Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
MicroRNA-488-3p Regulates Neuronal Cell Death in Cerebral Ischemic Stroke Through Vacuolar Protein Sorting 4B (VPS4B)
Authors Zhou L, Yang W, Yao E, Li H, Wang J, Wang K, Zhong X, Peng Z, Huang X
Received 26 March 2020
Accepted for publication 23 November 2020
Published 7 January 2021 Volume 2021:17 Pages 41—55
DOI https://doi.org/10.2147/NDT.S255666
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Li Zhou,1 Wanxin Yang,1 Enping Yao,1 Haiyan Li,2 Jihui Wang,2 Kun Wang,3 Xiaohua Zhong,1 Zhongxing Peng,4 Xuming Huang1
1Department of Rehabilitation, The First Affiliated Hospital of Guangdong Pharmaceutical University, Guangzhou 510080, People’s Republic of China; 2Department of Neurology, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou 510000, People’s Republic of China; 3School of Health Science, Guangdong Pharmaceutical University, Guangzhou 510310, People’s Republic of China; 4Department of Neurology, The First Affiliated Hospital of Guangdong Pharmaceutical University, Guangzhou 510080, People’s Republic of China
Correspondence: Xuming Huang
Department of Rehabilitation, The First Affiliated Hospital of Guangdong Pharmaceutical University, Guangzhou 510080, People’s Republic of China
Tel +86-20-82804660
Email [email protected]
Zhongxing Peng
Department of Neurology, The First Affiliated Hospital of Guangdong Pharmaceutical University, Guangzhou 510080, People’s Republic of China
Email [email protected]
Background: Ischemic stroke, which often occurs with high morbidity, disability, and mortality, is a main cause of brain disease. In various types of human diseases, it is found that microRNAs (miRNAs) are considered as gene regulators. Increasing studies have proved that fluctuation of miRNAs, in the pathologies of ischemic stroke, plays a vital role. However, the accurate regulatory mechanism of cerebral ischemic stroke by miRNAs is still unclear. In this research, we investigated the inhibition mechanism of miR-488-3p on neuronal death through targeting vacuolar protein sorting 4B (VPS4B) in cerebral ischemia/reperfusion (I/R) injury.
Methods: Western blot and qRT-PCR were utilized to detect the miR-488-3p level and VPS4B expression. The cell counting kit-8 (CCK-8) assay was utilized to measure the function of miR-488-3p in cell death induced by oxygen glucose deprivation/reoxygenation (OGD/R). After middle cerebral artery occlusion/reperfusion (MCAO/R), the impact of miR-488-3p on infarct volume in mouse brain was assessed. The targets of miR-488-3p were confirmed by luciferase analysis and bioinformatics software.
Results: The miR-488-3p level remarkably reduced in primary neuronal cells administrated with OGD/R. Similarly, it also decreased in the mouse brain administrated with MCAO/R. Additionally, the up-regulation of miR-488-3p expression suppressed the death of neuronal cells and restrained ischemic brain infarction in ischemia-stroked mice. Besides, the results showed that VPS4B, which could be inhibited by miR-488-3p, was a direct target of miR-488-3p. This research revealed that the inhibition of VPS4B protected the neuronal cells in ischemic stroke both in vitro as well as in vivo. Meanwhile, this inhibition strengthened positive impact generated by miR-488-3p on ischemic injury.
Conclusion: Overall, miR-488-3p played a critical role on neuroprotective function via reducing VPS4B protein level. These results performed a new underlying curative target for the treatment of cerebral ischemic stroke.
Keywords: miR-488-3p, VPS4B, ischemia/reperfusion, I/R, ischemic stroke
Introduction
Ischemic stroke (IS) is a kind of cerebrovascular event, which is induced by thrombosis or embolism. The vessels are rapidly occluded and blood flow is interrupted when it occurs in important or large cerebral vessels, with the subsequently irreversible or not irreversible neurologic dysfunction in a certain region of the brain.1,2 It is still one of the critical factors causing high-level debility and mortality on a global scale. Stroke initiates numerous incidents bringing about acute neuronal injury and cell death.3 Currently, a good deal of therapy approaches are managed clinically for IS, for instance, intravenous thrombolytics and endovascular thrombectomy. Other neurorestorative therapies are also applied to the therapy and prognosis of stroke, such as cell treatment, anti-inflammation treatment, and neuroprotection. These therapies have actually been proved to ameliorate the endings in appropriate patients, indicating significant improvements of stroke treatment, but there is still a poor prognosis in many stroke patients.2,4 The main causes of poor prognosis are rapid neuron damage and extensive cell death following the interrupted blood flow.5 Hence, there is a pressing need for novel therapies for ischemic stroke. Since stroke triggers rapid neuron damage and cell death, new therapy or neuroprotective strategies with the purpose of rescuing and restoring the damaged brain tissue are essential and imperative.
Lots of literatures have stated the essential roles of microRNAs (miRNAs) playing in physiological and pathological progressions of specific brain diseases as the critical mediators of gene silencing. There are also numerous studies that have reported that the level of miRNAs is up-regulated or down-regulated in quite a lot of neurodegenerative and neurological events, including IS.6,7 The alteration expression of miRNAs would be capable of considerably modulating the development of oxidative stress, cytotoxicity, inflammation, neuron apoptosis, and angiogenesis, and so on,8,9 suggesting that miRNAs are one of the profoundly notable molecules in ischemic stroke event progression. Other miRNAs have been noticeably studied as prospective therapy targets improving the prognosis of stroke.10 Massive studies have reported on distinguishing different miRNAs which can help to comprehend the ischemic stroke progression itself as well as identify the potential therapeutic schemes specifically.10,11 For example, studies have reported that miRNA 21 and miR-99a decrease the ischemic infarction volume, reduce neuronal apoptosis, and improve the neurological scores in ischemic stroke rats.12,13 Other studies presented that miR-367 and miR-132 could relieve neuropathological alterations through reducing neuroinflammation and protecting neurons in a stroke event.14,15 In addition, 16 miRNAs show significant deregulation in ischemic stroke serum samples, among which four miRNAs, including PC-5p-12969, PC-3p-57664, hsa/mmu-miR-211–5p, and hsa/mmu miR-122–5p, are up-regulated almost consistently and may be involved in stroke regulation.16 Up-regulation of PC-5P-12969 is observed at 4 hours and 1-day post-stroke, indicating PC-5P-12969 is a latent biomarker for ischemic stroke.17 A previous review exhibits the latest developments on identifying biomarkers in tissues of the peripheral and central nervous system and miRNAs alterations in stroke patients.18 Currently the probability of miRNAs to modulate physiological processes emphasizes their therapeutic value for stroke recovery.
