Back to Journals » International Journal of General Medicine » Volume 13
Metastases to the Breast from Extramammary Nonhematological Malignancies: Case Series
Authors Wan X, Zhang H, Zhang Y , Peng Y
Received 11 August 2020
Accepted for publication 21 October 2020
Published 12 November 2020 Volume 2020:13 Pages 1105—1114
DOI https://doi.org/10.2147/IJGM.S276602
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Xue Wan,1 Heqing Zhang,1 Yahan Zhang,2 Yulan Peng1
1Department of Ultrasound, West China Hospital of Sichuan University, Chengdu, Sichuan 610041, People’s Republic of China; 2Department of Pathology, West China Hospital of Sichuan University, Chengdu, Sichuan 610041, People’s Republic of China
Correspondence: Yulan Peng Tel/Fax +86 18980601606
Email [email protected]
Objective: This article aims to provide a better understanding of ultrasonography and immunohistochemistry of secondary nonhematological tumors of breast.
Methods: The study reviewed the ultrasound findings and immunohistochemical features of nonhematological metastatic breast tumor cases found in patients of West China Hospital, Sichuan University from 2007 to 2019. Each case was categorized as secondary breast malignancy using histopathological results.
Results: Fourteen cases were identified from West China Hospital database. Ten cases originated in the lung, 2 cases in the stomach, 1 case in the ovary and 1 case of neuroendocrine carcinomas. Fourteen masses were evaluated. Ultrasound findings showed that tumors were hypoechoic (14/14), irregular (13/14), indistinct margin (13/14), along a long axis parallel to the skin (11/14), lacked vascularity via color doppler flow imaging (9/14). Eight cases showed no posterior features. Calcification was found in 1 case of lung adenocarcinoma that had metastasized to the breast. Abnormal axillary lymph nodes were detected in 5 cases. Immunohistochemical analysis showed that estrogen receptor (ER) and progesterone receptor (PR) were both negative in 11 cases, including gastric and lung cancer metastasis. One case of ovarian metastasis was positive for ER and negative for PR. Six patients were positive for cytokeratin 7 (CK7) and negative for cytokeratin 20 (CK20), including lung and ovarian carcinoma metastasis. Thyroid transcription factor-1 (TTF-1) was positive in 9 of 10 pulmonary carcinoma metastases. The patient of ovarian metastasis was positive for Wilms’ tumour 1 (WT-1) and carbohydrate antigen 125 (CA125). Two cases from gastric metastasis were positive for caudal-type homeobox 2 (CDX2).
Conclusion: Although breast ultrasound is not useful in distinguishing metastases from primary breast cancer, it is helpful in diagnosing breast lesions as oncological diseases and provide evidence for further examination of patients. Immunohistochemistry plays an important role in distinguishing secondary breast cancer from primary, especially in patients without tumor history.
Keywords: breast secondary carcinomas, ultrasonography, immunohistochemistry, clinical history
Introduction
Breast carcinoma is one of the most common types of malignant tumor in women. In 2018, the incidence of breast cancer accounted for 24.2% of all malignant tumors in women across the world.1 However, metastases of extramammary malignancies to the breast are extremely scarce, accounting for only 0.3% to 2.7% of all malignant breast tumors.2,3 In addition to metastasis of contralateral mammary cancer, the most common primary tumors with breast metastases are hematologic malignancies, including leukemia and lymphoma.2,4–6 Breast metastases have also been reported in primary cancers such as melanoma, lung cancer, ovarian cancer, gastric cancer and neuroendocrine cancer.7–9 To avoid unnecessary and potentially harmful treatments, accurate diagnosis of secondary breast carcinoma is very important. Identification of the primary and secondary breast tumors is crucial. Therefore, we have summarized some features of metastatic neoplasms of the breast by reviewing our cases.
Materials and Methods
From January 2007 to December 2019, records of 14 cases of nonhematological metastases to the breast were collected from the Department of Ultrasound, West China Hospital, Sichuan University. All cases were confirmed by ultrasound-guided core needle biopsy or surgical pathology breast biopsies. All patients had undergone breast ultrasound (US). Breast nodules or masses were found in 9 of 11 cases of chest computed tomography. None of the patients had undergone mammography and mamma magnetic resonance imaging examination.
