFN α plus RBV therapy for 48 weeks. Compared with patients with unfavorable IL-28B genotypes, those with a favorable IL-28B genotype had greater SVR rates (96% vs 63%, respectively). Data are scarce on the role of IL-28 genotypes with regard to RGT in HCV-6 patients.70
BOC or TVR-based triple therapy in Asian patients with chronic HCV infection
In May 2011, two first-generation DAAs (BOC and TVR), which target the HCV NS3/4A serine protease, in combination with PEG-IFN α plus weight-based RBV were approved for use in treatment-naïve and treatment-experienced HCV-1 patients. In treatment-naïve HCV-1 patients, the SVR rates of BOC- or TVR-based triple therapy were higher than in PEG-IFN α plus RBV therapy patients (63%–66% vs 38%, respectively for BOC; 69%–76% vs 44%, respectively for TVR).12,14 In treatment-experienced HCV-1 patients, the SVR rates of BOC- or TVR-based triple therapy were also higher than for PEG-IFN α plus RBV therapy (59%–66% vs 21%, respectively for BOC; 64%–66% vs 17%, respectively for TVR).13,15
Although BOC- or TVR-based triple therapy has greatly improved overall SVR rates, data on the efficacy of BOC-based triple therapy in Asian patients are limited (Table 5). Hu et al retrospectively evaluated Asian HCV-1 patients who participated in three Phase II and Phase III BOC-based trials. Compared to patients receiving PEG-IFN α-2b plus RBV for 48 weeks, patients receiving BOC-based triple therapy for 24–48 weeks had a slightly higher SVR rate (79% vs 67%, respectively).106
At the time of this writing, almost all studies on TVR-based triple therapy were from Japan, and PEG-IFN α-2b was universally included in their treatment regimens. Data from these studies showed that the SVR rates of TVR-based triple therapy for 12 weeks were only 45% in treatment-naïve patients.107,108 The SVR rates increased to 70%–82% with an additional 12 weeks of PEG-IFN α plus RBV, except for one small trial112 enrolling the most difficult-to-treat partial responders and non-responders, which showed an SVR rate of 27%.107,109–115
In line with Western reports, IL-28B genotypes and RVR strongly affected the SVR rates in Asian HCV-1 patients receiving BOC- or TVR-based triple therapy.107,109,115 Because the numbers of Asian HCV-1 patients who received BOC were low, and no studies were designed for TVR-based RGT in Asian HCV-1 patients, the optimized strategies for shortening treatment duration on the basis of early viral kinetics and IL-28B genotypes have not yet been determined.
Implications of personalizing management of HCV in Asian patients
Epidemiologically, HCV-1 is dominant in most regions of Asia; while HCV-2, 3, 4, and 6 are common in East Asia, South Asia, Southwest Asia, and Southeast Asia, respectively. Currently, PEG-IFN α plus RBV is the SOC for the treatment of patients with all HCV genotypes in most Asian countries. Although BOC- or TVR-based triple therapy has become the SOC for HCV-1 patients since 2011, the recent approval of simeprevir-based and sofosbuvir-based triple therapy may change the landscape of HCV therapy in the coming years. However, few Asian countries have gained access to BOC and TVR. In addition, a recent cost-effectiveness study for BOC and TVR-based triple therapy indicated that such treatments would only be cost-effective in patients with advanced hepatic fibrosis who received IL-28B guided therapy.116
Asian populations tend to have higher frequencies of favorable IL-28B genotypes than other ancestries. In Asian HCV-1/4/6 patients, those with a favorable IL-28B genotype have higher SVR rates when treated with PEG-IFN α plus RBV. Moreover, Asian HCV-1/4 patients with low baseline viral load can further truncate the treatment duration to 24 weeks without compromising the SVR rate. In Asian HCV-1/4 patients who fail to achieve RVR, Wk-8R and EVR (including cEVR and pEVR), rather than IL-28B genotypes, are the key to determine the optimal treatment duration.
In line with HCV-1 patients, Asian HCV-2/3 patients with a favorable IL-28B genotype also tend to have a higher RVR rate in response to PEG-IFN α plus RBV therapy than those with unfavorable IL-28B genotypes. A truncated duration of PEG-IFN α plus RBV therapy from 24 weeks to 12–16 weeks may be considered in Asian HCV-2 patients with RVR, regardless of IL-28B genotypes. In contrast, Asian HCV-3 patients still require 24 weeks of PEG-IFN α plus RBV therapy even if they achieve RVR. Although existing evidence is limited, HCV-2/3 patients who fail to achieve RVR may extend the treatment duration to 48 weeks if they have unfavorable IL-28B genotypes.
In HCV-1 patients who have unfavorable factors to achieve RVR and SVR, including advanced hepatic fibrosis or cirrhosis, unfavorable IL-28B genotypes, high pretreatment viral load, or previous non-response to PEG-IFN α plus RBV therapy, the health care providers should allocate these patients to receive BOC or TVR-based triple therapy or novel DAA therapies on a case-by-case basis according to the anticipated RVR rate, SVR rate, patient tolerance, treatment duration, financial burdens, and drug accessibility.
