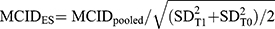Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Minimal Clinically Important Differences for Patient-Reported Outcome Measures of Cough and Sputum in Patients with COPD
Authors Rebelo P, Oliveira A , Paixão C , Valente C, Andrade L , Marques A
Received 5 July 2019
Accepted for publication 31 October 2019
Published 29 January 2020 Volume 2020:15 Pages 201—212
DOI https://doi.org/10.2147/COPD.S219480
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Patrícia Rebelo,1,2 Ana Oliveira,1,2 Cátia Paixão,1,2 Carla Valente,3 Lília Andrade,3 Alda Marques1,2
1Respiratory Research and Rehabilitation Laboratory (Lab3R), School of Health Sciences, University of Aveiro (ESSUA), Aveiro, Portugal; 2Institute of Biomedicine, School of Health Sciences (iBiMED), University of Aveiro (ESSUA), Aveiro, Portugal; 3Pulmonology Department, Centro Hospitalar Do Baixo Vouga (CHBV), Aveiro, Portugal
Correspondence: Alda Marques
Escola Superior de Saúde, Universidade de Aveiro - Edifício 30, Agras do Crasto - Campus Universitário de Santiago, Aveiro 3810-193, Portugal
Email [email protected]
Background: Cough and sputum are highly prevalent in patients with chronic obstructive pulmonary disease (COPD). Pulmonary rehabilitation (PR) has shown to be effective in managing these symptoms. However, the interpretation of the magnitude of PR effects is hindered by the lack of minimal clinically important differences (MCIDs).
Purpose: This study established MCIDs for the Leicester cough questionnaire (LCQ) and the cough and sputum assessment questionnaire (CASA-Q), in patients with COPD after PR.
Patients and Methods: An observational prospective study was conducted in patients with COPD who participated in a 12-weeks community-based PR program. Anchor- (mean change, receiver operating characteristic curves and linear regression analysis) and distribution-based methods [0.5*standard deviation; standard error of measurement (SEM); 1.96*SEM; minimal detectable change and effect size] were used to compute the MCIDs. The anchors used were: i) patients and physiotherapists global rating of change scale, ii) COPD assessment test, iii) St. George’s respiratory questionnaire and iv) occurrence of an exacerbation during PR. Pooled MCIDs were computed using the arithmetic weighted mean (2/3 for anchor- and 1/3 for distribution-based methods).
Results: Forty-nine patients with COPD (81.6% male, 69.8± 7.4years, FEV1 50.4± 19.4%predicted) were used in the analysis. The pooled MCIDs were 1.3 for LCQ and for CASA-Q domains were: 10.6 - cough symptoms; 10.1 - cough impact; 9.5 - sputum symptoms and 7.8 - sputum impact.
Conclusion: The MCIDs found in this study are potential estimates to interpret PR effects on cough and sputum, and may contribute to guide interventions.
Keywords: COPD, symptoms, pulmonary rehabilitation, patient health questionnaire, measurement characteristics, statistics
Introduction
Chronic obstructive pulmonary disease (COPD) is a growing global health concern that poses major burden on individuals, as well as, on economics and social systems.1–3 Cough and sputum are present in approximately 60% of patients with COPD4–6 and have been recognized to affect significantly and negatively patients’ health-related quality of life (HRQoL).7,8 Nevertheless, these symptoms have been scarcely explored and underappreciated in COPD research.9–12
Pulmonary rehabilitation (PR) is a well-established non-pharmacological intervention to manage patients with COPD.3,13,14 However, to interpret the magnitude of the results achieved with PR on symptoms relief, it is important to understand the minimal clinically important differences (MCIDs) of patient-reported outcome measures (PROMs), ie, the smallest change in a measure score that is subjectively perceived as relevant to the patient.15–17 Having MCIDs for symptoms-related PROMs will: aid to guide interventions;18,19 enhance judgement about the clinical relevance and magnitude of the PR effect15 allow samples size calculations; and contribute for defining expected endpoints in clinical trials.16,20,21 Thus, establishing MCIDs is of paramount importance for several stakeholders, from health professionals and researchers to guideline developers and policymakers.
We estimated the MCID of PROMs that assess symptoms of cough and sputum, ie, the Leicester cough questionnaire (LCQ)22 and the cough and sputum assessment questionnaire (CASA-Q)23 in patients with COPD, following a PR program.
Materials and Methods
Study Design and Participants
An observational prospective study, part of a larger trial (3R: Revitalising pulmonary rehabilitation – NCT03799666 on ClinicalTrials.gov) was conducted in accordance with the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee for Health of the Administração Regional de Saúde do Centro (Ref. 73/2016) and from the National Committee for Data Protection (no. 7295/2016). Prior to enrolment and data collection, a written description of the study was provided to every participant, who then signed an informed consent.