US examinations were performed by ultrasonic technicians or ultrasound doctors using a 5-to12-MHz linear probe and iU22 scanner (Philips Ultrasound, Bothell, WA), or a 6- to 14-MHz probe and a EUB-8500 scanner (Hitachi Medical, Tokyo, Japan) or a 6- to 15-MHz probe and a LOGIQ E9 scanner (GE Healthcare, Wauwatosa, WI, USA). The location, size, shape, margin, orientation, echogenicity, calcification, color doppler blood flow signal of the mass and axillary lymph node status were recorded. Two ultrasound doctors with more than five years of clinical experience retrospectively assessed the ultrasonic imaging features using the American college of Radiology-Breast imaging reporting and data system (ACR-BIRADS) 5th edition specifications, including shape, margin, orientation, echogenicity, posterior features, calcifications, vascularity, the presence of axillary lymphadenopathy and BIRADS category. Meanwhile, they collected additional ultrasound information about mass lesion or diffused infiltrative lesion, location, multiplicity, size, bilaterality. In the presence of multiple breast masses of one patient, they evaluated ultrasonic image characteristics of the largest mass.
The immunohistochemical staining of the data from the department of pathology in our hospital was analyzed retrospectively. These included ER, PR, CK7and CK20, TTF-1, WT-1, CA125 and CDX2. Other stains were pancytokeratin (PCK), chromogranin A (CgA), synaptophysin (Syn) and Desmin.
Results
Clinical Features
There were 12 women and 2 men aged between 31 and 81 years old (median age was 50 years old). These patients had an interval of 22 days to 6 years from the diagnosis of primary cancer to the discovery of breast metastases. Ten cases originated in the lung (9 cases in lung adenocarcinoma, 1 case in small cell lung carcinoma), 2 cases in the gastrointestinal tract (both in high-grade gastric adenocarcinoma), 1 case originated in the ovary (serous papillary ovarian adenocarcinoma) and 1 case in laryngeal neuroendocrine carcinomas. Among 14 patients, an ultrasound-guided core needle biopsy was performed in 10 patients, 2 patients were diagnosed by excisional biopsy and the rest were treated using both means.
Nine patients had a known history of a primary extramammary neoplasm and they had undergone operation and/or chemotherapy, radiotherapy previously. As for the remaining 5 patients, the breast lesions were diagnosed as simultaneous metastasis from extramammary malignancy. Three cases originated from lung adenocarcinoma, 1 from small cell carcinoma of lung and 1 from poorly differentiated adenocarcinoma of the stomach. Two cases only had breast symptom as the initial manifestation. One merely presented with a palpable breast mass while the other manifested as skin edema and erythema of breast. The primary lesions originated from lung adenocarcinoma and small cell carcinoma, respectively. Amidst all of our cases, breast was the only site of metastasis in two cases. Breast metastasis of the rest of the cases occurred as part of more widespread tumor dissemination. Other sites of widespread metastasis had bone, liver, brain, pleura, peritoneum or soft tissue. The final diagnosis was made by ultrasound-guided core needle biopsy or surgical resection biopsy. Four of these 14 patients underwent mastectomy for the breast mass.
The breast symptom was present as palpable lumps in 10 cases, of which 6 cases had hard breast texture and 2 cases had breast pain. One patient had diffused redness and swelling of the skin of one side of the breast with pain, but no breast lump was palpable. In one patient with bilateral breast involvement, the skin of both breasts displayed orange peel-like skin changes. No nipple discharge or depression was found in all patients.
