Back to Journals » Clinical Interventions in Aging » Volume 16
Mirabegron Alleviates the Degree of Burden Experienced by Caregivers of Older Females with Mixed or Urge Incontinence: A Prospective Study
Authors Zachariou A , Filiponi M, Kaltsas A , Dimitriadis F , Champilomatis I, Paliouras A, Tsounapi P, Mamoulakis C , Takenaka A, Sofikitis N
Received 25 October 2020
Accepted for publication 12 January 2021
Published 16 February 2021 Volume 2021:16 Pages 291—299
DOI https://doi.org/10.2147/CIA.S283737
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Athanasios Zachariou,1,2 Maria Filiponi,2 Aris Kaltsas,1,2 Fotios Dimitriadis,3 Ioannis Champilomatis,1 Athanasios Paliouras,1 Panagiota Tsounapi,4 Charalampos Mamoulakis,5 Atsushi Takenaka,4 Nikolaos Sofikitis1
1Urology Department, School of Medicine, Ioannina University, Ioannina, Greece; 2Incontinence Unit, Physical Medicine and Rehabilitation Centre EU PRATTEIN, Volos, Greece; 3 1st Urology Department, School of Medicine, Aristotle University, Thessaloniki, Greece; 4Urology Department, School of Medicine, Tottori University, Yonago, Japan; 5Urology Department, Medical School, University of Crete, Heraklion, Greece
Correspondence: Athanasios Zachariou
Urology Department, Ioannina University, 3 Spyridi Street, Volos, 38221, Greece
Tel +306937227778
Fax +302421026932
Email [email protected]
Purpose: Older people, especially women, have the highest known prevalence of urinary incontinence (UI) of any other age-group. Continual care provision for elderly incontinent females is an incredibly arduous process, yet only very few studies have investigated the issue. Aim of the study was to evaluate the impact of mirabegron’s treatment on the degree of burden experienced by caregivers of elderly female patients with UI.
Patients and Methods: A hundred and eighty-six caregivers of older females with mixed or urgency UI besides various conditions (strokes, post-operative recovery after major surgery, etc.) were included in the study. Group A comprised 91 patients that did not want to receive any treatment for UI. Group B consisted of 95 elderly females treated for UI with mirabegron 50 mg/daily for three months. All caregivers completed the Zarit Burden Scale (ZBS) questionnaire at the outset and after the three months. All patients completed a bladder diary at the beginning and at the end of the observation/medication period.
Results: Patients receiving mirabegron presented a statistically significant improvement in UI parameters. Their caregivers showed a statistically significant decrease in the ZBS total score as well as separate domains.
Conclusion: This pilot study confirms that mirabegron administration can improve the quality of life of older females suffering from UI while substantially relieving caregiver burden. Recognizing the physical and emotional reactions of caregivers may help health providers deliver better support and resources to meet the needs of caregivers and patients alike.
Keywords: mirabegron, caregiver, Zarit Burden Scale, aging, incontinence
Introduction
The life expectancy of older adults has significantly increased due to better living conditions and improved medical treatment.1 Despite the potential to live longer and with a good quality of life, many older patients, suffer from multiple diseases, such as Alzheimer’s disease, urinary incontinence (UI), etc. Such pathologic conditions are responsible for an ever-increasing demand for health and social care. Yet, these demands are increasingly more difficult to meet due to a corresponding reduction in the number of younger people available to care for the dependent elderly.
