Back to Journals » Infection and Drug Resistance » Volume 14
Maxing Shigan Decoction Mitigates Mycoplasma pneumonia-Induced Pyroptosis in A549 Cells via the NLRP3 Inflammasome
Authors Liu F, Liu TT, Sun M , Zhou JM, Xue F, Chen SH, Chen J, Zhang L
Received 20 November 2020
Accepted for publication 19 February 2021
Published 3 March 2021 Volume 2021:14 Pages 859—867
DOI https://doi.org/10.2147/IDR.S292413
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Héctor Mora-Montes
Fang Liu,1,2,* TianTian Liu,2,* Min Sun,2 JingMin Zhou,2 Feng Xue,2 ShuangHui Chen,2 Jia Chen,3 Lei Zhang1
1Department of Pediatrics, Pudong New Area People’s Hospital, Shanghai, 201200, People’s Republic of China; 2Department of Pediatrics, East Hospital Affiliated to Tongji University, Shanghai, 200123, People’s Republic of China; 3Department of Traditional Chinese Medicine, Shanghai Children’s Medical Center, Shanghai, 200127, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lei Zhang; Jia Chen Email [email protected]; [email protected]
Background: Mycoplasma pneumoniae is a predominant cause of community-acquired respiratory infections. We recently discovered the clinical efficacy of Maxing shigan decoction (MXSG) in M. pneumoniae infection and designed a study to explore the mechanism of action.
Methods: Serum IL-1β, IL-18, and TNF-α, and transcript expression of the NLR Family, Pyrin Domain Containing Protein 3 (NLRP3) were measured in the peripheral blood mononuclear cells (PBMCs) of 30 children with M. pneumoniae infection and 30 healthy donors. An in vitro model of M. pneumoniae infection in A549 cell culture was used to explore the curative effects and mechanisms of MXSG. Pyroptosis was measured by flow cytometry with activated caspase-1 and propidium iodide staining. IL-1β, IL-18, and TNF-α, and NLRP3 transcript expression were measured by qRT-PCR. Protein expression of NLRP3, Caspase-1, pro-caspase-1, IL-1β, pro-IL-1β, and GSDMD-N was determined by Western blotting. Experimental confirmation was performed in NLRP3-overexpressing A549 cells and in the presence of an NLRP3 inhibitor, INF39.
Results: M. pneumoniae infection-induced IL-1β, IL-18, TNF-α, and mRNA expression of NLRP3 in PBMCs and promoted pyroptosis in A549 cells. It also induced IL-1β, IL-18, TNF-α, and up-regulated NLRP3, ro-IL-1β, Caspase-1, Pro-Caspase-1, and GSDMD-N in culture. Similar to the NLRP3 inhibitor INF39, MXSG (0.1, 0.2, and 0.4 mg/mL) suppressed pyroptosis induced by M. pneumoniae infection and decreased IL-1β (P < 0.001), IL-18, TNF-α in culture. MXSG down-regulated NLRP3, pro-IL-1β, Caspase-1, pro-Caspase-1, and GSDMD-N in infected cultures and mitigated NLRP3 overexpression-induced pyroptosis.
Conclusion: MXSG mitigates M. pneumoniae-induced pyroptosis in A549 cells via the NLRP3 inflammasome.