Patients were recruited via clinicians at Centro Hospitalar do Baixo Vouga and primary healthcare centers of the center region of Portugal during January 2019 and enrolled a community-based PR program. Patients were eligible if diagnosed with COPD,3 and clinically stable over the previous month, ie, no hospital admissions or exacerbations and no change in medication for the cardiorespiratory system. Exclusion criteria included the presence of other respiratory diseases or any clinical condition that precluded participants of being involved in a community-based PR program, ie, signs of cognitive impairment or presence of a significant cardiovascular, neurological or musculoskeletal disease.
Data Collection
Data were collected before (T0) and after 12 weeks of PR (T1). Sociodemographic (age, gender), anthropometric (height and weight to compute body mass index-BMI) and clinical data (smoking status, medication, number of exacerbations, hospitalizations or emergency admissions in the past year) were first obtained. The severity of comorbid diseases was recorded and scored according to Charlson Comorbidity Index (CCI): i) scores of 1–2; ii) scores of 3–4; and iii) scores ≥5.24 The modified British medical research council questionnaire (mMRC) was used to assess functional dyspnea,25 the COPD assessment test (CAT)26 to evaluate the impact of the disease and the St. George’s respiratory questionnaire (SGRQ) to assess HRQoL.27
The LCQ was used to evaluate cough-related quality of life. The LCQ is a 19-items scale organized in 3 domains (physical, psychological and social).22 Each domain has a score ranging from 1 to 7 and the total score varies from 3 to 21.22 Higher scores express a better cough-related quality of life and less impact of cough.22 The LCQ has shown to be a valid, reliable and responsive instrument, namely in COPD.22,28–30
The CASA-Q was used to assess cough and sputum symptoms, based on its reported frequency and severity, and impact on daily life activities.23 CASA-Q is a 20-item questionnaire containing 4 domains: cough symptoms, cough impact, sputum symptoms and sputum impact.23 All items are rescored and summed, achieving a score ranging from 0 to 100 for each domain, with higher scores indicating fewer symptoms and less cough and sputum impact.23 The CASA-Q has shown to be valid, reliable and responsive in patients with COPD.23,31,32
The global rating of change scale (GRC)33 was administered using an 11-point Likert scale ranging from −5 (much worse) to +5 (much better).33 Participants were asked to rate their perceived amount of change in cough and sputum after the PR program, compared to the initial assessment.
Intervention
All participants completed a 12-weeks community-based PR program, with two exercise training sessions per week and one psychoeducational session every two weeks, in 6 primary healthcare centers and in the Lab3R-Respiratory Research and Rehabilitation laboratory of the School of Health Sciences, University of Aveiro. Further information regarding the intervention has been published elsewhere.34
Data Analysis
Statistical analysis was performed using IBM SPSS Statistics, version 24 (IBM Corporation, Armonk, NY, USA) and plots created using GraphPad Prism, version 7 (GraphPad, San Diego, CA, USA) and MetaXL 5.3 (EpiGear International, Queensland, Australia). The level of significance was set at 0.05. The analysis included only participants that adhered to at least 65% of PR sessions (ie, participated in at least 8 weeks of PR).3,35
Changes in PROMs from T0 to T1 were analyzed with paired t-test or Wilcoxon signed-rank tests, accordingly to data normality verified with the Kolmogorov–Smirnov test. Floor or ceiling effects (more than 15% of the patients scoring at the bottom or top)36 were checked. Outlier’s analysis was performed (ie, inspection of extreme points on the plotted graphs of the variables studied) and, when present, were excluded.37 MCIDs were calculated for the LCQ and CASA-Q.
Since a gold standard to determine the MCID has not been established, concurrent comparisons of different methods were performed, integrating both anchor-based and distribution-based approaches.19,38 The final MCID for each measure was pooled by calculating the arithmetic weighted mean with the MCID generated by each anchor- and distribution-based method, which were then introduced into the MetaXL39 to create the MCIDs'plots. Anchor-based methods were weighed more than distribution methods (ie, 2/3 against 1/3).19,21,38,40
Minimal Important Clinical Differences
Anchor-Based Methods
Four anchor-based approaches were applied: i) patients referencing – GRC; ii) physiotherapists referencing – GRC; iii) questionnaire referencing – CAT and SGRQ and iv) criterion referencing – occurrence of an exacerbation, as following described.
- A change of 2 points or more in the patients’ GRC scale was considered clinically meaningful change.33 Thus, patients were categorized into two groups, those rating ≥2 and those rating <2 points in the GRC.
- Physiotherapists that conducted the exercise sessions were asked to judge about patients’ change in cough and sputum using a GRC. A change ≥2 was used as the cut-off point for improvement.33 Physiotherapists answered the GRC questions prior to assessing patients.
