Back to Journals » OncoTargets and Therapy » Volume 13
MiR-195-5p Inhibits Malignant Progression of Cervical Cancer by Targeting YAP1
Authors Liu X, Zhou Y, Ning Y, Gu H, Tong Y, Wang N
Received 19 August 2019
Accepted for publication 20 January 2020
Published 30 January 2020 Volume 2020:13 Pages 931—944
DOI https://doi.org/10.2147/OTT.S227826
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjay Singh
Xiaomin Liu,1 Yi Zhou,1 Yu-e Ning,1 Hui Gu,2 Yuxin Tong,3 Ning Wang1
1Department of Obstetrics and Gynecology, Shengjing Hospital of China Medical University, Shenyang 110000, People’s Republic of China; 2Key Laboratory of Health Ministry for Congenital Malformation, Shengjing Hospital, China Medical University, Shenyang 110004, People’s Republic of China; 3Medical Research Center, Shengjing Hospital of China Medical University, Shenyang, Liaoning 110004, People’s Republic of China
Correspondence: Ning Wang
Department of Obstetrics and Gynecology, Shengjing Hospital of China Medical University, Shenyang 110000, People’s Republic of China
Email [email protected]
Purpose: Our previous studies have shown that miR-195 is reduced in cervical cancer tissues, and that upregulation of miR-195 suppressed cervical cancer cell growth and induced a cell cycle block. In this study, we aimed to further elucidate the mechanism of action between miR-195-5p and Yes-associated protein 1 (YAP1) in the malignant progression of cervical cancer.
Methods: MiR-195-5p and YAP1 were detected using qRT-PCR in cervical cancer cells transfected with miR-195-5p mimics or inhibitor. Cell proliferation, migration, and invasion ability were detected using MTT, wound healing, and transwell invasion assays. Dual luciferase reporter assay, qRT-PCR, and Western blot analysis were used to demonstrate that YAP1 was a target of miR-195-5p.
Results: Our results showed that miR-195-5p is negatively correlated with YAP1 protein levels but not with mRNA expression. Moreover, upregulation of miR-195-5p by transient transfection with miR-195-5p mimics in HeLa and SiHa cells inhibited cell proliferation, migration ability, invasiveness, and the EMT. Conversely, miR-195-5p downregulation produced opposite results. In addition, multiple miRNA target prediction sites showed that YAP1 was a potential target gene; this was confirmed by dual luciferase assay. Rescue experiments further confirmed that YAP1 is involved in miR-195-5p-mediated inhibition of proliferation, migration ability, invasiveness, and the EMT of cervical cancer cells.
Conclusion: Taken together, our data suggest that miR-195-5p may act as a tumor suppressor which could provide a theoretical basis for cervical cancer patient targeted therapy.
Keywords: cervical cancer, miR-195-5p, YAP1, epithelial to mesenchymal transition
Introduction
According to GLOBOCAN 2018, cervical cancer ranks fourth in women’s morbidity and mortality in developing countries due to the lack of screening programs, vaccine awareness, and effective therapies, which have led to more than 500,000 newly diagnosed cases and nearly 300,000 deaths in 2018.1 Despite significant advances in preventive measures, diagnostic methods, and treatment of cervical cancer in recent years, the prognosis for patients with advanced stage cervical cancer remains poor; many patients die from recurrence and metastasis following surgical resection and standardized radiotherapy and chemotherapy.2 Thus, a greater understanding of the mechanisms underlying the occurrence and metastasis and indicators or biomarkers contributing to early diagnosis and therapies of cervical cancer are urgently required.
MicroRNAs (miRNAs), single-stranded small molecule RNAs composed of 20–25 nucleotides, regulate post-transcriptional gene expression via binding to the 3ʹ untranslated region (3ʹ-UTR) of a target gene. Moreover, they can also act as oncogenes or tumor suppressor genes, participating in many biological processes, such as embryonic development, cell differentiation, apoptosis, drug resistance, angiogenesis, and tumor metastasis, through specific targets or signaling pathways.3,4 Recently, there is increasing evidence that many miRNAs are abnormally expressed in cervical cancer, such as miRNA-944,5 miRNA-155-5p,6 and miR-449b-5p,7 demonstrating miRNAs may act as novel prognostic markers or as prospective drug therapy targets for patients with cervical cancer. Significantly, recent studies have found that abnormal downregulation of miR-195 is associated with the occurrence of cancer and malignant progression. For example, miR-195 has been reported to suppress the proliferation, migration ability, and invasiveness of cervical cancer via downregulation of DCUN1D1, HDGF, CCND1, CCND2, MYB, and SMAD3.8–12 In this study, we identified YAP1 as a potential target gene of miR-195-5p using miRNA prediction websites. YAP1 is a major transcriptional regulator of the Hippo pathway and is highly correlated with the development of malignant tumors in humans.13 YAP1 has been reported to play both carcinogenic and anti-carcinogenic roles in different tumors.14 The carcinogenic effect of YAP1 has been reported in colorectal cancer cells, where it could promote proliferation, invasion, migration, and angiogenesis, as well as the epithelial to mesenchymal transition (EMT),15 while several studies have reported YAP1 as a tumor suppressor gene in hematological cancers. For example, low expression of YAP1 is predictive of poor outcome in acute myeloid leukemia and multiple myeloma.16 He et al17 found that YAP was highly expressed in cervical cancer tissues and overexpression of YAP promoted malignant progression of cervical cancer cells. Based on the above results, we hypothesized that miR-195-5p may affect the malignant progression of cervical cancer by downregulating YAP1.