Ultrasound Findings
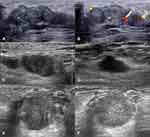
A total of 14 masses were evaluated (Table 1). Unilateral, left-sided and outer upper quadrant of the breast were more frequently affected. Among 14 cases, 13 patients’ breast masses were located on a single side of the breast and 8 cases had a single mass, while 6 cases had multiple masses. The mass occurred on the left breast in 8 cases, on the right breast in 5 cases and on both sides in 1 case. The maximum diameter of the breast mass was about 6 mm to 78 mm (mean diameter was 22 mm), of which 8 cases were less than 20 mm and 6 cases were more than 20 mm. Of the 14 cases, 13 cases showed irregular and ill-defined breast masses and 11 cases showed that the long axis of the mass was parallel to the skin (Figure 1A and B and D). Only one case from metastasis of lung small cell carcinoma showed a clear, regular and round-like breast nodule. All cases presented with hypoechoic masses, including homogeneous hypoechoes in 11 cases and heterogeneous hypoechoes in 3 cases. There were no posterior features in 8 cases (Figure 1D) and posterior enhancement in 2 cases. Four cases of metastases from lung adenocarcinoma had posterior shadowing or combined pattern (Figure 1E and F). Two cases of enhancement posterior features were, respectively, metastasized to the breast from pulmonary adenocarcinoma and lung small cell carcinoma. Calcification was found in 1 case. To the best of our knowledge, this is the first-ever report of calcification in breast metastasis of lung adenocarcinoma. Vascularity was found in 5 cases, including internal blood flow signals in 2 cases, vessels in rim in 1 case, and 2 cases with internal blood flow and vessels in rim (Figure 1B). Nine masses were detected with no vascularity on color doppler examination (Figure 1F). Affected side or bilateral abnormal axillary lymph node could be detected in 5 patients (Figure 1C). The final assessment categories were BIRADS 3 in 1 case, BIRADS 4 in 12 cases and BIRADS 5 in 1 case, including BIRADS 4A in 2 cases, BIRADS 4B in 2 cases and BIRADS 4C in 8 cases.
 |
Table 1 Ultrasound Findings |
Immunohistochemical Characteristics
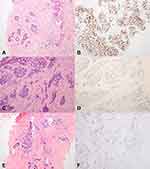
All patients performed immunohistochemical staining (Table 2). With the exception of the one case of ovarian cancer metastatic to the breast, which was positive for ER and negative for PR, the other 11 cases were negative for both biomarkers, including 2 cases of stomach cancer metastasis and 9 cases of pulmonary carcinoma metastasis. CK 7 and CK 20 were assessed in 6 patients with primary tumors originating from the lung and ovary. All of them were positive for CK7 and negative for CK20. Nine cases were TTF-1 positive and 1 case was negative in the cases of breast metastasis from lung cancer (Figure 2A–D). WT-1 and CA125 were positive in patients with ovarian metastasis. CDX2 was positive in two cases of primary gastric origin (Figure 2E and F). The immunohistochemical markers showed that the tissue was positive for PCK, CgA and Syn, negative for Desmin of neuroendocrine carcinomas metastatic to the breast.
 |
Table 2 Immunohistochemical Staining Features |
Discussion
The breast is an uncommon metastatic site of malignancies.10 In this study, there was a high incidence of pulmonary cancer, which is probably due to a high prevalence of lung cancer in China. But the relevant evidence needs to be further studied. In the reported literature, men are less likely to be involved than women.5,11 This may be due to differences in breast size, blood vessels or hormone levels between men and women.11 It is also possible that angiogenic factors and immunological determinants also contribute to this.11 Two male patients in our study had metastases to the breast, and the ratio of male to female was 1:6.
Clinically, the characteristic of breast metastasis is a palpable nontender movable lump. The symptom of peaud’orange, nipple retraction, or nipple discharge is uncommon.8,11–14 Most breast metastatic masses grow rapidly and close to the skin, which may induce skin edema or erythema. This manifestation can often be mistaken for inflammatory breast cancer.11,12 In our study, most mammary symptoms of metastatic tumor were painless breast masses.
As per our research, malignant tumors of lung and stomach are more likely to be detected at the same time as breast metastasis. This may be related to the pathway of tumor metastasis. The specific mechanism needs to be further studied. The diagnosis of breast masses becomes more complicated in the absence of malignancy history, especially with breast symptoms being the first manifestation. In this case, pathological examination is particularly important for these patients. Imaging examination is also essential for indicating extra-breast tumors.