Peginterferon α with or without RBV therapy in a special population of Asian patients with chronic HCV infection
In addition to ordinary patients with HCV infection, patients with HBV co-infection and patients with end-stage renal diseases are also at high risk of HCV infection. This is of particular importance because Asian people have high prevalence rates of HBV infection and end-stage renal disease (Table 6).
  | Table 6 Treatment responses to peginterferon α with or without ribavirin in special Asian populations with chronic HCV infection |
The long-term prognosis of patients with HBV and HCV co-infection is inferior to those with HBV or HCV mono-infection.21,117 However, data on the efficacy of PEG-IFN α plus RBV on HCV treatment in HBV and HCV co-infected patients were limited in Asian patients. Liu et al evaluated the efficacy of SVR rates in HBV/HCV dually infected East Asian patients with HCV-1 or HCV-2/3 who received 48 and 24 weeks of PEG-IFN α-2a plus RBV therapy, taking HCV-1 or HCV-2/3 mono-infected patients as the reference controls. The overall SVR rates in HCV-1 and HCV-2/3 patients were 72% and 83% in dually infected patients, respectively, and were comparable to mono-infected patients (77% and 84%), suggesting that dually infected patients can be treated as effectively as HCV mono-infected patients.55 Because PEG-IFN α-2a is also effective in the treatment of HBV infection, dually infected patients also had improved HBV seroclearance after long-term follow-up. In addition, the HCV viral clearance was durable in these patients who achieved SVR.3
Because of high rates of nosocomial transmission, patients with end-stage renal disease who are on maintenance dialysis are at an increased risk of HCV infection. The natural history of HCV-infected dialysis patients showed that their long-term prognosis was worse than non-HCV-infected dialysis patients. It is estimated that the prevalence rate of HCV infection in dialysis patients is about ten times that in the general population. Therefore, the treatment of HCV infection in this special population is important, and it is relevant in Asian patients where the prevalence rates of end-stage renal disease are high.118 Diabetes, hypertension, hyperlipidemia, cardiovascular disease, obesity, smoking, and probably the use of herbal medicines are attributed to the development of end-stage renal disease. Based on the pharmacokinetics studies, the optimal dose of PEG-IFN α-2a and PEG-IFN α-2b for patients with end-stage renal disease who are on hemodialysis is 135 μg per week and 0.75–1.0 μg per kilogram of body weight per week, respectively.36,119
Many Asian experts treated dialysis patients with HCV infection by using PEG-IFN α-2a or PEG-IFN α-2b monotherapy for 24–48 weeks.120–129 However, the SVR rates varied widely (from 0%–100%) because of small sample size, heterogeneous population, and divergent regimens. Recently, two large-scale trials evaluated East Asian HCV-1 and HCV-2 hemodialysis patients receiving PEG-IFN α-2a for 48 and 24 weeks, and the SVR rates were 34% and 44%, respectively, which was similar to the results from the meta-analysis.130–133 RBV has been considered contraindicated for treating dialysis patients because of concerns of life-threatening hemolytic anemia. However, the pharmacokinetics study of RBV in hemodialysis patients showed that the daily dose of 200 mg may be suitable for these patients.134,135 Liu et al further evaluated PEG-IFN α-2a at a dose of 135 μg per week plus RBV at a dose of 200 mg per day for 48 and 24 weeks in treatment-naïve and -experienced East Asian HCV-1 and HCV-2 patients on hemodialysis.130,131,136 The SVR rates for treatment-naïve HCV-1 and HCV-2 patients were 64% and 74%, respectively; those for treatment-experienced HCV-1 and HCV-2 patients were 52% and 80%, respectively. Furthermore, Tseng et al evaluated PEG-IFN α-2b at a dose of 1.0 μg per kg of body weight per week plus RBV at a dose of 200 mg three times per week for 48 and 24 weeks in treatment-naïve East Asian HCV-1 and non-HCV-1 patients on hemodialysis, and the overall SVR rate was 62%.129 Compared to PEG-IFN α monotherapy, combination therapy had higher SVR rates. Furthermore, the safety profiles were comparable between monotherapy and combination therapy groups.129–131 Two recent pilot studies used TVR in combination with PEG-IFN α plus low-dose RBV to treat Western HCV-1 patients on hemodialysis, and the SVR rates were superior to those using PEG-IFN α plus low-dose RBV.137,138 Whether triple therapy or all oral DAA regimen could benefit Asian patients on hemodialysis deserves further investigation.
Conclusion
HCV infection is prevalent in Asia, and the distribution of HCV genotypes varies across different Asian regions. Currently, PEG-IFN α plus RBV therapy remains the SOC in most Asian countries. PEG-IFN α-2a seems to have superior pharmacokinetics profiles to PEG-IFN α-2b, and therefore has better response rates than PEG-IFN α-2b to treat HCV infection. The optimized dose and duration of PEG-IFN α plus RBV therapy for Asian HCV patients should consider HCV genotypes, baseline viral load, and the early viral decline during the first 12 weeks of treatment. Although IL-28B genotyping is an important baseline predictor of SVR, its role in predicting SVR and determining the optimized treatment duration is less mandatory when on-treatment viral kinetics are taken into consideration. More studies are needed to personalize HCV treatment in Asian patients in terms of the use of IFN-containing triple therapy and IFN-free all oral regimens.
