Back to Journals » OncoTargets and Therapy » Volume 13
MicroRNA-423 Drug Resistance and Proliferation of Breast Cancer Cells by Targeting ZFP36
Authors Xia W, Liu Y, Du Y, Cheng T, Hu X, Li X
Received 30 May 2019
Accepted for publication 23 December 2019
Published 24 January 2020 Volume 2020:13 Pages 769—782
DOI https://doi.org/10.2147/OTT.S217745
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
This paper has been retracted.
Wenfei Xia,1,* Yun Liu,2,* Yaying Du,1 Teng Cheng,1 Xiaopeng Hu,1 Xingrui Li1
1Department of Breast and Thyroid Surgery, Division of General Surgery, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430030, People’s Republic of China; 2Department of ENT, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430030, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xingrui Li
Department of Breast and Thyroid Surgery, Division of General Surgery, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, No. 095 Jiefang Avenue, Wuhan, Hubei 430030, People’s Republic of China
Email [email protected]
Background/Aims: The effects of microRNA-423 on proliferation and drug resistance of breast cancer cells were explored, the downstream target genes of miR-423 and the targeted regulatory relationship between them were studied.
Methods: RT-qPCR was used to detect the expression of miR-423 in breast cancer tissues and cell lines, and the transfection efficiency of miR-423 inhibitory vector miR-423-inhibitor was constructed and verified. CCK-8 and colony formation assays were used to examine the effect of miR-423 on tumor cell proliferation. Target gene prediction and screening and luciferase reporter assay were used to verify downstream target genes of miR-432. The mRNA and protein expression of miR-423target gene ZFP36 was detected by RT-qPCR and Western blotting.
Results: The expression of miR-423 was significantly higher than that in normal tissues. Compared to the non-malignant mammary epithelial cell line MCF-10A, the expression of miR-423 was significantly raised in MCR-7 and MCF-7/ADR cells. ZFP36 was a downstream target gene of miR-423 and negatively correlated with the expression of miR-423 in breast cancer. The knockdown of miR-423 can significantly enhance the cytotoxicity of the drug, increase the apoptotic rate of MCF-7/ADR cells. miR-423 was capable of activating the Wnt/β-catenin signaling pathway leading to chemoresistance and proliferation, whereas overexpression of ZFP36 reduced drug resistance and proliferation.
Conclusion: miR-423 acted as an oncogene to promote tumor cell proliferation and migration. ZFP36 was a downstream target gene of miR-423, and miR-423 inhibited the expression of ZFP36 via Wnt/β-catenin signaling pathway of breast cancer cells.
Keywords: micro RNAs, miR-423, ZFP36, Wnt/β-catenin, breast cancer
Introduction
Breast cancer (BC) is one of the most common cancers in women, accounting for 22.9% of all cancers in women.1 At present, surgery, radiotherapy, chemotherapy, endocrine therapy and other means have made great progress, and breast cancer patients can obtain higher survival rate by receiving the above treatment.2–4 However, once breast cancer metastasizes or recurs after surgery, it is rarely cured completely, so the prognosis is poor. Although many researchers have initially confirmed that part of the signaling pathway is involved in the development of breast cancer, a deeper understanding of its underlying pathogenesis remains challenging.5,6 Therefore, it is extremely urgent to study and explore the molecular biological mechanism of breast cancer development and deterioration. In recent years, there is increasing evidence that mi RNAs, as proto-oncogenes or tumor-suppressor genes, participate in tumor cell proliferation, differentiation, apoptosis, mutation, DNA methylation, and angiogenesis by negatively regulating target gene expression. The imbalance of these processes is a prerequisite for the occurrence of various diseases, especially tumors.7,8 New research showed that mi RNA is closely related to the development of tumors and plays an important role in the malignant characteristics of tumors.9,10 At present, various miRNAs such as miR-25, miR-98, miR-34a, miR-194, mi R-495 and miR-365 have been confirmed to be involved in proliferation, differentiation, apoptosis and drug resistance of breast cancer cells.11–13 Therefore, based on the regulation of miRNAs, further research on breast cancer-related miRNA and its mechanism of action can provide new ideas and directions for targeted therapy.
miR-423 is a member of the miRNAs family, which is found to play a role in hepatocellular carcinoma, neuroblastoma, tumor gene regulation in ovarian cancer, malignant melanoma, gastric cancer and colorectal cancer.14,15 Studies have found that miR-423 is overexpressed in breast cancer, and the expression level of miR-423 is significantly increased in metastasis.16 However, studies on the expression characteristics of miR-423 downstream target genes in breast cancer and how miR-423 and target genes regulate tumorigenesis, development and specific mechanisms have not been reported. With the approval of our ethics committee, the relationship between miR-423 and breast cancer tissue grading and tumor TNM staging was investigated by analyzing the differential expression of miR-423 in breast cancer and its adjacent tissues in this study. On the basis of this, the effects of micro RNA-423 on breast cancer cell proliferation and drug resistance were investigated, the target genes were searched and verified, and the targeting relationship between mR-423 and ZFP36 was explored, which will provide new strategies and directions for the treatment of breast cancer.