- Changes in the LCQ and CASA-Q scores were anchored against changes in the CAT total score and in the SGRQ total score. The MCIDs of CAT (2 points) and SGRQ (4 points) were used to discriminate between patients.41,42
- Having had an exacerbation during PR.18
The presence of a significant and moderate association (≥0.3) between the change in the PROM and the anchor was a requirement to proceed with the MCID calculation.38 Correlations were assessed using Pearson’s or Spearman’s coefficients and scatter plots were generated.
To calculate the MCID according to patients, physiotherapists and questionnaire referencing, three methods were used: i) the mean change, ie, the absolute difference between the two means of the PROM score (T1 and T0), calculated for patients who achieved the MCID established for the anchor17,19 ii) receiver operating characteristic (ROC) curves [the area under the curve (AUC) of a ROC≥0.7 was considered adequate43–45 and the closest point to the left corner, where specificity (SP) and sensitivity (SN) are both optimized was considered the optimal cut-off point] and iii) linear regression analysis, with the anchor change score being used as an independent variable.19
As for the criterion referencing, the difference in the baseline score between patients who experienced an exacerbation and those who did not was considered the MCID.18,46 Independent t-tests or Mann–Whitney tests, depending on data normality, were used.
Distribution-Based Methods
To calculate the MCID, five distribution-based methods were used: i) 0.5 times standard deviation (SD);47 ii) standard error of measurement (SEM);16 iii) 1.96 times SEM;47 iv) minimal detectable change (MDC)47 and v) effect size (ES)48 (Table 1).
 |
Table 1 Distribution-Based Methods to Estimate the Minimal Clinically Important Difference |
ES were interpreted as small (≥0.2), medium (≥0.5) or large (≥0.8).48 ES greater than 0.2 were considered to be minimally clinically/subjectively important.48 After combining both anchor- and distribution-based methods and pooling the final MCID for each PROM, the corresponding percentage of change was calculated. Furthermore, we used the pooled MCID value to compute the matching ES,19 using this formula:
Results
Sample Characterization
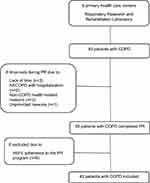
A flow diagram of the forty-nine patients included in the study is provided in Figure 1.
At baseline, no differences were observed between the included patients and drop-outs (p>0.05). Patients’ characteristics are summarized in Table 2.
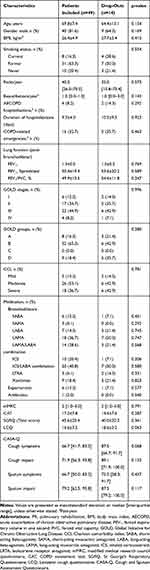 |
Table 2 Sample Characterization (n=63) |
At baseline, all PROMs were completed by the forty-nine participants, except for the CASA-Q. Data for CASA-Q was not possible to collect from eight participants, due to data collection commencement prior to obtaining the authorization to use the scale from the author. Additionally, one participant failed to complete the LCQ at the follow-up appointment. As missing data from CASA-Q and LCQ were completely unrelated to questionnaires scores, disease and symptoms severity, or patients’ adherence to PR (no statistical significant differences were present between the 8 patients with missing data on CASA-Q and the remaining 41 patients for GOLD stages/groups, adherence to PR, SGRQ and CAT scores), they were considered missing-completely-at-random.49,50 Thus, we chose to use the listwise deletion method to handle missing data, since this is the most frequently used method and is known for producing unbiased results.49,50 After the PR programme, significant improvements were found for CAT, SGRQ, LCQ and CASA-Q cough impact dimension. Baseline and post-PR scores can be found in Table S.1. Thirty-seven (75.5%) patients improved beyond the MCID of 2 points established for the CAT and 31 (63.3%) above the 4 points in the SGRQ. Only cough and sputum impact dimensions of CASA-Q demonstrated a ceiling effect, at T0 and T1. After the PR programs, 56.2% and 60.4% of the participants perceived improvements (GRC) in their cough (2.0, [0.0–3.0]) and sputum (2.0, [0.0–4.0]), respectively. Physiotherapists reported improvements in cough for 51% (2.0, [0.0–3.0]), and in sputum for 55.1% (2.0, [0.0–2.0]) of their patients.
Minimal Clinically Important Differences
After checking for outliers, three participants were excluded from the LCQ analysis. No differences were found between the baseline characteristics of the included patients and the outliers (p>0.05). No outliers were found in the CASA-Q analysis.
Resume tables of the correlation values between changes in the PROM and changes in the anchors (Table S.2) and the MCID achieved with the mean change method (Table S.3) can be found in Supplementary materials. It was not possible to use the criterion referencing method to compute the MCIDs since no significant differences were observed between patients who experienced an exacerbation and those who did not (Table S.4). In our sample, only mild to moderate exacerbations occurred.