Actually, it has been identified that miR-488-3p is involved in the progression of several cancers, such as esophageal squamous cell carcinoma,19 glioma,20 and retinoblastoma.21 Besides, miR-488-3p also participates in other disorders to regulate cell proliferation, migration, and apoptosis. For example, circular RNA CCDC66 targets DCX to regulate cell migration and proliferation in Hirschsprung’s disease through functioning as a sponge for miR-488-3p.22 LncRNA PVT1 modulates apoptosis of chondrocyte via sponging miR-488-3p in osteoarthritis.23 miRNA profiling of small intestinal neuroendocrine tumors identifies reduced level of miR-488-3p in tumors of cluster M1.24 The expression levels of rno-miR-488-3p is verified significantly downregulated in L3–L6 dorsal root ganglion in neuropathic pain rats caused by spared nerve injury (SNI).25 A study has mapped GluR2+ extracellular vesicle (EV) miRNA across many injury types and produced a panel of eight miRNAs, including miR-488-3p, for injured mice versus sham mice, to classify specific states of injury for traumatic brain injury (TBI).26 However, it remains unclear about the expression and the underlying pathophysiology mechanism of miR-488-3p in ischemic stroke.
The vacuolar protein sorting 4 (VPS4) is one of the members of a group of genes encoding the vacuolar protein sorting (VPS) proteins and is a component of the endosomal sorting complex required for transport (ESCRT) machinery. ESCRT also includes escrt-I, escrt-II, and escrt-III, which are recruited into the cell membrane to mediate cell fission and cell scission.27,28 VPS4 consists of VPS4A and VPS4B, which are two parallel hexametric rings and are usually located in the cytoplasm.29 VPS4B, also known as vacuolar protein sorting 4 homolog B, reduces the efficient transfer of vacuolar hydrolases when the cell function is impaired.30,31 Previous studies have reported that VPS4B is up-regulated in many diseases and is closely related to disease progression and diagnosis. VPS4B is highly expressed in mouse dental pulp cells of the molar tooth germ, and significantly increased in the odontoblastic differentiation of human dental pulp stem cells (hDPSCs).32 In Parkinson’s Disease, VPS4 acts as the target site of alpha-synuclein (α-SYN) in regulation of lysosomal or extracellular secretion, leading to intercellular propagation of Lewy pathology.33 It was found that VPS4B level was significantly increased in the hippocampus of adult mice with Middle Cerebral Artery Occlusion (MCAO).34 However, the pathophysiological alterations and underlying mechanisms of VPS4B in neurons of ischemic stroke are still unclear.
This study used oxygen glucose deprivation/reoxygenation (OGD/R) of cultured cortical neurons and mouse transient middle cerebral artery occlusion/reperfusion (MCAO/R) model of ischemic stroke, to explore the expression and role of miR-488-3p in the development of ischemic stroke, and then to elucidate the pathological mechanism of miR-488-3p in ischemic stroke by examining whether there was a targeting relationship between miR-488-3p and the predicted target gene VPS4B. The results may provide therapeutic targets for controlling progression or improving prognosis in ischemic stroke.
Materials and Methods
Reagents
The miR-488-3p mimic and its negative control (NC), miR-488-3p inhibitor and its negative control (NC), and lentivirus-mediated VPS4B shRNA (sh-VPS4B) and its negative control (sh-NC) were obtained from Ribobio (Guangzhou, China). Trizol reagent, the Reverse Transcriptase kit, and MTT Cell Viability Assay Kit were bought from Invitrogen (Carlsbad, CA, USA). The primary antibody including anti-VPS4B (ab224736, Abcam) and anti-GAPDH (ab8245, Abcam) were applied from Abcam. Other reagents and buffers used in this research were all obtained from Sigma-Aldrich.
Cell Culture in vitro and Administration of Oxygen Glucose Deprivation/Reoxygenation (OGD/R)
Cortical Neurons were cultured at a concentration of 1×106 cells/mL. They were separated from the cerebral cortex of postnatal 10–17 days old mice according to the earlier guidelines. These neurons were cultured using neurobasal medium (Invitrogen, Carlsbad, CA, USA) supplied with 2% B27 (Invitrogen), keeping at 37°C with 95% air and 5% CO2 in a humidified incubator. Fresh medium was replaced every 2 days. Experiments related with the cell culture were all operated 24 hours later following neurons planted.
To administrate the oxygen glucose deprivation/reoxygenation (OGD/R) simulating ischemic conditions in vitro, then the cultured cells were removed to Dulbecco’s Modified Eagle’s Medium (DMEM) with glucose-free at 37°C, combining with the mixture of 1% O2, 5% CO2, and 94% N2 in a humidified incubator for a total 2 hours. After that, these cells were then incubated in DMEM containing glucose within 95% air and 5% CO2 conditions, again lasting for total 24 hours to perform reoxygenation. The neurons of the control group were cultured in DMEM containing glucose in normal conditions, which is 37°C with 95% air and 5% CO2 in a humidified incubator, without OGD/R.