Incontinence in elderly adults is costly to manage, a significant risk factor for falls, depression, social isolation, skin breakdown, admission to a nursing home or hospital, and is independently associated with mortality.2 Although the etiology of UI in older adults is multifaceted, there are some functional causes, eg, the inability to reach and use the toilet due to cognitive or physical impairments.2
Care provision for the elderly imposes physical, psychological, financial and social burden on caregivers.3–6 Susceptible to a host of variables such as gender, resource availability, social status, and personal expectations, it is a complex issue which demands a better understanding. Caregivers are an indispensable, yet often unrecognized, social asset. Reducing their burden should be the subject of greater scrutiny and effort by researchers and health policymakers alike.7,8
Elderly adults with incontinence often require changes in family structure, depending on the severity of the condition or the coexistence of other diseases.9,10 Moreover, caring for older adults has not only financial implications for families but it also affects the society as a whole. Nonetheless, constant care provision requires arduous effort which can lead to numerous problems for the caregiver such as increased psychiatric morbidity (ie, depression), strain on interpersonal relationships, social isolation and a loss of self-worth.11 Caregivers subjected to inordinate levels of sustained burden invariably display a deterioration in the quality of their work. This often results in their patients being institutionalize in long-term care facilities and all the social and financial issues that this entails.12,13
Mirabegron, the first b3 adrenoceptor agonist, approved for the treatment of urgency and urge incontinence. It provides an alternative to antimuscarinic therapy and has a unique mechanism of action and side effect profile.14 It facilitates bladder filling and prolongs the storage phase by activating the b3 adrenoceptors thereby improving the functional performance of UI patients, and delays the onset of behavioral symptoms. Mirabegron, alone or in combination with solifenacin, is also a well tolerated and effective treatment for patients with overactive bladder (OAB) symptoms, irrespective of their age.15
This is a pilot study, aiming to test the hypothesis that UI treatment of elderly female patients could be associated with a decrease in caregivers’ burden. The primary objective was to implement and evaluate the impact of mirabegron’s treatment on the burden, experienced by caregivers of elderly females with mixed or urges UI. Secondary objective was to identify which outcomes are being affected by the intervention.
Patients and Methods
The study was carried out between January 2016 and May 2019 in urban areas of Volos, the capital city of the Magnesia prefecture, Greece. Two hundred and twenty-four caregivers and their female patients were initially selected from the records of a Physical Medicine and Rehabilitation Centre EU PRATTEIN to enroll in the study. Participants represented a convenience sample. In other words, inclusion criteria were not entirely random and involved easy access in terms of patient homes and patient/caregiver communication. Social workers, aiming to enhance overall well-being of female patients, recorded data during their scheduled visits.
The Zarit Burden Scale (ZBS) was used to evaluate all caregivers, both at the beginning of the three-month-long study and its conclusion. The ZBS is established as one of the most widely used scales for burden assessment in caregivers of elderly patients with dementia and related disorders.16 It was originally developed for clinical and research purposes, with a focus on elderly patients with dementia and their relatives. ZBS items are comprehensive and deal with many dimensions, common to several other mental and physical illnesses. Accordingly, the ZBS has been used to assess the burden of caregivers of elderly patients, suffering from stroke, chronic diseases, incontinence and so forth.5,17–19 The ZBS is used in many countries besides the USA, where it was initially developed and is translated as well as validated in several languages, including Greek.12 Scoring ranges from 0 to 88, with higher scores corresponding to higher levels of caregiver burden. The burden threshold was set at a score of 29. Caregivers registering a score below this value were not considered to have a burden (Supplementary Figure 1).
Caregivers staying with their patients and providing care for at least ten hours per day, willing to engage in study procedures and speaking the Greek language fluently were recruited for the study. Caregivers with major diseases or mental disorders, planning to miss more than one week during the study period, or issue incomplete questionnaires and/or exhibit inadequate diligence due to communication or access obstacles were excluded.
The elderly female patients were evaluated with the Mini-Mental State Examination (MMSE) to measure their cognitive impairment and screen for dementia. All patients were evaluated by a urologist. To confirm and classify the diagnosis of urge or mixed diagnosis, a simple 3-item, self-administered questionnaire (the 3 Incontinence Questions – 3IQ) was administered to all female patients. The 3IQ has demonstrated good sensitivity and specificity in distinguishing between different types of incontinence, compared with an extended classical evaluation.20
The inclusion criteria for affected individuals included: an MMSE score >24 or more, absence of cognitive impairment or dementia, willingness/ability to engage in study procedures and Greek language fluency. Patients planning to miss more than one week during the study period, or who presented behavioral and physical issues that would be disruptive or dangerous to themselves or others excluded from the study. Patients who had a terminal illness (life expectancy less than a year), unable to walk with help to reach and use the toilet, or presented urinary tract infection were also excluded.
Concerning medication in both groups, the concurrent use of 6 medications or more per person was added as an extra exclusion criterion. All patients reported that they were using five or fewer drugs concomitantly. Depending on their popularity, mention can be made of beta-adrenergic blockers, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor antagonists (ARAIIs), diuretics, calcium channel blockers, and vasodilators. As expected, the more common medications of our female patients were concerning cardiovascular diseases. There were no significant differences concerning the treatment that was delivered to both groups.