Metastasis may show single or multiple and unilateral or bilateral lesions.11,14 In this study, a single and unilateral breast mass from non-breast metastatic disease are more prevalent and masses are more common than diffused infiltrative lesions. The mass was located in the left breast in most cases. There was a wide range in the size of the mass. However, masses smaller than 20 mm in diameter are relatively more common.
The common manifestations of ultrasonography were round to oval shaped, well-defined hypoechoic solid masses.11,14–18 Some of our results were different. The sonogram of breast masses is more common with irregular shape and unclear boundaries (13/14). But the mass of metastasis from lung small cell carcinoma was circumscribed, regular and round-like. The metastasis of lung small cell carcinoma is easier to perform as benign nodules than other origins of tumors. The long axis of most masses was parallel to the skin (11/14). All masses of the nonparallel growing to the skin were metastasized from lung adenocarcinoma. In other words, the masses of the breast metastases from pulmonary adenocarcinoma were more likely to appear nonparallel growth than other sources of mammary metastases. All masses are hypoechoic, but the echotexture is often homogeneous. No posterior features of the mass are more common, accounting for 57.1% (8/14). The masses of shadowing or combined pattern of posterior features were more likely to occur in metastases of lung adenocarcinoma. Although the ultrasound images of masses from different metastatic sites are different, it is difficult to distinguish them from one another. The discovery of calcification in metastatic tumors is rare. It may occur in metastasis from ovarian cancer, hepatocellular carcinoma, medullary thyroid carcinoma and gastric cancer in reported literatures.8,13,15,16,18 In our study, we found one case of calcifications of metastatic lung adenocarcinoma. To the best of our knowledge, this is the first-ever report of calcification in breast metastasis of lung adenocarcinoma. It also highlights the diversity and complexity of the characteristics of metastatic lesions. Color Doppler examination showed that no blood flow signal of the mass accounted for 64.3% (9/14). This is different from the abundant blood supply of primary breast carcinoma. These common features of secondary breast carcinoma are not easy to differentiate from primary malignant tumors of the breast, but they can suggest that the breast mass is malignant. In Mun et al report, according to breast metastases routes from extramammary malignancies, they summarized typical and atypical radiological features of metastatic diseases of the breast.15 Compared with primary breast cancer, metastatic breast cancer from hematogenous routes has less axillary lymph node involvement.15 But abnormal enlargement of axillary or internal mammary lymph nodes are associated with metastases from lymphatic routes. These metastases originate from contralateral breast cancer, as well as in gastric and ovarian cancer.8,15,19,20 There were 5 cases of abnormal axillary lymph node in the series of our study. Original lesions originated from lung in 4 cases and stomach in 1 case. This may be related to lymphatic metastasis.
The pathologic and immunohistochemical studies become quite helpful when clinical and imaging features of non-breast metastatic tumors are difficult to identify from primary breast cancer. The point of identification of primary breast cancer from secondary cancer is that the former should be able to find the composition of the catheter in situ or the small leaf in situ on HE section.21 It is an important hint for the diagnosis of metastasis to the breast if the histological characteristics are rare for primary breast cancers. Meanwhile, the diagnosis of secondary breast carcinoma depends on the features of primary malignancies.3,22 For example, small cell carcinoma suggests lung origin, clear cell carcinoma suggests renal origin, papillary carcinoma may be serous papillary carcinoma of the ovary, pigment and intranuclear inclusion bodies suggest melanoma. However, not every metastatic lesion has these characteristics, and about 1/3 of the lesions have no special histological features.22 A history plays an important role in the correct diagnosis of these patients. A diagnosis can usually be made by comparing the pathologic characteristics of primary tumor.