Leicester Cough Questionnaire
Changes in the LCQ correlated significantly with changes in patients’ GRC for cough symptoms (r=0.340). No other correlations were found (Table S.2).
The MCID established for the LCQ using the mean change according to patients’ GRC was 1.4 (Table 3). It was not possible to use ROC statistics to compute the MCID, since the AUC generated was not significant. Using linear regression, the estimated MCID for the LCQ was 0.7 (Figure S.1).
 |
Table 3 Anchor and Distribution-Based Methods Used to Compute the Minimal Clinically Important Difference of Patient-Reported Outcome Measures Assessing Cough and Sputum |
The distribution-based methods for the LCQ, and the overall MCID pooled statistics are presented in Table 3. The pooled MCID for the LCQ was 1.3 (Figure 2).
Cough and Sputum Assessment Questionnaire
Changes in CASA-Q cough symptoms domain correlated significantly with changes in SGRQ (s=−0.322), in CAT (r=−0.378) and with patients’ GRC for cough (s=0.317). Changes in CASA-Q cough impact domain correlated significantly with patients’ GRC for cough (s=0.464). Changes in CASA-Q sputum domains, both symptoms and impact, correlated significantly with changes in SGRQ (s=−0.398 and r=−0.407, respectively). No other correlations were found (Table S.2).
The MCID derived from the mean change methods were: i) 9.3, 9.1 and 9.9 for cough symptoms with SGRQ, CAT and patients’ GRC, respectively; ii) 11.8 for cough impact; iii) 7.7 for sputum symptoms, and iv) 6 for sputum impact (Table 3).
Using ROC statistics, the AUCs generated for CASA-Q cough symptoms domain showed adequate discrimination between those improving above and below the MCID for SGRQ (AUC=0.70; 95% CI 0.54 to 0.86; p=0.031) and for CAT (AUC=0.79; 95% CI 0.62 to 0.97; p=0.005) (Figure 3). The AUCs obtained for the CASA-Q cough impact (patient’s GRC: AUC=0.74; 95% CI 0.59 to 0.90; p=0.008) and sputum symptoms (SGRQ: AUC=0.72; 95% CI 0.56 to 0.88; p=0.019) were also able to distinguish between patients who improved from those who did not (Figure 3). The AUCs’ discrimination ability was not acceptable for CASA-Q sputum impact using SGRQ and for CASA-Q cough symptoms using patients’ GRC for cough as anchors (ie, AUC<0.7). According to the ROC analysis, the MCID found were 4.2 for both cough and sputum symptoms and 4.7 for cough impact.
Using linear regression, the estimated MCID for the cough symptoms domain was 1.6 and for sputum impact domain was 2.2 (Figure S.2).
The distribution-based methods for the CASA-Q and the overall MCID pooled statistics are presented in Table 3. Pooled MCID for the CASA-Q subscales were 10.6 for cough symptoms; 10.1 for cough impact; 9.5 for sputum symptoms and 7.8 for sputum impact (Figure 4).
Discussion
This study estimated a pooled MCID of 1.3 for the LCQ. The pooled MCIDs established for CASA-Q domains were: 10.6 for cough symptoms; 10.1 for cough impact; 9.5 for sputum symptoms and 7.8 for sputum impact.
The pooled MCID found for LCQ matched previous estimates for patients with chronic cough, ie, 1.3 points.51 Nevertheless, higher MCIDs (from 2 to 3 points) have been suggested,52–54 using a GRC with a period recall of 6 months,52 increasing the recall risk of bias,19,46 and including patients with acute cough only.53,54 Higher levels of baseline severity (e.g., acute cough) usually lead to greater improvements,19,20,47 which result in larger MCIDs. Moreover, studies have involved pharmacological interventions only, whilst our study reports on PR. Since PR demands more from patients, expectations of benefits and improvements are often higher, producing larger effect sizes when compared to medication, thus, generating larger MCIDs.55
The pooled MCIDs for each CASA-Q domain were similar. Although CASA-Q emerged as a good tool to discriminate between patients above and below the anchor’s MCIDs, the ceiling effect observed was notorious; thus, its MCIDs should be interpreted with caution. Scores close to the end of the scale, limit the amount of potential change, affect responsiveness and consequently the establishment of the MCIDs.19 Presence of chronic cough was not an inclusion criterion of our study. This may explain the observed ceilings effects and why impact of cough and sputum in the HRQoL of our participants was comparable to other studies enrolling patients with COPD,31,56 but different (our sample scored better) from studies considering patients with chronic cough only.28,57,58
Although previous research has showed a relationship between CAT and cough and sputum,59,60 in our study an association was verified only with cough. However, association was explored with mean changes, while previous studies have focused on absolute scores.59,60 Nevertheless, correlations between changes in SGRQ and changes in CASA-Q cough and sputum dimensions were found (Table S.2). These findings further establish the impact of cough and sputum on HRQoL of patients with COPD, as previously demonstrated,7,59–64 and emphasise the urge for assessing and implementing tailored interventions to manage these symptoms.