The sample size was calculated using the probability of prevalence of urge and mixed urinary incontinence in Greece which was assumed up to 14%.21 This yielded 159 pairs but it was added 10% for lost on follow-up and 10% for non-response. Thus, the final sample size worked out to be more than 192 pairs (patients and caregivers) and we collected 224 pairs.
The control population (Group Α) comprised 91 caregivers of elderly females presenting urge or mixed urinary incontinence and suffering from various medical conditions (strokes, post-operative recovery after major orthopedic surgery, Parkinson’s disease, etc.). All patients of this group had no intention of receiving UI treatment owing to polypharmacy, coexistent diseases, etc.
Group B consisted of 95 caregivers whose elderly female patients also presented with mixed or urge UI. Their underlying medical conditions varied in a similar way to those of Group A. However, a UI treatment of mirabegron 50 mg/daily was administered to these patients for three months. An appropriate non-random convenience sampling was undertaken.
All-female patients used hypoallergenic absorbent products and pads. In an attempt to ameliorate the quality of life for patients and caregivers alike, comprehensive information about UI and commercially available incontinence products was provided to all participants.
Patients in control group A were required to complete a 3-day micturition diary at the beginning and end of the three-month period. Group B patients were required to complete a 3-day micturition diary before and after mirabegron treatment. Voiding frequency and volume, nocturia, urgency episodes, incontinence episodes and the number of incontinence pads were all parameters recorded.
Additional information about the caregivers in each group, including sociodemographic characteristics such as age, body weight, gender, accommodation, employment status and length of time acting as a caregiver were obtained. We considered all variables in sociodemographic characteristics as potential factors that might affect ZBS scores.
Ethical and access approvals for study procedures were obtained from the Institutional Review Board, and all participants provided written informed consent before the interviews.
All outcome variables were tested for normality using the Shapiro–Wilk W-test. Concretely, the following continues/ordinal variables were tested per group: age of caregiver, body weight, caregiving duration; pre- and post-study period ZBS domains (personal intensity, intensity of role, deprivation of relationships and management of care), total ZBS score and percentage (%) improvement in total score of burden; pre- and post-study period episodes of frequency, urgency, nocturia, incontinence; number of pads; and voided volumes. The potential presence of any relationship was examined between the % of improvement in UI and in caregiver burden (independently for each parameter) in order to assess the outcome of UI improvement on caregivers’ burden. Data were analyzed using SPSS Statistics for Windows, Version 24.0. Two-tailed p<0.050 was considered significant.
Results
All tested variables showed significant departures from the normal distribution. There were two exceptions; the pre- and post-study period total scores of ZBS and the voided volumes. Therefore, non-parametric tests (Mann Whitney U-test and Wilcoxon signed-rank test) were accomplished for all evaluations (for uniformity purposes) between and within groups, respectively; all data are presented as medians (interquartile ranges; IQRs).
Of the 115 elderly females from Group A, 17 failed to fill a three-day micturition test at the end of observation period. Consequently, they were excluded from the study. Ten patients were excluded from Group B (Mirabegron Group) for the same reason. As 11 caregivers (7 from Group A, 4 from Group B) failed to complete their ZBS questionnaire at the end of the study, they too were excluded. No adverse reactions or effects from the administration of mirabegron were reported in any of the Group B patients (Figure 1).
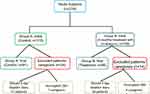 |
Figure 1 Study Flow Diagram Illustrating the Process of Screening and Selecting Patients and Caregivers. |
Demographic characteristics of caregivers and baseline UI parameters had no significant differences between groups (Table 1, Table 3). Twenty percent of females in Group B that were incontinent at primary evaluation became continent by the study-endpoint and used pads occasionally due to psychological reasons. Moreover, a significant improvement in all urinary outcomes, all ZBS domain scores and the total score was recorded in Group B after a 3-month study period. This was not presented in patients of Group A (Tables 2–4). Spearman’s rank-order correlation test produced statistically significant (positive) correlations between % improvement in incontinence parameters and % improvement in burden scale. Such correlations (frequency-deprivation of relationships, urgency-management of care, incontinence-management of care, pads-intensity of role/management of care; Table 5) were exclusively in Group B.