Immunohistochemical analysis is helpful in the diagnosis of secondary tumor in the absence of medical history. In general, one patient should use a group of antibodies of immunohistochemical study because no marker is 100% specific or sensitive.23 The examination of CK 7 and CK 20 contributed to the recognition source of tumors. Breast cancer is typically CK 7+ and CK 20-. In addition, CK7+ and CK 20- are also found in non-mucinous ovarian cancer, lung adenocarcinoma and thyroid cancer.22,24,25 CK20 is commonly expressed in colorectal cancers and other gastrointestinal cancers and is infrequently positive in breast cancers.25,26 In our study, CK7+ and CK20- were found in metastasis lung adenocarcinoma and papillary adenocarcinoma of the ovary. The positive expression of mammaglobin, ER, PR, GATA binding protein 3 (GATA3) and gross cystic disease fluid protein 15 (GCDFP-15) helps to support the origin of breast.23 GCDFP-15 is expressed in about 60% of mammary carcinomas, including 10–20% of triple-negative mammary carcinomas.25,27 GCDFP-15 may also be expressed in salivary glands, sweat glands, ovaries, endometrium and pulmonary carcinomas.25,27 Triple-negative carcinomas do not usually express GCDFP-15, GATA-3 or Mammaglobin. In such cases, SOX-10 may be helpful.28 Primary small cell carcinoma of the breast is extremely rare. The negative expression of ER and PR in the small cell carcinoma favors metastasis since the very rare examples of primary small cell carcinoma of the breast express these markers in 33% to 50% of cases.29 Biomarkers of TTF-1 for lung cancer, CDX2 for gastric cancer, CD56(+), Syn, CgA for neuroendocrine cancer and WT-1 and CA125 for ovarian cancer are common markers for identification of the origin of metastatic lesions.3,9,22,30 TTF-1 is expressed in most lung adenocarcinomas and small cell carcinomas accounting for 70–80%, and usually most cells are positive.25,31,32 TTF-1 is rarely expressed in other types of lung cancers.25 It is very rarely expressed in breast cancers, mostly in grade 3 non-special type25,33 and in rare primary small cell carcinoma of the breast.25,34 CDX2 is sparsely expressed in colorectal cancer and is often positive in gastric and esophageal adenocarcinomas.35–37 CDX2 is occasionally positive in other sites, including breast.25 WT1, CA125 and ER are usually diffusely expressed in ovarian serous papillary carcinoma.38 WT1 is also often expressed in pure or mixed breast mucinous carcinoma and occasionally focally expressed in other histological types of mammary carcinoma.39 CA125 is also positive in mammary carcinoma.25 ER is positive for about 80% of breast cancers.25 Therefore, a group of antibodies is needed to comprehensively consider the metastasis. As a type of nerve cell adhesion molecule, CD56 is highly sensitive to the identification of neuroendocrine phenotype of tumor.40,41 However, it has low specificity because anti-CD56 antibodies also located other tumors, including well-differentiated thyroid carcinoma, lung squamous cell carcinoma, hepatocellular carcinoma, renal cell carcinoma, ovarian carcinoma, endometrial carcinoma, neuroblastoma, etc.40,42,43 CgA and Syn are the preferred markers to verify the neuroendocrine origin of tumors.40 CgA represents strongly positive in normal neuroendocrine cells and is a specific neuroendocrine marker.40 Syn is the most sensitive marker of general neuroendocrine tumor, which is positive both in well-differentiated and in poorly differentiated tumors.40 But it is also positive in olfactory neuroblastoma, neuroblastoma, Ewing’s sarcoma and adrenocortical carcinoma.40,44,45 Therefore, Syn is not a specific neuroendocrine marker.40 The combination of CgA and Syn is always desirable.
Conclusion
Timely and accurate diagnosis of metastases to the breast is significant for treatment and to avoid unnecessary surgery for these patients. The sonographic characteristics of secondary breast involvements help to indicate that breast disease is a malignant tumor, despite it being difficult to distinguish between secondary and primary breast cancer. It can also provide a basis for further examination of patients. If the history of extramammary tumor is known, it is of great significance with regard to indicating breast metastasis. Pathological examination is very critical to the diagnosis and differential diagnosis of metastatic carcinoma. Immunohistochemistry is very helpful for diagnosis, especially for differential diagnosis of patients without tumor history.
