Back to Journals » Clinical Ophthalmology » Volume 14
Management Strategies of Ocular Chemical Burns: Current Perspectives
Authors Soleimani M , Naderan M
Received 9 July 2020
Accepted for publication 20 August 2020
Published 15 September 2020 Volume 2020:14 Pages 2687—2699
DOI https://doi.org/10.2147/OPTH.S235873
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Mohammad Soleimani, Morteza Naderan
Ocular Trauma and Emergency Department, Farabi Eye Hospital, Tehran University of Medical Sciences, Tehran, Iran
Correspondence: Mohammad Soleimani; Morteza Naderan
Ocular Trauma and Emergency Department, Farabi Eye Hospital, Tehran University of Medical Sciences, Tehran 1336616351, Iran
Tel +98 912 1096496
Email [email protected]; [email protected]
Abstract: Ocular chemical burns are absolute ophthalmic emergencies and require immediate management to minimize devastating sequelae. Management of alkali and acid burns is started at the scene of the accident by copious irrigation. Treatment is directed at improving epithelial integrity and stromal stability, reduction of undue inflammation, and prevention or timely management of complications. To ascertain the best possible outcome, numerous biological medications and surgical interventions have been merged into conventional therapeutic regimens. These include autologous and umbilical cord serum preparations, platelet-rich plasma, amniotic membrane transplantation, limbal stem-cell transplantation, and anti-angiogenic agents.
Keywords: ocular chemical burn, amniotic membrane transplantation, autologous serum, limbal stem cell transplantation
Introduction
It is inevitable for an ophthalmologist to be consulted for an acute chemical burn. Many of such cases are mild and may be managed easily without remarkable sequelae. On the other hand, some chemical burns are so severe that they require frequent ophthalmic examinations and surgical operations for the management of structural, functional, and cosmetic complications.1 Despite what seems appropriate and timely management, further damage may ensue and result in severe loss of vision.
Epidemiology
From an epidemiological point of view, ocular chemical burns comprise up to 22% of all ocular injuries.2,3 Whether ocular insult occurs inadvertently (for instance, following unnoticed exposure or accidental contact at home or workplace) or as a result of a purposeful crime,4–6 it is more common in populations with low socioeconomic status.2 Large scale studies have demonstrated that the incidence of chemical burn is higher in men, though women are affected at an earlier age.2,5 A noticeable finding is that children of 1 to 2 years old are afflicted twice as much as adults.2
In general, two-thirds of chemical burns result from an alkali, and the remainder from acids and alcohols.7 Sulfuric acid is the most common agent responsible for acid burns. This is most commonly found in industrial cleansers and automobile batteries. Ammonia is the most common agent leading to alkaline burns and is commonly found in various industrial and household fertilizers and refrigerants.5
Pathophysiology
Any liquid or solid material with alkaline or acidic ingredients may cause an ocular burn. An alkali is especially notorious for severe damage to ocular tissues.5,7,8 The difference in the ocular effects between acids and alkali resides in their mechanism of action. Acids and alkali cause coagulative and liquefactive necroses respectively. Tissue surface proteins are sacrificed to neutralize acid pH and the connective tissue collagens are shrunk so that the resulting coagulum prevents further penetration of the insulting agent into deeper ocular structures. In contrast, alkaline agents contain hydroxyl moieties that saponify the fatty acids enclosed in the superficial cell membranes. Once the function of the cell membrane is disturbed, cell death ensues and the insulting agent more efficiently reaches the underlying connective tissue where the matrix proteoglycans are readily hydrolyzed leaving collagen fibrils especially susceptible to enzymatic degradation. These enzymes are produced by damaged or regenerating epithelial cells and immune cells which would have invaded the tissue immediately after exposure.3
It is noteworthy, however, that acids are equally as devastating as alkalis in severe burns. The chemical structure of the material is not the only determinant of the severity of the damage. Chemicals are usually more active at a higher temperature.9 Liquids are more easily irrigated and expelled from the eye, in contrast to solid particles that may become entrapped in the conjunctival papillae and steadily resolve in the tear meniscus, thus leading to prolonged exposure and irritation of the ocular tissues.9 Forceful impact may also add to the influence of chemical agents.10 Finally, the higher the concentration and the more prolonged the exposure, the more severe the damage.11
When the chemical agent reaches the anterior chamber, it circulates through the aqueous and reaches the angle structure. In trabecular meshwork (TM), the collagen fibrils are denatured and shrunken so that the outflow tract is obstructed and the IOP is elevated acutely. The damaged collagen in the TM may leave scar tissue despite treatment, which is the main reason for glaucoma in the long-term.10
Clinical Course: Grading and Staging
Efforts have been made to propose universal classifications of clinical findings that can be used reliably to predict outcomes. Grading refers to the severity of the damage and should be documented at the very first ophthalmic examination. The grading systems unify the corneal, limbal, and conjunctival changes to prognosticate the clinical condition and are very helpful to acquire a general idea of the interventions that may be recommended in later stages.
The first widely accepted grading system was published by Roper and Hall in 196512 (Table 1). This grading system was based on the amount of corneal haziness and the proportion of ischemic perilimbal conjunctiva. According to the 4-step Roper-Hall classification, the prognosis of all patients with >50% limbal ischemia was poor.
 |
Table 1 Roper-Hall Classification for the Severity of Ocular Surface Burns12 |
Later, Dua et al presented a new classification which, although in essence was based on the Ropper-Hall classification, had a number of advantages (Table 2).13 In the Dua’s classification, corneal haziness was abandoned as an important prognostic factor; instead, the extent of limbal involvement (in clock hours) and the percentage of conjunctival involvement were incorporated. According to Dua’s classification, limbal ischemia is not the sole determinant of limbal stem cell deficiency and total epithelium loss at the limbus may occur in the presence of apparently insignificant ischemia with poor outcomes. Thus, they proposed that frequent observation of the stained limbus in order to document the extent of the epithelial defects is better correlated with outcomes rather than the extent of limbal ischemia (blanching). In this way, grade IV in Roper-Hall classification was revised as grades IV, V, and VI in Dua’s classification with different prognoses. Dua’s classification also provides an analog scale that is more flexible and allows for combining features of the different grades which is more compatible with real clinical situations.13
 |
Table 2 Dua Classification for the Severity of Ocular Surface Burns13 |
Chronologically, an ocular chemical burn can be classified into four stages. This staging demonstrates the potential healing responses of the ocular surface tissue to a chemical insult. Originally described by Culley in 1990 in animal models, this staging is well known in clinical practice.14 Management strategies should be based not only on the severity of the burn but also on the stage at which the condition is being evaluated. These stages include the following:
- immediate (day 0). This is right after the exposure has occurred.
- Acute (days 1–7). This is the 1st week following the immediate phase. In the acute stage, the limbal stem cell remnants try to repopulate the epithelial defects over denuded corneal stroma. This stage is crucial because tear-soluble proteolytic enzymes and immune-cell-derived enzymes may be conveyed to the stroma through epithelial defects and result in stromal thinning and perforation in later stages.
- Early reparative (days 8–21). The 2nd and 3rd weeks following exposure represent a transition window in which chronic inflammation supersedes acute inflammation, hence, leading to stromal hyperplasia and scar formation. This is the most common stage at which corneal ulcers or thinning are noted.