 |
Table 1 Demographic Parameters and Baseline Characteristics of Participants (P<0.05) |
 |
Table 2 Urinary Parameters of the Older Female Patients (P<0.05) |
 |
Table 3 Distribution of the Individual Scales and the Overall ZBS Total Score in Observation Group A (P<0.05) |
 |
Table 4 Comparison of Pre-Treatment and Post-Treatment ZBS in Group B (P<0.05) |
 |
Table 5 Correlation Between Improvements (%) in Incontinence Parameters and ZBS Domains and Total Score (P<0.05) |
A significant ratio of caregivers suffering from a “severe burden” (p<0.05), which corresponds to a score of 61–88 in the Zarit scale, was noted in Group A after the 3 months’ observation study. Therefore, while the rates in Group B are 26.3%, in Group A the rate reaches approximately 37.4%. In practical terms, this means that 1 out of 3 caregivers experience a severe burden. The elderly females without UI treatment required greater care and constant cleaning. The higher burden rates of caregivers in group A suggest that there is probably a negative synergistic effect between incontinence and other factors making difficult for caregivers to deal with the ordeals of UI.
Discussion
The aim of the current study was to determine the impact of mirabegron treatment of elderly females with incontinence on their caregiver’s burden. Our data demonstrate that the simple strategy of UI treatment with mirabegron 50 mg per day has a significant mitigating effect. It is noteworthy that caregivers of Group A, at the end of the observation, demonstrated high rates of ZBS, thereby indicating that incontinence augments caregivers’ burden. Our data is in agreement with Tamanini et al and with our own previous findings.13,19
There are social taboos associated with incontinence, and the level of shame or embarrassment among incontinent patients are much higher than those reported for depression and cancer.22 Many elderly patients are still nowadays not correctly informed about the causes of and treatments for UI. Furthermore, affected individuals continue to believe that incontinence is an inevitable consequence of age and only seek help when the situation becomes untenable.23 The prevalence of UI in residential settings ranges between 50% and 80%. UI is a significant predictor of institutionalization and of death among the institutionalized elderly people.24
Caregivers attending UI patients are generally responsible for cleaning and washing them, along with their clothes and bed sheets. Caregivers work in intimate settings such as the bedroom or bathroom, and their assistance is required at varying frequencies. Much depends on the mobility of incontinent patients and whether they are capable of making a visit to the toilet unassisted or getting out of bed on their own accord.
As a result of their caring role, many caregivers reported physical, emotional and practical challenges in managing UI topics. The more common physical problems described by caregivers worldwide are back pain, tendinitis, and tiredness. These are symptomatic of the physical efforts required to move and clean their elderly disabled patients.25 Furthermore, in comparison with control subjects of the same age and gender, caregivers have a higher risk of premature death.26 Fatigue, along with sleep and leisure deprivation are among the most commonly cited consequences of incontinence management.27,28 Emotional problems were often the result of anxiety, undue tension, sadness, and strain. These are also associated with the unceasing need for continuous watchfulness and a substantial effort to keep clean the old incontinent patients, which make caregivers feel stressed and tired.29,30 Practical issues include, for example, having to stand the reek of urine and having to contend with unsuitable or inadequate continence devices. Managing incontinence also causes considerable disruption to caregiver work patterns as they are obliged to schedule all other tasks around pad changes.
There are specific strategies to help caregivers deal with such issues. Some advocate the use of high-quality absorbent products to prevent bedwetting and embarrassment during social interaction.27 Others underline the importance of affording caregivers the opportunity to ask confidential questions to nursing specialists, counseling agencies, support groups or consult online educational resources to protect caregiver wellbeing.
Only a few papers describe the effect of incontinence on caregiver quality of life.3,5,13,19,29,31–34 Most are qualitative studies referring to a small number of caregivers, typically less than 25. Our study evaluated 186 pairs (caregivers and older female patients) and is one of the few, according to our knowledge, proposing that incontinence treatment with mirabegron will alleviate the caregivers’ burden. Our data come in agreement with findings made by Cassells and Watt.29 They stated that simple nursing intervention or minimal medical care could make a substantial difference in reducing caregiver burden and improving patient quality of life.29
Prior to this study, most of the informal caregivers recruited had no information or third party support specific to the needs of UI patients. Given the physical and psychological risks caregivers face, if no proper support is provided, the consequences could be dire. Additionally, our findings suggest that if an awareness program was instigated so as to make UI patients more aware of the condition, it would also help relieve caregiver burden. Policymakers should be made more aware of the issues involved, especially for caregivers, upon whom the responsibility for looking after our ageing population rests. Pharmacologic therapy of UI in older and frail patients seems to be an exciting alternative.