In the current study, we show that miR-195-5p could suppress cervical cancer progression by inhibiting proliferation, migration ability, invasiveness, and the EMT via YAP1 in vitro. We also determined that upregulation of YAP1 can partially reverse the inhibitory effect of miR-195-5p in cervical cancer cells HeLa and SiHa. These results reveal a key role of miR-195-5p/YAP1 axis in the pathogenesis of malignant progression of cervical cancer.
Materials and Methods
Cell Culture and Cell Transfection
Human cervical cancer cells HeLa and SiHa were purchased from the Shanghai Institute of Biochemistry and Cell Biology (Shanghai, China) and cultured in Roswell Park Memorial Institute (RPMI) 1640 medium (BioInd, Kibbutz Beit Haemek, Israel) supplemented with 10% fetal bovine serum (FBS) (BioInd) and 1% penicillin-streptomycin solution (BioInd) at 37°C, 5% CO2, and 50% relative humidity. The medium was replaced according to the growth rate of the cells. Chemosynthetic Has-miR-195-5p mimics, negative control (NC), Has-miR-195-5p inhibitor, and NC inhibitor were purchased from Shanghai GenePharma Co., Ltd. (Shanghai, China). Cells were seeded at 70–90% density and transfected with LipofectamineTM 2000 reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
Bioinformatics Analysis
TargetScan (http://www.targetscan.org/vert_72/), MiRwalk 3.0 (http://mirwalk.umm.uni-heidelberg.de/), and Starbase 2.0 (http://starbase.sysu.edu.cn/starbase2/index.php) were used to predict target genes of miR-195-5p according to the manufacturers’ instructions.
Luciferase Reporter Assay
DNA fragments of the YAP1 3ʹ-UTR with the predicted binding site of miR-195-5p and its mutant sequence were amplified and inserted into pmirGLO luciferase reporter vector (Promega, Madison, WI, USA) to construct the wild-type and mutant plasmids pmirGLO-YAP1-Luc-WT and pmirGLO-YAP1-Luc-MUT, respectively. HeLa and SiHa cells were seeded into 24-well plates at 2 × 105 cells/well until the confluency rate was 70–90% at the time of transfection. After 24 h, luciferase activity was measured with the Dual-Luciferase Reporter Assay System (Promega) according to the manufacturer’s instructions and normalized with Renilla luciferase activity. The co-transfections were performed as follows: miR-195-5p mimics + YAP1-WT-Luc, mimics-NC + YAP1-WT-Luc, miR-195-5p mimics + YAP1-MUT-Luc, mimics-NC + YAP1-MUT-Luc, miR-195-5p mimics + Control, and mimics-NC + Control.
Methods
Fluorescence in situ Hybridization (FISH)
To identify co-localization of miR-195-5p and YAP1 in cervical cancer cells HeLa and SiHa, the YAP1 probe (green-labeled, genepharma, Shanghai, China) and miR-195-5p probe (red-labeled, genepharma). Briefly, slides were treated with 1% paraformaldehyde and blocked with prehybridization buffer (genepharma) for 4 h at 37°C. Then add probe mixture and incubate at 37°C for 16 h. The next day, the slide was washed three times with PBS, stained with DAPI working solution, and observed under a confocal microscope as soon as possible. All fluorescence images (630×, oil lens) were captured using a fluorescence microscope (Nikon, Japan).