Keywords: Mycoplasma pneumoniae, pyroptosis, Maxing shigan decoction, NLRP3 inflammasome
Introduction
Mycoplasma pneumoniae is one of the smallest self-replicating microorganisms. A harmful human pathogen, M. pneumoniae is one of the primary causes of community-acquired respiratory infections, leading to life-threatening tracheobronchitis and pneumonia.1,2 Although the diagnosis of M. pneumoniae has dramatically improved, the available treatments (macrolides antibiotics, quinolones, and glucocorticoids) are not fully effective against this pathogen in the respiratory tract.2–5 Alternative remedies for M. pneumoniae respiratory infections are urgently needed. In recent years, numerous pharmacological studies have shown that traditional Chinese medicine (TCM) can effectively treat several intractable diseases, particularly inflammatory diseases and infections caused by drug-resistant bacteria.6,7 Viral and bacterial infections result in an inflammasome-mediated immune response. The inflammasome induces the release of pro-inflammatory cytokines such as IL-1β, TNF-α, and IL-18, leading to Caspase-1 activation and pyroptosis.8,9
Maxing shigan decoction (MXSG) is a classic TCM formula composed of whole Ephedra sinica herb, Prunus armeniaca seeds, Glycyrrhiza uralensis roots, and gypsum. MXSG is commonly used to treat respiratory tract diseases, including upper respiratory tract infection, acute bronchitis, pneumonia, chronic obstructive pulmonary disease, and bronchial asthma.10–12 The active components of MXSG are ephedrine, pseudoephedrine, methylephedrine, amygdalin, amygdalin prunasin, glycyrrhizic acid, liquiritin, glycyrrhizin, and CaSO4.10,13,14 MXSG is also an effective therapy for M. pneumoniae infection,15,16 although its mechanism of action is unknown. This study aimed to explore the mechanism of MXSG action in M. pneumoniae infection to support further pharmaceutical development of Maxing shigan decoction. A549 cell cultures have been used to explore the effects of M. pneumonia infection on IL-8 release and pyroptosis in human lung epithelial cells, so the same cell line was used in the current study.
Materials and Methods
Ethics Statement
Ethical approval for the study was provided by the independent Ethics Committee of Pudong New Area People’s Hospital. Written informed consent was obtained from the guardians of all participants prior to enrollment. This study was conducted in accordance with the Declaration of Helsinki.
Serum Samples and Isolation of Peripheral Blood Mononuclear Cells
We enrolled 30 children (mean age 31 months, 17 male, 13 female) with M. pneumoniae infection admitted to the Department of Pediatrics, Pudong New Area People’s Hospital (Shanghai, China). Nasopharyngeal aspirate samples were collected for M. pneumonia detection. Patients were excluded if they had chronic lung disease, immunodeficiency, congenital heart disease, or bronchopulmonary dysplasia. Fasting blood samples were obtained before treatment. Fasting blood samples were collected from 30 age-matched healthy donors as controls. Serum samples were obtained by centrifugation and stored at–80°C. Peripheral blood mononuclear cells (PBMCs) were isolated by centrifugation at 300 × g for 30 min with lymphocyte isolation reagent.
Real-Time Quantitative PCR
Total RNA was extracted using Trizol reagent according to the manufacturer’s protocol (Invitrogen Life Technologies), and 1 μg of each sample was reverse transcribed using the Revert AidTM First Strand cDNA Synthesis Kit (#K1622; Thermo) with random primers. NLRP3 transcript expression was measured by qRT-PCR with SYBR Green PCR Master Mix (#K0223; Thermo) according to manufacturer instructions with GAPDH as an internal control on an ABI 7300 thermocycler (Applied Biosystems, Foster City, CA, USA). The 2−ΔΔCt relative quantification method was applied. Primer sequences were NLRP3 (5′-AGCCTCAACAAACGCTACAC-3′ and 5′-CATCATCGGGGTCAAACAG-3′), GAPDH (5′-AATCCCATCACCATCTTC-3′ and 5′-AGGCTGTTGTCATACTTC-3′).
Measurement of IL-1β, IL-18, and TNF-α
Serum and supernatant IL-1β, IL-18, and TNF-α content was measured by enzyme-linked immunosorbent assay (ELISA; Jiancheng, Nanjing, China).
Mycoplasma Culture
M. pneumoniae strain 29,342 (American Type Culture Collection, Rockville, MD) was grown in broth containing mycoplasma broth base CM403 and selective supplement G SR59 (Oxoid, Hampshire, UK), 0.5% glucose, and 0.002% phenol red at 37°C in the presence of 5% CO2 for 3 days. Viable M. pneumoniae was quantified by colony counting in 10-fold serial dilutions on mycoplasma agar.
Cell Culture and Infection
A549 and 293T cells obtained from the Shanghai Cell Bank, Chinese Academy of Sciences (Shanghai, China) were grown in Dulbecco’s modified Eagle’s medium (DMEM, Hyclone) supplemented with 10% fetal bovine serum (Gibco, Grand Island, NY, USA) at 37°C in the presence of humidified 5% CO2.