Animal Model of Ischemic Stroke: Middle Cerebral Artery Occlusion/Reperfusion (MCAO/R)
For the reason that estrogen secreted by female rats/mice was not conducive to the stability of the I/R model, male mice were selected for the study. Mice (male, 8–10 weeks, 25±3 g) were all purchased from the Laboratory Animal Department (Sun Yat-sen University, China). And these mice were all raised and kept in a pathogen-free apparatus. All mouse experimental operations have been approved by the Animal Care Committee of The First Affiliated Hospital of Guangdong Pharmaceutical University. All the procedures involved in the study were conducted in accordance with the guidelines published by China Animal Protection Association. The mice were individually housed with a 12/12 hour light/dark cycle in a room. They could obtain food and water ad libitum freely. The room temperature and humidity were respectively kept in 24±1°C and 50–60%. We adopted the improved method of MCAO/R with slight modifications to perform an ischemic stroke mice model as indicated before, with the purpose of extended survival time of the animals following the MCAO/R surgery. In brief, after being anesthetized with sodium pentobarbital using the concentration of 40 mg/kg per mouse, animals were placed on the operating table. Two blood vessels were fully exposed, including the left external carotid artery and the left common artery. The cusp of 5–0 monofilament nylon suture was smoothed by heat to some extent, and then the left internal carotid artery was occluded with it to the bottom of the middle cerebral artery. Animals were housed individually and recovered for a total 24 hours after 2 hours of the MCAO surgery. Mice in the sham group were only exposed to the same arteries with no artery occlusion. These animals were sacrificed following analgesia and brains were obtained for the next biochemical detection. The injury areas of brains were applied to evaluate the level of protein and mRNA. Two pathologists needed to calculate the animal mortality rate and the data is ~20%, owing to the variant personal resistance to the vascular occlusion mainly and the operation itself.
Neurons Transfected with the miR-488-3p Mimic or miR-488-3p Inhibitor
Six-well or 24-well plates were applied to plant the neurons obtained from the cerebral cortex in the culture medium with antibiotic-free and cells were incubated for a total 24 hours. After that, applying Lipofectamine 2000 (Invitrogen), the cells were transfected with the miR-488-3p mimic, the miR-488-3p inhibitor, or the sh-VPS4B with a concentration of 20 nM in accordance with the manufacturer’s protocols.
In brief, 250 μL serum-free Opti-MEM medium (Gibco Company, Grand Island, NY, USA) was used to dilute the miR-488-3p mimic, miR-488-3p inhibitor, sh-VPS4B (final concentration=20 nM) and then gently mixed and incubated at room temperature for 5 minutes. Then 250 μL serum-free Opti-MEM was used to dilute 5 μL lipofectamine 2000, gently mixed, and incubated at room temperature for 5 min. This was followed by mixing these two above-mentioned dilutions and incubating at room temperature for 20 minutes before adding them to plate wells. After being cultured with 5% CO2 at 37°C for 6~8 hours, the mixture were continuously cultured in complete medium. Twenty-four hours later the neurons were performed with OGD/R. Then we collected samples for detecting the mRNA level with qRT-PCR, the proteins level with Western blot, the cell viability with MTT, and cell apoptosis with flow cytometry.
Treatment with miR-488-3p Mimic, miR-488-3p Inhibitor, or sh-VPS4B in Mice
We injected miR-488-3p mimics, miR-488-3p inhibitor, or sh-VPS4B into the mice cortex and the animals in the sham group were performed with corresponding negative controls as described before. Briefly, mice were anesthetized with 40 mg/kg pentobarbital sodium, and then fixed in a stereotactic frame (Stoelting, Dublin, Ireland), equipped with a warming pad and temperature feedback control for efficient control of the physiological state (medres GmbH, Cologne, Germany). The skull was exposed by a small incision and one hole (0.5 mm diameter, F.S.T.) was drilled for injection on the ipsilateral hemisphere. The stereotaxic coordinates were 0.5 mm posterior, 1.0 mm lateral to the bregma, and 2.5–3.0 mm ventral to the bregma. Then mixing miR-488-3p mimic, miR-488-3p inhibitor, or the controls with siRNA-Mate (GenePharma) at a concentration of 100 μM. Placing the mixtures for a total 20 minutes at room temperature. Lastly, 7 µL reagent per animal was injected into the cortex, lasting 10 minutes. Similarly, sh-VPS4B at a concentration of 109 TU per mL was mixed with 4 μg/μL cationic lipid polybrene (GenePharma) and the mixture placed for a total 15 minutes at room temperature. Then the mixture was injrcted into the cortex lasting for 10 minutes. The wound was sutured and the animal woke up under controlled conditions in an incubation chamber at 30°C. Following the injection, animals were suffered from MCAO/R operation.
Fluorescence in situ Hybridization (FISH)
A directly labeled fluorescent probe was used for FISH assay. According to the manufacturer’s procedure, miR-488-3p-specific probe obtained from SpectrumGreen (Biosense, Science City, Guangzhou, China) was applied. Then FISH assay was operated, including the preparation of a probe and slide, hybridization, and washing post-hybridization. In brief, mice brains were first cut within a cryostat for collecting coronal sections (5 μm). Sections were thawed on slides and fixed using 4% ice-cold paraformaldehyde. Dehydration was performed by using a graded ethanol series. Slides were kept at 4°C within 95% ethanol, before hybridization they needed to be air-dried. The blending of the probe diluents and FITC fluorescent tag probe (Biosense, Science City, Guangzhou, China) were mixed to harvest the probe hybridization mixture. Then the mixture was denatured for 5 minutes at 78°C, and reannealed for 30 minutes at 37°C. After that, the mixture was used immediately under a coverslip for sample hybridization in a humidified chamber at 42°C overnight. After being washed three times at 43°C in 50% formamide/2×SSC (pH 7.0) solution and washed two times at 43°C in 2×SSC (pH 7.0) solution (each time lasted 5 minutes), antifade solution containing DAPI was applied for counterstaining. Then a coverslip was covered onto the slide. An inverted fluorescence microscope (DMI6000B, Leica, Zhengzhou, China) was used to capture fluorescence images at ×200 and ×400 magnification.