Total RNA Extraction and Quantitative Real-Time RT-PCR (qRT-PCR)
Total RNA including miRNAs was extracted from HeLa and SiHa cells using the miRNeasy Mini Kit (Qiagen, Hilden, Germany). Then, the extracted miRNAs needed to add Escherichia coli Poly(A) Polymerase Reaction Buffer (NEB, Ipswich, MA, USA) to reverse all miRNAs using the GoScriptTM Reverse Transcription System (Promega). Next, the GoScript™ Reverse Transcript-ion System (Promega) was used to synthesize first-strand cDNA optimized for quantitative PCR amplification. Gotaq® qPCR Master Mix (Promega) was subsequently applied for qRT-PCR according to the manufacturer’s instructions. The reactions were performed in an ABI 7500 Real-time PCR system (Applied Biosystems, Foster City, CA, USA). All primers were designed and synthesized by Guangzhou RiboBio Co., Ltd. (Guangzhou, China). The primer sequences were as follows: miR-195-5p, F: 5ʹ-GCGTAGCAGCACAGAAATATTGGC-3ʹ and R: 5ʹ-CTGTCGTCGTAGAGCCAGGGAA-3ʹ; U6, F: 5ʹ-CTCGCTTCGGCAGCACA-3ʹ and R: 5ʹ-AACGCTTCACGAATTTGCGT-3ʹ; YAP1, F: 5ʹ-TGACCCTCGTTTTGCCATGA-3ʹ and R: 5ʹ’GTTGCTGCTGGTTGGAGTTG-3ʹ; and GAPDH, F: 5ʹ-TGCACCACCAACTGCTTAGC-3ʹ and R: 5ʹ-GGCATGGACTGTGGTCATGAG-3ʹ. U6 and GAPDH were used as internal controls for miR-195-5p and YAP1, respectively. The relative expression levels of miR-195-5p and YAP1 were analyzed using the 2−∆∆Ct method.
Western Blot Analysis
After transfection, 50–100 μL of RIPA buffer (Beyotime, Shanghai, China) was added to HeLa and SiHa cells to extract whole cell protein. A BCA protein quantitative kit (Beyotime) was used to determine protein concentration according to the manufacturer’s instructions. Following separation of 50 μg of protein by 8% SDS-PAGE (80 V for 2 h), the protein was transferred to PVDF membrane (Millipore, Burlington, MA, USA) by wet transformation. Then, 5% skimmed milk was added and the blots were incubated at room temperature for 2 h followed by three washes with Tris-buffered saline + Tween-20 (TBST). Specific antibodies against the target proteins were added and incubated overnight at 4°C. The primary antibodies used were as follows: YAP1, E-cadherin, Snail, and Vimentin, which were all obtained from Cell Signaling Technology (Danvers, MA, USA). After washing with TBST, secondary antibody labeled with horseradish peroxidase (Proteintech, Rosemont, IL, USA) was added and the blots were incubated at room temperature for 2 h. Then, the protein bands were developed by enhanced chemiluminescence (ECL, Hercules, CA, USA), and the gray ratio of target protein to internal reference protein was calculated using ImageJ software. Experiments were performed in triplicate followed by statistical analysis.
MTT Assay
HeLa and SiHa cells (2 × 103 cells/well) were seeded in 96-well plates, transfected, and cultured for 24, 48, 72, and 96 h with 50 nM miR-195-5p mimics and NC, or 100 nM miR-195-5p inhibitor and NC inhibitor according to the manufacturer’s instructions, with six repetitive wells for each group. The cells were then cultured with serum-free medium containing 10 μL MTT (Sigma-Aldrich, St. Louis, MO, USA) at regular time intervals for 4 h. The supernatant was removed, 100 μL dimethyl sulfoxide (Sigma-Aldrich) was added to each well, and the absorbance at 490 nm was measured using a microplate reader (ThermoFisher Scientific, Waltham, MA, USA) to examine cell viability. Experiments were performed in triplicate.
Transwell Invasion Assay
The invasiveness of HeLa and SiHa cells was detected by Transwell assay. After 48 h of transfection, matrigel (4.0 μg/μL, 60 μL) was added into Transwell chambers (Corning, Corning, NY, USA) and incubated for 2–3 h at 37°C for gel solidification. The assay was performed using 24-well transwell plates with polycarbonate membrane and 8.0 μm pores (Corning). RPMI 1640 medium containing 10% FBS was added to the lower chambers. HeLa and SiHa cells were seeded in the upper chambers with serum-free RPMI 1640 medium at a density of 4 × 103 cells/per well. After 24 h, the medium from the lower chamber was removed and the cells in the upper chambers were wiped with a cotton swab. The migrated cells were fixed in 4% paraformaldehyde and stained with crystal violet for 30 min at room temperature. Migrating cell numbers were observed in five random fields under an inverted phase microscope (Nikon, Japan). Cells numbers were counted using ImageJ software for statistical analysis.
Wound Healing Assay
HeLa and SiHa cells (1 × 105 cells) were seeded in a 6-well plate in an incubator at 37°C and 5% CO2. When the cells reached to 90% confluency, the cell monolayer was scraped using a 200 μL sterile pipette tip to generate a wound, and washed twice with phosphate-buffered saline. HeLa and SiHa cells were cultured in serum-free medium and photographed at 0 and 48 h following generation of the wound using an inverted phase microscope (Nikon). The percentage of wound healing was determined; larger wound healing percentages indicated stronger cell migration ability. Assays were performed in triplicate followed by statistical analysis.
Statistical Analysis
SPSS 17.0 software (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. All data are presented as the mean ± standard deviation. All statistical data satisfy the normal distribution and homogeneity of variance, and the difference between the two groups should be analyzed by Student’s t-test. P < 0.05 was accepted as indicative of significant differences.