Infection was induced by adding 1 mL M. pneumoniae to 10 mL cell medium (approximately 1 × 107 CFU/106 A549 cells) unless otherwise specified and incubated for 12 h, 24 h, or 48 h. Negative controls were A549 cell cultures with 1 mL bacteria-free mycoplasma broth.
Preparation of Freeze-Dried MXSG Powder
The herbal medicines (whole Ephedra sinica, Prunus armeniaca seeds, Glycyrrhiza uralensis roots, and gypsum) were powdered and soaked in purified water for 30 min for 1.5 h. The extracts were, then decocted filtered and concentrated at 50°C using a rotary vacuum evaporator. The water extracts were then freeze-dried.
Lentiviral Overexpression of NLRP3
The full-length human NLRP3 was cloned into pLVX-puro (Clontech, Palo Alto, CA, USA), then pLVX-NLRP3 (oeNLRP3) and pLVX-puro (vector control) were propagated in 293T cells along with packaging plasmids psPAX2 and pMD2.G.
Pyroptosis Measurement by PI Staining
The treated cells were collected and resuspended in staining buffer, then incubated with FLICA 660-YVAD-FMK (FLICA 660 Caspase-1 Assay, Catalog number: 9122) and propidium iodide (PI, Thermo Fisher) for 25 min at 4°C. The ratios of activates caspase-1 and PI-positive cells were analyzed by flow cytometry (BD Biosciences).
Protein Extraction and Western Blotting
A549 cells were lysed in ice-cold RIPA lysis buffer (Beyotime Biotechnology), and total protein was quantified by BCA protein assay (Thermo Fisher) according to standard protocols. Protein extracts (25 μg) were resolved by 10% or 12% SDS polyacrylamide gel electrophoresis and electroblotted onto nitrocellulose membranes (Millipore). After blocking with 5% nonfat milk, the membranes were probed with primary antibodies of NLRP3 (Abcam, cat. Ab214185, Shanghai, China), IL-1β (Abcam, cat. Ab2105, Shanghai, China), pro-IL-1β (NOVUS, cat. NB600-633, Saint Louis, MO, USA), Caspase 1 (Abcam, cat. Ab74279, Shanghai, China), pro-caspase 1 (Abcam, cat. Ab179515, Shanghai, China), GSDMD-M (NOVUS, cat. NBP2-80427, Saint Louis, MO, USA), and GAPDH (Proteintech, cat. 60004-1-1G, Shanghai, China), followed by incubation with horseradish peroxidase-conjugated secondary antibody (1:2000, Beyotime Biotechnology, Haimen, China). Results were visualized using ECL chromogenic substrate (Bio-Rad Laboratories).
Statistical Analysis
Each experiment was conducted at least three times. Statistical analysis between more than two groups and between two groups was performed using ANOVA and Student’s t-test, respectively. A P-value <0.05 was considered significant. Data are presented as means ± standard deviations.
Results
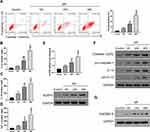
M. pneumoniae Infection Increased Serum Levels of IL-1β, IL-18, and TNF-α, and mRNA Expression of NLRP3 in PBMCs
Serum levels of IL-1, IL-18, and TNF-α, and mRNA expression of NLRP3 in PBMCs were significantly higher in patients than in healthy controls (Figure 1, P < 0.01).
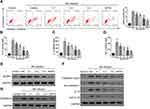
M. pneumoniae Infection Promoted Pyroptosis in A549 Cells
Flow cytometry showed that M. pneumoniae infection-induced incubation-time-dependent pyroptosis in A549 cells and induced expression of pro-inflammatory cytokines IL-1β, IL-18, and TNF-α (Figure 2A–D; P < 0.05). M. pneumoniae infection also up-regulated expression of NLRP3, pro-IL-1β, Caspase-1, pro-Caspase-1, and GSDMD-N (Figure 2E–G; P < 0.05). All measured indices showed a significant difference by 24 h infection (P < 0.05), which was used as the standard infection duration for all subsequent studies.