- Late reparative (after day 21). Except for those who had good prognostic features and who had managed appropriately, this stage represents the most severe complications of ocular chemical burns. Severe dry eye, poor visual acuity due to corneal scarring and neovascularization, increased IOP and glaucoma, restricted ocular motility due to symblepharon, and lid abnormalities including entropion, ectropion, trichiasis, and incomplete closure may ensue and lead to a vicious cycle.5
Management Strategies
Appropriate management of ocular chemical burn requires caring for epithelial defects, inflammatory response, and complications through steps that promote epithelialization, reduce inflammation, and prevent complications.8,10 This will break a vicious cycle that each of these factors, if left untreated, may bring about, and thus, may adversely affect outcomes (Figure 1).
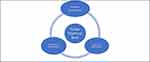 |
Figure 1 Management strategies of ocular chemical burns have three facets: promotion of epithelialization, reduction of inflammation, and prevention of complications. |
An ophthalmologist taking care of ocular chemical burns must be familiar with both medical and surgical management of complications. While lower grades may demonstrate continued healing with an excellent prognosis, higher grades may deteriorate rapidly and require surgical interventions at earlier stages. Table 3 provides stage-specific treatment options in ocular chemical burns.
 |
Table 3 Summary of Interventions in Different Stages of Ocular Chemical Burns |
Non-Operative Management
Management of Immediate Stage
The very first step after exposure of the ocular surface to chemical agents is to institute continuous irrigation.10,15 Prompt irrigation takes priority over looking for the chemical composition of the offending agent or waiting for specific fluids.5,8 Although some authors have proposed a certain duration or amount of irrigation, it is generally accepted that irrigation should be continued until the ocular surface pH has been neutralized.7,8,10 Frequent application of litmus paper should be commenced until stable neutralized pH results are ensured.16
There are also arguments against using hypo-osmolar fluids for irrigation because they may increase the epithelial permeability and result in greater diffusion of the chemicals.17 However, this has been debated in alkali burns, and currently, delaying irrigation for a proper solution to become available is not recommended.18 This is especially the case in the scene of an accident, where even tap water should be used until the patient is transferred to a medical facility.11,19–21
In experimental alkali burns, where intracameral pH is relatively rapidly affected, hyperosmolar amphoteric solutions are more efficacious than iso-osmolar solutions in normalizing intracameral pH.18,22 In a 30-year longitudinal study published by Wiesner et al, it was shown that pre-hospital rinsing of the ocular surface with Previn (an amphoteric polyvalent agent) followed by secondary rinsing in the hospital setting reduced the severity of chemical burns in comparison to all solutions.23 However, using tap water in the pre-hospital setting was as efficacious as Previn.
The other important step is removing any chemical particles from the ocular surface (especially fornices) using cotton tip swab or forceps; sometimes it may be necessary to double evert the upper eyelid even in the operation room.
Management of Epithelial Defects
Several topical and systemic medications have been proposed to promote re-epithelialization. Intact epithelium plays an important role in preserving stromal stability because it can effectively inhibit digestive enzymes from reaching underlying stroma. It is also critical in smoothing the ocular surface and expediting visual rehabilitation. Here, we summarize the measures that are used to enhance epithelial growth and stromal stability.
Artificial Tears
Frequent administration of preservative-free artificial tears is currently incorporated into many treatment protocols so that it is virtually considered routine clinical practice by most authorities.8 It will reduce the chance of persistent epithelial defects and recurrent epithelial erosions, both of which are disabling complications.3
Fibronectin and Laminin
Fibronectin and laminin are essential components of connective tissue. Experimental animal studies have shown that these factors naturally increase in the acute stage and correlate with re-epithelialization.24–26 Nonetheless, their effect in human chemical burns is still undiscovered.
Epidermal Growth Factor
As its name suggests, this is a growth factor that promotes epithelial and keratocyte proliferation. In previous animal models, it accelerated wound healing and epithelial growth but the evidence in human patients is lacking.27,28
Retinoic Acid
Retinoic acid (vitamin A) is essential to normal epithelial growth and development. However, both hyper- and hypovitaminosis A can be deleterious for epithelial tissues. In experimental animal studies, the administration of retinol palmitate eyedrops (1500 IU/mL) has been shown to improve wound healing and impression cytology findings.29,30 The possible explanation for the effects of retinoic acid is that it may provoke dormant limbal stem cells and conjunctival stem cells to proliferate and differentiate into corneal epithelial cells by affecting intracellular signaling pathways.31 It may also prevent corneal pannus formation by inhibiting vascular endothelial growth factor type A (VEGF-A) and stimulating thrombospondin-2.29 However, the evidence for the efficacy of retinoic acid in human subjects is largely anecdotal and well-controlled human studies are lacking.
Hyaluronic Acid
The administration of topical 1% and 2% hyaluronic acid in experimental animal studies has been associated with improved corneal epithelialization.32–34 It provokes the process of wound healing, possibly through induction of formation of hemidesmosomes.35 However, the evidence for benefit in the human chemical burn is sparse.
Tetracycline Medications
These antibiotics exert matrix metalloproteinase inhibition (and thus prevent enzymatic proteolysis of the corneal stroma) via mechanisms independent of their bactericidal action.36,37 Both systemic and topical preparations have been effective in animal studies.36 Due to its irreversible action, doxycycline has been postulated to be more effective than other agents of this class.38
N-Acetyl-Cysteine (NAC)
NAC is a synthetic inhibitor of matrix metalloproteinases. It also inhibits neutrophil migration to the site of injury, which is a potential source of many degrading enzymes.39 However, the clinical role of these agents is not known.
Ascorbate
The observation that aqueous ascorbate levels fall in response to alkali burns has led some investigators to propose that early vitamin C supplements may improve collagen synthesis, prevent the development of corneal ulcers, and accelerate healing.40–43 Although the evidence was mainly derived from animal studies, it appears that the efficiency of topical ascorbate outweighs that of systemic supplementation.44 Some authors have recommended the administration of both preparations simultaneously (oral ascorbate 2 g in 4 divided doses and topical 10% eyedrops every 2 hours).