It was not possible to use the physiotherapists’ GRC, probably due to the well-known lack of agreement on symptoms perception between patients with COPD and health professionals.65 Moreover, the non-significant differences in baseline symptoms between patients who experienced an AECOPD and those who did not, hindered the use of this variable in the anchor-based approach. In our sample, only mild to moderate exacerbations3 occurred and during PR, patients were closely monitored; therefore, exacerbations were promptly identified and tackled, thus enhancing a faster recovery.66 Exacerbations were managed as follows: patients were referred to their clinician, who adjusted their pharmacological therapy, and were instructed to follow the symptoms management strategies taught during educational sessions of PR (energy conservation techniques, postures to relieve dyspnoea and active cycle of breathing techniques).34 As soon as the contagious risk was controlled and patients felt capable they were encouraged to return to PR (around 7 to 15 days).67 When patients re-integrated the PR programme the training load was readjusted to their physical condition.
This study has some limitations that need to be acknowledged. First, the presence of ceiling effects in the CASA-Q might have biased the results. Secondly, our sample was mainly composed of patients with moderate to severe COPD. Therefore, the established MCIDs might not be generalizable to all patients and should be interpreted with caution in patients with mild or very severe COPD. Since MCIDs are influenced by disease severity,19,20,47 we recommend interpreting PROMs changes within the MCIDs ranges provided by the different methods and not limiting it to the absolute proposed value. Finally, this study is integrated in a larger trial,34 therefore, a specific sample size calculation to establish MCIDs was not performed, which might have underpowered this study for its goal. Nevertheless, similar samples sizes have been used to establish MCIDs in other studies,40,68,69 and the fact that all MCIDs fell within the recommended range of 6 to 10% change in the scale range, which corresponded to a desirable effect size of 0.2 to 0.5,19,38,48 strengthens the validity of our estimates. Moreover, to our best knowledge, this is the first study to provide MCIDs estimations for LCQ and CASA-Q in patients with COPD; thus, we believe that our MCIDs estimates can be useful for health professional and policy makers, ensuring they are used with caution and in accordance with each clinical context. To confirm our MCIDs estimates, future studies in this area with larger sample sizes are still required.
An important strength of our study is that MCIDs were computed through a combination of methodologies, including a wide range of anchor and distribution-based approaches. In addition, the pooled method selected allowed to attribute a higher weight to anchor than distribution-based methods, following the recommendations for establishing MCIDs.19,38 Standardization of community-based PR programs in terms of structure, intensity, frequency, duration and progression, as recommended,13 minimised the heterogeneity of intervention, assuring that the MCIDs proposed are valid and suitable for PR.
Conclusion
In summary, this study suggests that improvements of 1.3 in the LCQ, 10.6 in the cough symptoms, 10.1 in cough impact, 9.5 in sputum symptoms and 7.8 points in the sputum impact dimensions of CASA-Q are clinically relevant for patients with COPD, following a PR program. These estimates have the potential to be used to interpret clinical relevance, as thresholds for the intervention effectiveness and to inform future studies regarding sample calculation.
Data Sharing Statement
Data sets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Author Contributions
PR and CP were responsible for data acquisition. PR also performed formal data analysis and drafted the manuscript. AM and AO were responsible for the conception and design of the study. CV and LA provided a substantial contribution for data acquisition. AM was responsible for obtaining the funding and ensured project administration and resources. All authors critically revised the manuscript, ensured accuracy and integrity of the work, approved the final version to be published, and agree to be accountable for all aspects of the work.
Funding
This work was funded by Fundo Europeu de Desenvolvimento Regional (FEDER) - Comissão Diretiva do Programa Operacional Regional do Centro, by Fundação para a Ciência e Tecnologia - FCT (SAICT-POL/23926/2016, PTDC/DTP-PIC/2284/2014, PTDC/SAU-SER/28806/2017, UIDB/04501/2020) and by Programa Operacional Competitividade e Internacionalização (COMPETE), through COMPETE 2020 (POCI-01-0145-FEDER-016701, POCI-01-0145-FEDER-007628, POCI-01-0145-FEDER-028806).
Disclosure
The authors report no financial, or non-financial, conflicts of interest in this work.
References
1. Forum of International Respiratory Societies. The Global Impact of Respiratory Disease.
2. López‐Campos JL, Tan W, Soriano JB. Global burden of COPD. Respirology. 2016;21(1):14–23. doi:10.1111/resp.12660
3. Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2019 Report). 2019.