Disclosure
The authors report no conflicts of interest in this work.
References
Rosen HR. Clinical practice. Chronic hepatitis C infection. N Engl J Med. 2011;364(25):2429–2438. | |
Lauer GM, Walker BD. Hepatitis C virus infection. N Engl J Med. 2001;345(1):41–52. | |
Swain MG, Lai MY, Shiffman ML, et al. A sustained virologic response is durable in patients with chronic hepatitis C treated with peginterferon alfa-2a and ribavirin. Gastroenterology. 2010;139(5):1593–1601. | |
Yu ML, Lee CM, Chen CL, et al. Sustained hepatitis C virus clearance and increased hepatitis B surface antigen seroclearance in patients with dual chronic hepatitis C and B during posttreatment follow-up. Hepatology. 2013;57(6):2135–2142. | |
Manns MP, McHutchison JG, Gordon SC, et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet. 2001;358(9286):958–965. | |
Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347(13):975–982. | |
Hadziyannis SJ, Sette H Jr, Morgan TR, et al; PEGASYS International Study Group. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med. 2004;140(5):346–355. | |
Mangia A, Andriulli A. Tailoring the length of antiviral treatment for hepatitis C. Gut. 2010;59(1):1–5. | |
Di Martino V, Richou C, Cervoni JP, et al. Response-guided peg-interferon plus ribavirin treatment duration in chronic hepatitis C: meta-analyses of randomized, controlled trials and implications for the future. Hepatology. 2011;54(3):789–800. | |
Hayes CN, Imamura M, Aikata H, Chayama K. Genetics of IL28B and HCV – response to infection and treatment. Nat Rev Gastroenterol Hepatol. 2012;9(7):406–417. | |
Liu CH, Kao JH. IL28B genotype on HCV infection in Asia. Curr Hepat Rep. 2013;12(3):149–156. | |
Poordad F, McCone J Jr, Bacon BR, et al; SPRINT-2 Investigators. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364(13):1195–1206. | |
Bacon BR, Gordon SC, Lawitz E, et al; HCV RESPOND-2 Investigators. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med. 2011;364(13):1207–1217. | |
Jacobson IM, McHutchison JG, Dusheiko G, et al; ADVANCE Study Team. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med. 2011;364(25):2405–2416. | |
Zeuzem S, Andreone P, Pol S, et al; REALIZE Study Team. Telaprevir for retreatment of HCV infection. N Engl J Med. 2011;364(25):2417–2428. | |
Mohd Hanafiah K, Groeger J, et al. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57(4):1333–1342. | |
Nguyen LH, Nguyen MH. Systematic review: Asian patients with chronic hepatitis C infection. Aliment Pharmacol Ther. 2013;37(10):921–936. | |
Shiratori Y, Omata M. Predictors of the efficacy of interferon therapy for patients with chronic hepatitis C before and during therapy: how does this modify the treatment course? J Gastroenterol Hepatol. 2000;15(Suppl):E141–E151. | |
Buti M, Casado MA, Fosbrook L, et al. Cost-effectiveness of combination therapy for naive patients with chronic hepatitis C. J Hepatol. 2000;33(4):651–658. | |
Poynard T, Bedossa P, Opolon P. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet. 1997;349(9055):825–832. | |
Liu CJ. Treatment of patients with dual HCV and HBV infection: resolved and unresolved issues. J Gastroenterol Hepatol. 2014;29(1):26–30. | |
Naggie S, Sulkowski MS. Management of patients coinfected with HCV and HIV: a close look at the role for direct-acting antivirals. Gastroenterology. 2012;142(6):1324–1334. e3. | |
Lee MH, Yang HI, Lu SN, et al. Hepatitis C virus seromarkers and subsequent risk of hepatocellular carcinoma: long-term predictors from a community-based cohort study. J Clin Oncol. 2010;28(30):4587–4593. | |
Raimondi S, Bruno S, Mondelli MU, et al. Hepatitis C virus genotype 1b as a risk factor for hepatocellular carcinoma development: a meta-analysis. J Hepatol. 2009;50(6):1142–1154. | |
Lee MH, Yang HI, Lu SN, et al. Chronic hepatitis C virus infection increases mortality from hepatic and extrahepatic diseases: a community-based long-term prospective study. J Infect Dis. 2012;206(4):469–477. | |
Cammà C, Di Bona D, Schepis F, et al. Effect of peginterferon alfa-2a on liver histology in chronic hepatitis C: a meta-analysis of individual patient data. Hepatology. 2004;39(2):333–342. | |
Singal AG, Volk ML, Jensen D, et al. A sustained viral response is associated with reduced liver-related morbidity and mortality in patients with hepatitis C virus. Clin Gastroenterol Hepatol. 2010;8(3):280–288, 288. e1. | |