Materials and Methods
Tissue Specimens and Cells
This study is based on relevant international norms and ethical standards. From January 2016 to March 2018, 40 pairs of BC tissues and adjacent non-cancer tissues were collected from patients who underwent surgical resection in Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology. Clinical data were obtained from patients diagnosed by two independent and experienced pathologists. All patients were signed written informed consent. The investigation project and informed consent had been approved by the Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology Ethics Committee. According to the International Union Against Cancer (UICC), pathological results confirmed that the extracted samples were BC tissues.
BC cells MCF-7 and MCF-7/ADR were obtained from Nan Jing key GEN Company. MCF-7 cells were cultured in 90% DMEM containing 1% penicillin-streptomycin, 10% fetal bovine serum (Gibco, Grand Island, NY, USA). MCF-7/ADR cell lines were subcultured in RPMI 1640 medium (Gibco) containing 1 mg/L adriamycin (ADR) to maintain drug resistance. Cells were cultured in a 37°C, 5% CO2 cell culture incubator. Doxorubicin was removed from the culture medium 48 hrs before any experiment.
CCK-8 Method
Cells (1 × 104) were inoculated on 96-well plates, each hole containing 100-μL medium, and treated with adriamycin (ADR) and vincristine (VCR), respectively. It was incubated with Taxel for 48 hrs (Sigma, St. Louis, MO, USA). The cells were treated with CCK-8 (Cell Counting Kit-8) 11 μL. After 24 hrs, the plate was read at 450 nm using a microplate reader (Bio-Rad Laboratories Inc, Hercules, CA, USA). Drug resistance was determined by comparing IC50 values of growth curve (50% of drug concentration for inducing cell proliferation).
Colony Formation
Cells (1 × 103 cells/well) were inoculated on 96-well plates (GraphPad Sofware, Inc., San Diego, CA, USA) allowed to adhere for 24 hrs, and then exposed to ADR for 24 hrs. Each well was washed and the culture was updated. Then, the cells were cultured in an incubator for 8 days. Finally, colonies were counted after fixation for 10 mins with 10% formaldehyde. Cell viability was measured by GraphPad Prism 6 (Costar, Charlotte, NC, USA).
Oligonucleotides, Plasmids, siRNA and Transfection
miR-423 simulation, miR-423 inhibitor (miR-423 inhibitor), negative control oligonucleotide (miR-NC), negative control oligonucleotide (NC), control vector (Vector), ZFP36 pEGFP-N2 vector (ZFP36), small interfering RNA of ZFP36 (siZFP36), scramble siRNA of ZFP36 (si-NC) were obtained from RiboBio (Shanghai, China). Overexpressed miRNA-423 was using the miRNA-423 mimetic, ZFP36 was overexpressed by transfection of ZFP36. miRNA-423 and ZFP36 were knocked out using miRNA-423 inhibitor and siZFP36, respectively. According to the specific requirements of the instructions, Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA, USA) was used for blood transfusion. After 48 hrs of transfection, the cells were prepared for further analysis. Finally, the transfection efficiency was detected by qRT-PCR.
Luciferase Assay
PMIRGLO Dual Luciferase Small RNA Targeted Expression Vector for Luciferase Analysis in 3ʹ-untranslated Region (UTR) (Shanghai, China). Both hsa-miR-423 mimic and negative control oligonucleotides were obtained from RiboBio Co. Ltd. (GenePharma, Shanghai, China). Cells were inoculated into 24-well plates with 5 × 104 cells per well. The hsa-mir-423 simulator and wild or mutant target sequences were co-transfected into each well using Lipofectamine 2000 (Invitrogen). After 48 hrs of transfection, the activities of freon and glomerular luciferase were determined by double luciferase reporting system, normalized into the activity for Renilla luciferase. The average result of mir-control transfection was set to 1.0.