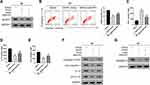
Maxing Shigan Decoction Mitigated M. pneumoniae Infection-Induced Pyroptosis in A549 Cells
Compared to the control, 24 h M. pneumoniae infection (vehicle group) induced pyroptosis in A549 cells (Figure 3A, P < 0.01). Treatment with NLRP3 inhibitor INF39 (P < 0.01) or MXSG (0.1, P < 0.05; 0.2 and 0.4 mg/mL, P < 0.01) provided dose-dependent suppression of pyroptosis induced by M. pneumoniae infection versus the vehicle control. MXSG (0.1, 0.2, and 0.4 mg/mL, P < 0.05) also suppressed expression of pro-inflammatory cytokines IL-1β, IL-18, and TNF-α and down-regulated NLRP3, pro-IL-1β, Caspase-1, Pro-Caspase-1, and GSDMD-N in M. pneumoniae-infected A549 cells in a dose-dependent manner (P < 0.05; Figure 3B–G). MXSG significantly reduced all measured infection-induced indices at 0.2 mg/mL, which was selected as the standard dosage for subsequent experiments.
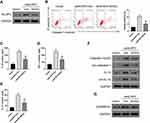
Maxing Shigan Decoction Mitigated M. pneumoniae Infection-Induced Pyroptosis in A549 Cells by Downregulating NLRP3
We then explored whether NLRP3 mediated the anti-pyroptosis effects of MXSG in A549 cells. A549 cells overexpressing NLRP3 were exposed to M. pneumonia and 0.2 mg/mL MXSG. The results showed that NLRP3 overexpression significantly suppressed the inhibitory effects of MXSG on NLRP3 expression, pyroptosis, pro-inflammatory cytokines IL-18, TNF-α, and IL-1β, as well as pro-IL-1β, Caspase-1, pro-Caspase-1, and GSDMD-N (Figure 4).
Maxing Shigan Decoction Mitigated NLRP3 Overexpression-Induced Pyroptosis in A549 Cells
NLRP3 overexpressing A549 cells were used to determine the anti-pyroptotic effects of MXSG (Figure 5A). NLRP3 overexpression-induced significant pyroptosis in A549 cells compared to the vector group (P < 0.01); however, MXSG counteracted this effect (Figure 5B). Our results also indicated that MXSG (P < 0.05) suppressed the induction of pro-inflammatory cytokines such as IL-18, TNF-α, and IL-1β induced by NLRP3 overexpression (Figure 5C–F). MXSG also down-regulated NLRP3 (Figure 5A), pro-IL-1β, Caspase-1, pro-Caspase-1 (Figure 5F), and GSDMD-N (Figure 5G) in NLRP3-overexpressing A549 (P < 0.05).
Discussion
M. pneumoniae is one of the most common causes of community-acquired pneumonia and is closely related to the development of asthma. It is widely believed that an outbreak of M. pneumoniae would bring about a public health emergency.1,17 We explored the curative effect and mechanisms of the classic TCM formula Maxing shigan decoction on M. pneumoniae infection.
Pyroptosis, closely related to the inflammatory response, is a recently identified pathway of programmed cell death.18–20 Unlike apoptosis, which is physiological suicide, pyroptosis is pathological suicide commonly caused by a viral or bacterial infection. Pyroptosis results in rupture of the cellular membrane and leakage of cytoplasm, triggering an inflammatory response.21,22 Pyroptosis induces pro-inflammatory cytokines such as IL-1β, TNF-α, and IL-18, exacerbating the inflammatory response.23,24 NLRP3 has been associated with the development of pyroptosis.25,26 Here, we found increased serum levels of IL-1β, IL-18, TNF-α, and mRNA expression of NLRP3 in PBMCs of children with M. pneumoniae infection compared to healthy children. In the future, we will determine whether pyroptosis occurs in PBMCs of children with M. pneumonia infection. We found that M. pneumoniae infection induces time-dependent pyroptosis in A549 lung cancer cells as well as serum IL-1β, TNF-α, and IL-18, and NLRP3 expression. The NLRP3 inflammasome is an important member of the NLR family and comprises NLRP3, ASC, and pro-caspase 1.27 Pyroptosis is mediated by activation of the NLRP3 inflammasome and Caspase-1, as well as Gasdermin D (GSDMD-N).28–30 Importantly, our results suggest that M. pneumoniae infection causes time-dependent upregulation of NLRP3, Caspase-1, pro-caspase-1, and GSDMD-N in A549 cells. We thus speculated that M. pneumoniae induces pyroptosis in these cultures via regulation of the NLRP3 inflammasome. Thus, we determined the effects of the NLRP3 inhibitor INF39 on M. pneumoniae infection-induced pyroptosis in A549 cells. Indeed, INF39 abrogated infection-induced pyroptosis and NLRP3 overexpression induced pyroptosis in A549 cells.