4. Kessler R, Partridge MR, Miravitlles M, et al. Symptom variability in patients with severe COPD – a pan-European cross-sectional study. Eur Respir J. 2011;37(2):264–272. doi:10.1183/09031936.00051110
5. Miravitlles M, Worth H, Cataluña JJS, et al. Observational study to characterise 24-hour COPD symptoms and their relationship with patient-reported outcomes: results from the ASSESS study. Respir Res. 2014;15(1):122. doi:10.1186/s12931-014-0122-1
6. Crooks MG, Brown T, Morice AH. Is cough important in acute exacerbations of COPD? Respir Physiol Neurobiol. 2018;257:30–35. doi:10.1016/j.resp.2018.02.005
7. Miravitlles M, Ribera A. Understanding the impact of symptoms on the burden of COPD. Respir Res. 2017;18(1):67. doi:10.1186/s12931-017-0548-3
8. Satia I, Badri H, Lahousse L, Usmani OS, Spanevello A. Airways diseases: asthma, COPD and chronic cough highlights from the European Respiratory Society Annual Congress 2018. J Thorac Dis. 2018;10(Suppl 25):S2992. doi:10.21037/jtd.2018.08.23
9. Crooks MG, Hayman Y, Innes A, Williamson J, Wright CE, Morice AH. Objective measurement of cough frequency during COPD exacerbation convalescence. Lung. 2016;194(1):117–120. doi:10.1007/s00408-015-9782-y
10. Smith J, Woodcock A. Cough and its importance in COPD. Int J Chron Obstruct Pulmon Dis. 2006;1(3):305.
11. van Buul AR, Kasteleyn MJ, Chavannes NH, Taube C. Morning symptoms in COPD: a treatable yet often overlooked factor. Expert Rev Respir Med. 2017;11(4):311–322. doi:10.1080/17476348.2017.1305894
12. Calverley PM. Cough in chronic obstructive pulmonary disease: is it important and what are the effects of treatment? Cough. 2013;9(1):17. doi:10.1186/1745-9974-9-17
13. Spruit MA, Singh SJ, Garvey C, et al. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13–e64. doi:10.1164/rccm.201309-1634ST
14. McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015;23(2):CD003793.
15. Johnston BC, Ebrahim S, Carrasco-Labra A, et al. Minimally important difference estimates and methods: a protocol. BMJ Open. 2015;5(10):e007953. doi:10.1136/bmjopen-2015-007953
16. Guyatt GH, Osoba D, Wu AW, Wyrwich KW, Norman GR. Methods to explain the clinical significance of health status measures. Mayo Clin Proc. 2002;77(4):371–383. doi:10.4065/77.4.371
17. Jaeschke R, Singer J, Guyatt GH. Measurement of health status: ascertaining the minimal clinically important difference. Control Clin Trials. 1989;10(4):407–415. doi:10.1016/0197-2456(89)90005-6
18. Jones P. Interpreting thresholds for a clinically significant change in health status in asthma and COPD. Eur Respir J. 2002;19(3):398–404. doi:10.1183/09031936.02.00063702
19. Angst F, Aeschlimann A, Angst J. The minimal clinically important difference raised the significance of outcome effects above the statistical level, with methodological implications for future studies. J Clin Epidemiol. 2017;82:128–136. doi:10.1016/j.jclinepi.2016.11.016
20. Wright A, Hannon J, Hegedus EJ, Kavchak AE. Clinimetrics corner: a closer look at the minimal clinically important difference (MCID). J Man Manip Ther. 2012;20(3):160–166. doi:10.1179/2042618612Y.0000000001
21. Alma H, de Jong C, Tsiligianni I, Sanderman R, Kocks J, van der Molen T. Clinically relevant differences in COPD health status: systematic review and triangulation. Eur Respir J. 2018;52(3):1800412. doi:10.1183/13993003.00412-2018
22. Birring S, Prudon B, Carr A, Singh S, Morgan M, Pavord I. Development of a symptom specific health status measure for patients with chronic cough: Leicester Cough Questionnaire (LCQ). Thorax. 2003;58(4):339–343. doi:10.1136/thorax.58.4.339
23. Crawford B, Monz B, Hohlfeld J, et al. Development and validation of a cough and sputum assessment questionnaire. Respir Med. 2008;102(11):1545–1555. doi:10.1016/j.rmed.2008.06.009
24. Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. doi:10.1016/0895-4356(94)90129-5
25. Bestall J, Paul E, Garrod R, Garnham R, Jones P, Wedzicha J. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax. 1999;54(7):581–586. doi:10.1136/thx.54.7.581
26. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD assessment test. Eur Respir J. 2009;34(3):648–654. doi:10.1183/09031936.00102509
27. Jones PW, Quirk FH, Baveystock CM, Littlejohns P. A self-complete measure of health status for chronic airflow limitation. Am Rev Respir Dis. 1992;145(6):1321–1327. doi:10.1164/ajrccm/145.6.1321