Real-Time PCR
Total RNA containing miRNA was collected from the cells by the RNeasy Mini Kit (Shanghai, China). According to the specifications of the instructions, we used Quantitec reverse transcription kit (Shanghai, China) to synthesize the cDNA. The primer sequence is described as follows: miR-NC, sense: 5ʹ-UCCUUCCGUGGAAUCACGUTT-3ʹ; hsa-miR-423 mimics, 5ʹ-CAGUGCAAUGAUGAAAGGGCAu-3ʹ; miR-423 inhibitor, 5ʹ-CCCAUGCUU CACUGCCAAUUGU-3ʹ; NC, 5ʹ-GUACUUUCACGAAGUGGGAA-3ʹ; siRNA-ZFP36, 5ʹ-GGCGAAACAAUAGCCCUACTT-3ʹ; and si-NC: 5ʹ-ACTCTATGCAACTGCACAC-3ʹ. SYBR Green qRT-PCR master mix and GAPDH were used as internal controls. According to the instructions (Ambion Inc., Austin, TX), mirVana qRT-PCR microRNA detection kit was used to measure the expression of mir-423 and normalized by 2−ΔΔCT method relative to U6 small nuclear RNA.
Western Blot
Cells were decomposed with protein extract for 30 mins and centrifuged for 5 mins at 4°C for 12,000 r/min. Equivalent supernatant (protein) was mixed with SDS Loading Buffer and heated in a boiling water bath at 100°C, for 5 mins to fully denaturate the protein. 1 μL was taken for protein quantification. Cells were treated with protein lysate and protein samples were collected. The protein samples were quantified by BCA protein quantitative kit. SDS-PAGE electrophoresis was carried out with 50μg protein samples added to each well. After the electrophoresis, the protein was transferred to PVDF membrane, and the transfer membrane was sealed with 1% BSA. After that, 1:1000 rabbit anti-GAPDH was added (1:2000, Cell Signaling Technology, Beverly, MA, USA), and ZFP36, β-catenin, p-β-catenin (Ser675), and polyclonal antibody were added and placed overnight in the refrigerator at 4°C. After washing with TBST 3 times, 1:5000 labeled anti-rabbit secondary antibody was added, incubated for 1 hr at room temperature, washed 3 times with TBS. After that, the gray values of the target bands and the internal reference bands were recorded by ECL chemiluminescence.
Immunohistochemistry
Embedding breast cancer tissue specimens: tissue was immersed in neutral formic acid for fixation. After dehydration, the absolute ethanol was used to be transparent once; the xylene was used to be transparent twice. The tissue was immersed in the melted paraffin, embedded in the embedding machine, and the excess paraffin on the outside of the wax block was trimmed. The specimens were sectioned continuously on a paraffin slicer. The paraffin slices were dewaxed in an oven, hydrated with gradient alcohol and repaired with 0.1% citric acid buffer, sealed and removed with 0.5% sheep serum. Each paraffin slice was added to the corresponding diluted anti-coated tissues. After overnight, the first anti-body was washed once, and the second antibody was added. After incubation, the second antibody was removed, washed and observed under a microscope. After re-dyeing with hematoxylin, differentiation with hydrochloric acid alcohol solution, dehydration and drying, xylene transparent, the neutral gum was added to seal.
Antitumor Activity Assay in vivo
Animal experiments were performed in accordance with approved laboratory animal care and international guidelines. All animal procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals of “Tianjin University of Science and Technology” University and approved by the Animal Ethics Committee of “Animal Ethical and Welfare Committee (AEWC).” Four-week-old male nude mice were purchased from the animal facility of Dalian Medical University. In order to establish BC xenografts, MCF-7 cells with a density of about 2 × 106 were inoculated into the right abdomen of each nude mouse. About a week after inoculation, mice were randomly divided into two groups, control group and treatment group (n=6/group). Then, miR-423 mimic or miR-423 inhibitor was injected into the tumors 3 times a week for 4 weeks. In order to detect the chemosensitivity of breast tumors, 7 mg/kg adriamycin or saline was injected intraperitoneally every week. The tumors were separated after execution in mice, weighed and photographed. Tumors were weighed in 4% paraformaldehyde and the growth curve was plotted. The experiment was approved by the Animal Experimental Ethics Committee of the Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology of China.
Statistical Methods
The monitoring data were analyzed by SPSS19.0 statistical software. The data analysis results were expressed as mean ± standard deviation (mean ± SD). The data analysis between the two groups was performed by t-test. One-way variance analysis (ANOVA) was used for data analysis among multiple groups, and LSD test was used for subsequent analysis. P < 0.05 indicated that the difference was statistically significant.