Limitation of the study is the non-random sampling method used. A deliberate and non-random convenience sample allows the easy identification and use of all available populations for the study. Furthermore, as the questionnaire employed in the study was of a self-assessment type, we cannot objectively verify the validity and reliability of responses. Thus, the scoring was dependent on the caregiver’s perception of burden, which may or may not be an accurate indicator of the actual burden caused by the UI patient. The researcher cannot be sure about the reliability of responses on sensitive issues that constitute health care provision.
The lack of a general homogeneous, appropriate support and the widespread, stigma-driven silence on UI, confirms just how difficult the situation can be for caregivers. In most cases, they cannot count on strong public or private support network or appropriate information.
Conclusion
This pilot study confirms the hypothesis that UI treatment of older female patients would be associated with a decrease in caregivers’ burden. Mirabegron administration can improve the quality of life of elderly females suffering from UI while substantially relieving caregiver burden. Recognizing the physical and emotional reactions of caregivers caring for patients with UI may help health providers deliver better support and resources to meet the needs of caregivers and patients alike.
Abbreviations
UI, urinary incontinence; ZBS, Zarit Burden Scale; OAB, overactive bladder; MMSE, Mini-Mental State Examination; IQRs, interquartile ranges; 3IQ, 3 Incontinence Questions.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Ethics Approval
Ethical and access approvals for study procedures were obtained from the Institutional Review Board of the Rehabilitation Centre EU PRATTEIN. The study was conducted in accordance with the declaration of Helsinki, and participants provided written informed consent before the interviews.
Disclosure
The authors report no conflicts of interest for this work. The abstract of preliminary data of this paper was presented at the 47th Annual Meeting of the International Continence Society (abstract 503) as a poster presentation with interim findings, https://www.ics.org/2017/abstract/503.
References
1. Crimmins EM. Lifespan and Healthspan: past, Present, and Promise. Gerontologist. 2015;55(6):901–911.
2. Wagg AS, Kung Chen L, Johnson T, et al. Incontinence in frail older persons. Abrams P, Cardozo L, Wagg A, Wein A, editors. Continence, Arnheim.
3. Di Rosa M, Lamura G. The impact of incontinence management on informal caregivers’ quality of life. Aging Clin Exp Res. 2016;28:89–97.
4. Noimark D, Steventon N, Wagg A. A qualitative study of the impact of caring for a person with urinary incontinence. Neurour Urodyn. 2009;28:637–638.
5. Gotoh M, Matsukawa Y, Yoshikawa Y, et al. Impact of urinary incontinence on the psychological burden of family caregivers. Neurour Urodyn. 2009;28:492–496.
6. Pinquart M, Sorensen S. Differences between caregivers and non-caregivers in psychological health and physical health: a meta-analysis. Psychology and Ageing. 2003;18:250–267.
7. Gaugler JE, Given WC, Linder J, et al. Work, gender and stress in family cancer caring. Support Care Cancer. 2008;16:347–357.
8. Schulz R, Martire LM. Caregiver burden. Family caregiving of persons with dementia: prevalence, health effects, and support strategies. Am J Geriatr Psychiatry. 2004;12:240–249.
9. Ahn S, Hochhalter AK, Moudouni DKM, et al. Self-reported physical and mental health of older adults: the roles of caregiving and resources. Maturitas. 2012;71:62–69.
10. Alberts NM, Hadjistavropoulos HD, Pugh NE, et al. Dementia anxiety among older adult caregivers: an exploratory study of older adult caregivers in Canada. Int Psychogeriatrics. 2011;23:880–886.
11. Bachner YG, O’Rourke N, Davidov E, et al. Mortality communication as a predictor of psychological distress among family caregivers of home hospice and hospital in patients with terminal cancer. Aging Ment Health. 2009;13:54–63.
12. Papastavrou E, Kalokerinou-Anagnostopoulou A, Alevizopoulos G, et al. The reliability and validity of the burden scale (Zarit Burden Interview) in Greek Cypriot caregivers of patients with dementia. Nosileftiki. 2006;45:439–449.