Iacobellis A, Perri F, Valvano MR, et al. Long-term outcome after antiviral therapy of patients with hepatitis C virus infection and decompensated cirrhosis. Clin Gastroenterol Hepatol. 2011;9(3):249–253. | |
Harris JM, Martin NE, Modi M. Pegylation: a novel process for modifying pharmacokinetics. Clin Pharmacokinet. 2001;40(7):539–551. | |
Delgado C, Francis GE, Fisher D. The uses and properties of PEG-linked proteins. Crit Rev Ther Drug Carrier Syst. 1992;9(3–4):249–304. | |
Wang YS, Youngster S, Grace M, et al. Structural and biological characterization of pegylated recombinant interferon alpha-2b and its therapeutic implications. Adv Drug Deliv Rev. 2002;54(4):547–570. | |
Foser S, Schacher A, Weyer KA, et al. Isolation, structural characterization, and antiviral activity of positional isomers of monopegylated interferon alpha-2a (PEGASYS). Protein Expr Purif. 2003;30(1):78–87. | |
Glue P, Fang JW, Rouzier-Panis R, et al. Pegylated interferon-alpha2b: pharmacokinetics, pharmacodynamics, safety, and preliminary efficacy data. Hepatitis C Intervention Therapy Group. Clin Pharmacol Ther. 2000;68(5):556–567. | |
Hoffman-La Roche Pharmaceuticals. Copegus [package insert] Nutley NJ: Hoffman-La Roche Inc, 2011. Accessed at http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/021511s023lbl.pdf on 10 April, 2014. | |
Schering Corporation. Pegintron [package insert] Kenilworth NJ: Schering Corporation 2011, Accessed at http://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/ucm094490.pdf on 10 April 2014. | |
Lamb MW, Marks IM, Wynohradnyk L, et al. 40 kDa peginterferon alfa-2a can be administered safely in patients with end-stage renal disease. Hepatology. 2001;34(Suppl):326A. | |
Xu ZX, Patel I, Joubert P. Single-dose safety/tolerability and pharmacokinetic/pharmacodynamics (PK/PD) following administrating of ascending subcutaneous doses of pegylated-interferon (PEG-IFN) and interferon α-2a (IFN α-2a) to healthy subjects. Hepatology. 1998;28(Suppl):702A. | |
Ghany MG, Strader DB, Thomas DL, et al. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology. 2009;49(4):1335–1374. | |
Martinot-Peignoux M, Stern C, Maylin S, et al. Twelve weeks posttreatment follow-up is as relevant as 24 weeks to determine the sustained virologic response in patients with hepatitis C virus receiving pegylated interferon and ribavirin. Hepatology. 2010;51(4):1122–1126. | |
Veldt BJ, Heathcote EJ, Wedemeyer H, et al. Sustained virologic response and clinical outcomes in patients with chronic hepatitis C and advanced fibrosis. Ann Intern Med. 2007;147(10):677–684. | |
van der Meer AJ, Veldt BJ, Feld JJ, et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. JAMA. 2012;308(24):2584–2593. | |
Hsu YC, Lin JT, Ho HJ, et al. Antiviral treatment for hepatitis C virus infection is associated with improved renal and cardiovascular outcomes in diabetic patients. Hepatology. Epub October 12, 2013. | |
Fried MW, Buti M, Dore GJ, et al. Once-daily simeprevir (TMC435) with pegylated interferon and ribavirin in treatment-naïve genotype 1 hepatitis C: The randomized PILLAR study. Hepatology. 2013;58(6):1918–1929. | |
Lawitz E, Mangia A, Wyles D, et al. Sofosbuvir for previously untreated chronic hepatitis C infection. N Engl J Med. 2013;368(20):1878–1887. | |
Awad T, Thorlund K, Hauser G, et al. Peginterferon alpha-2a is associated with higher sustained virological response than peginterferon alfa-2b in chronic hepatitis C: systematic review of randomized trials. Hepatology. 2010;51(4):1176–1184. | |
Chou R, Hartung D, Rahman B, Wasson N, Cottrell EB, Fu R. Comparative effectiveness of antiviral treatment for hepatitis C virus infection in adults: a systematic review. Ann Intern Med. 2013;158(2):114–123. | |
Miyase S, Haraoka K, Ouchida Y, et al. Randomized trial of peginterferon α-2a plus ribavirin versus peginterferon α-2b plus ribavirin for chronic hepatitis C in Japanese patients. J Gastroenterol. 2012;47(9):1014–1021. | |
McHutchison JG, Lawitz EJ, Shiffman ML, et al; IDEAL Study Team. Peginterferon alfa-2b or alfa-2a with ribavirin for treatment of hepatitis C infection. N Engl J Med. 2009;361(6):580–593. | |
Yu JW, Wang GQ, Sun LJ, et al. Predictive value of rapid virological response and early virological response on sustained virological response in HCV patients treated with pegylated interferon alpha-2a and ribavirin. J Gastroenterol Hepatol. 2007;22(6):832–836. | |
Kuboki M, Iino S, Okuno T, et al. Peginterferon alpha-2a (40 KD) plus ribavirin for the treatment of chronic hepatitis C in Japanese patients. J Gastroenterol Hepatol. 2007;22(5):645–652. | |