Biological Medications
These are concentrates of growth factors that are normally present in patients’ autologous serum or umbilical cord serum.45,46 There is a good body of evidence in humans that these preparations are superior to conventional treatment.47–52 Serum is a rich source of various growth factors, cytokines, and vitamins, and has been shown to accelerate the process of wound healing and re-epithelialization and decelerate corneal vascularization and limbal damage.47,51,53,54 It may also provide pain relief and cause inflammation to subside through local immune modulation.49
The efficacy of umbilical cord serum is proposed to be superior to the pure autologous serum, though it has more accessibility issues. In a randomized clinical trial with moderate to severe chemical burns, the efficacy of 20% umbilical cord serum was shown to be superior to 20% autologous serum or artificial tears in terms of acute symptom relief, healing of epithelial defects, corneal clarity, and vascularization but not symblepharon formation.51
The addition of platelets to serum creates platelet-rich plasma (PRP) which may be more efficient because platelets possess manufacturing properties and thus may provide continued production of growth factors.52,55,56 In a randomized clinical trial on moderate to severe chemical burns, Panda et al reported that autologous PRP eyedrops resulted in better corneal clarity and faster healing of the epithelial defects at three months, in comparison to conventional treatment. However, the sample size was not large enough and the study lacked comparison with pure autologous serum.52
Recently, amniotic membrane extract has been used in the treatment of ocular chemical burns with promising results.48,57 In a human study by Liang et al, a small series of patients with various severities of ocular chemical burns were prescribed frequent instillation of amniotic membrane extract for 6 weeks in addition to conventional therapy. The results were promising in terms of symptom relief, reduced inflammation, and improved visual acuity.48
Currently, it is recommended that all eyes with grades III to VI Dua’s classification receive at least one type of biological eyedrops every 2 hours for a month starting in the acute stage and continued with slow taper until the inflammation has completely resolved.8
Control of Inflammation
The institution of anti-inflammatory treatment should be considered as early as possible whenever there are no contraindications. Inflammatory transmitters are produced in the acute stage by the summoned immune cells, especially neutrophils, and regenerating epithelial cells. Inflammation presents two facets: it provides resources for activating repair mechanisms; on the other hand, these mechanisms may go uncontrolled and result in further damage. Hence, the balance between these two potentials of the same mechanism determines whether the healing process is efficiently accomplished. The anti-inflammatory options include the following.
Corticosteroids
The benefits of topical corticosteroids in the acute stage of ocular chemical burn are invaluable. It has been shown that steroids can prevent the reduction in the number of goblet cells, enhance the stability of the basement membrane and endothelial cells, reduce the migration of immune cells into the wound, and prevent degranulation of neutrophils.58–61 Conversely, prolonged treatment with corticosteroids has been associated with corneal and scleral sterile ulcers and melting because it interferes with collagen synthesis.62 Some investigators have postulated that co-administration of topical steroids and vitamin C preparations would prevent steroid-induced corneal ulceration.63 Most of the evidence regarding the efficacy of corticosteroids in ocular chemical burns was derived from animal models or retrospective human studies.
Frequent administration of topical steroids (prednisolone acetate 1% or Dexamethasone 0.1% every 2 hours) is recommended in the acute stage. It should be tapered in the early reparative stage in order to avoid complications.8 They may be continued thereafter, if the epithelial defects have been healed, but the inflammation is still a concern.
Progesterone-Derivatives
Progesterone inhibits collagenase activity, in addition to having anti-inflammatory characteristics. In comparison to corticosteroids, their prolonged use is less associated with inhibition of collagen production and wound healing.64,65 These properties make progesterone derivatives attractive, especially in the early and late reparative stages. Firm evidence in human subjects is lacking.
Citrate
Sodium citrate is a potent inhibitor of collagenase and leukocyte migration.66,67 Its mechanism of action is related to the chelation of calcium ions. Brodovsky et al reported that topical citrate administration was associated with better outcomes in patients with a guarded prognosis.40 In animal experiments topical 10% citrate was shown to be more effective than ascorbate and NAC.68 Currently, frequent administration of topical 10% citrate is recommended in the acute stage of chemical burns.8
NSAIDs
The effectiveness of topical or systemic nonsteroidal anti-inflammatory medications, whether as adjuvants to corticosteroids or as corticosteroid-sparing agents, needs to be explored. Animal studies have yielded promising results regarding prophylaxis against IOP rise.69,70
Prevention of Complications
Chemical burn injury may induce devastating complications at any stage of the disease. It is critical to specifically look for complications at each stage irrespective of their absence in prior stages.
Corneas with epithelial defects are especially vulnerable to a secondary infection. Therefore, prophylactic broad-spectrum topical antibiotics are recommended in the acute stage and thereafter until re-epithelialization has been completed. Any suspicious stromal infiltration should be examined for culture and sensitivity immediately and empirical treatment should not be postponed while waiting for lab results.
Measurement of intraocular pressure (IOP) is mandatory during the course of management. IOP may rise primarily due to chemical-induced disruption of the trabecular meshwork or contracture of the sclera, or it may rise secondarily due to aggressive corticosteroid treatment or surgical interventions.71–73 The higher the initial grading, the higher the chance of glaucoma.72 If elevated IOP is detected, initiation of topical anti-glaucoma medications will be prudent. However, the treatment of such cases may be challenging as many IOP-lowering agents may cause epithelial toxicity and delay epithelialization. Systemic IOP-lowering substitutes or surgical interventions may be beneficial for more severe and/or uncontrolled cases.
Severe intraocular inflammation may cause posterior synechia and secondary glaucoma. Cycloplegic medications may prevent or resolve synechia.
Pain may be severe in the acute stage and merits special consideration at all stages. Topical cycloplegia may remedy mild pain due to spasm of the ciliary body, but systemic medications may be necessary in more severe cases.
Surgical Management
Although surgical management is reserved for higher grades (generally grades III to VI Dua’s classification), it should not be considered longitudinal to medical management. Instead, at any stage of the chemical burn, surgical interventions may be recommended to accelerate the healing process and to reduce the burden of complications. The following section discusses various surgical options that are recommended in the current clinical practice.
Amniotic Membrane Transplantation (AMT)
The amniotic membrane is a fetal membrane that covers the amniotic fluid around the fetus. It consists of a single cuboidal epithelium overlying a basement membrane. The basement membrane contains collagen (types 4 and 7), laminin (types 1 and 5), and fibronectin which provide a bed for extension of the native corneal epithelial cells over the membrane.74 Beneath the basement membrane, the so-called stroma, is cellular connective tissue that supports the epithelium and produces multiple growth factors that are essential for the proliferation and maintenance of corneal stem cells while inhibiting the production of fibrotic tissue by local fibroblasts.75 Furthermore, it has been demonstrated that amniotic membrane possesses anti-angiogenic, anti-inflammatory, anti-protease, and anti-microbial characteristics which are postulated to result from local modulation of signaling pathways.75–78
Amniotic membrane may be transplanted as single or multiple layers and with the stromal side up or down. It may be sutured or attached by tissue adhesives onto the overlying ocular surface.8 It may be used as a permanent graft which is left indefinitely or as a transient support that is removed following stabilization of the ocular surface. It may cover the whole ocular surface, the fornices, and the lid margins or only cover the de-epithelialized area. Amniotic membrane may be attached to a polycarbonate ring (PROKERA [Bio-Tissue, Inc., Miami, USA]) and inserted over the ocular surface in the office under topical anesthesia.79 Amniotic membrane may be left uncovered or under a bandage contact lens for a long time.80
Many human studies have demonstrated the benefits of AMT. Patient satisfaction was increased due to the reduction of pain, photophobia, and epiphora.47,80,81 Symptomatic relief was more common after acid burns in comparison to alkali burns.