Histopathological Observation
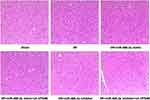
The mice (n=6/per group) were anesthetized with 40 mg/kg sodium pentobarbital and perfused transitorily with physiological saline until the liver appeared to be white, followed by 4% paraformaldehyde in the phosphate buffered saline (PBS) (0.1 M, PH 7.4). Each brain was rapidly removed and immersed in 10% formalin for 2 hours, then was washed three times using the PBS. After being dehydrated and embedded with paraffin, the brain tissues were cut into 5-µm-thick coronal sections for the HE staining. The sections were counterstained with hematoxylin in nucleuses and differentiated by eosin in the cytoplasm. The area of interest in the cortex was scanned under high-power magnification for histopathological observation in a blinded manner.
Infarct Volume Evaluation
Animals were anesthetized 24 hours following MCAO/R and sacrificed to obtain the brains. For infarct volume evaluation, brains were sliced into coronal sections with the thickness of 1.0 mm. Brain sections were treated at 37°C with 2,3,5-triphenyl tetrazolium chloride (TTC) solution (0.5%) for 15 minutes. The stained sections were then checked to get images with a computer and evaluated in Image Pro 6.0 software (Media Cybernetics, Silver Spring, MD, USA). Thirty-six animals were used in behavioral procedures.
Neurological Function Test
The neurological function test in mice suffering from cerebral I/R was assessed by applying the scales according to guidelines earlier. The standard is: 0 point, animals perform normally; 1 point, animals are not able to totally stretch the left front legs; 2 points, animals turn around into a circle; 3 points, animals fall down to the left side; 4 points, animals are not able to move by their own efforts, without consciousness. Thirty-six animals were used in behavioral procedures.
qRT-PCR Detection
Total RNA was obtained from brain tissues in vivo and the cultured neurons in vitro applying Trizol (BBI, Toronto, Canada) following the procedure. We used a TaqMan™ miRNA assay kit (Applied Biosystems, Foster City, CA, USA) and a Prime Script™ RT reagent kit (Takara, Shiga, Japan) to conduct the reverse transcription procedure according to the manufacturer’s guidelines, We then performed the qRT-PCR detection with a standard SYBR Green PCR kit (Agilent Technologies, Santa Clara, CA, USA) and a Rotor-Gene RG-3000A (Corbett Research, Mortlake, Australia) according to the manufacturer’s guidelines. The miRNA expression was standardized using small nuclear RNA U6. The mRNA expression was standardized using GAPDH. Every sample was examined in triplicate. The mRNA or miRNA expression in tissues and cells were analyzed using the 2−ΔΔCt method. All primers in this study are listed in Table 1.
 |
Table 1 Sequences of Primers Used in qRT-PCR |
Dual-Luciferase Reporter Assay System
Luciferase reporter plasmids containing the mutant (MT) or wild-type (WT) VPS4B-3′-UTR reporter vectors were produced by Promega (Madison, WI, USA).Briefly, the wild-type VPS4B-3ʹ-UTR (WT) and mutant VPS4B-3ʹ-UTR (MT) containing the putative binding site of miR-488-3p were constructed and cloned in the psiCHECKTM-2 dual luciferase reporter vector (Promega, Madison, WI, USA) to construct psiCHECKTM-2-VPS4B-WT or psiCHECKTM-2-VPS4B-MT reporter vectors. Cortical neurons (ATCC, Manassas, VA, USA) were co-transfected with the MT or WT VPS4B 3′-UTR reporter vectors and miR-488-3p mimic or control (NC) utilizing Lipofectamine 2000 (Invitrogen) in line with the manufacturer’s procedure. Following incubation of 48 hours, the neurons were collected and lysed. We used the dual-luciferase detect system (Promega) to evaluate the activities of luciferase.
Analysis of Western Blot
Samples were split by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis. After that, they were transferred to polyvinylidene difluoride (PVDF) membrane (Invitrogen, Carlsbad, CA, USA). Then the membranes were incubated with PBST (phosphate-buffered saline with 0.1% Tween 20) containing 5% BSA and blocked for 1 hour. Then the membranes were incubated with primary antibodies, that is anti-GAPDH (ab8245, 1:1,000, Abcam) or anti-VPS4B (ab224736, 1:500, Abcam), diluted, at 4°C overnight, with PBST containing 5% BSA. After that, with Tris buffered saline containing Tween-20 and the membranes were washed three times. Then secondary antibodies containing horseradish peroxidase (HRP) (1:5,000, Abcam) were used to incubate the membranes. Finally, enhanced chemiluminescence (Pierce, Rockford, IL, USA) was applied to detect the proteins of interest.
Cell Viability Assay
MTT Cell Viability Assay Kit (Invitrogen; Carlsbad, CA, USA) was applied to measure the cell viability following the manufacturer’s protocol. Briefly, after being planted into 96-well plates, cells were cultured overnight. After transfection and treatment, the medium was refreshed, and we added MTT stock solution (10 μL) into each well at 37°C for 4 hours. After that, dissolution reagent (100 μL) was added to each well and treated at 37°C for 4 hours. An optical density measurement at 570 nm using a microplate absorbance reader (Bio-Rad, Sunnyvale, CA, USA) was utilized to analyze the absorbance.