Lee H, Choi MS, Paik SW, et al. Peginterferon alfa-2a plus ribavirin for initial treatment of chronic hepatitis C in Korea. Korean J Hepatol. 2006;12(1):31–40. | |
Park SH, Park CK, Lee JW, et al. Efficacy and tolerability of peginterferon alpha plus ribavirin in the routine daily treatment of chronic hepatitis C patients in Korea: a multi-center, retrospective observational study. Gut Liver. 2012;6(1):98–106. | |
Yu ML, Dai CY, Huang JF, et al. Rapid virological response and treatment duration for chronic hepatitis C genotype 1 patients: a randomized trial. Hepatology. 2008;47(6):1884–1893. | |
Liu CH, Liu CJ, Lin CL, et al. Pegylated interferon-alpha-2a plus ribavirin for treatment-naive Asian patients with hepatitis C virus genotype 1 infection: a multicenter, randomized controlled trial. Clin Infect Dis. 2008;47(10):1260–1269. | |
Liu CJ, Chuang WL, Lee CM, et al. Peginterferon alfa-2a plus ribavirin for the treatment of dual chronic infection with hepatitis B and C viruses. Gastroenterology. 2009;136(2):496–504. e3. | |
Kogure T, Ueno Y, Fukushima K, et al. Pegylated interferon plus ribavirin for genotype Ib chronic hepatitis C in Japan. World J Gastroenterol. 2008;14(47):7225–4230. | |
Hashimoto Y, Ochi H, Abe H, et al. Prediction of response to peginterferon-alfa-2b plus ribavirin therapy in Japanese patients infected with hepatitis C virus genotype 1b. J Med Virol. 2011;83(6):981–988. | |
Hepburn MJ, Hepburn LM, Cantu NS, et al. Differences in treatment outcome for hepatitis C among ethnic groups. Am J Med. 2004;117(3):163–168. | |
Yu ML, Dai CY, Huang JF, et al. A randomised study of peginterferon and ribavirin for 16 versus 24 weeks in patients with genotype 2 chronic hepatitis C. Gut. 2007;56(4):553–559. | |
Kanda T, Imazeki F, Azemoto R, et al. Response to peginterferon-alfa 2b and ribavirin in Japanese patients with chronic hepatitis C genotype 2. Dig Dis Sci. 2011;56(11):3335–3342. | |
Sato K, Hashizume H, Yamazaki Y, et al. Response-guided peginterferon-alpha-2b plus ribavirin therapy for chronic hepatitis C patients with genotype 2 and high viral loads. Hepatol Res. 2012;42(9):854–863. | |
Kagawa T, Kojima S, Shiraishi K, et al. Weight-based high- and low-dose ribavirin in combination with peginterferon α-2b therapy for genotype 2 chronic hepatitis C: a randomized trial. Hepatol Res. 2012;42(4):351–358. | |
Varghese R, Al-Khaldi J, Asker H, et al. Treatment of chronic hepatitis C genotype 4 with peginterferon alpha-2a plus ribavirin. Hepatogastroenterology. 2009;56(89):218–222. | |
Derbala MF, El Dweik NZ, Al Kaabi SR, et al. Viral kinetic of HCV genotype-4 during pegylated interferon alpha 2a: ribavirin therapy. J Viral Hepat. 2008;15(8):591–599. | |
Shobokshi OA, Serebour FE, Skakni L, et al. Combination therapy of peginterferon alfa-2a (40KD) (PEGASYS®) and ribavirin (COPEGUS®) significantly enhance sustained virological and biochemical response rate in chronic hepatitis C genotype 4 patients in Saudi Arabia. Hepatology. 2003;38(4 Suppl 1):636A. | |
Alfaleh FZ, Hadad Q, Khuroo MS, et al. Peginterferon alpha-2b plus ribavirin compared with interferon alpha-2b plus ribavirin for initial treatment of chronic hepatitis C in Saudi patients commonly infected with genotype 4. Liver Int. 2004;24(6):568–574. | |
Hasan F, Asker H, Al-Khaldi J, et al. Peginterferon alfa-2b plus ribavirin for the treatment of chronic hepatitis C genotype 4. Am J Gastroenterol. 2004;99(9):1733–1737. | |
Zhou YQ, Wang XH, Hong GH, et al. Twenty-four weeks of pegylated interferon plus ribavirin effectively treat patients with HCV genotype 6a. J Viral Hepat. 2011;18(8):595–600. | |
Fung J, Lai CL, Hung I, et al. Chronic hepatitis C virus genotype 6 infection: response to pegylated interferon and ribavirin. J Infect Dis. 2008;198(6):808–812. | |
Seto WK, Tsang OT, Liu K, et al. Role of IL28B and inosine triphosphatase polymorphisms in the treatment of chronic hepatitis C virus genotype 6 infection. J Viral Hepat. 2013;20(7):470–477. | |
Tangkijvanich P, Komolmit P, Mahachai V, et al. Response-guided therapy for patients with hepatitis C virus genotype 6 infection: a pilot study. J Viral Hepat. 2012;19(6):423–430. | |
Lam KD, Trinh HN, Do ST, et al. Randomized controlled trial of pegylated interferon-alfa 2a and ribavirin in treatment-naive chronic hepatitis C genotype 6. Hepatology. 2010;52(5):1573–1580. | |