Results
miR-423 Was Upregulated in Breast Cancer Tissues and Cell Lines
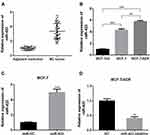
In order to investigate the functional role of miR-423 in breast cancer, our study included 40 patients with BC. As shown in Figure 1A, miR-423 expression was significantly upregulated in BC (P<0.05), compared with normal breast tissue. And miR-423 was significantly associated with tumor size, T stage, and cancer chemosensitivity, but not with lymph node metastasis (Table 1).
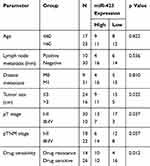 |
Table 1 The Correlation Between miR-423 Expression and Its Clinicopathological Correlation in Breast Cancer Patients |
The results in Figure 1B showed that miR-423 expression was significantly raised in MCR-7 and MCF-7/ADR cells compared with the non-malignant mammary epithelial cell line MCF-10A cells (p <0.05). In addition, compared with the MCF-7 cell line, miR-423 was significantly raised in the MCF-7/ADR cell line. The results of Figure 1C and D showed that miR-423 overexpression in MCF-7 cells and knockout of miR-423 in MCF-7/ADR were achieved by transfection with miR-423 mimic or miR-423 inhibitor.
ZFP36 Was a Direct Target Gene of miR-423
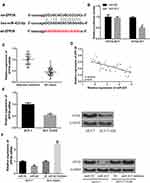
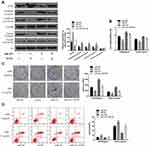
In order to determine the underlying mechanism of action of miR-423 in BC, the bioinformatics were predicted and ZFP36 was identified as a potential target for miR-423 (Figure 2A). Subsequently, the miRNA-423 binding region was mutated in the ZFP36 3ʹ-UTR to verify the interaction between miR-423 and ZFP36 3ʹ-UTR. The wild-type ZFP36 luciferase vector (ZFP36-WT) and the mutant ZFP36 luciferase vector (ZFP36-MUT) were co-transfected into HEK293T cells with miR-423 mime. As shown in the results of Figure 2B, miR-423 mimics significantly reduced relative luciferase activity in HEK293T cells transfected with ZFP36 (WT) vector, while mutations in the 3ʹ-utr of ZFP36 matched fluorescein Enzyme activity had no significant effect, suggesting that the interaction between miR-423 and the binding site of ZFP36 3ʹ-UTR can directly regulate the expression of the luciferase reporter gene.
In Figure 2C, compared with BC tissues, the expression level of ZFP36 in BC adjacent non-tumor tissues was significantly higher. Furthermore, as shown in Figure 2D, there was a negative correlation between the expression of miR-423 and ZFP36. Figure 2E shows that ZFP36 in MCF-7/ADR cells was significantly reduced in mRNA and protein expression levels compared with MCF-7 cells. As shown in Figure 2E and F, the upregulation of miR-423 expression resulted in a significant decrease in ZFP36 expression. In contrast, inhibition of expression of miR-423 resulted in a significant upregulation of ZFP36 expression. These results indicated that ZFP36 was a direct target of miR-423.
The Effect of Overexpression of miR-423 on Chemotherapeutic Resistance and Proliferation
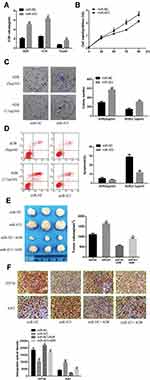
The IC50 values of chemotherapeutic agents (ADR, VCR, Taxel) were calculated by the CCK-8 method. As shown in Figure 3A, the IC50 values of these chemotherapeutic agents were significantly increased in miR-423 mimetic-transfected MCF-7 cells compared with miR-NC cells.
Figure 3B shows that transfection of miR-423 mimics significantly increased MCF-7 cell viability. Colony formation assay results showed that colonies in miR-423 overexpressing cells were significantly more than in miR-NC cells (P < 0.05).As shown in Figure 3C, compared with control cells, the rate of colony formation induced by ADR increased significantly by overexpression of miR-423 (P<0.05).
As shown in Figure 3D, compared with control cells, the apoptotic rate in miR-423 overexpression was significantly lower (P<0.05). These results indicated that miR-423 can promote the proliferation of MCF-7 cells.
In vivo animal experiments were conducted to determine whether miR-423 could inhibit ADR-induced apoptosis of MCF-7 cells. Compared with the miR-423 simulation group, the tumor volume combined with ADR was significantly smaller (P<0.05; Figure 3E). In addition, immunohistochemical staining showed that the expression level of ZFP36 was lower in the miR-423 overexpression group and higher in Ki67 than the control group (Figure 3F). In conclusion, studies demonstrated that overexpression of miR-423 promoted ADR resistance in MCF-7 cells and overexpression of miR-423 promoted proliferation in MCF-7 cells.