TCM provides a broad spectrum of therapies derived from natural active compounds that mitigate disease.31,32 Indeed, we found that the effects of Maxing shigan decoction on M. pneumoniae infection-induced pyroptosis were similar to that of INF39, inhibiting the production of pro-inflammatory cytokines. This effect was abrogated by NLRP3 overexpression. MXSG treatment also down-regulated the expression of NLRP3, Pro-IL-1β, Caspase-1, pro-caspase-1, and GSDMD-N in M. pneumoniae-infected and NLRP3-overexpressing A549 cells.
In conclusion, our findings suggest Maxing shigan decoction mitigates M. pneumoniae-induced pyroptosis in A549 cells via the NLRP3 inflammasome, supporting the further pharmaceutical development of Maxing shigan decoction.
Funding
This work was supported by the Important Weak Subject Construction Project of Pudong Health and Family Planning Commission of Shanghai (Grant No. PWZbr2017-23).
Disclosure
The authors declare that they have no competing interests.
References
1. Waites KB, Xiao L, Liu Y, Balish MF, Atkinson TP. Mycoplasma pneumoniae from the respiratory tract and beyond. Clin Microbiol Rev. 2017;30:747–809.
2. He J, Liu M, Ye Z, et al. Insights into the pathogenesis of Mycoplasma pneumoniae (review). Mol Med Rep. 2016;14(5):4030–4036. doi:10.3892/mmr.2016.5765
3. Lee H, Yun KW, Lee HJ, Choi EH. Antimicrobial therapy of macrolide-resistant Mycoplasma pneumoniae pneumonia in children. Expert Rev Anti Infect Ther. 2018;16:23–34.
4. Tanaka T, Oishi T, Miyata I, et al. Macrolide-resistant Mycoplasma pneumoniae infection, Japan, 2008–2015. Emerg Infect Dis. 2017;23:1703–1706.
5. Wang BJ, Zhang H. Research progress of macrolide-resistant Mycoplasma pneumonia. Lab Med. 2020;35:1070–1074.
6. Long Y, Yang Q, Xiang Y, et al. Nose to brain drug delivery - a promising strategy for active components from herbal medicine for treating cerebral ischemia reperfusion. Pharmacol Res. 2020;159:104795.
7. Ren JL, Zhang AH, Wang XJ. Traditional Chinese medicine for COVID-19 treatment. Pharmacol Res. 2020;155:104743.
8. Sugiyama M, Saeki A, Hasebe A, et al. Activation of inflammasomes in dendritic cells and macrophages by Mycoplasma salivarium. Mol Oral Microbiol. 2016;31(3):259–269.
9. Susan L, Fink L, Brad T. Apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect Immun. 2005;73(4):1907–1916.
10. Wang WG, Shi XG, Zhai LX, Wang PJ, Peng XF, Xu YY. Influence of compatibility on content of berberine in Maxingganshi decoction. J Chin Med Mater. 2007;30:602–605.
11. Zuo H, Qian WD. Effect of maxingganshi decoction on the expression of STAT4 and STAT6 in lung tissues of rats with chronic obstructive pulmonary disease. J Hainan Med Univ. 2019;25:892–900.
12. Jiang S, Shi S, Shan WT, Ju AX, Li QH. Study on active compounds from Maxingganshi decoction for treatment of novel coronavirus pneumonia (COVID-19) based on network pharmacology and molecular docking method. Chin Pharmacol Bull. 2020;36:1309–1316.