Thu Thuy PT, Bunchorntavakul C, Tan Dat H, et al. A randomized trial of 48 versus 24 weeks of combination pegylated interferon and ribavirin therapy in genotype 6 chronic hepatitis C. J Hepatol. 2012;56(5):1012–1018. | |
Zeuzem S, Herrmann E, Lee JH, et al. Viral kinetics in patients with chronic hepatitis C treated with standard or peginterferon alpha2a. Gastroenterology. 2001;120(6):1438–1447. | |
Fried MW, Hadziyannis SJ, Shiffman ML, et al. Rapid virological response is the most important predictor of sustained virological response across genotypes in patients with chronic hepatitis C virus infection. J Hepatol. 2011;55(1):69–75. | |
Jensen DM, Morgan TR, Marcellin P, et al. Early identification of HCV genotype 1 patients responding to 24 weeks peginterferon alpha-2a (40 kd)/ribavirin therapy. Hepatology. 2006;43(5):954–960. | |
Moreno C, Deltenre P, Pawlotsky JM, et al. Shortened treatment duration in treatment-naive genotype 1 HCV patients with rapid virological response: a meta-analysis. J Hepatol. 2010;52(1):25–31. | |
Khattab MA, Ferenci P, Hadziyannis SJ, et al. Management of hepatitis C virus genotype 4: recommendations of an international expert panel. J Hepatol. 2011;54(6):1250–1262. | |
Mangia A, Minerva N, Bacca D, et al. Individualized treatment duration for hepatitis C genotype 1 patients: a randomized controlled trial. Hepatology. 2008;47(1):43–50. | |
Liu CH, Liang CC, Liu CJ, et al. Interleukin 28B genetic polymorphisms play a minor role in identifying optimal treatment duration in HCV genotype 1 slow responders to pegylated interferon plus ribavirin. Antivir Ther. 2012;17(6):1059–1067. | |
Omata M, Kanda T, Yu M-L, et al. APASL consensus statements and management algorithms for hepatitis C virus infection. Hepatol Int. 2012;6(2):409–435. | |
Farnik H, Lange CM, Sarrazin C, et al. Meta-analysis shows extended therapy improves response of patients with chronic hepatitis C virus genotype 1 infection. Clin Gastroenterol Hepatol. 2010;8(10):884–890. | |
Cheinquer H, Shiffman ML, Zeuzem S, et al. The outcome of 24 vs 48 weeks of peginterferon alfa-2a (40 KD) plus ribavirin on sustained virologic response rates in patients infected with genotype 2 or 3 hepatitis C virus who do not achieve a rapid viral response: the N-CORE study. Hepatology. 2012;56(Suppl 1):271A–272A. | |
Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461(7262):399–401. | |
Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet. 2009;41(10):1100–1104. | |
Tanaka Y, Nishida N, Sugiyama M, et al. Genome-wide association of IL28B with response to pegylated interferon-alpha and ribavirin therapy for chronic hepatitis C. Nat Genet. 2009;41(10):1105–1109. | |
Rauch A, Kutalik Z, Descombes P, et al. Genetic variation in IL28B is associated with chronic hepatitis C and treatment failure: a genome-wide association study. Gastroenterology. 2010;138(4):1338–1345, 1345. e1–e7. | |
Chen Y, Xu HX, Wang LJ, et al. Meta-analysis: IL28B polymorphisms predict sustained viral response in HCV patients treated with pegylated interferon-α and ribavirin. Aliment Pharmacol Ther. 2012;36(2):91–103. | |
Rangnekar AS, Fontana RJ. Meta-analysis: IL-28B genotype and sustained viral clearance in HCV genotype 1 patients. Aliment Pharmacol Ther. 2012;36(2):104–114. | |
Schreiber J, Moreno C, Garcia BG, et al. Meta-analysis: the impact of IL28B polymorphisms on rapid and sustained virological response in HCV-2 and -3 patients. Aliment Pharmacol Ther. 2012;36(4):353–362. | |
Balagopal A, Thomas DL, Thio CL. IL28B and the control of hepatitis C virus infection. Gastroenterology. 2010;139(6):1865–1876. | |
Thomas DL, Thio CL, Martin MP, et al. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nature. 2009;461(7265):798–801. | |
Thompson AJ, Muir AJ, Sulkowski MS, et al. Interleukin-28B polymorphism improves viral kinetics and is the strongest pretreatment predictor of sustained virologic response in genotype 1 hepatitis C virus. Gastroenterology. 2010;139(1):120–129. e18. | |
Lin CY, Chen JY, Lin TN, et al. IL28B SNP rs12979860 is a critical predictor for on-treatment and sustained virologic response in patients with hepatitis C virus genotype-1 infection. PLoS One. 2011;6(3):e18322. | |
Huang CF, Huang JF, Yang JF, et al. Interleukin-28B genetic variants in identification of hepatitis C virus genotype 1 patients responding to 24 weeks peginterferon/ribavirin. J Hepatol. 2012;56(1):34–40. | |