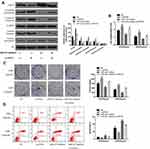
Downregulation of miR-423 Induced Chemosensitivity and Proliferation of MCF-7/ADR Cells
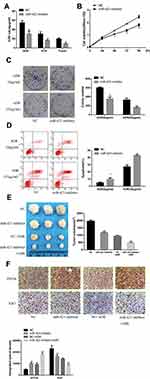
The IC50 values of chemotherapeutic agents (ADR, VCR, Taxel) were calculated by the CCK-8 method. The results showed that the IC50 value of the chemotherapeutic agent in the miR-423 inhibitor transfected cells was significantly reduced (Figure 4A). The CCK-8 method found that the growth rate of the miR-423 inhibitor group gradually decreased with the extension of the action time (Figure 4B). The results of Figure 4C and D demonstrated that miR-423 inhibitors were capable of inhibiting cell proliferation and increasing apoptosis rates in MCF-7/ADR cells (Figure 4C and D).
As shown in Figure 4C, compared with the control cells, the downregulated miR-423 caused gradually decreased in the colony formation rate. As shown in Figure 4D, flow cytometry assay showed that the apoptotic rate in MCF-7/ADR cells transfected with miR-423 inhibitor was significantly higher than that of negative control cells.
As shown in Figure 4E, compared with the control group, the average volume of MCF-7/ADR tumors transfected with miR-423 inhibitor was significantly smaller. In addition, by immunohistochemical staining, compared with the control group, the expression level of ZFP36 in the MCF-7/ADR tumor cells was significantly increased. And the expression level of Ki67 was significantly decreased (Figure 4F). These results indicated that downregulation of miR-423 restrained ADR resistance and proliferation in MCF-7/ADR cells.
Overexpression of miR-423 Stimulated Drug Resistance and Inhibited Proliferation via the ZFP36- Wnt/β-Catenin Signaling Pathway
As shown in Figure 5A, compared with the control group, ZFP36 can decrease the expressions of nuclear β-catenin, cytoplasmic β-catenin, cytoplasmic p-β-catenin and raised expression of ZFP36 in MCF-7 cells. miR-423 can raise the expressions of nuclear β-catenin, cytoplasmic β-catenin and cytoplasmic p-β-catenin and decreased the expression of ZFP36 in MCF-7 cells. And the miR-423+ ZFP36 can reverse the effect of miR-423 on the expressions of nuclear β-catenin, cytoplasmic β-catenin, cytoplasmic p-β-catenin and ZFP36. These results indicated that miR-423 can activate Wnt/β-catenin signaling by inhibiting ZFP36.
In MCF-7 cells, cells were treated with a concentration gradient of ADR (2.1 μg/mL). The CCK8 method results (Figure 5B) showed that increased expression of Zfp36 can increase the resistance induced by miR-423.When cells were treated with ADR, similar results were found in colony formation analysis and flow cytometry analysis. CCK8 (Figure 5B), colony formation (Figure 5C) and flow cytometry (Figure 5D) assay showed that miR-423 mimic + vector cell proliferation ability was significantly enhanced and the rate of apoptosis was significantly reduced compared with miR-423 mimic + ZFP36 transfected cells. In conclusion, in miR-423-mediated drug resistance and proliferation, the ZFP36-Wnt/β-catenin pathway played an important role in BC.
Downregulation of miR-423 Inhibited Drug-Resistance and Proliferation via the ZFP36- Wnt/β-Catenin Pathway in MCF-7/ADR Cells
As shown in Figure 6A, compared with the control group, si-ZFP36 can raise the expressions of nuclear β-catenin, cytoplasmic β-catenin, and cytoplasmic p-β-catenin and decrease expression of ZFP36 in MCF-7 cells. miR-423 inhibitor can decrease the expressions of nuclear β-catenin, cytoplasmic β-catenin, and cytoplasmic p-β-catenin and raise the expression of ZFP36 in MCF-7 cells. And the miR-423 inhibitor +si-ZFP36 can reverse the effect of miR-423 on the expressions of nuclear β-catenin, cytoplasmic β-catenin, cytoplasmic p-β-catenin and ZFP36.
CCK-8, colony formation and flow cytometry results showed that siRNA ZFP36 can reverse the reduced chemical resistance and proliferation induced by the transfection of miR-423 inhibitors (induced by induction of Figure 6B–D with miRNA-423 inhibitors).