Flow Cytometry Analysis of Apoptosis
At room temperature, the cells were washed with cold PBS and stained with Annexin V conjugated with PE (Thermo Fisher, Waltham, MA, 1:20 diluted) for 20 minutes. After incubation, flow cytometry analysis was conducted to analyze the stained cells. Briefly, cortical neurons were transfected with miR-488-3p mimic, miR-488-3p inhibitors, or sh-VPS4B utilizing Lipofectamine 2000 (Invitrogen) as stated by the procedures. Neurons were administered with OGD/R. After the treatment, the cells were treated with 0.05% trypsin (Sigma-Aldrich) at 37°C. Following that, these cells were centrifuged at a speed of 200g for 3 minutes and then washed three times with 0.01 M PBS, and the cells were resuspended with binding buffer at a density of 1×106 cells/mL. The neurons were treated with Annexin V-FITC and propidium iodide (PI) maintaining darkness for 20 minutes. Finally, a FACScan flow cytometer was purchased from Becton Dickinson (Mountain View, USA) and was adopted to analyze apoptotic cells. We assessed the figures by the Cell Quest (Becton Dickinson).
Statistical Analysis
We adopted the SPSS 22.0 for data analysis. To obtain the mean value and reduce errors, each experiment was conducted separately three times. All data were performed by means±SD. Student’s t-test was used to analyze the data between two groups, while one-way ANOVA was used to analyze the data among more than two groups. Post-hoc test was used by Tukey’s method. A value of P<0.05 was measured to be statistically significant.
Results
Down-Regulation of miR-488-3p Following OGD/R in vitro or I/R in vivo
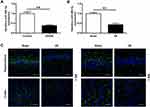
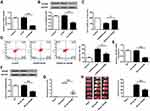
Stroke model was adopted to assess the impact of miR-488-3p on ischemic brain injury modulation. In the beginning, qRT-PCR was adopted to evaluate the miR-488-3p expression in mice brains with I/R and cortical neurons with OGD/R. The figures revealed that, in comparison to control, miR-488-3p in the neurons significantly reduced at 24 hours post-OGD/R (P<0.01) (Figure 1A). Consistently, the miR-488-3p expression apparently dropped in the I/R group compared with that in the sham group (P<0.01) (Figure 1B). FISH assay was performed to observe the alteration of miR-488-3p in the hippocampus and cortex after I/R. The relative fluorescence intensity of miR-488-3p was significantly declined in the I/R group both in the hippocampus and cortex, in comparison with Sham mice (Figure 1C). These results indicated that there was a correlation between the ischemic brain injury and the reduction of miR-488-3p.
Up-Regulation of miR-488-3p Protects Neurons Against the Injury Induced by OGD/R in vitro
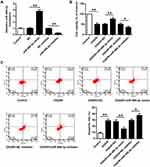
To explore the role of miR-488-3p in the survival of neuronal cells, we analyzed the neuronal cells treated with miR-488-3p mimic or its inhibitor. We observed that miR-488-3p expression was apparently raised in neurons transfected with miR-488-3p mimic and down-regulated by miR-488-3p inhibitor (P<0.01) (Figure 2A). After OGD/R for 24 hours, there was a growth in neuronal cell death rate (P<0.01) (Figure 2B). Additionally, the neuronal cell death was suppressed by the up-regulation of miR-488-3p (P<0.01) (Figure 2B). Conversely, it was promoted by the down-regulated miR-488-3p (P<0.05) (Figure 2B). Moreover, after OGD/R, the neuronal cell apoptotic rate was apparently raised (P<0.01) (Figure 2C). Up-regulated miR-488-3p inhibited the neuronal apoptotic rate (P<0.01) (Figure 2C). On the contrary, down-regulated miR-488-3p promoted the neuronal apoptotic rate (P<0.05) (Figure 2C). These data suggest that the increase of miR-488-3p protected neurons against injury induced by OGD/R in vitro.
Up-Regulated miR-488-3p Diminished Ischemic Brain Injury in vivo
The cerebral cortex was injected with miR-488-3p inhibitor and miR-488-3p mimic to explore the effect of miR-488-3p in ischemic brain injury. We adopted the qRT-PCR to verify the fluctuant of miR-488-3p. It was revealed that miR-488-3p expression was obviously raised in the miR-488-3p mimic group in MCAO/R mice. Conversely, it was remarkably dropped in the miR-488-3p inhibitor group (Figure 3A). Additionally, miR-488-3p overexpression diminished the neurological scores and infarct volume (P<0.01), however reduction of miR-488-3p, after MCAO/R for 24 hours, remarkably increased the ischemic injury (P<0.05) (Figure 3B and C).
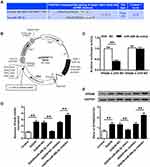
VPS4B Was a Critical Target Gene of miR-488-3p
The TargetScan 7.2 (http://www.targetscan.org/vert_72/) miRNA database was utilized to identify the target of miR-488-3p. It was proved that VPS4B was a vital target of miR-488-3p (Figure 4A). The vector map for the reporter clone was exhibited (Figure 4B). Additionally, a firefly luciferase reporter (including mutant 3′-UTR of VPS4B and wild 3′-UTR of VPS4B) was constructed. The results revealed that the luciferase activity in the miR-488-3p mimic+VPS4B 3′-UTR (WT) group was evidently inhibited (P<0.01); meanwhile, the activity of miR-488-3p mimic+VPS4B 3′-UTR (MT) was similar with the NC group (Figure 4C). Moreover, it was presented that miR-488-3p mimic and miR-488-3p inhibitor apparently reduced and elevated the mRNA level of VPS4B in neuronal cells, respectively (P<0.01). Consistently they had a similar effect on the protein level of VPS4B (P<0.01) (Figure 4D and E). These data indicated that VPS4B was a target of miR-488-3p and the VPS4B expression was reduced induced by binding to the 3ʹ-UTR.