Results
MiR-195-5p Inhibited YAP1 Translation
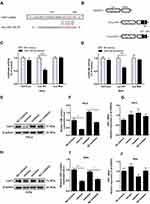
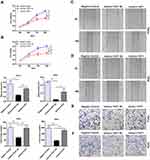
In our previous study, Wang et al10 determined that expression of miR-195 was lower in cervical cancer than in adjacent normal tissues, and that upregulation of miR-195 inhibited cervical cancer cell growth and induced a cell cycle block. This study, we further explored the molecular mechanism of miR-195-5p in the malignant progression of cervical cancer. Based on bioinformatics analysis, TargetScan, and miRwalk, YAP1 was predicted to contain a binding site for miR-195-5p (Figure 1A). YAP1, a key effector of the Hippo pathway in humans, has been shown to play a carcinogenic role by promoting cell proliferation and colony formation in cervical cancer cells.17
To confirm the functional correlation between miR-195-5p and YAP1, we constructed a luciferase reporter vector to detect whether miR-195-5p can directly regulate the expression of YAP1. The specific binding sites (Fraction-F) of miR-195-5p in the YAP1 3ʹ-UTR were cloned into the pmirGLO dual-luciferase vector to generate the F-Luc reporter construct (Figure 1B). HeLa and SiHa cells were transfected with miR-195-5p mimics or NC mimics as well as the pmirGLO-YAP1-Luc-WT or pmirGLO-YAP1-Luc-MUT plasmid. As illustrated in Figure 1C and D, HeLa and SiHa cells co-transfected with miR-195-5p mimics and YAP1-Luc-WT plasmid presented a statistically significant decline in luciferase activity compared with cells co-transfected with NC mimics and YAP1-Luc-WT plasmid, while HeLa and SiHa cells co-transfected with miR-195-5p mimics and pmirGLO-YAP1-Mut or NC mimics with pmirGLO-YAP1-MuT revealed no considerable change in reporter activity. These results suggested that miR-195-5p inhibits the expression of YAP1 by interacting with a specific binding site of the YAP1 3ʹ-UTR in HeLa and SiHa cells. At the same time, FISH experiments verified the colocalized expression of miR-195-5p and YAP1 in cervical cancer cells HeLa and SiHa, both of which are distributed in the cytoplasm, which further confirmed the interaction between miR-195-5p and YAP1 (Supplementary Figure 1A and B).
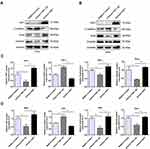
The inhibitory effect of miR-195-5p on YAP1 was further confirmed. Upregulation of miR-195-5p in HeLa cells led to substantial downregulation of the protein levels of YAP1 (Figure 1E and F), while there was no noticeable difference in YAP1 mRNA levels (Figure 1G). Conversely, when miR-195-5p expression levels were inhibited following transfection of miR-195-5p inhibitor, protein levels of YAP1 increased accordingly (Figure 1E and F) and YAP1 mRNA levels did not change significantly (Figure 1G). Similarly, overexpression of miR-195-5p in SiHa cells resulted in declining protein levels of YAP1 (Figure 1H and I), on the contrary, the inhibition of miR-195-5p suppressed YAP1 protein levels (Figure 1H and I). Similar to HeLa cells, regardless of miR-195-5p overexpression or inhibition, the mRNA level of YAP1 was not statistically significant (Figure 1J).
MiR-195-5p Inhibited Cell Proliferation in Cervical Cancer Cells
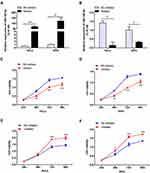
To further study the effect of miR-195-5p on the proliferation of cervical cancer cells, miR-195-5p mimics or inhibitor transient transfection was used to analyze the effects of gain or loss of miR-195-5p function. QRT-PCR was performed to confirm the upregulation or downregulation effects of miR-195-5p mimics and inhibitors. Following transfection of miR-195-5p mimics into HeLa and SiHa cells, the expression level of miR-195-5p increased significantly (Figure 2A). By contrast, miR-195-5p expression was obviously reduced following transfection with inhibitors in HeLa and SiHa cells compared with NC-inhibitors (Figure 2B). The MTT assay demonstrated that proliferation of HeLa and SiHa cells was significantly decreased following transfection with miR-195-5p mimics compared with NC mimics (Figure 2C and D), whereas inhibition of miR-195-5p promoted cell proliferation (Figure 2E and F).
MiR-195-5p Inhibited Migration and Invasion of Cervical Cancer Cells
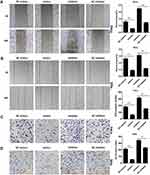
Next, wound healing was performed to verify the role of miR-195-5p in the migratory abilities of cervical cancer cells. Wound healing experiment results demonstrated that the migration of cervical cancer cells was attenuated following transfection with miR-195-5p mimics compared with NC mimics (Figure 3A and B). Conversely, the migration of HeLa and SiHa cells was evidently increased following transfection with miR-195-5p inhibitor (Figure 3A and B).