Discussion
Chemotherapy is the most common method for the treatment of malignant tumors. However, with the use of chemotherapeutic drugs, many tumor cells inevitably develop drug resistance, both intrinsic and acquired.17,18 Chemotherapy plays a key role in the treatment of breast cancer.19 However, almost half of patients develop multidrug resistance (MDR) during chemotherapy, which increases clinical challenges for breast cancer treatment.20 Studies have shown that the mechanism of tumor drug resistance is caused by multiple factors such as oncogene activation and inactivation of tumor-suppressor genes, and multiple steps.21 There are a number of mechanisms that can explain the mechanism of breast cancer resistance. Recently, many studies have shown that drug-induced microRNA (miRNA) dysregulation plays an important role in the mechanism of acquired resistance.22
Research evidence suggests that miRNAs play a crucial role in tumor formation and progression.23 Although many miRNAs are abnormally expressed in breast cancer tissues or cells, the molecular regulatory functions and signal regulation mechanisms for the development and evolution of breast cancer have not been fully elucidated. Therefore, by analyzing miRNA-related targets and the signaling pathways involved in breast cancer, it is helpful to fully understand the relationship between mRNAs and tumor development, thus providing new target genes for the diagnosis and treatment of breast cancer.
Researchers found that miR-423 has a strong carcinogenic effect on the occurrence and development of breast cancer.16 In this study, the results showed that miR-423 was highly expressed in breast cancer. Moreover, miR-423 was significantly associated with tumor size, T stage and cancer chemosensitivity, but was not associated with lymph node metastasis, which is in a good agreement with other literatures.24
In breast cancer, miRNAs can be used to differentiate benign and malignant breast tissues. And some miRNAs have been shown to be closely related to chemosensitivity (5-FU).25 The result indicated that overexpression of miR-423 can significantly promote the proliferation and resistance of breast cancer cells, and inhibition of the expression of miR-423 can inhibit the proliferation and resistance of breast cancer cells. The experimental results were further confirmed by mouse experiments. These data highlighted the important role of MI R-423 as an oncogene in breast cancer.
Studies have reported that many miRNAs affected the development of tumors by targeting the regulation of mRNA expression. As a tumor-suppressor gene, miR-124 inhibited the proliferation, invasion and metastasis of colorectal tumor cells by targeting PRRX1 expression.26 miR-101 inhibited the development of liver cancer by downregulating EZH2 and increased the sensitivity of drugs that inhibited cell growth.27 In this study, bioinformatics software was first used to predict that ZFP36 might be a downstream target gene of miR-423. ZFP36 was a transcription factor with a finger-like domain.28 In recent years, there have been more and more reports of ZFP36 in tumors, which were shown to inhibit tumorigenesis and affect the malignant phenotype of tumors.29 However, the current report of ZFP36 in breast cancer was rare, and its expression regulation and specific mechanism of action were still unclear. The results indicated that downregulation of miR-423 significantly enhanced wt-ZFP36-3ʹUTR luciferase activity, confirming the existence of a targeted relationship between miR-423 and ZFP36. Simultaneous inhibition of miR-423 expression significantly enhanced the expression of ZFP36 mRNA and protein, indicating a negative correlation between DACH1 and mi R-552.
It had been previously reported that ZFP36 inhibited tumor proliferation and migration by inhibiting Wnt/β-catenin signaling pathway.30 The Wnt/β-catenin signaling pathway was an evolutionarily relatively conserved signaling pathway that played an important role in initiating and regulating a variety of cellular activities such as cell proliferation, cell polarity, and calcium channel homeostasis.31 β-catenin was a downstream pivotal signal molecule in this signaling pathway.32 GSK3 is the phosphorylated kinase of β-catenin. The phosphorylation of GSK3 will cause the degradation of β-catenin and keep the free amount of the latter in the cytoplasm at a low level. When GSK3 is inhibited, the degradation of β-catenin will be interrupted immediately, and the affinity of the phosphorylated β-catenin with APC will be reduced, so that the content of free β-catenin in the cell will be increased and transported into the nucleus. In order to further investigate the molecular mechanisms by which mi R-423 and ZFP36 regulated the biological functions of colorectal tumors, the Wnt/β-catenin signaling pathway was investigated in MCF-7 cells and miR-NC cells. The study revealed that the protein level of β-catenin expression was enhanced in miR-423 mimic-transfected MCF-7 cells compared with miR-NC cells. In contrast to the miR-423 mock group, increased expression of β-catenin and decreased expression of ZFP36 were not observed in MCF-7 cells transfected with miR-423 mimic + ZFP36. These results demonstrated that miR-423 activated Wnt/β-catenin signaling by silencing ZFP36, and that the miR-423 mimic + vector enhanced cell proliferation and reduced cell apoptosis compared with miR-423 mimic + ZFP36 transfected cells. In MCF-7/ADR cells, expression of β-catenin was decreased in MCF-7/ADR cells treated with miR-423 inhibitor, whereas siRNA ZFP36 was able to increase chemical resistance and proliferation induced by miR-423 inhibitor.