Although some authors have reported improvement of visual acuity especially in lower grades,80–82 randomized clinical trials have failed to prove such findings.47,83 Conversely, some studies reported poorer visual outcomes after AMT.84–86
Two randomized clinical studies have shown faster epithelialization of the ocular surface. Sharma et al compared AMT with conventional treatment in patients with mild to moderate chemical burns and found faster healing.47 Tamhane et al compared adjuvant AMT or umbilical cord serum and reported superiority over traditional treatment alone.86 Despite the improvement in epithelial reconstruction, the results are not predictable. It may take as long as a week or it may take several months to observe a smooth epithelialized ocular surface.8 The re-epithelialization is found to occur faster in lower grades and suture-less techniques.80,86,87
Symblepharon was found to be less common after AMT compared to conventional medical treatment. Nonetheless, the efficacy of AMT may be reduced in higher grades and with sutured techniques.83,87
The AMT is very popular in current clinical practice and many authors recommend it immediately in the acute stage of grades III to VI Dua’s classification.8 Despite all its benefits, AMT has been shown to fail in some studies.81,86,88,89 Persistent epithelial defect may remain and may require repeat AMT.79,81 It is also less predictable in grades IV to VI Dua’s classification. Finally, results of a systematic review published in 2012 by Clare et al demonstrated that AMT in the first week post-chemical burn did not have sufficient high-quality evidence.90
Tenonplasty
In cases with severe chemical burns where limbal vasculature is significantly compromised, tenoplasty or tenonplasty remains a logical approach. Originally described by Teping et al in 1989, this procedure includes sufficient debridement of necrotic tissues, followed by the advancement of the remaining healthy Tenon’s capsule adjacent to the limbus.91 This may be sutured alone or combined with AMT or may be augmented with tissue adhesives.92,93 The goal is to provide the ischemic limbus with healthy vascular connective tissue thus preventing anterior segment necrosis and/or corneoscleral ulceration and melting while tissue repair and wound healing are promoted. In a series of 6 patients with severe chemical burns (Dua’s classification V and VI), we performed selectively localized tenoplasty in the first week based on anterior segment fluorescein angiography (FA) findings. The repeat FA demonstrated that reperfusion had occurred in all patients.94
Debridement of Necrotic Tissue
Obviously, necrotic tissue is a source of attracting immune cells and augmenting tissue instability. Therefore, it is prudent to remove devitalized tissues.8
Limbal Stem-Cell Transplantation (LSCT)
Limbal stem cell deficiency (LSCD) is a disabling long-term complication of ocular chemical burn. It may be partial or total; center-involving or non-center-involving.73 The sequelae include conjunctivalization of the cornea, recurrent epithelial erosions, scars, persistent epithelial defects, ulcers, melting, and perforations.95
LSCT is an option when the corneal conjunctivalization is total or center-involving.96–98 As inflammation is detrimental to a successful transplant, it is wise to wait until the inflammation has been perfectly controlled. Correction of structural abnormalities such as symblepharon or cicatricial entropion and/or ectropion take priority over LSCT.99,100
In unilateral cases, the healthy eye is used to acquire conjunctival limbal autograft (CLAU) which brings about excellent outcomes in terms of re-epithelialization and visual acuity, as described originally by Pellegrini et al.101–103 If bilateral but asymmetric involvement is the case, cultivated limbal epithelial transplantation (CLET) is an option.104–106
When bilateral LSCD is severe and symmetric, limbal stem-cells can be harvested from allograft donors either as living-related conjunctival limbal allograft (lr-CLAL) or keratolimbal allograft (KLAL).107–111 The former is obtained from a first-degree relative which also provides healthy conjunctival tissue to replace the recipient’s defects, but the number of stem-cells obtained by this technique is relatively small. The latter is obtained from a cadaver with more technical ease and more stem-cells available.111,112 All allografts need systemic immunosuppression and a multidisciplinary medical team if rejection and side effects are to be prevented.113,114
Keratoplasty and Keratoprosthesis
Corneal transplantation is considered for those with visually significant stromal scars and perforations. Both penetrating keratoplasty (PK) and deep anterior lamellar keratoplasty (DALK) are acceptable but the latter is a better option due to the lesser chance of rejection.115 Keratoplasty may be performed alone or in combination with AMT. Keratoplasty should not be scheduled if LSCT is anticipated.116 It is advised to delay keratoplasty for at least three months after LSCT. This approach is proposed to increase graft survival in chemical burn patients.117 An exception is when a large perforation is encountered so that urgent tectonic graft is required to provide support. The fate of the graft in such cases is failure.8 Larger corneoscleral grafts are preferable because they may provide limbal stem-cells in addition to tectonic support.118,119
Keratoprosthesis is indicated when multiple keratoplasty procedures have failed and the chance of a new successful keratoplasty is extremely low.120,121 In patients with minimal tear production, Boston type 1 keratoprosthesis (B1-KPro) may be tolerated especially in combination with LSCT and other reconstructive procedures.122 Finally, in extremely dry eyes or severe surface keratinization, or select cases with non-functional lids, Boston type 2 (B2-KPro), or osteo-odonto-keratoprosthesis (OOKP) may be recommended as a salvage treatment.123,124
Miscellaneous Therapies
Oxygen Therapy
Administration of hyperbaric oxygen has yielded contradicting results in animal models of ocular chemical burn.125,126 A human study reported that 100% oxygen administered for 1 hour every 12 hours resulted in faster epithelialization and resolution of limbal ischemia in comparison to the control group.127
Tissue Adhesives
If a small corneal perforation (<3 mm) occurs at any stage of a chemical burn, fibrin glue and cyanoacrylate adhesive may be applied to close the site of perforation. The fibrin glue is preferred as it induces less inflammation and vascularization.128–130 If the cornea has not been perforated but the thinning is severe, cyanoacrylate adhesive with overlying bandage contact lens may provide support and prevent melting.8 It may be left in place until it dislodges spontaneously, or it may be peeled off after enough neovascularization has ensured corneal stability. Tissue adhesives have been used with AMT successfully.131
Anti-Angiogenic Therapy
Inflammation results in elevation of numerous angiogenic factors that end in corneal neovascularization unless they are subsided.132 Angiogenesis not only impairs visual acuity but also increases the risk of corneal graft rejections due to disturbance of the immune privilege mechanisms.132 Multiple agents including anti-vascular endothelial growth factors (ANTI-VEGF) have been tried in animal and human studies.62,133–137 However, their clinical correlation needs further studies. However, in severe burns, we paradoxically like to enhance angiogenesis for fear of perforation in ischemic areas.
Conclusion
An ocular chemical burn is an ophthalmic emergency. Treatment should be started immediately at the scene of the accident with continued irrigation. Proper management requires the promotion of epithelialization, suppression of inflammation, and prevention of complications. Artificial tears, topical steroid therapy, ascorbate, and biological medications are the mainstay of treatment. AMT at an early stage reduces inflammation, supports stroma, and provides a bed for expansion of epithelial tissue. LSCT is a valuable option when corneal conjunctivalization is extensive. Finally, keratoplasty may be considered to improve visual function.
In our center, after proper management of the immediate stage, we classify patients according to Dua classification. Treatment of acute stage for grade I and II chemical burns includes frequent lubrication, topical corticosteroids, topical antibiotics, systemic doxycycline, and systemic vitamin C. Topical medications are prescribed as preservative-free preparations. Patients are advised to return within 48 hours for a follow-up visit.
For burns classified as grade III, we recommend frequent administration of autologous serum 20% in addition to the standard treatment. Patients are advised to choose between hospitalization or outpatient management. If they choose the latter, daily follow-up visits are scheduled.