13. Huang L, Ynag H, Wei Y, Fu Y, Xu G. Dose-response relationship of main components of Ma Xing Shi Gan decoction with relaxing tracheal smooth muscle. Tradit Chin Drug Res Clin Pharmacol. 2015;26:468–471.
14. He GM, Luo WA, Cheng XR, Wei M. UPLC fingerprint chromatography of Maxingshigan decoction. Chin Tradit Pat Med. 2020;42:2792–2795.
15. Deng MH. Treating refractory Mycoplasma pneumonia pneumonia with Maxin Shigan decoction. Clin J Chin Med. 2015;7:90–91.
16. Li GL, Zheng TT, Li LJ. Clinical observation and analysis of Maxing Shigan decoction combined with Azithromycin in the treatment of pediatric Mycoplasma pneumoniae pneumonia. Chin Arch Tradit Chin Med. 2020.
17. Atkinson TP, Balish MF, Waites KB. Epidemiology, clinical manifestations, pathogenesis and laboratory detection of Mycoplasma pneumoniae infections. FEMS Microbiol Rev. 2008;32(6):956–973. doi:10.1111/j.1574-6976.2008.00129.x
18. Xia X, Wang X, Cheng Z, et al. The role of pyroptosis in cancer: pro-cancer or pro-“host”? Cell Death Dis. 2019;10(9):650. doi:10.1038/s41419-019-1883-8
19. Frank D, Vince JE. Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ. 2019;26(1):99–114. doi:10.1038/s41418-018-0212-6
20. Tao HH, Tao XR. Pyroptosis, a novel form of programmed cellular necrosis. J Path Biol. 2017;12:1219–1221.
21. Lee S, Hirohama M, Noguchi M, Nagata K, Kawaguchi A. Influenza A virus infection triggers pyroptosis and apoptosis of respiratory epithelial cells through the Type I interferon signaling pathway in a mutually exclusive manner. J Virol. 2019;26(14):e00396–18. doi:10.1128/JVI.00396-18
22. Taabazuing CY, Okondo MC, Bachovchin DA. Pyroptosis and apoptosis pathways engage in bidirectional crosstalk in monocytes and macrophages. Cell Chem Biol. 2017;24:507–514.
23. Hong L, Cha YJ, Qiao SG, An JZ. Study on protective mechanism of resveratrol against pyroptosis induced by lipopolysaccharide in human lung epithelial cells. Anti Infect Pharm. 2020;17:160–164.
24. Zhang FJ, Ma P. Related research progress of NLRP3 inflammatory corpuscle and pyroptosis. World Latest Med Inform. 2017;17:33–34.
25. Han R, Wu D, Deng S, Liu T, Zhang T, Xu Y. NLRP3 inflammasome induces pyroptosis in lung tissues of radiation-induced lung injury in mice. Clin J Cell Mol Immunol. 2017;33:1206–1211.
26. Segovia JA, Chang TH, Winter VT, et al. NLRP3 is a critical regulator of inflammation and innate immune cell response during Mycoplasma pneumoniae infection. Infect Immun. 2018;86:e00548–17.
27. Zahid A, Li B, Kombe AJK, Jin T, Tao J. Pharmacological inhibitors of the NLRP3 inflammasome. Front Immunol. 2019;10:2538.
28. Miao EA, Rajan JV, Aderem A. Caspase-1-induced pyroptotic cell death. Immunol Rev. 2011;243:206–214.
29. Shi J, Gao W, Shao F. Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem Sci. 2017;42:245–254.
30. Qiu Z, Lei S, Zhao B, et al. NLRP3 inflammasome activation-mediated pyroptosis aggravates myocardial ischemia/reperfusion injury in diabetic rats. Oxid Med Cell Longev. 2017;2017:9743280.
31. Zhang Q, Liu J, Li R, et al. A network pharmacology approach to investigate the anticancer mechanism and potential active ingredients of Rheum palmatum L. against lung cancer via induction of apoptosis. Front Pharmacol. 2020;11:528308.
32. Sun JH, Sun F, Yan B, Li JY, Xin L. Data mining and systematic pharmacology to reveal the mechanisms of traditional Chinese medicine in Mycoplasma pneumoniae pneumonia treatment. Biomed Pharmacother. 2020;125:109900.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.