Conclusion
miR-423 was upregulated in breast tumor tissues and cell lines; miR-423 promoted proliferation and resistance of breast cancer cells; ZFP36 was a downstream target gene of miR-423; miR-423 inhibited the expression of ZFP36 by Wnt/β-catenin signaling pathway, thus promoting the proliferation and migration of breast cancer cells.
Ethical Approval
All applicable institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Veronesi U, Boyle P, Goldhirsch A, Orecchia R, Viale G. Breast cancer. Lancet. 2005;365(9472):1727–1741. doi:10.1016/S0140-6736(05)66546-4
2. Kã¤Rki A, Simonen R, Mã¤Lki㤠E, Selfe J. Impairments, activity limitations and participation restrictions 6 and 12 months after breast cancer operation. J Rehabil Med. 2005;37(3):180–188. doi:10.1080/16501970410024181
3. Ahn HS, Yoon SJ, Jo HY, Lee HY, Lee J, Seo HJ. Association between unplanned readmission rate and volume of breast cancer operation cases. Int J Clin Pract. 2010;60(1):32–35. doi:10.1111/j.1368-5031.2006.00736.x
4. Wirtgen M, Nahayo A, Linden A, et al. Breast cancer following radiotherapy and chemotherapy among young women with hodgkin disease—correction. J Am Med Assoc. 2003;290(10):1318.
5. Makoto K, Masafumi N, Akira T, et al. Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res. 2004;64(17):6071–6074. doi:10.1158/0008-5472.CAN-04-0416
6. Jin Q, Esteva FJ. Cross-talk between the ErbB/HER family and the type I insulin-like growth factor receptor signaling pathway in breast cancer. J Mammary Gland Biol Neoplasia. 2008;13(4):485–498. doi:10.1007/s10911-008-9107-3
7. Spizzo R, Almeida MI, Colombatti A, Calin GA. Long non-coding RNAs and cancer: a new frontier of translational research? Oncogene. 2012;31(43):4577–4587. doi:10.1038/onc.2011.621
8. Oliveras-Ferraros C, Cufí S, Vazquez-Martin A, et al. Micro(mi)RNA expression profile of breast cancer epithelial cells treated with the anti-diabetic drug metformin: induction of the tumor suppressor miRNA let-7a and suppression of the TGFβ-induced oncomiR miRNA-181a. Cell Cycle. 2011;10(7):1144–1151. doi:10.4161/cc.10.7.15210
9. Yusuke G, Satoko K, Rika N, et al. MicroRNA expression signature of castration-resistant prostate cancer: the microRNA-221/222 cluster functions as a tumour suppressor and disease progression marker. Br J Cancer. 2015;113(7):1055–1065. doi:10.1038/bjc.2015.300
10. Aslam MI, Venkatesh J, Jameson JS, et al. Identification of high-risk Dukes B colorectal cancer by microRNA expression profiling: a preliminary study. Colorectal Dis. 2015;17(7):578–588. doi:10.1111/codi.12886
11. Xiao-Feng L, Almeida MI, Weiqun M, et al. Modulation of MicroRNA-194 and cell migration by HER2-targeting trastuzumab in breast cancer. PLoS One. 2012;7(7):e41170. doi:10.1371/journal.pone.0041170
12. Cao M, Nie W, Li J, et al. MicroRNA-495 induces breast cancer cell migration by targeting JAM-A. Protein Cell. 2014;5(11):862–872. doi:10.1007/s13238-014-0088-2
13. Wang MJ, Zhang H, Li J, Zhao HD. microRNA-98 inhibits the proliferation, invasion, migration and promotes apoptosis of breast cancer cells by binding to HMGA2. Biosci Rep. 2018;38:BSR20180571. doi:10.1042/BSR20180571
14. Tang X, Zeng X, Huang Y, et al. miR-423-5p serves as a diagnostic indicator and inhibits the proliferation and invasion of ovarian cancer. Exp Ther Med. 2018;15(6):4723–4730.