Furthermore, to demonstrate the effect of miR-195-5p on the invasion of cervical cancer cells, miR-195-5p mimics, NC-mimics, miR-195-5p inhibitor, and NC-inhibitor were transfected into HeLa and SiHa cell for 24h. Then the cell invasion ability was detected by Transwell assay, which indicated that overexpression of miR-195-5p inhibited the invasiveness of HeLa and SiHa cells (Figure 3C and D), while inhibition of miR-195-5p can significantly increase the invasive ability of cervical cancer cells. (Figure 3C and D). These results demonstrated that overexpression of miR-195-5p suppressed migration and invasiveness, and vice versa.
MiR-195-5p Inhibited the EMT in Cervical Cancer Cells
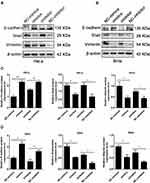
Cancer cells usually regulate invasion and migration through EMT. In this study, Western blotting was used to analyze the expression levels of epithelial and mesenchymal markers. Expression of the epithelial marker E-cadherin was remarkably increased, while expression of the mesenchymal markers Snail and Vimentin was decreased. These results were consistent with miR-195-5p overexpression by transfection of miR-195-5p (Figure 4A and B). By contrast, following transfection of HeLa and SiHa cells with miR-195-5p inhibitor, the expression levels of E-cadherin protein decreased, while expression levels Snail and Vimentin protein increased (Figure 4A and B). Statistical analyses are shown in Figure 4C and D. Overall, our results suggest that miR-195-5p negatively regulates proliferation, migration ability, invasiveness, and the EMT of cervical cancer cells. In other words, upregulation of miR-195-5p may inhibit the metastasis of cervical cancer cells.
YAP1 Reversed miR-195-5p-Induced Proliferation in Cervical Cancer Cells
To further confirm whether miR-195-5p regulated proliferation, migration ability, invasiveness, and the EMT by targeting YAP1 in cervical cancer cells, we performed miR-195-5p and YAP1 recovery experiments. HeLa and SiHa cells were treated as follows: Negative Control (miR-195-5p mimics+YAP1 NC); miR-195-5p mimics + YAP1 NC; and miR-195-5p mimics + YAP1. Relative wound closure and migration rates were determined for each group. First of all, the MTT assay showed that overexpression of YAP1 eliminated the inhibitory effect of miR-195-5p on HeLa proliferation. Similarly, MTT analysis on SiHa cells showed the same results (Figure 5A and B). Secondly, wound healing analysis showed that the upregulation of YAP1 restored the inhibition of miR-195-5p on the migration of cervical cancer cells, and there was significant statistical significance between the mimics + YAP1 group and mimics + YAP1 NC group (Figure 5C and D). In addition, Transwell analysis showed that overexpression of YAP1 significantly promoted the invasiveness of HeLa and SiHa cells. In other words, it also successfully reversed the inhibition of miR-195-5p on the invasion of cervical cancer cells (Figure 5E and F). These results suggest that the inhibitory effect of miR-195-5p on cervical cancer progression can be attributed, at least in part, to the suppression of YAP1 expression.
MiR-195-5p Inhibited the EMT by Targeting of YAP1 in Cervical Cancer Cells
To further explore whether miR-195-5p affects the metastasis of cervical cancer cells by regulating the expression of YAP1, EMT-related proteins in the transfected Negative Control, miR-195-5p mimics + YAP1 NC, and miR-195-5p + YAP1 groups were detected by Western blotting. Upregulation of miR-195-5p resulted in a remarkable decrease in YAP1 expression in HeLa and SiHa cells; however, YAP1 protein levels are significantly elevated by transfection of the YAP1 overexpression plasmid (Figure 6A and B). E-cadherin protein levels in the miR-195-5p mimics group were higher than those in the NC group, while Snail and Vimentin levels were lower than those in the NC group. At the same time, E-cadherin levels in the miR-195-5p mimics + YAP1 group were lower than that in the miR-195-5p mimics + YAP1 NC group, while Snail and Vimentin levels were significantly higher than those in the miR-195-5p mimics + YAP1 NC group. Statistical analyses are shown in Figure 6C and D. These results demonstrated that overexpression of miR-195-5p suppressed the EMT, while upregulation of YAP1 levels promoted the EMT.
Discussion
The occurrence and development of cervical cancer is a complex, multi-step and multi-gene process. Patients with early cervical squamous cell carcinoma have a better prognosis, while the treatment of advanced cervical adenocarcinoma is almost in vain due to local recurrence and metastasis diffusion.18 In this study, we concentrated on the effects of miR-195-5p and YAP1 in vitro following bioinformatics prediction. Our results showed that miR-195-5p act as a tumor suppressor gene in cervical cancer and its target gene Yap1 plays a role of oncogene in cervical cancer. Therefore, it is imperative to identify potential prognostic predictors and develop treatment strategies for improving the survival rate of patients with cervical cancer.