For burns classified as grades IV to VI, we recommend AMT with or without tenonplasty in addition to the aforementioned therapies. It is highly recommended that these patients continue their treatment in an inpatient setting.
Disclosure
The authors report no funding and no conflicts of interest for this work.
References
1. Hall AH. Epidemiology of Ocular Chemical Burn Injuries. In: Sharge N, Burgher F, Blomet J, et al. editors. Chemical Ocular Burns. Berlin, Heidelberg: Springer; 2011:9–15
2. Haring RS, Sheffield ID, Channa R, Canner JK, Schneider EB. Epidemiologic trends of chemical ocular burns in the United States. JAMA Ophthalmol. 2016;134(10):1119e24. doi:10.1001/jamaophthalmol.2016.2645
3. Wagoner MD. Chemical injuries of the eye: current concepts in pathophysiology and therapy. Surv Ophthalmol. 1997;41(4):275e313. doi:10.1016/S0039-6257(96)00007-0
4. Beare JDL. Eye injuries from assault with chemicals. Br J Ophthalmol. 1990;74:514–518. doi:10.1136/bjo.74.9.514
5. Singh P, Tyagi M, Kumar Y, Gupta KK, Sharma PD. Ocular chemical injuries and their management. Oman J Ophthalmol. 2013;6:83–86. doi:10.4103/0974-620X.116624
6. Macdonald E, Cauchi P, Azuara-Blanco A, Foot B. Surveillance of severe chemical corneal injuries in the UK. Br J Ophthalmol. 2009;93:1177–1180. doi:10.1136/bjo.2008.154831
7. Said DG, Dua HS. Chemical burns acid or alkali, what’s the difference? Eye. 2019. doi:10.1038/s41433-019-0735-1
8. Sharma N, Kaur M, Agarwal T, Sangwan VS, Vajpayee RB. Treatment of acute ocular chemical burns. Surv Ophthalmol. 2018;63:214–235. doi:10.1016/j.survophthal.2017.09.005
9. Schrage NF, Langefeld S, Zschocke J, Kuckelkorn R, Redbrake C, Reim M. Eye burns: an emergency and continuing problem. Burns. 2000;26(8):689–699. doi:10.1016/S0305-4179(00)00044-9
10. Eslani M, Baradaran-Rafii A, Movahedan A, Djalilian AR. The ocular surface chemical burns. Hindawi J Ophthalmol. 2014;2014:1–9. doi:10.1155/2014/196827
11. Bunker DJL, George RJ, Kleinschmidt A, Kumar RJ, Maitz P. Alkali-related ocular burns: a case series and review. J Burn Care Res. 2014;35(3):261–268. doi:10.1097/BCR.0b013e31829b0037
12. Roper-Hall MJ. Thermal and chemical burns. Trans Ophthalmol Soc U K. 1965;85:
13. Dua HS, King AJ, Joseph A. A new classification of ocular surface burns. Br J Ophthalmol. 2001;85:1379–1383. doi:10.1136/bjo.85.11.1379
14. McCulley JP. Ocular hydrofluoric acid burns: animal model, mechanism of injury and therapy. Trans Am Ophthalmol Soc. 1990;88:649e84.
15. Kubota M, Shimmura S, Kubota S, et al. Hydrogen and Nacetyl-L-cysteine rescue oxidative stress-induced angiogenesis in a mouse corneal alkali-burn model. Invest Ophthalmol Vis Sci. 2011;52(1):427e33. doi:10.1167/iovs.10-6167
16. Monaghan MT, Brogan K, Lockington D, Rotchford A, Ramaesh K. Variability in measuring pH using litmus paper and the relevance in ocular chemical injury. Eye. 2019. doi:10.1038/s41433-019-0737-z
17. Kuckelkorn R, Schrage N, Keller G, Redbrake C. Emergency treatment of chemical and thermal eye burns. Acta Ophthalmol Scand. 2002;80:4–10. doi:10.1034/j.1600-0420.2002.800102.x
18. Kompa S, Redbrake C, Hilgers C, Wustemeyer H, Schrage N, Remky A. Effect of different irrigating solutions on aqueous humour pH changes, intraocular pressure and histological findings after induced alkali burns. Acta Ophthalmol Scand. 2005;83(4):467e70. doi:10.1111/j.1600-0420.2005.00458.x
19. Rihawi S, Frentz M, Becker J, Reim M, Schrage NF. The consequences of delayed intervention when treating chemical eye burns. Graefes Arch Clin Exp Ophthalmol. 2007;245(10):1507e13. doi:10.1007/s00417-007-0597-2
20. Ikeda N, Hayasaka S, Hayasaka Y, Watanabe K. Alkali burns of the eye: effect of immediate copious irrigation with tap water on their severity. Ophthalmologica. 2006;220(4):225e8. doi:10.1159/000093075
21. Kompa S, Schareck B, Tympner J, Wustemeyer H, Schrage NF. Comparison of emergency eye-wash products in burned porcine eyes. Graefes Arch Clin Exp Ophthalmol. 2002;240(4):308e13. doi:10.1007/s00417-001-0406-2
22. Langefeld S, Press UP, Frentz M, Kompa S, Schrage N. Use of lavage fluid containing diphoterine for irrigation of eyes in first aid emergency treatment. Ophthalmologe. 2003;100(9):727e31.
23. Wiesner N, Dutescu RM, Uthoff D, Kottek A, Reim M, Schrage N. First aid therapy for corrosive chemical eye burns: results of a 30-year longitudinal study with two different decontamination concepts. Graefes Arch Clin Exp Ophthalmol. 2019;257(8):1795–1803. doi:10.1007/s00417-019-04350-x
24. Berman M, Manseau E, Law M, Aiken D. Ulceration is correlated with degradation of fibrin and fibronectin at the corneal surface. Invest Ophthalmol Vis Sci. 1983;24(10):1358e66.
25. Ren G, Song C, Liang P, Zhang Z. The effect of fibronectin on re-epithelialization of rabbits’ cornea after alkali burn. Yan Ke Xue Bao. 1994;10(3):138e43.
26. Spigelman AV, Vernot JA, Deutsch TA, Peyman GA, Molnar J. Fibronectin in alkali burns of the rabbit cornea. Cornea. 1985;4(3):169e72. doi:10.1097/00003226-198503000-00004
27. Schultz GS, Davis JB, Eiferman RA. Growth factors and corneal epithelium. Cornea. 1988;7(2):96e101. doi:10.1097/00003226-198802000-00002
28. Singh G, Foster CS. Epidermal growth factor in alkali-burned corneal epithelial wound healing. Am J Ophthalmol. 1987;103(6):802e7. doi:10.1016/S0002-9394(14)74397-1
29. Kim EC, Kim TK, Park SH, Kim MS. The wound healing effects of vitamin A eye drops after a corneal alkali burn in rats. Acta Ophthalmol. 2012;90(7):e540e6. doi:10.1111/j.1755-3768.2012.02496.x
30. Toshida H, Odaka A, Koike D, Murakami A. Effect of retinol palmitate eye drops on experimental keratoconjunctival epithelial damage induced by n-heptanol in rabbit. Curr Eye Res. 2008;33(1):13e8. doi:10.1080/02713680701827696
31. Kruse FE, Tseng SC. Retinoic acid regulates clonal growth and differentiation of cultured limbal and peripheral corneal epithelium. Invest Ophthalmol Vis Sci. 1994;35(5):2405e20.