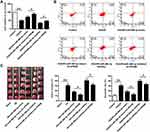
Reduction of VPS4B Level Inhibited Ischemic Brain Injury in vivo and Neuronal Cell Death in vitro
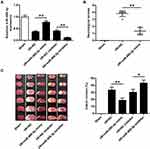
To evaluate the impact of VPS4B on ischemic brain injury and neuronal cell death, the knockdown of VPS4B was utilized. mRNA and protein expression of VPS4B in neuronal cells were both remarkably down-regulated in the VPS4B silencing group. (Figure 5A and B) (P<0.01). Furthermore, when VPS4B was inhibited, cell viability was increased, and apoptosis was reduced (P<0.05) (Figure 5C and D). It was presented that sh-VPS4B injected into the cerebral cortex evidently reduced the mRNA expression and protein level of VPS4B in ischemic brains, in comparison with the I/R+sh-NC group (P<0.01) (Figure 5E and F). Then the neurobehavioral outcomes and infarct volume of ischemic brain were detected. The data showed that sh-VPS4B noticeably declined the neurological scores as well as the infarction volume compared with the sh-NC following I/R treatment (P<0.01) (Figure 5G and H). These data demonstrated that sh-VPS4B suppressed ischemic brain injury in vivo and neuronal cell death in vitro.
Up-Regulated miR-488-3p Attenuated Ischemic Brain Injury by Targeting VPS4B
To verify whether miR-488-3p suppressed ischemic brain injury via adjusting VPS4B expression, several experiments were then performed. As shown in Figure 6A and B, cell viability and apoptosis were detected in groups of miR-488-3p mimic and sh-VPS4B or miR-488-3p inhibitor and sh-VPS4B. The data revealed that neuronal cell death and apoptosis were reversed by miR-488-3p, and the protective effect of miR-488-3p was strengthened by the sh-VPS4B addition (P<0.05) (Figure 6A and B). Instead, neuronal cell death and apoptosis was obviously elevated by inhibiting miR-488-3p; these impacts, however, were reversed by sh-VPS4B (P<0.05) (Figure 6A and B). Additionally, we injected the sh-VPS4B lentivirus into the cerebral cortex of mice. The results presented that miR-488-3p remarkably declined the cerebral infarction volume, meanwhile the infarction volume was further declined by VPS4B reduction (P<0.05) (Figure 6C). The MiR-488-3p inhibitor significantly aggravated cerebral infarction, which was reversed by sh-VPS4B (P<0.05) (Figure 6C). HE staining showed that I/R induced-cell death was remarkably reduced by miR-488-3p overexpression and was further reduced by VPS4B inhibition (P<0.05) (Figure 7). Besides, miR-488-3p inhibition considerably exacerbated cell death, and this effect could be antagonized by sh-VPS4B (P<0.05) (Figure 7). These data represented that the miR-488-3p inhibited the ischemic brain injury, partly via suppressing VPS4B expression.
Discussion
A number of stroke institutions around the world regularly review useful literature and update guidelines for the management of ischemic stroke. Over the years, the definition of stroke has been redefined, and there have been major advances in the treatment and prevention of stroke. A growing body of evidence indicates that the expression of miRNAs in ischemic stroke is significantly abnormal and meaningfully affects the progression and prognosis of the disease.6,8,10 Our results showed that the expression levels of miR-488-3p were notably reduced in OGD/R-treated neurons and I/R-treated mice brain tissues. Previous studies have shown that miR-488 affects the pathophysiological processes of various diseases.35–37 In this study, it was found that over-expression of miR-488-3p inhibited neuron apoptosis and alleviated cerebral infarction caused by ischemic stroke in mice, indicating that miR-488-3p might be a potential curative target for ischemic stroke. Previous studies have shown that VPS4 affects extracellular secretion and lysosomal and cell proliferation and differentiation through regulating specific signaling pathways.32,33 Some scholars found that the level of VPS4B in the brain tissues was significantly increased after MCAO and the activity of caspase-3 protein was up-regulated to contribute for neuron apoptosis.34 Our study further demonstrated that VPS4B was a direct target of miR-488-3p and verified that miR-488-3p could inhibit the expression of VPS4B in OGD/R-treated neurons. We proved the neuroprotective effects of miR-488-3p in ischemic stroke. And in vivo experiments confirmed that silencing VPS4B could further enhance the neuroprotective effects of miR-488-3p.
MiRNAs typically mediate the pathophysiological processes of many diseases, such as neurodegenerative diseases and ischemic stroke, by targeting specific genes.6,7 Studies have shown that miR-488 is abnormally expressed in several diseases. Some researchers have provided data showing that miR-488 expression is negatively regulated by inhibiting ROS and AKT/NF κB pathways in LPS-induced inflammation of intimal epithelial cells.35 In osteosarcoma (OS) cells, miR-488 is down-regulated by targeting aquaporin 3 (AQP3) to inhibit cell proliferation and invasion,37 indicating that miR-488 is a miRNA of tumor suppressor. Besides, miR-488 participates in drug tolerance of anti-cancer drug cisplatin mediated by eukaryotic translation initiation factor 3a (eIF3a) in non-small cell lung cancer cells (NSCLC).36 At present, the expression and mechanism of miR-488-3p in ischemic stroke is still uncertain. Our findings suggest that inhibition of miR-488-3p can aggravate cell apoptosis and the decreased cell viability induced by OGD/R-treatment, as well as the abnormalities cerebral infarction induced by cerebral I/R operation. However, supplying miR-488-3p alone can partially alleviate neuron apoptosis and the declined cell viability following OGD/R-treatment, as well as the effects following cerebral I/R treatment. Inhibiting the expression of VPS4B can further alleviate the above effects, indicating that miR-488-3p plays its role via VPS4B partially.