Over the past few years, accumulating evidence has indicated that miRNAs have become the focus of cancer research especially in malignant tumors, and this has appealed to interest from numerous researchers hoping to reveal the underlying mechanisms.19 MiR-195-5p has been shown to exhibit different levels of expression in different tumor types, and its role in tumorigenesis and cancer development cannot be simply summarized as a tumor suppressor gene or oncogene. For instance, Dai et al found miR-195-5p was down-regulated in ovarian cancer, which could inhibit tumor angiogenesis and reduce chemotherapy resistance by targeting the expression of PAST1.20 What’s more, Li et al also observed that the prognosis of cervical cancer patients is better in the relatively high expression of miR-195-5p, which may inhibit the invasion and metastasis of cervical cancer cells via targeting MMP-14 and acting on the TNF signal pathway.21 In contrast, miR-195, which is significantly up-regulated in human osteoarthritis, promotes the further development of OA by targeting PTHrP.22 So, miR-195 plays different roles in different tissues.
On the other hand, YAP1, a major downstream effector of the Hippo pathway, functions as a transcriptional regulator and plays a key role to control cell proliferation and growth.23 It is well accepted nowadays that when the Hippo pathway is activated, the YAP/TAZ transcriptional activator is inactivated by hosphorylation of LATS1/2, and the phosphorylated TAZ and YAP1 promote their interaction with the 14-3-3 protein and cause its retention in the cytosol.24,25 It can be seen that the Hippo signal pathway can not only inhibit the carcinogenic activity of YAP/TAZ in the nucleus, but also limit the growth of tumor by enhancing its growth inhibitory activity in the cytoplasm. Furthermore, numerous studies have shown that elevated YAP1 activity and/or YAP1 overexpression is observed in some primary human cancers, including hepatocellular carcinoma, pancreatic cancer, gastric carcinoma,26–28 and elevated YAP1 protein expression and nuclear localization are associated with poor prognosis.27 More importantly, YAP1 was reportedly highly expressed in cervical cancer and is considered to be a tumor suppressor in this type of cancer,17 and which is also highly accumulated in the nucleus of HPV-positive oropharyngeal squamous cell carcinoma.29 Our study shows that YAP1 was a direct target gene of miR-195-5p and plays an important carcinogenic role in cervical cancer. These results are supported by previous studies.30
Accumulating evidence has shown that miR-195 is decreased in multiple tumors, including cervical cancer,11 prostate cancer,12 glioma,31 and colon cancer,32 and acts as a tumor suppressor gene. In this study, we have demonstrated a new regulatory axis of miR-195-5p/YAP1 in the metastasis of cervical cancer according to strong in vitro evidence based on a series of classical research methods. In addition, many researches have offered more similar examples, for instance, it has been reported that miR-591 expression in breast cancer is lower than that in normal tissues, and its ectopic overexpression inhibited cell proliferation, invasiveness, and the Hippo-YAP1 signaling pathway by downregulating the YAP1 gene.33 Additionally, the low expression of miR-375 is closely related to the overall survival of CRC patients, and miR-375 enhances the sensitivity of CRC cells to chemotherapeutic drugs by directly targeting YAP1.34 Another important finding of this study was that the restoration of YAP1 could partially inhibit the tumor-suppression effect induced by miR-195-5p. These findings further confirmed that miR-195-5p could target the YAP1 gene to regulate the malignant behavior of cervical cancer cells including proliferation, migration ability, invasiveness, and the EMT.
The EMT is defined as a transcriptionally regulated phenotypic transition process that renders cancer cells more metastatic and invasive, which then makes it more difficult to treat cancer.35 The EMT involves the loss of expression of some epithelial markers, such as E-cadherin and α-catenin, as well as an increase in the expression of a number of mesenchymal markers, such as N-cadherin, Snail, Vimentin, and matrix metalloproteinases.36 Many miRNAs have been reported in recent years to regulate the EMT, thereby inhibiting tumor progression. MiR-34 can negatively regulate the EMT in different ways, such as via TGF-β signaling, the EMT-related transcription factor Snail, and the tumor suppressor gene p53.37,38 Similarly, in vitro cell transfection experiments in this study showed that E-cadherin protein expression was increased, while Snail and Vimentin expression was decreased, consistent with miR-195-5p overexpression and vice versa. In summary, miR-195-5p may be a negative regulator of the EMT and thus regulate cancer metastasis.
Conclusion
In conclusion, our results suggest that miR-195-5p overexpression may inhibit proliferation potential, migration, invasiveness, and the EMT of cervical cancer cells by targeting the 3ʹ-UTR of YAP1. Therefore, the miR-195-5p/YAP1 axis is expected to become a promising molecular target for targeted therapy of cervical cancer. Although we strive to make the experiment comprehensive and accurate, there are inevitably some limitations in our research, which including lack of in vivo experimental verification and similar experimental results after clinical sampling of tumor cells. Thus, further studies are needed to determine the exact role of miR-195-5p in the proliferation and metastasis of cervical cancer as well as downstream signaling pathways in vivo to provide a theoretical basis for clinical practice.