32. Asari A, Morita M, Sekiguchi T, Okamura K, Horie K, Miyauchi S. Hyaluronan, CD44 and fibronectin in rabbit corneal epithelial wound healing. Jpn J Ophthalmol. 1996;40(1):18e25.
33. Chung JH, Kim HJ, Fagerholmb P, Cho BC. Effect of topically applied Na-hyaluronan on experimental corneal alkali wound healing. Korean J Ophthalmol. 1996;10(2):68e75. doi:10.3341/kjo.1996.10.2.68
34. Yang G, Espandar L, Mamalis N, Prestwich GD. A crosslinked hyaluronan gel accelerates healing of corneal epithelial abrasion and alkali burn injuries in rabbits. Vet Ophthalmol. 2010;13(3):144e50. doi:10.1111/j.1463-5224.2010.00771.x
35. Chung JH, Kim WK, Lee JS, Pae YS, Kim HJ. Effect of topical Na-hyaluronan on hemidesmosome formation in nheptanol- induced corneal injury. Ophthalmic Res. 1998;30(2):96e100. doi:10.1159/000055460
36. Ralph RA. Tetracyclines and the treatment of corneal stromal ulceration: a review. Cornea. 2000;19(3):274e7. doi:10.1097/00003226-200005000-00003
37. Uitto VJ, Firth JD, Nip L, Golub LM. Doxycycline and chemically modified tetracyclines inhibit gelatinase A (MMP-2) gene expression in human skin keratinocytes. Ann N Y Acad Sci. 1994;732:140e51. doi:10.1111/j.1749-6632.1994.tb24731.x
38. Perry HD, Hodes LW, Seedor JA, Donnenfeld ED, McNamara TF, Golub LM. Effect of doxycycline hyclate on corneal epithelial wound healing in the rabbit alkali-burn model. Preliminary observations. Cornea. 1993;12(5):379e82. doi:10.1097/00003226-199309000-00002
39. Pfister RR, Haddox JL, Sommers CI. Effect of synthetic metalloproteinase inhibitor or citrate on neutrophil chemotaxis and the respiratory burst. Invest Ophthalmol Vis Sci. 1997;38(7):1340e9.
40. Brodovsky SC, McCarty CA, Snibson G, et al. Management of alkali burns: an 11-year retrospective review. Ophthalmology. 2000;107(10):1829e35. doi:10.1016/S0161-6420(00)00289-X
41. Mackway-Jones K, Marsden J. Ascorbate for alkali burns to the eye. Emerg Med J. 2003;20(5):465e6. doi:10.1136/emj.20.5.465
42. Risa O, Saether O, Midelfart A, Krane J, Cejkova J. Analysis of immediate changes of water-soluble metabolites in alkaliburned rabbit cornea, aqueous humor and lens by highresolution 1H-NMR spectroscopy. Graefes Arch Clin Exp Ophthalmol. 2002;240(1):49e55. doi:10.1007/s00417-001-0403-5
43. Saika S, Uenoyama K, Hiroi K, Tanioka H, Takase K, Hikita M. Ascorbic acid phosphate ester and wound healing in rabbit corneal alkali burns: epithelial basement membrane and stroma. Graefes Arch Clin Exp Ophthalmol. 1993;231(4):221e7. doi:10.1007/BF00918845
44. Pfister RR, Paterson CA, Spiers JW, Hayes SA. The efficacy of ascorbate treatment after severe experimental alkali burns depends upon the route of administration. Invest Ophthalmol Vis Sci. 1980;19(12):1526e9.
45. Yoon KC, Im SK, Park YG, Jung YD, Yang SY, Choi J. Application of umbilical cord serum eyedrops for the treatment of dry eye syndrome. Cornea. 2006;25(3):268e72. doi:10.1097/01.ico.0000183484.85636.b6
46. Yoon KC, You IC, Im SK, Jeong TS, Park YG, Choi J. Application of umbilical cord serum eyedrops for the treatment of neurotrophic keratitis. Ophthalmology. 2007;114(9):1637e42. doi:10.1016/j.ophtha.2006.12.014
47. Sharma N, Singh D, Maharana PK, et al. Comparison of amniotic membrane transplantation and umbilical cord serum in acute ocular chemical burns: a randomized controlled trial. Am J Ophthalmol. 2016;168:157e63. doi:10.1016/j.ajo.2016.05.010
48. Liang L, Li W, Ling S, et al. Amniotic membrane extraction solution for ocular chemical burns. Clin Exp Ophthalmol. 2009;37(9):855e63. doi:10.1111/j.1442-9071.2009.02159.x
49. Sharma N, Lathi SS, Sehra SV, et al. Comparison of umbilical cord serum and amniotic membrane transplantation in acute ocular chemical burns. Br J Ophthalmol. 2015;99(5):669e73. doi:10.1136/bjophthalmol-2014-305760
50. Semeraro F, Forbice E, Braga O, Bova A, Di Salvatore A, Azzolini C. Evaluation of the efficacy of 50% autologous serum eye drops in different ocular surface pathologies. Biomed Res Int. 2014;2014:826970. doi:10.1155/2014/826970
51. Sharma N, Goel M, Velpandian T, Titiyal JS, Tandon R, Vajpayee RB. Evaluation of umbilical cord serum therapy in acute ocular chemical burns. Invest Ophthalmol Vis Sci. 2011;52(2):1087e92. doi:10.1167/iovs.09-4170
52. Panda A, Jain M, Vanathi M, Velpandian T, Khokhar S, Dada T. Topical autologous platelet rich plasma eyedrops for acute corneal chemical injury. Cornea. 2012;31(9):989e93. doi:10.1097/ICO.0b013e3182114661
53. Matsumoto Y, Dogru M, Goto E, et al. Autologous serum application in the treatment of neurotrophic keratopathy. Ophthalmology. 2004;111(6):1115e20. doi:10.1016/j.ophtha.2003.10.019
54. Poon AC, Geerling G, Dart JK, Fraenkel GE, Daniels JT. Autologous serum eyedrops for dry eyes and epithelial defects: clinical and in vitro toxicity studies. Br J Ophthalmol. 2001;85(10):1188e97. doi:10.1136/bjo.85.10.1188
55. Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10(4):225e8. doi:10.1097/00008505-200110000-00002
56. Alio JL, Arnalich-Montiel F, Rodriguez AE. The role of “eye platelet rich plasma” (E-PRP) for wound healing in ophthalmology. Curr Pharm Biotechnol. 2012;13:1257–1265. doi:10.2174/138920112800624355
57. Choi JA, Jin HJ, Jung S, et al. Effects of amniotic membrane suspension in human corneal wound healing in vitro. Mol Vis. 2009;15:2230e8.
58. Brent BD, Karcioglu ZA. Effect of topical corticosteroids on goblet-cell density in an alkali burn model. Ann Ophthalmol. 1991;23(6):221e3.
