Back to Archived Journals » Reports in Medical Imaging » Volume 2
Mangafodipir trisodium: review of its use as an injectable contrast medium for magnetic resonance imaging
Authors Regge D, Cirillo S, Macera, Galatola G
Published 10 July 2009 Volume 2009:2 Pages 55—68
DOI https://doi.org/10.2147/RMI.S4472
Review by Single anonymous peer review
Peer reviewer comments 3
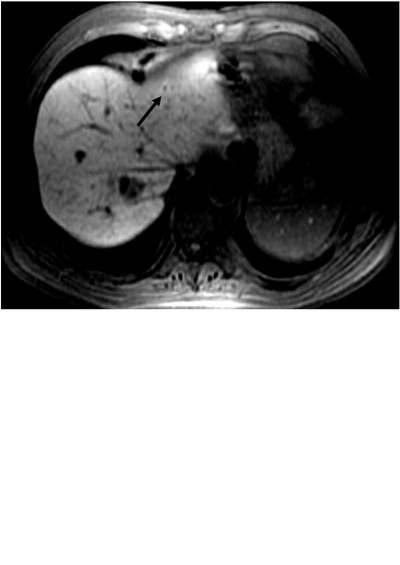
Daniele Regge,1 Stefano Cirillo,1 Annalisa Macera,1 Giovanni Galatola2
1Radiology Unit, 2Gastroenterology Unit, Institute for Cancer Research and Treatment, Candiolo, Turin, Italy
Abstract: Mangafodipir trisodium is a hepatobiliary contrast agent, taken up by the hepatocytes and largely excreted via the bile ducts. The agent increases the signal intensity of the normal liver, and to a lesser extent of the pancreas, adrenal glands, kidneys and myocardium, on T1-weighted imaging. The increase of the signal intensity on the T1 images allows better visualization of focal lesions, especially of those that are of non-hepatocitary origin such as metastases. For this reason the most important indication for the use of mangafodipir trisodium is in detecting liver metastases, especially when the information may influence therapeutic planning, which in many cases is surgical resection. New data show that this liver-specific contrast agent is accurate in detecting small lesions and in assessing the liver status following neoadjuvant chemotherapy, where other imaging techniques, such as CT and PET, fail. Other lesser indications for studies with mangafodipir trisodium are: in characterizing liver and pancreatic lesions; in identifying biliary leakage following bile duct and/or liver surgery; and possibly in the future in the assessment of the extent of myocardial damage.
Keywords: mangafodipir trisodium, magnetic resonance imaging, contrast agent
 © 2009 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2009 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