To validate the accurate role of miR-488-3p in ischemic brain injury, the target genes of miR-488-3p were predicted and identified. Gene analysis of the dual luciferase reporter indicated that VPS4B was a target of miR-488-3p, which negatively regulated VPS4B expression through binding to 3ʹ-UTR site. VPS4B is a member of the VPS family (which still includes VPS4A and VPS4B) and is responsible for coding the vacuolar sorting proteins.30,38 Studies have shown that VPS4B regulates the proliferation and odontoblastic differentiation of human dental pulp stem cells through the Wnt-β-catenin pathway.32 VPS4 impacts the lysosomal or extracellular secretion and impacts Parkinson’s disease through targeting alpha-synuclein (alpha SYN).33 Previous studies found that Middle Cerebral Artery Occlusion (MCAO) caused significant up-regulation of VPS4B in the hippocampus, but the mechanism was rarely studied.6 This study further verified that VPS4B was significantly up-regulated in OGD/R-treated neurons, consistent with previous studies.34 Inhibition of VPS4B can partially relieve cell apoptosis and the decreased cell viability of OGD/R-treated neurons, and partially relieve the increased neurological score and cerebral infarction caused by cerebral I/R, which can be changed by up-regulation or down-regulation of miR-488-3p, suggesting that the role of VPS4B in ischemic stroke is modulated by its upstream molecule miR-488-3p.
Over the years, the management of IS has undergone numerous changes, with patients receiving more effective treatments to diminish long-term disability. Appropriate use of existing therapies is critical to optimize the prognosis of stroke in patients. In this study, the expression of miR-488-3p decreased in cerebral I/R mice and OGD/R-treated neurons. Over-expression of miR-488-3p significantly improved neuronal viability, inhibited cell apoptosis, improved the neurological scores, and decreased the cerebral infarction, which was achieved by the combined effect of miR-488-3p and VPS4B. In addition, we verified that VPS4B was the target gene of miR-488-3p.
Conclusion
In conclusion, miR-488-3p regulates cell viability, apoptosis, neurological function, and cerebral infarction through targeting VPS4B, which would affect the progression and prognosis of ischemic stroke, suggesting that miR-488-3p might be a potential therapy for ischemic stroke.
Ethical Approval
The experiment methods have been approved by the Animal Care Committee of The First Affiliated Hospital of Guangdong Pharmaceutical University. All the procedures involved in the study were conducted in accordance with the guidelines published by China Animal Protection Association.
Funding
This work is supported by Guangdong Provincial Natural Science Foundation (NO. 2018A0303130323).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Hankey GJ. Stroke. Lancet. 2017;389(10069):641–654. doi:10.1016/S0140-6736(16)30962-X
2. Venkat P, Shen Y, Chopp M, Chen J. Cell-based and pharmacological neurorestorative therapies for ischemic stroke. Neuropharmacology. 2018;134(PtB):310–322. doi:10.1016/j.neuropharm.2017.08.036
3. Barakat W, Safwet N, El-Maraghy NN, Zakaria MNM. Candesartan and glycyrrhizin ameliorate ischemic brain damage through downregulation of the TLR signaling cascade. Eur J Pharmacol. 2014;724:43–50. doi:10.1016/j.ejphar.2013.12.032
4. Phipps MS, Cronin CA. Management of acute ischemic stroke. BMJ. 2020;368:l6983. doi:10.1136/bmj.l6983
5. Fonarow GC, Zhao X, Smith EE, et al. Door-to-needle times for tissue plasminogen activator administration and clinical outcomes in acute ischemic stroke before and after a quality improvement initiative. JAMA. 2014;311(16):1632–1640. doi:10.1001/jama.2014.3203
6. Eyileten C, Wicik Z, De Rosa S, et al. MicroRNAs as Diagnostic and Prognostic Biomarkers in Ischemic Stroke—A Comprehensive Review and Bioinformatic Analysis. Cells. 2018;7(12):12. doi:10.3390/cells7120249
7. Juzwik CA, S. Drake SD, Zhang Y, et al. microRNA dysregulation in neurodegenerative diseases: A systematic review. Prog Neurobiol. 2019;182:101664. doi:10.1016/j.pneurobio.2019.101664
8. Li G, Morris-Blanco KC, Lopez MS, et al. Impact of microRNAs on ischemic stroke: from pre- to post-disease. Prog Neurobiol. 2018;163-164:59–78. doi:10.1016/j.pneurobio.2017.08.002
9. Chen Z, Lai T-C, Jan Y-H, et al. Hypoxia-responsive miRNAs target argonaute 1 to promote angiogenesis. J Clin Invest. 2013;123(3):1057–1067. doi:10.1172/JCI65344
10. Xu W, Gao L, Zheng J, et al. The Roles of MicroRNAs in Stroke: possible Therapeutic Targets. Cell Transplantation. 2018;27(12):1778–1788. doi:10.1177/0963689718773361
11. Kaur H, Sarmah D, Saraf J, et al. Noncoding RNAs in ischemic stroke: time to translate. Ann N Y Acad Sci. 2018;1421(1):19–36. doi:10.1111/nyas.13612
12. Buller B, Liu X, Wang X, et al. MicroRNA-21 protects neurons from ischemic death. FEBS J. 2010;277(20):4299–4307. doi:10.1111/j.1742-4658.2010.07818.x
13. Tao Z, Zhao H, Wang R, et al. Neuroprotective effect of microRNA-99a against focal cerebral ischemia–reperfusion injury in mice. J Neurol Sci. 2015;355(1–2):113–119. doi:10.1016/j.jns.2015.05.036
14. Yuan B, Shen H, Lin L, Su T, Zhong L, Yang Z. MicroRNA367 negatively regulates the inflammatory response of microglia by targeting IRAK4 in intracerebral hemorrhage. J Neuroinflammation. 2015;12:206.