Huang CF, Yeh ML, Huang JF, et al. Host interleukin-28B genetic variants versus viral kinetics in determining responses to standard-of-care for Asians with hepatitis C genotype 1. Antiviral Res. 2012;93(2):239–244. | |
Yu ML, Huang CF, Huang JF, et al. Role of interleukin-28B polymorphisms in the treatment of hepatitis C virus genotype 2 infection in Asian patients. Hepatology. 2011;53(1):7–13. | |
Liu CH, Liang CC, Liu CJ, et al. Interleukin 28B genetic polymorphisms and viral factors help identify HCV genotype-1 patients who benefit from 24-week pegylated interferon plus ribavirin therapy. Antivir Ther. 2012;17(3):477–484. | |
Chen MY, Liu CH, Chen TC, et al. Value of interleukin-28B genetic polymorphism on retreatment outcomes of chronic hepatitis C genotype 1 relapsers by peginterferon alfa plus ribavirin. J Gastroenterol Hepatol. 2014;29(1):102–109. | |
Yu ML, Liu CH, Huang CF, et al. Revisiting the stopping rule for hepatitis C genotype 1 patients treated with peginterferon plus ribavirin. PLoS One. 2012;7(12):e52048. | |
Huang CF, Yu ML, Kao JH, et al. Profound week 4 interferon responsiveness is mandatory for hepatitis C genotype 1 patients with unfavorable IL-28B genotype. J Clin Virol. 2013;56(4):293–298. | |
Mangia A, Thompson AJ, Santoro R, et al. An IL28B polymorphism determines treatment response of hepatitis C virus genotype 2 or 3 patients who do not achieve a rapid virologic response. Gastroenterology. 2010;139(3):821–827, 827.e1. | |
Sarrazin C, Susser S, Doehring A, et al. Importance of IL28B gene polymorphisms in hepatitis C virus genotype 2 and 3 infected patients. J Hepatol. 2011;54(3):415–421. | |
Huang CF, Dai CY, Yeh ML, et al. Virological predictors of response to retreatment in hepatitis C genotype 2 infected patients. PLoS One. 2013;8(3):e58882. | |
Stättermayer AF, Stauber R, Hofer H, et al. Impact of IL28B genotype on the early and sustained virologic response in treatment-naïve patients with chronic hepatitis C. Clin Gastroenterol Hepatol. 2011;9(4):344–350. e2. | |
Hu KQ, Alvarez Bognar F, Thompson S, et al. Boceprevir (BOC) plus peginterferon alfa-2b/ribavirin (PR) in the treatment of chronic hepatitis C virus genotype-1 (HCV-G1) infected Asian patients in the SPRINT-1, SRPINT-2, and RESPOND-2 trials. Hepatol Int. 2012;6(1):164. | |
Akuta N, Suzuki F, Hirakawa M, et al. Amino acid substitution in hepatitis C virus core region and genetic variation near the interleukin 28B gene predict viral response to telaprevir with peginterferon and ribavirin. Hepatology. 2010;52(2):421–429. | |
Suzuki F, Suzuki Y, Sezaki H, et al. Exploratory study on telaprevir given every 8 h at 500 mg or 750 mg with peginterferon-alpha-2b and ribavirin in hepatitis C patients. Hepatol Res. 2013;43(7):691–701. | |
Chayama K, Hayes CN, Abe H, et al. IL28B but not ITPA polymorphism is predictive of response to pegylated interferon, ribavirin, and telaprevir triple therapy in patients with genotype 1 hepatitis C. J Infect Dis. 2011;204(1):84–93. | |
Kumada H, Toyota J, Okanoue T, et al. Telaprevir with peginterferon and ribavirin for treatment-naive patients chronically infected with HCV of genotype 1 in Japan. J Hepatol. 2012;56(1):78–84. | |
Hayashi N, Okanoue T, Tsubouchi H, et al. Efficacy and safety of telaprevir, a new protease inhibitor, for difficult-to-treat patients with genotype 1 chronic hepatitis C. J Viral Hepat. 2012;19(2):e134–e142. | |
Akuta N, Suzuki F, Seko Y, et al. Determinants of response to triple therapy of telaprevir, peginterferon, and ribavirin in previous non-responders infected with HCV genotype 1. J Med Virol. 2012;84(7):1097–1105. | |
Furusyo N, Ogawa E, Nakamuta M, et al. Telaprevir can be successfully and safely used to treat older patients with genotype 1b chronic hepatitis C. J Hepatol. 2013;59(2):205–212. | |
Ogawa E, Furusyo N, Nakamuta M, et al. Telaprevir-based triple therapy for chronic hepatitis C patients with advanced fibrosis: a prospective clinical study. Aliment Pharmacol Ther. 2013;38(9):1076–1085. | |
Tsubota A, Shimada N, Atsukawa M, et al. Impact of IL28B polymorphisms on 24-week telaprevir-based combination therapy for Asian chronic hepatitis C patients with HCV genotype 1b. J Gastroenterol Hepatol. 2014;29(1):144–150. | |
Liu S, Cipriano LE, Holodniy M, et al. New protease inhibitors for the treatment of chronic hepatitis C: a cost-effectiveness analysis. Ann Intern Med. 2012;156(4):279–290. | |
Huang YT, Jen CL, Yang HI et al. Lifetime risk and sex difference of hepatocellular carcinoma among patients with chronic hepatitis B and C. J Clin Oncol. 2011;29(27):3643–3650. | |