Acknowledgments
This study was funded by grants from the National Natural Science Foundation of China (No. 81202047, 81771595).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.v68.6
2. Li H, Wu X, Cheng X. Advances in diagnosis and treatment of metastatic cervical cancer. J Gynecol Oncol. 2016;27(4):e43. doi:10.3802/jgo.2016.27.e43
3. Acunzo M, Romano G, Wernicke D, Croce CM. MicroRNA and cancer–a brief overview. Adv Biol Regul. 2015;57:1–9. doi:10.1016/j.jbior.2014.09.013
4. Musavi Shenas MH, Eghbal-Fard S, Mehrisofiani V, et al. MicroRNAs and signaling networks involved in epithelial-mesenchymal transition. J Cell Physiol. 2019;234(5):5775–5785. doi:10.1002/jcp.27489
5. Park S, Kim J, Eom K, et al. microRNA-944 overexpression is a biomarker for poor prognosis of advanced cervical cancer. BMC Cancer. 2019;19(1):419. doi:10.1186/s12885-019-5620-6
6. Li N, Cui T, Guo W, Wang D, Mao L. MiR-155-5p accelerates the metastasis of cervical cancer cell via targeting TP53INP1. Onco Targets Ther. 2019;12:3181–3196. doi:10.2147/OTT.S193097
7. Cheng L, Shi X, Huo D, Zhao Y, Zhang H. MiR-449b-5p regulates cell proliferation, migration and radioresistance in cervical cancer by interacting with the transcription suppressor FOXP1. Eur J Pharmacol. 2019;856:172399. doi:10.1016/j.ejphar.2019.05.028
8. Zhong J, Yuan H, Xu X, Kong S. MicroRNA195 inhibits cell proliferation, migration and invasion by targeting defective in cullin neddylation 1 domain containing 1 in cervical cancer. Int J Mol Med. 2018;42(2):779–788. doi:10.3892/ijmm.2018.3660
9. Song R, Cong L, Ni G, et al. MicroRNA-195 inhibits the behavior of cervical cancer tumors by directly targeting HDGF. Oncol Lett. 2017;14(1):767–775. doi:10.3892/ol.2017.6210
10. Wang N, Wei H, Yin D, et al. MicroRNA-195 inhibits proliferation of cervical cancer cells by targeting cyclin D1a. Tumour Biol. 2016;37(4):4711–4720. doi:10.1007/s13277-015-4292-3
11. Du X, Lin LI, Zhang L, Jiang J. microRNA-195 inhibits the proliferation, migration and invasion of cervical cancer cells via the inhibition of CCND2 and MYB expression. Oncol Lett. 2015;10(4):2639–2643. doi:10.3892/ol.2015.3541
12. Zhou Q, Han LR, Zhou YX, Li Y. MiR-195 suppresses cervical cancer migration and invasion through targeting Smad3. Int J Gynecol Cancer. 2016;26(5):817–824. doi:10.1097/IGC.0000000000000686
13. Pan D. The hippo signaling pathway in development and cancer. Dev Cell. 2010;19(4):491–505. doi:10.1016/j.devcel.2010.09.011
14. Lorenzetto E, Brenca M, Boeri M, et al. YAP1 acts as oncogenic target of 11q22 amplification in multiple cancer subtypes. Oncotarget. 2014;5(9):2608–2621. doi:10.18632/oncotarget.1844
15. Sun Z, Ou C, Liu J, et al. YAP1-induced MALAT1 promotes epithelial-mesenchymal transition and angiogenesis by sponging miR-126-5p in colorectal cancer. Oncogene. 2019;38(14):2627–2644. doi:10.1038/s41388-018-0628-y
16. Cottini F, Hideshima T, Xu C, et al. Rescue of Hippo coactivator YAP1 triggers DNA damage-induced apoptosis in hematological cancers. Nat Med. 2014;20(6):599–606. doi:10.1038/nm.3562
17. He C, Mao D, Hua G, et al. The Hippo/YAP pathway interacts with EGFR signaling and HPV oncoproteins to regulate cervical cancer progression. EMBO Mol Med. 2015;7(11):1426–1449. doi:10.15252/emmm.201404976
18. Yoshida K, Suzuki S, Sakata J, et al. The upregulated expression of vascular endothelial growth factor in surgically treated patients with recurrent/radioresistant cervical cancer of the uterus. Oncol Lett. 2018;16(1):515–521. doi:10.3892/ol.2018.8610
19. Ruksha TG. MicroRNAs’ control of cancer cell dormancy. Cell Div. 2019;14:11. doi:10.1186/s13008-019-0054-8
20. Dai J, Wei R, Zhang P, Kong B. Overexpression of microRNA-195-5p reduces cisplatin resistance and angiogenesis in ovarian cancer by inhibiting the PSAT1-dependent GSK3beta/beta-catenin signaling pathway. J Transl Med. 2019;17(1):190. doi:10.1186/s12967-019-1932-1