Abbreviations
ACR-BIRADS, American college of Radiology-Breast imaging reporting and data system; ER, estrogen receptor; PR, progesterone receptor; GATA3, GATA binding protein 3; GCDFP-15, gross cystic disease fluid protein 15; CK7, cytokeratin 7; CK20, cytokeratin 20; TTF-1, thyroid transcription factor-1; CDX2, caudal-type homeobox 2; WT-1, Wilms’ tumour 1; CA125, carbohydrate antigen 125; PCK, pancytokeratin; CgA, chromograninA; Syn, synaptophysin.
Ethics Approval and Consent to Participate
The Research Ethics Committee of West China Hospital of Sichuan University approved the retrospective report and waived the requirement for written informed consent because of the loss of follow-up of patients. This retrospective study of 14 patients in the West China Hospital of Sichuan University complies with the Declaration of Helsinki. The author ensures the confidentiality of patient information and data.
Acknowledgments
This work was supported by the Technology Innovation Project of New Industry Technology Research Institute in Chengdu in 2017 (Project 2017-CY02-00027-GX).
Disclosure
The authors affirm that there is no conflict of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
2. Ota T, Hasegawa Y, Okimura A, et al. Breast metastasis from EGFR-mutated lung adenocarcinoma: a case report and review of the literature. Clin Case Rep. 2018;6(8):1510–1516.
3. Lee SK, Kim WW, Kim SH, et al. Characteristics of metastasis in the breast from extramammary malignancies. J Surg Oncol. 2010;101(2):137–140. doi:10.1002/jso.21453
4. Magnoni F, Di Tonno C, Accardo G, et al. Breast cancer with rare metastatic manifestation. Future Oncol. 2019;15(21):2437–2440. doi:10.2217/fon-2019-0263
5. DeLair DF, Corben AD, Catalano JP, Vallejo CE, Brogi E, Tan LK. Non-mammary metastases to the breast and axilla: a study of 85 cases. Mod Pathol. 2013;26(3):343–349.
6. Sun P, Chen J, Lu J, Luo R, Li M, He J. Characteristics of breast metastases from non-breast solid tumors in 22 patients from a southern Chinese population. Oncol Lett. 2018;15(3):3685–3693.
7. Vizcaino I, Torregrosa A, Higueras V, et al. Metastasis to the breast from extramammary malignancies: a report of four cases and a review of literature. Eur Radiol. 2001;11(9):1659–1665. doi:10.1007/s003300000807
8. Lee SH, Park JM, Kook SH, Han BK, Moon WK. Metastatic tumors to the breast: mammographic and ultrasonographic findings. J Ultrasound Med. 2000;19(4):257–262. doi:10.7863/jum.2000.19.4.257
9. Ali RH, Taraboanta C, Mohammad T, Hayes MM, Ionescu DN. Metastatic non-small cell lung carcinoma a mimic of primary breast carcinoma-case series and literature review. Virchows Arch. 2018;472(5):771–777. doi:10.1007/s00428-017-2262-4
10. Kim HJ, Kim SY, Huh S. Multimodality imaging findings of metaplastic breast carcinomas: a report of five Cases. Ultrasound Q. 2018;34(2):88–93. doi:10.1097/RUQ.0000000000000340
11. Noguera J, Martinez-Miravete P, Idoate F, et al. Metastases to the breast: a review of 33 cases. Australas Radiol. 2007;51(2):133–138. doi:10.1111/j.1440-1673.2007.01681.x
12. Feder JM, de Paredes ES, Hogge JP, Wilken JJ. Unusual breast lesions: radiologic-pathologic correlation. Radiographics. 1999;19:
13. Yang WT, Muttarak M, Ho LW. Nonmammary malignancies of the breast: ultrasound, CT, and MRI. Semin Ultrasound CT MR. 2000;21(5):375–394.