28. Berkhof FF, Boom LN, Ten Hertog NE, Uil SM, Kerstjens HA, van den Berg JW. The validity and precision of the Leicester Cough Questionnaire in COPD patients with chronic cough. Health Qual Life Outcomes. 2012;10(1):4. doi:10.1186/1477-7525-10-4
29. Faruqi S, Thompson R, Wright C, Sheedy W, Morice AH. Quantifying chronic cough: objective versus subjective measurements. Respirology. 2011;16(2):314–320. doi:10.1111/res.2011.16.issue-2
30. Kelsall A, Decalmer S, Webster D, et al. How to quantify coughing: correlations with quality of life in chronic cough. Eur Respir J. 2008;32(1):175–179. doi:10.1183/09031936.00101307
31. Deslee G, Burgel P-R, Escamilla R, et al. Impact of current cough on health-related quality of life in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2016;11:2091. doi:10.2147/COPD
32. Monz BU, Sachs P, McDonald J, Crawford B, Nivens MC, Tetzlaff K. Responsiveness of the cough and sputum assessment questionnaire in exacerbations of COPD and chronic bronchitis. Respir Med. 2010;104(4):534–541. doi:10.1016/j.rmed.2009.10.026
33. Kamper SJ, Maher CG, Mackay G. Global rating of change scales: a review of strengths and weaknesses and considerations for design. J Man Manip Ther. 2009;17(3):163–170. doi:10.1179/jmt.2009.17.3.163
34. Marques A, Jácome C, Rebelo P, et al. Improving access to community-based pulmonary rehabilitation: 3R protocol for real-world settings with cost-benefit analysis. BMC Public Health. 2019;19:676. doi:10.1186/s12889-019-7045-1
35. Alison JA, McKeough ZJ, Johnston K, et al. Australian and New Zealand pulmonary rehabilitation guidelines. Respirology. 2017;22(4):800–819. doi:10.1111/resp.2017.22.issue-4
36. McHorney CA, Tarlov AR. Individual-patient monitoring in clinical practice: are available health status surveys adequate? Qual Life Res. 1995;4(4):293–307. doi:10.1007/BF01593882
37. Aggarwal R, Ranganathan P. Common pitfalls in statistical analysis: the use of correlation techniques. Perspect Clin Res. 2016;7(4):187. doi:10.4103/2229-3485.192046
38. Revicki D, Hays RD, Cella D, Sloan J. Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. J Clin Epidemiol. 2008;61(2):102–109. doi:10.1016/j.jclinepi.2007.03.012
39. Doi SA, Thalib L. A quality-effects model for meta-analysis. Epidemiology. 2008;19(1):94–100. doi:10.1097/EDE.0b013e31815c24e7
40. Oliveira A, Machado A, Marques A. Minimal important and detectable differences of respiratory measures in outpatients with AECOPD. COPD. 2018;1–10.
41. Jones PW. St. George’s respiratory questionnaire: MCID. COPD. 2005;2(1):75–79. doi:10.1081/COPD-200050513
42. Kon SS, Canavan JL, Jones SE, et al. Minimum clinically important difference for the COPD assessment test: a prospective analysis. Lancet Respir Med. 2014;2(3):195–203. doi:10.1016/S2213-2600(14)70001-3
43. Terwee CB, Bot SD, de Boer MR, et al. Quality criteria were proposed for measurement properties of health status questionnaires. J Clin Epidemiol. 2007;60(1):34–42. doi:10.1016/j.jclinepi.2006.03.012
44. Carter JV, Pan J, Rai SN, Galandiuk S. ROC-ing along: evaluation and interpretation of receiver operating characteristic curves. Surgery. 2016;159(6):1638–1645. doi:10.1016/j.surg.2015.12.029
45. Mandrekar JN. Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol. 2010;5(9):1315–1316. doi:10.1097/JTO.0b013e3181ec173d
46. Alma H, De Jong C, Jelusic D, et al. Health status instruments for patients with COPD in pulmonary rehabilitation: defining a minimal clinically important difference. NPJ Prim Care Respir Med. 2016;26:16041. doi:10.1038/npjpcrm.2016.41
47. Copay AG, Subach BR, Glassman SD, Polly DW, Schuler TC. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 2007;7(5):541–546. doi:10.1016/j.spinee.2007.01.008
48. Cohen J. A power primer. Psychol Bull. 1992;112(1):155. doi:10.1037/0033-2909.112.1.155
49. Kang H. The prevention and handling of the missing data. Korean J Anesthesiol. 2013;64(5):402. doi:10.4097/kjae.2013.64.5.402
50. Wells BJ, Chagin KM, Nowacki AS, Kattan MW. Strategies for handling missing data in electronic health record derived data. EGEMS. 2013;1:3. doi:10.13063/2327-9214.1035
51. Raj A, Pavord D, Birring S. Clinical cough IV: what is the minimal important difference for the Leicester Cough Questionnaire? Pharmacol Ther Cough. 2009;311–320.