59. Chung JH, Kang YG, Kim HJ. Effect of 0.1% dexamethasone on epithelial healing in experimental corneal alkali wounds: morphological changes during the repair process. Graefes Arch Clin Exp Ophthalmol. 1998;236(7):537e45. doi:10.1007/s004170050118
60. Chung JH, Paek SM, Choi JJ, Park YK, Lee JS, Kim WK. Effect of topically applied 0.1% dexamethasone on endothelial healing and aqueous composition during the repair process of rabbit corneal alkali wounds. Curr Eye Res. 1999;18(2):110e6. doi:10.1076/ceyr.18.2.110.5375
61. Kenyon KR. Inflammatory mechanisms in corneal ulceration. Trans Am Ophthalmol Soc. 1985;83:610e63.
62. Ge HY, Xiao N, Yin XL, et al. Comparison of the antiangiogenic activity of modified RGDRGD endostatin to endostatin delivered by gene transfer in vivo rabbit neovascularization model. Mol Vis. 2011;17:1918e28.
63. Davis AR, Ali QK, Aclimandos WA, Hunter PA. Topical steroid use in the treatment of ocular alkali burns. Br J Ophthalmol. 1997;81(9):732e4. doi:10.1136/bjo.81.9.732
64. Huang Y, Meek KM, Ho MW, Paterson CA. Analysis of birefringence during wound healing and remodeling following alkali burns in rabbit cornea. Exp Eye Res. 2001;73(4):521e32. doi:10.1006/exer.2001.1057
65. Phillips K, Arffa R, Cintron C, et al. Effects of prednisolone and medroxyprogesterone on corneal wound healing, ulceration, and neovascularization. Arch Ophthalmol. 1983;101(4):640e3. doi:10.1001/archopht.1983.01040010640024
66. Parker AV, Williams RN, Paterson CA. The effect of sodium citrate on the stimulation of polymorphonuclear leukocytes. Invest Ophthalmol Vis Sci. 1985;26(9):1257e61.
67. Paterson CA, Williams RN, Parker AV. Characteristics of polymorphonuclear leukocyte infiltration into the alkali burned eye and the influence of sodium citrate. Exp Eye Res. 1984;39(6):701e8. doi:10.1016/0014-4835(84)90069-1
68. Pfister RR, Nicolaro ML, Paterson CA. Sodium citrate reduces the incidence of corneal ulcerations and perforations in extreme alkali-burned eyes- acetylcysteine and ascorbate have no favorable effect. Invest Ophthalmol Vis Sci. 1981;21(3):486e90.
69. Paterson CA, Pfister RR. Prostaglandin-like activity in the aqueous humor following alkali burns. Invest Ophthalmol. 1975;14(3):177e83.
70. Struck HG, Giessler S, Giessler C. [Effect of non-steroidal anti-inflammatory drugs on inflammatory reaction. An animal experiment study]. Ophthalmologe. 1995;92(6):849e53.German
71. Lin A, Patel N, Yoo D, Demartelaere S, Bouchard C. Management of ocular conditions in the burn unit: thermal and chemical burns and Stevens-Johnson syndrome/toxic epidermal necrolysis. J Burn Care Res. 2011;32(5):547–560. doi:10.1097/BCR.0b013e31822b0f29
72. Lin MP, Eksioglu U, Mudumbai RC, Slabaugh MA, Chen PP. Glaucoma in patients with ocular chemical burns. Am J Ophthalmol. 2012;154(3):481–485. doi:10.1016/j.ajo.2012.03.026
73. Baradaran-Rafii A, Eslani M, Haq Z, Shirzadeh E, Huvard MJ, Djalilian AR. Current and upcoming therapies for ocular surface chemical injuries. Ocul Surf. 2017;15(1):48–64. doi:10.1016/j.jtos.2016.09.002
74. Champliaud MF, Lunstrum GP, Rousselle P, Nishiyama T, Keene DR, Burgeson RE. Human amnion contains a novel laminin variant, laminin 7, which like laminin 6, covalently associates with laminin 5 to promote stable epithelialstromal attachment. J Cell Biol. 1996;132(6):1189e98. doi:10.1083/jcb.132.6.1189
75. Hao Y, Ma DH, Hwang DG, Kim WS, Zhang F. Identification of antiangiogenic and antiinflammatory proteins in human amniotic membrane. Cornea. 2000;19(3):348e52. doi:10.1097/00003226-200005000-00018
76. Kim JS, Kim JC, Na BK, Jeong JM, Song CY. Amniotic membrane patching promotes healing and inhibits proteinase activity on wound healing following acute corneal alkali burn. Exp Eye Res. 2000;70(3):329e37. doi:10.1006/exer.1999.0794
77. Rao TV, Chandrasekharam V. Use of dry human and bovine amnion as a biological dressing. Arch Surg. 1981;116(7):891e6. doi:10.1001/archsurg.1981.01380190029007
78. Robson MC, Krizek TJ. The effect of human amniotic membranes on the bacteria population of infected rat burns. Ann Surg. 1973;177(2):144e9. doi:10.1097/00000658-197302000-00003
79. Suri K, Kosker M, Raber IM, et al. Sutureless amniotic membrane ProKera for ocular surface disorders: short-term results. Eye Contact Lens. 2013;39(5):341e7. doi:10.1097/ICL.0b013e3182a2f8fa
80. Ucakhan OO, Koklu G, Firat E. Nonpreserved human amniotic membrane transplantation in acute and chronic chemical eye injuries. Cornea. 2002;21(2):169e72. doi:10.1097/00003226-200203000-00008
81. Tejwani S, Kolari RS, Sangwan VS, Rao GN. Role of amniotic membrane graft for ocular chemical and thermal injuries. Cornea. 2007;26(1):21e6. doi:10.1097/ICO.0b013e31802b4201
82. Westekemper H, Figueiredo FC, Siah WF, Wagner N, Steuhl KP, Meller D. Clinical outcomes of amniotic membrane transplantation in the management of acute ocular chemical injury. Br J Ophthalmol. 2017;101:103e7. doi:10.1136/bjophthalmol-2015-308037
83. Tandon R, Gupta N, Kalaivani M, Sharma N, Titiyal JS, Vajpayee RB. Amniotic membrane transplantation as an adjunct to medical therapy in acute ocular burns. Br J Ophthalmol. 2011;95(2):199e204. doi:10.1136/bjo.2009.173716
84. Arora R, Mehta D, Jain V. Amniotic membrane transplantation in acute chemical burns. Eye. 2005;19(3):273e8. doi:10.1038/sj.eye.6701490
85. Joseph A, Dua HS, King AJ. Failure of amniotic membrane transplantation in the treatment of acute ocular burns. Br J Ophthalmol. 2001;85(9):1065e9. doi:10.1136/bjo.85.9.1065
86. Tamhane A, Vajpayee RB, Biswas NR, et al. Evaluation of amniotic membrane transplantation as an adjunct to medical therapy as compared with medical therapy alone in acute ocular burns. Ophthalmology. 2005;112(11):1963e9. doi:10.1016/j.ophtha.2005.05.022
87. Liang X, Liu Z, Lin Y, Li N, Huang M, Wang Z. A modified symblepharon ring for sutureless amniotic membrane patch to treat acute ocular surface burns. J Burn Care Res. 2012;33(2):e32e8. doi:10.1097/BCR.0b013e318239f9b9
88. Prabhasawat P, Tesavibul N, Prakairungthong N, Booranapong W. Efficacy of amniotic membrane patching for acute chemical and thermal ocular burns. J Med Assoc Thai. 2007;90(2):319e26.