21. Li M, Ren CX, Zhang JM, et al. The effects of miR-195-5p/MMP14 on proliferation and invasion of cervical carcinoma cells through TNF signaling pathway based on bioinformatics analysis of microarray profiling. Cell Physiol Biochem. 2018;50(4):1398–1413. doi:10.1159/000494602
22. Cao X, Duan Z, Yan Z, et al. miR-195 contributes to human osteoarthritis via targeting PTHrP. J Bone Miner Metab. 2019;37(4):711–721. doi:10.1007/s00774-018-0973-5
23. Li L, Liu T, Li Y, et al. The deubiquitinase USP9X promotes tumor cell survival and confers chemoresistance through YAP1 stabilization. Oncogene. 2018;37(18):2422–2431. doi:10.1038/s41388-018-0134-2
24. Huang J, Wu S, Barrera J, Matthews K, Pan D. The Hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila Homolog of YAP. Cell. 2005;122(3):421–434. doi:10.1016/j.cell.2005.06.007
25. Varelas X, Miller BW, Sopko R, et al. The Hippo pathway regulates Wnt/beta-catenin signaling. Dev Cell. 2010;18(4):579–591. doi:10.1016/j.devcel.2010.03.007
26. Liu J, Zhao X, Wang K, et al. A novel YAP1/SLC35B4 regulatory axis contributes to proliferation and progression of gastric carcinoma. Cell Death Dis. 2019;10(6):452. doi:10.1038/s41419-019-1674-2
27. Wu Y, Hou Y, Xu P, et al. The prognostic value of YAP1 on clinical outcomes in human cancers. Aging (Albany NY). 2019;11(19):8681–8700. doi:10.18632/aging.v11i19
28. Yoo W, Lee J, Jun E, et al. The YAP1-NMU axis is associated with pancreatic cancer progression and poor outcome: identification of a novel diagnostic biomarker and therapeutic target. Cancers (Basel). 2019;11:10. doi:10.3390/cancers11101477
29. Alzahrani F, Clattenburg L, Muruganandan S, et al. The Hippo component YAP localizes in the nucleus of human papilloma virus positive oropharyngeal squamous cell carcinoma. J Otolaryngol Head Neck Surg. 2017;46(1):15. doi:10.1186/s40463-017-0187-1
30. Sun M, Song H, Wang S, et al. Integrated analysis identifies microRNA-195 as a suppressor of Hippo-YAP pathway in colorectal cancer. J Hematol Oncol. 2017;10(1):79. doi:10.1186/s13045-017-0445-8
31. Chen LP, Zhang NN, Ren XQ, He J, Li Y. miR-103/miR-195/miR-15b regulate SALL4 and inhibit proliferation and migration in glioma. Molecules. 2018;23:11. doi:10.3390/molecules23112938
32. Li B, Wang S, Wang S. MiR-195 suppresses colon cancer proliferation and metastasis by targeting WNT3A. Mol Genet Genomics. 2018;293(5):1245–1253. doi:10.1007/s00438-018-1457-y
33. Huang X, Tang F, Weng Z, Zhou M, Zhang Q. MiR-591 functions as tumor suppressor in breast cancer by targeting TCF4 and inhibits Hippo-YAP/TAZ signaling pathway. Cancer Cell Int. 2019;19:108. doi:10.1186/s12935-019-0818-x
34. Xu X, Chen X, Xu M, et al. miR-375-3p suppresses tumorigenesis and partially reverses chemoresistance by targeting YAP1 and SP1 in colorectal cancer cells. Aging (Albany NY). 2019;11(18):7357–7385. doi:10.18632/aging.102214
35. Jakobsen KR, Demuth C, Sorensen BS, Nielsen AL. The role of epithelial to mesenchymal transition in resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small cell lung cancer. Transl Lung Cancer Res. 2016;5(2):172–182. doi:10.21037/tlcr
36. Nie D, Fu J, Chen H, Cheng J, Fu J. Roles of microRNA-34a in epithelial to mesenchymal transition, competing endogenous RNA sponging and its therapeutic potential. Int J Mol Sci. 2019;20:4. doi:10.3390/ijms20040861
37. Kim NH, Kim HS, Li XY, et al. A p53/miRNA-34 axis regulates Snail1-dependent cancer cell epithelial-mesenchymal transition. J Cell Biol. 2011;195(3):417–433. doi:10.1083/jcb.201103097
38. Huang G, Du MY, Zhu H, et al. MiRNA-34a reversed TGF-beta-induced epithelial-mesenchymal transition via suppression of SMAD4 in NPC cells. Biomed Pharmacother. 2018;106:217–224. doi:10.1016/j.biopha.2018.06.115
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.