52. Brokkaar L, Uil SM, Van Den Berg JWK. Minimal Clinically Important Difference (MCID) of the Dutch version of the Leicester Cough Questionnaire and baseline predictors of reaching the MCID after six months. Chest. 2007;132(4):468B. doi:10.1378/chest.132.4_MeetingAbstracts.468b
53. Lee KK, Matos S, Evans DH, White P, Pavord ID, Birring SS. A longitudinal assessment of acute cough. Am J Respir Crit Care Med. 2013;187(9):991–997. doi:10.1164/rccm.201209-1686OC
54. Yousaf N, Lee KK, Jayaraman B, Pavord ID, Birring SS. The assessment of quality of life in acute cough with the Leicester Cough Questionnaire (LCQ-acute). Cough. 2011;7(1):4. doi:10.1186/1745-9974-7-4
55. Houchen-Wolloff L, Evans RA. Unravelling the mystery of the ‘minimum important difference’ using practical outcome measures in chronic respiratory disease. Chron Respir Dis. 2019;16:1479973118816491. doi:10.1177/1479973118816491
56. Arikan H, Savci S, Calik-Kutukcu E, et al. The relationship between cough-specific quality of life and abdominal muscle endurance, fatigue, and depression in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2015;10:1829.
57. Polley L, Yaman N, Heaney L, et al. Impact of cough across different chronic respiratory diseases: comparison of two cough-specific health-related quality of life questionnaires. Chest. 2008;134(2):295–302. doi:10.1378/chest.07-0141
58. Reychler G, Schinckus M, Fremault A, Liistro G, Pieters T. Validation of the French version of the Leicester Cough Questionnaire in chronic obstructive pulmonary disease. Chron Respir Dis. 2015;12(4):313–319. doi:10.1177/1479972315602618
59. Koo H-K, Park S-W, Park J-W, et al. Chronic cough as a novel phenotype of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2018;13:1793. doi:10.2147/COPD
60. Jones PW, Brusselle G, Dal Negro RW, et al. Patient-centred assessment of COPD in primary care: experience from a cross-sectional study of health-related quality of life in Europe. Prim Care Respir J. 2012;21(3):329. doi:10.4104/pcrj.2012.00065
61. Lee H, Jhun BW, Cho J, et al. Different impacts of respiratory symptoms and comorbidities on COPD-specific health-related quality of life by COPD severity. Int J Chron Obstruct Pulmon Dis. 2017;12:3301. doi:10.2147/COPD
62. Monteagudo M, Rodríguez-Blanco T, Llagostera M, et al. Factors associated with changes in quality of life of COPD patients: a prospective study in primary care. Respir Med. 2013;107(10):1589–1597. doi:10.1016/j.rmed.2013.05.009
63. Miravitlles M. Cough and sputum production as risk factors for poor outcomes in patients with COPD. Respir Med. 2011;105(8):1118–1128. doi:10.1016/j.rmed.2011.02.003
64. Stephenson JJ, Cai Q, Mocarski M, Tan H, Doshi JA, Sullivan SD. Impact and factors associated with nighttime and early morning symptoms among patients with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2015;10:577. doi:10.2147/COPD
65. Miravitlles M, Ferrer J, Baró E, Lleonart M, Galera J. Differences between physician and patient in the perception of symptoms and their severity in COPD. Respir Med. 2013;107(12):1977–1985. doi:10.1016/j.rmed.2013.06.019
66. Oliveira A, Munhá J, Bugalho A, Guimarães M, Reis G, Marques A. Identification and assessment of COPD exacerbations. Pulmonology. 2018;24(1):42–47. doi:10.1016/j.rppnen.2017.10.006
67. Oliveira A, Afreixo V, Marques A. Enhancing our understanding of the time course of acute exacerbations of COPD managed on an outpatient basis. Int J Chron Obstruct Pulmon Dis. 2018;13:3759. doi:10.2147/COPD
68. Beauchamp MK, Harrison SL, Goldstein RS, Brooks D. Interpretability of change scores in measures of balance in people with COPD. Chest. 2016;149(3):696–703. doi:10.1378/chest.15-0717
69. Hanson LC, Taylor NF, McBurney H. Interpreting meaningful change in the distance walked in the 10-metre ISWT in cardiac rehabilitation. Heart Lung Circ. 2018.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.