15. Zhang Y, Han B, He Y, et al. MicroRNA-132 attenuates neurobehavioral and neuropathological changes associated with intracerebral hemorrhage in mice. Neurochem Int. 2017;107:182–190. doi:10.1016/j.neuint.2016.11.011
16. Vijayan M, Kumar S, Yin X, et al. Identification of novel circulatory microRNA signatures linked to patients with ischemic stroke. Hum Mol Genet. 2018;27(13):2318–2329. doi:10.1093/hmg/ddy136
17. Vijayan M, Alamri FF, Al Shoyaib A, Karamyan VT, Reddy PH. Novel miRNA PC-5P-12969 in Ischemic Stroke. Mol Neurobiol. 2019;56(10):6976–6985. doi:10.1007/s12035-019-1562-x
18. Vijayan M, Reddy PH. Peripheral biomarkers of stroke: focus on circulatory microRNAs. Biochim Biophys Acta. 2016;1862(10):1984–1993. doi:10.1016/j.bbadis.2016.08.003
19. Yang Y, Li H, He Z, Xie D, Ni J, Lin X. MicroRNA-488-3p inhibits proliferation and induces apoptosis by targeting ZBTB2 in esophageal squamous cell carcinoma. J Cell Biochem. 2019;120(11):18702–18713.
20. Xue W, Chen J, Liu X, et al. PVT1 regulates the malignant behaviors of human glioma cells by targeting miR-190a-5p and miR-488-3p. Biochim Biophys Acta Mol Basis Dis. 2018;1864(5):1783–1794. doi:10.1016/j.bbadis.2018.02.022
21. Wu XZ, Cui HP, Lv HJ, Feng L. Knockdown of lncRNA PVT1 inhibits retinoblastoma progression by sponging miR-488-3p. Biomed Pharmacother. 2019;112:108627.
22. Wen Z, Shen Q, Zhang H, et al. Circular RNA CCDC66 targets DCX to regulate cell proliferation and migration by sponging miR-488-3p in Hirschsprung’s disease. J Cell Physiol. 2019;234(7):10576–10587. doi:10.1002/jcp.27733
23. Li Y, Li S, Luo Y, Liu Y, Yu YN. LncRNA PVT1 Regulates Chondrocyte Apoptosis in Osteoarthritis by Acting as a Sponge for miR-488-3p. DNA Cell Biol. 2017;36(7):571–580. doi:10.1089/dna.2017.3678
24. Arvidsson Y, Rehammar A, Bergstrom A, et al. miRNA profiling of small intestinal neuroendocrine tumors defines novel molecular subtypes and identifies miR-375 as a biomarker of patient survival. Mod Pathol. 2018;31(8):1302–1317.
25. Dai D, Wang J, Jiang Y, et al. Small RNA sequencing reveals microRNAs related to neuropathic pain in rats. Braz J Med Biol Res. 2019;52(10):e8380. doi:10.1590/1414-431x20198380
26. Ko J, Hemphill M, Yang Z, et al. Multi-dimensional mapping of brain-derived extracellular vesicle microrna biomarker for traumatic brain injury diagnostics. J Neurotrauma. 2019.
27. Elia N, Sougrat R, Spurlin TA, Hurley JH, Lippincott-Schwartz J. Dynamics of endosomal sorting complex required for transport (ESCRT) machinery during cytokinesis and its role in abscission. Proc Natl Acad Sci U S A. 2011;108(12):4846–4851. doi:10.1073/pnas.1102714108
28. Davies BA, Azmi IF, Katzmann DJ. Regulation of Vps4 ATPase activity by ESCRT-III. Biochem Soc Trans. 2009;37(1):143–145. doi:10.1042/BST0370143
29. Scheuring S, Rohricht RA, Schoning-Burkhardt B, et al. Mammalian cells express two VPS4 proteins both of which are involved in intracellular protein trafficking. J Mol Biol. 2001;312(3):469–480. doi:10.1006/jmbi.2001.4917
30. Beyer A, Scheuring S, Muller S, Mincheva A, Lichter P, Kohrer K. Comparative sequence and expression analyses of four mammalian VPS4 genes. Gene. 2003;305(1):47–59. doi:10.1016/S0378-1119(02)01205-2
31. Bishop N, Woodman P. ATPase-defective mammalian VPS4 localizes to aberrant endosomes and impairs cholesterol trafficking. Mol Biol Cell. 2000;11(1):227–239. doi:10.1091/mbc.11.1.227
32. Pan Y, Lu T, Peng L, et al. Vacuolar protein sorting 4B regulates the proliferation and odontoblastic differentiation of human dental pulp stem cells through the Wnt-beta-catenin signalling pathway. Artif Cells Nanomed Biotechnol. 2019;47(1):2575–2584. doi:10.1080/21691401.2019.1629950
33. Hasegawa T, Konno M, Baba T, et al. The AAA-ATPase VPS4 regulates extracellular secretion and lysosomal targeting of alpha-synuclein. PLoS One. 2011;6(12):e29460. doi:10.1371/journal.pone.0029460
34. Cui G, Wang Y, Yu S, et al. The expression changes of vacuolar protein sorting 4B (VPS4B) following middle cerebral artery occlusion (MCAO) in adult rats brain hippocampus. Cell Mol Neurobiol. 2014;34(1):83–94. doi:10.1007/s10571-013-9989-5
35. Liu J, Guo S, Jiang K, et al. miR-488 mediates negative regulation of the AKT/NF-κB pathway by targeting Rac1 in LPS-induced inflammation. J Cell Physiol. 2020;235(5):4766–4777. doi:10.1002/jcp.29354
36. Fang C, Chen Y-X, Wu N-Y, et al. MiR-488 inhibits proliferation and cisplatin sensibility in non-small-cell lung cancer (NSCLC) cells by activating the eIF3a-mediated NER signaling pathway. Sci Rep. 2017;7(1):40384. doi:10.1038/srep40384
37. Qiu J, Zhang Y, Chen H, Guo Z. MicroRNA-488 inhibits proliferation, invasion and EMT in osteosarcoma cell lines by targeting aquaporin 3. Int J Oncol. 2018;53(4):1493–1504.
38. Fujita H, et al. A dominant negative form of the AAA ATPase SKD1/VPS4 impairs membrane trafficking out of endosomal/lysosomal compartments: class E vps phenotype in mammalian cells. J Cell Sci. 2003;116(2):401–414. doi:10.1242/jcs.00213
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.