15. Liu J, Wang X, Yang X, et al. miRNA423-5p regulates cell proliferation and invasion by targeting trefoil factor 1 in gastric cancer cells. Cancer Lett. 2014;347(1):98–104. doi:10.1016/j.canlet.2014.01.024
16. Zhao H, Gao A, Zhang Z, et al. Genetic analysis and preliminary function study of miR-423 in breast cancer. Tumor Biol. 2015;36(6):4763–4771. doi:10.1007/s13277-015-3126-7
17. Zhou J, Giannakakou P. Targeting microtubules for cancer chemotherapy. Curr Med Chem. 2005;5(1). doi:10.2174/1568011053352569
18. Ohtsukasa S, Okabe S, Yamashita H, Iwai T, Sugihara K. Increased expression of CEA and MHC class I in colorectal cancer cell lines exposed to chemotherapy drugs. J Cancer Res Clin Oncol. 2003;129(12):719–726. doi:10.1007/s00432-003-0492-0
19. Smith IC, Heys SD, Hutcheon AW, et al. Neoadjuvant chemotherapy in breast cancer: significantly enhanced response with docetaxel. J Clin Oncol. 2002;20(6):1456–1466. doi:10.1200/JCO.20.6.1456
20. Batrakova EV, Kelly DL, Li S, et al. Alteration of genomic responses to doxorubicin and prevention of MDR in breast cancer cells by a polymer excipient: pluronic P85. Mol Pharm. 2006;3(2):113–123. doi:10.1021/mp050050g
21. Ana-Maria F, Dietrich B. Cisplatin as an anti-tumor drug: cellular mechanisms of activity, drug resistance and induced side effects. Cancers. 2011;3(1):1351–1371. doi:10.3390/cancers3011351
22. Miller TE, Kalpana G, Bhuvaneswari R, et al. MicroRNA-221/222 confers tamoxifen resistance in breast cancer by targeting p27Kip1. J Biol Chem. 2008;283(44):29897–29903. doi:10.1074/jbc.M804612200
23. Rika N, Yusuke G, Satoko K, et al. Tumor-suppressive microRNA-29s inhibit cancer cell migration and invasion via targeting LAMC1 in prostate cancer. Int J Oncol. 2014;45(1):401–410. doi:10.3892/ijo.2014.2437
24. Smith RA, Jedlinski DJ, Gabrovska PN, Weinstein SR, Haupt L, Griffiths LR. A genetic variant located in miR-423 is associated with reduced breast cancer risk. Cancer Genomics Proteomics. 2012;9(3):115–118.
25. Kovalchuk O, Filkowski J, Meservy J, et al. Involvement of microRNA-451 in resistance of the MCF-7 breast cancer cells to chemotherapeutic drug doxorubicin. Mol Cancer Ther. 2008;7(7):2152–2159. doi:10.1158/1535-7163.MCT-08-0021
26. Yuqin Z, Lin Z, Jing H, et al. MiR-124 radiosensitizes human colorectal cancer cells by targeting PRRX1. PLoS One. 2014;9(4):e93917. doi:10.1371/journal.pone.0093917
27. Wu X, Dinglin X, Wang X, et al. Long noncoding RNA XIST promotes malignancies of esophageal squamous cell carcinoma via regulation of miR-101/EZH2. Oncotarget. 2017;8(44):76015–76028. doi:10.18632/oncotarget.18638
28. Shah S, Mostafa MM, Mcwhae A, Traves SL, Newton R. Negative feed-forward control of tumor necrosis factor (TNF) by tristetraprolin (ZFP36) is limited by the mitogen-activated protein kinase phosphatase, dual-specificity phosphatase 1 (DUSP1) IMPLICATIONS FOR REGULATION BY GLUCOCORTICOIDS. J Biol Chem. 2016;291(1):110. doi:10.1074/jbc.M115.697599
29. Wei F, Zhang T, Yang Z, et al. Gambogic acid efficiently kills stem-like colorectal cancer cells by upregulating ZFP36 expression. Cell Physio Biochem. 2018;46(2):829–846. doi:10.1159/000488740
30. Montorsi L, Guizzetti F, Alecci C, et al. Loss of ZFP36 expression in colorectal cancer correlates to wnt/ß-catenin activity and enhances epithelial-to-mesenchymal transition through upregulation of ZEB1, SOX9 and MACC1. Oncotarget. 2016;7(37):59144–59157. doi:10.18632/oncotarget.10828
31. MacDonald BT, Tamai K, Xi H. Wnt/β-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17(1):9–26. doi:10.1016/j.devcel.2009.06.016
32. Desheng L, Liu JX, Tomoyuki E, et al. Ethacrynic acid exhibits selective toxicity to chronic lymphocytic leukemia cells by inhibition of the Wnt/beta-catenin pathway. PLoS One. 2009;4(12):e8294. doi:10.1371/journal.pone.0008294
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.