Liu CH, Kao JH. Treatment of hepatitis C virus infection in patients with end-stage renal disease. J Gastroenterol Hepatol. 2011;26(2):228–239. | |
Rostaing L, Chatelut E, Payen JL, et al. Pharmacokinetics of alphaIFN-2b in chronic hepatitis C virus patients undergoing chronic hemodialysis or with normal renal function: clinical implications. J Am Soc Nephrol. 1998;9(12):2344–2348. | |
Chan TM, Ho SK, Tang CS, et al. Pilot study of pegylated interferon-alpha 2a in dialysis patients with chronic hepatitis C virus infection. Nephrology (Carlton). 2007;12(1):11–17. | |
Kojima A, Kakizaki S, Hosonuma K, et al. Interferon treatment for patients with chronic hepatitis C complicated with chronic renal failure receiving hemodialysis. J Gastroenterol Hepatol. 2013;28(4):690–699. | |
Alsaran K, Sabry A, Shaheen N. et al. Pegylated interferon alpha-2a for treatment of chronic HCV infection in hemodialysis patients: a single Saudi center experience. Int Urol Nephrol. 2011;43(3):865–873. | |
Kokoglu OF, Uçmak H, Hosoglu S, et al. Efficacy and tolerability of pegylated-interferon alpha-2a in hemodialysis patients with chronic hepatitis C. J Gastroenterol Hepatol. 2006;21(3):575–580. | |
Ayaz C, Celen MK, Yuce UN, et al. Efficacy and safety of pegylated-interferon alpha-2a in hemodialysis patients with chronic hepatitis C. World J Gastroenterol. 2008;14(2):255–259. | |
Kose S, Gurkan A, Akman F, et al. Treatment of hepatitis C in hemodialysis patients using pegylated interferon alpha-2a in Turkey. J Gastroenterol. 2009;44(4):353–358. | |
Liu CH, Liang CC, Lin JW, et al. Pegylated interferon alpha-2a versus standard interferon alpha-2a for treatment-naive dialysis patients with chronic hepatitis C: a randomised study. Gut. 2008;57(4):525–530. | |
Liu CH, Liang CC, Liu CJ, et al. Pegylated interferon alfa-2a monotherapy for hemodialysis patients with acute hepatitis C. Clin Infect Dis. 2010;51(5):541–549. | |
Tan SS, Abu Hassan MR, Abdullah A, Ooi BP, Korompis T, Merican MI. Safety and efficacy of an escalating dose regimen of pegylated interferon alpha-2b in the treatment of haemodialysis patients with chronic hepatitis C. J Viral Hepat. 2010;17(6):410–418. | |
Tseng PL, Chen TC, Chien YS, et al. Efficacy and safety of pegylated interferon alfa-2b and ribavirin combination therapy versus pegylated interferon monotherapy in hemodialysis patients: a comparison of 2 sequentially treated cohorts. Am J Kidney Dis. 2013;62(4):789–795. | |
Liu CH, Huang CF, Liu CJ, et al. Pegylated interferon-α2a with or without low-dose ribavirin for treatment-naive patients with hepatitis C virus genotype 1 receiving hemodialysis: a randomized trial. Ann Intern Med. 2013;159(11):729–738. | |
Liu CH, Huang CF, Liu CJ, et al. Peginterferon alfa-2a with or without low dose ribavirin for hemodialysis patients with hepatitis C virus genotype 2 infection: a randomized trial. Hepatology. 2013;58(Suppl 1):1096A. | |
Gordon CE, Uhlig K, Lau J, et al. Interferon treatment in hemodialysis patients with chronic hepatitis C virus infection: a systematic review of the literature and meta-analysis of treatment efficacy and harms. Am J Kidney Dis. 2008;51(2):263–277. | |
Fabrizi F, Dixit V, Messa P, et al. Pegylated interferon monotherapy of chronic hepatitis C in dialysis patients: Meta-analysis of clinical trials. J Med Virol. 2010;82(5):768–775. | |
Gupta SK, Kantesaria B, Glue P. Pharmacokinetics, safety, and tolerability of ribavirin in hemodialysis-dependent patients. Eur J Clin Pharmacol. 2012;68(4):415–418. | |
Brennan BJ, Wang K, Blotner S, et al. Safety, tolerability, and pharmacokinetics of ribavirin in hepatitis C virus-infected patients with various degrees of renal impairment. Antimicrob Agents Chemother. 2013;57(12):6097–6105. | |
Liu CH, Liang CC, Liu CJ, et al. Pegylated interferon alpha-2a plus low-dose ribavirin for the retreatment of dialysis chronic hepatitis C patients who relapsed from prior interferon monotherapy. Gut. 2009;58(2):314–316. | |
Dumortier J, Guillaud O, Gagnieu MC, et al. Anti-viral triple therapy with telaprevir in haemodialysed HCV patients: is it feasible? J Clin Virol. 2013;56(2):146–149. | |
Basu PP, Siriki R, Shah NJ, et al. Telaprevir with adjusted dose of ribavirin in naïve CHC-G1: efficacy and treatment in CHC in hemodialysis population. J Hepatol. 2013;58(Suppl 1):S30–S31. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.