14. Lee JH, Kim SH, Kang BJ, Cha ES, Kim HS, Choi JJ. Metastases to the breast from extramammary malignancies-sonographic features. J Clin Ultrasound. 2011;39(5):248–255. doi:10.1002/jcu.20815
15. Mun SH, Ko EY, Han BK, Shin JH, Kim SJ, Cho EY. Breast metastases from extramammary malignancies: typical and atypical ultrasound features. Korean J Radiol. 2014;15(1):20–28. doi:10.3348/kjr.2014.15.1.20
16. Bitencourt AGV, Gama RRM, Graziano L, et al. Breast metastases from extramammary malignancies: multimodality imaging aspects. Br J Radiol. 2017;90(1077):20170197. doi:10.1259/bjr.20170197
17. Shen YW, Sui YX, Zhang XM, et al. Ipsilateral breast metastasis from a pulmonary adenocarcinoma: a case report and a focused review of the literature. Int J Clin Exp Pathol. 2015;8(8):9647–9654.
18. Sippo DA, Kulkarni K, Carlo PD, et al. Metastatic disease to the breast from extramammary malignancies: a multimodality pictorial review. Curr Probl Diagn Radiol. 2016;45(3):225–232. doi:10.1067/j.cpradiol.2015.07.001
19. Muttarak M, Nimmonrat A, Chaiwun B. Metastatic carcinoma to the male and female breast. Australas Radiol. 1998;42(1):16–19. doi:10.1111/j.1440-1673.1998.tb00556.x
20. Krishnan EU, Phillips AK, Randell A, Taylor B, Garg SK. Bilateral metastatic inflammatory carcinoma in the breast from primary ovarian cancer. Obstet Gynecol. 1980;55(3Suppl):94s–96s. doi:10.1097/00006250-198003001-00029
21. Harris GC, Denley HE, Pinder SE, et al. Correlation of histologic prognostic factors in core biopsies and therapeutic excisions of invasive breast carcinoma. Am J Surg Pathol. 2003;27(1):11–15. doi:10.1097/00000478-200301000-00002
22. Lee AH. The histological diagnosis of metastases to the breast from extramammary malignancies. J Clin Pathol. 2007;60(12):1333–1341. doi:10.1136/jcp.2006.046078
23. Bonacho T, Rodrigues F, Liberal J. Immunohistochemistry for diagnosis and prognosis of breast cancer: a review. Biotech Histochem. 2020;95(2):71–91. doi:10.1080/10520295.2019.1651901
24. Chu PG, Weiss LM. Keratin expression in human tissues and neoplasms. Histopathology. 2002;40(5):403–439. doi:10.1046/j.1365-2559.2002.01387.x
25. Lee AHS, Hodi Z, Soomro I, et al. Histological clues to the diagnosis of metastasis to the breast from extramammary malignancies. Revised version. Histopathology. 2020. doi:10.1111/his.14141
26. Chu P, Wu E, Weiss LM. Cytokeratin 7 and cytokeratin 20 expression in epithelial neoplasms: a survey of 435 cases. Mod Pathol. 2000;13(9):962–972. doi:10.1038/modpathol.3880175
27. Gown AM, Fulton RS, Kandalaft PL. Markers of metastatic carcinoma of breast origin. Histopathology. 2016;68(1):86–95. doi:10.1111/his.12877
28. Kriegsmann K, Flechtenmacher C, Heil J, et al. Immunohistological expression of SOX-10 in triple-negative breast cancer: a descriptive analysis of 113 samples. Int J Mol Sci. 2020;21(17):6407. doi:10.3390/ijms21176407
29. Mirza IA, Shahab N. Small cell carcinoma of the breast. Semin Oncol. 2007;34(1):64–66. doi:10.1053/j.seminoncol.2006.10.029
30. Lin Q, Cai GP, Yang KY, Yang L, Chen CS, Li YP. Case report: small cell transformation and metastasis to the breast in a patient with lung adenocarcinoma following maintenance treatment with epidermal growth factor receptor tyrosine kinase inhibitors. BMC Cancer. 2016;16:593. doi:10.1186/s12885-016-2623-4
31. Kaufmann O, Dietel M. Expression of thyroid transcription factor-1 in pulmonary and extrapulmonary small cell carcinomas and other neuroendocrine carcinomas of various primary sites. Histopathology. 2000;36(5):415–420. doi:10.1046/j.1365-2559.2000.00890.x