13. Zachariou A, Filiponi M. Investigation of the factors affecting the burden on caregivers of elderly patients with urinary incontinence: a cross-sectional study in Greece. Hellenic Urology. 2018;28:45–54.
14. Andersson KE, Choudhury N, Cornu JN, et al. The efficacy of mirabegron in the treatment of urgency and the potential utility of combination therapy. Ther Adv Urol. 2018;10(8):243–256.
15. Mueller ER, van Maanen R, Chapple C, et al. Long-term treatment of older patients with overactive bladder using a combination of mirabegron and solifenacin: a prespecified analysis from the randomized, Phase III SYNERGY II study. Neurourol Urodyn. 2019;38(2):779–792.
16. Zarit SH, Reever KE, Back-Peterson J. Relatives of the impaired elderly: correlates of feelings of burden. Gerontologist. 1980;20:649–655.
17. Hu P, Yang Q, Kong L, et al. Relationship between the anxiety/depression and care burden of the major caregiver of stroke patients. Medicine. 2018;97(40):e12638.
18. Tessitore A, Marano P, Modugno N, et al. Caregiver burden and its related factors in advanced Parkinson’s disease: data from the PREDICT study. J Neurol. 2018;265:1124–1137.
19. Tamanini JT, Santos JL, Lebrão ML, et al. Association between urinary incontinence in elderly patients and caregiver burden in the city of Sao Paulo/Brazil: health, Wellbeing, and Ageing Study. Neurourol Urodyn. 2011;30:1281–1285.
20. Brown JS, Bradley CS, Subak LL, et al. The sensitivity and specificity of a simple test to distinguish between urge and stress urinary incontinence. Ann Intern Med. 2006;144:715–723.
21. Liapis A, Bakas P, Liapi S, Sioutis D, Creatsas G. Epidemiology of female urinary incontinence in the Greek population: EURIG study. Int Urogynecol J. 2010;21(2):217–222.
22. Elenskaia K, Haidvogel K, Heidinger C, et al. The greatest taboo: urinary incontinence as a source of shame and embarrassment. Wien Klin Wochenschr. 2011;123:607–610.
23. Teunissen D, van Weel C, Lagro-Janssen T. Urinary incontinence in older people lining in the community: examining help-seeking behavior. Br J Gen Practice. 2005;55:776–782.
24. Nuotio M, Tammela TL, Luukkaala T, et al. Predictors of institutionalization in an older population during a 13-year period: the effect of urge UI. J Gerontol a Biol Sci Med Sci. 2003;58:756–762.
25. Langa KM, Fultz NH, Saint S, et al. Informal caring time and costs for urinary UI in older individuals in the United States. J Am Geriatr Soc. 2002;50:733–737.
26. Zarit SH. Family care and burden at the end of the life. CMAJ. 2004;170:1811.
27. Lane P, McKenna H, Ryan A, et al. The experience of the family carers’ role: a qualitative study. Res Theory Nurs Pract. 2003;17:137–151.
28. Brittain KR, Shaw C. The social consequences of living with and dealing with incontinence-a carer’s perspective. Soc Sci Med. 2007;65:1274–1283.
29. Cassells C, Watt E. The impact of incontinence on older spousal carers. J Adv Nurs. 2003;42:607–616.
30. Hayder D, Schnepp W. Urinary incontinence-the family caregivers' perspective. Gerontology Geriatrics. 2008;41:261–266.
31. Carsughi A, Santini S, Lamura G. Impact of the lack of integrated care for older people with urinary incontinence and their family caregivers: results from a qualitative pilot study in two large areas of the Marche Region. Ann Ist Super Sanita. 2019;55:26–33.
32. Lobchuc M, Rosenberg F. A qualitative analysis of individual and family caregiver responses to the impact of urinary incontinence on quality of life. J Wound Ostomy Continence Nurs. 2014;41:589–596.
33. Santini S, Andersson G, Lamura G. Impact of incontinence on the quality of life of caregivers of older persons with incontinence: a qualitative study in four European countries. Arch Gerontol Geriatr. 2016;63:92–101.
34. Tseng CN, Huang GS, Yu PJ, et al. A qualitative study of family caregiver experiences of managing incontinence in Stroke survivors. PLoS One. 2015;10:e0129540.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