32. Kaufmann O, Dietel M. Thyroid transcription factor-1 is the superior immunohistochemical marker for pulmonary adenocarcinomas and large cell carcinomas compared to surfactant proteins A and B. Histopathology. 2000;36(1):8–16. doi:10.1046/j.1365-2559.2000.00801.x
33. Robens J, Goldstein L, Gown AM, Schnitt SJ. Thyroid transcription factor-1 expression in breast carcinomas. Am J Surg Pathol. 2010;34(12):1881–1885. doi:10.1097/PAS.0b013e3181f884e8
34. Shin SJ, DeLellis RA, Rosen PP. Small cell carcinoma of the breast–additional immunohistochemical studies. Am J Surg Pathol. 2001;25(6):831–832. doi:10.1097/00000478-200106000-00023
35. Werling RW, Yaziji H, Bacchi CE, Gown AM. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. Am J Surg Pathol. 2003;27(3):303–310. doi:10.1097/00000478-200303000-00003
36. Moskaluk CA, Zhang H, Powell SM, Cerilli LA, Hampton GM, Frierson HF. Cdx2 protein expression in normal and malignant human tissues: an immunohistochemical survey using tissue microarrays. Mod Pathol. 2003;16(9):913–919. doi:10.1097/01.MP.0000086073.92773.55
37. Panarelli NC, Yantiss RK, Yeh MM, Liu Y, Chen YT. Tissue-specific cadherin CDH17 is a useful marker of gastrointestinal adenocarcinomas with higher sensitivity than CDX2. Am J Clin Pathol. 2012;138(2):211–222. doi:10.1309/AJCPKSHXI3XEHW1J
38. Moritani S, Ichihara S, Hasegawa M, et al. Serous papillary adenocarcinoma of the female genital organs and invasive micropapillary carcinoma of the breast. Are WT1, CA125, and GCDFP-15 useful in differential diagnosis? Hum Pathol. 2008;39(5):666–671. doi:10.1016/j.humpath.2007.09.009
39. Domfeh AB, Carley AL, Striebel JM, et al. WT1 immunoreactivity in breast carcinoma: selective expression in pure and mixed mucinous subtypes. Mod Pathol. 2008;21(10):1217–1223. doi:10.1038/modpathol.2008.69
40. Uccella S, La Rosa S, Volante M, Papotti M. Immunohistochemical biomarkers of gastrointestinal, pancreatic, pulmonary, and thymic neuroendocrine neoplasms. Endocr Pathol. 2018;29(2):150–168. doi:10.1007/s12022-018-9522-y
41. Jin L, Hemperly JJ, Lloyd RV. Expression of neural cell adhesion molecule in normal and neoplastic human neuroendocrine tissues. Am J Pathol. 1991;138(4):961–969.
42. Bösmüller HC, Wagner P, Pham DL, et al. CD56 (neural cell adhesion molecule) expression in ovarian carcinomas: association with high-grade and advanced stage but not with neuroendocrine differentiation. Int J Gynecol Cancer. 2017;27(2):239–245. doi:10.1097/IGC.0000000000000888
43. Wachowiak R, Rawnaq T, Metzger R, et al. Universal expression of cell adhesion molecule NCAM in neuroblastoma in contrast to L1: implications for different roles in tumor biology of neuroblastoma? Pediatr Surg Int. 2008;24(12):1361–1364. doi:10.1007/s00383-008-2264-z
44. Gould VE, Lee I, Wiedenmann B, Moll R, Chejfec G, Franke WW. Synaptophysin: a novel marker for neurons, certain neuroendocrine cells, and their neoplasms. Hum Pathol. 1986;17(10):979–983. doi:10.1016/S0046-8177(86)80080-6
45. Komminoth P, Roth J, Schröder S, Saremaslani P, Heitz PU. Overlapping expression of immunohistochemical markers and synaptophysin mRNA in pheochromocytomas and adrenocortical carcinomas. Implications for the differential diagnosis of adrenal gland tumors. Lab Invest. 1995;72(4):424–431.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.