89. Meller D, Pires RT, Mack RJ, et al. Amniotic membrane transplantation for acute chemical or thermal burns. Ophthalmology. 2000;107(5):980e9. doi:10.1016/S0161-6420(00)00024-5
90. Clare G, Suleman H, Bunce C, Dua H. Amniotic membrane transplantation for acute ocular burns. Cochrane Database Syst Rev. 2012;9:CD009379.
91. Teping C, Reim M. Tenonplasty as a new surgical principle in the early treatment of the most severe chemical eye burns. Klin Monatsbl Augenheilkd. 1989;194:1–5. doi:10.1055/s-2008-1046325
92. Sharma A, Pandey S, Sharma R, Mohan K, Gupta A. Cyanoacrylate tissue adhesive augmented tenoplasty: a new surgical procedure for bilateral severe chemical eye burns. Cornea. 1999;18(3):366e9. doi:10.1097/00003226-199905000-00020
93. Casas VE, Kheirkhah A, Blanco G, Tseng SC. Surgical approach for scleral ischemia and melt. Cornea. 2008;27:196–201. doi:10.1097/ICO.0b013e31815ba1ae
94. Tabatabaei SA, Soleimani M, Mirshahi R. Selective localized tenonplasty for corneal burns based on the findings of ocular surface fluorescein angiography. Cornea. 2017;36(8):104–117. doi:10.1097/ICO.0000000000001256
95. Utheim TP. Limbal epithelial cell therapy: past, present, and future. Methods Mol Biol. 2013;1014:3–43.
96. Fernandes M, Sangwan VS, Rao SK, et al. Limbal stem cell transplantation. Indian J Ophthalmol. 2004;52:5–22.
97. Liang L, Sheha H, Li J, Tseng SC. Limbal stem cell transplantation: new progresses and challenges. Eye (Lond). 2009;23:1946–1953. doi:10.1038/eye.2008.379
98. Crawford AZ, McGhee CN. Management of limbal stem cell deficiency in severe ocular chemical burns. Clin Exp Ophthalmol. 2012;40:227–229. doi:10.1111/j.1442-9071.2012.02775.x
99. Welder JD, Pandya HK, Nassiri N, Djalilian AR. Conjunctival limbal autograft and allograft transplantation using fibrin glue. Ophthalmic Surg Lasers Imaging. 2012;43:323–327. doi:10.3928/15428877-20120618-04
100. Baradaran-Rafii A, Eslani M, Djalillian AR. Complications of keratolimbal allograft surgery. Cornea. 2013;32:561–566. doi:10.1097/ICO.0b013e31826215eb
101. Pellegrini G, Traverso CE, Franzi AT, et al. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet. 1997;349(9057):990–993. doi:10.1016/S0140-6736(96)11188-0
102. Eslani M, Baradaran-Rafii A, Djalilian A. Conjunctival-limbal autograft (CLAU). In: Thomsen WL, editor. Advances in Eye Research. Vol. 1. Hauppauge, NY: Nova Biomedical Press; 2011;989–993.
103. Ozdemir O, Tekeli O, Ornek K, et al. Limbal autograft and allograft transplantations in patients with corneal burns. Eye (Lond). 2004;18:241–248. doi:10.1038/sj.eye.6700640
104. Higa K, Shimazaki J. Recent advances in cultivated epithelial transplantation. Cornea. 2008;27:S41–7. doi:10.1097/ICO.0b013e31817f358e
105. Basu S, Ali H, Sangwan VS. Clinical outcomes of repeat autologous cultivated limbal epithelial transplantation for ocular surface burns. Am J Ophthalmol. 2012;153:643–650. doi:10.1016/j.ajo.2011.09.016
106. Holland EJ. Management of limbal stem cell deficiency: A historical perspective, past, present, and future. Cornea. 2015;34:S9–S15. doi:10.1097/ICO.0000000000000534
107. Espana EM, Di Pascuale M, Grueterich M, et al. Keratolimbal allograft in corneal reconstruction. Eye (Lond). 2004;18:406–417. doi:10.1038/sj.eye.6700670
108. Javadi MA, Baradaran-Rafii A. Living-related conjunctival-limbal allograft for chronic or delayed-onset mustard gas keratopathy. Cornea. 2009;28:51–57. doi:10.1097/ICO.0b013e3181852673
109. Solomon A, Ellies P, Anderson DF, et al. Long-term outcome of keratolimbal allograft with or without penetrating keratoplasty for total limbal stem cell deficiency. Ophthalmology. 2002;109:1159. doi:10.1016/S0161-6420(02)00960-0
110. Dua HS, Azuara-Blanco A. Allo-limbal transplantation in patients with limbal stem cell deficiency. Br J Ophthalmol. 1999;83:414–419. doi:10.1136/bjo.83.4.414
111. Daya SM, Ilari FA. Living related conjunctival limbal allograft for the treatment of stem cell deficiency. Ophthalmology. 2001;108:126–133. doi:10.1016/S0161-6420(00)00475-9
112. Fish R, Davidson RS. Management of ocular thermal and chemical injuries, including amniotic membrane therapy. Curr Opin Ophthalmol. 2010;21:317–321. doi:10.1097/ICU.0b013e32833a8da2
113. Djalilian AR, Mahesh SP, Koch CA, et al. Survival of donor epithelial cells after limbal stem cell transplantation. Invest Ophthalmol Vis Sci. 2005;46:803–807. doi:10.1167/iovs.04-0575
114. Krakauer M, Welder JD, Pandya HK, et al. Adverse effects of systemic immunosuppression in keratolimbal allograft. J Ophthalmol. 2012;2012:576712. doi:10.1155/2012/576712
115. Singh G, Singh Bhinder H. Evaluation of therapeutic deep anterior lamellar keratoplasty in acute ocular chemical burns. Eur J Ophthalmol. 2008;18(4):517e28. doi:10.1177/112067210801800403
116. Chan CC, Biber JM, Holland EJ. The modified Cincinnati procedure: combined conjunctival limbal autografts and keratolimbal allografts for severe unilateral ocular surface failure. Cornea. 2012;31:1264–1272. doi:10.1097/ICO.0b013e31823f8e95
117. Nassiri N, Djalilian AR. Keratoplasty: moving to the front. J Ophthalmic Vis Res. 2009;4:5–7.
118. Kuckelkorn R, Redbrake C, Schrage NF, Reim M. Keratoplasty with 11-12 mm diameter for management of severely chemical-burned eyes. Ophthalmologe. 1993;90(6):683e7.
119. Redbrake C, Buchal V. Keratoplasty with scleral rim after the most severe chemical eye burns of the anterior eye segment. Klin Monbl Augenheilkd. 1996;208(3):145e51.
120. Hou JH, de la Cruz J, Djalilian AR. Outcomes of Boston keratoprosthesis implantation for failed keratoplasty after keratolimbal allograft. Cornea. 2012;31:1432–1435. doi:10.1097/ICO.0b013e31823e2ac6
121. Zerbe BL, Belin MW, Ciolino JB. Boston Type 1 Keratoprosthesis Study G. Results from the multicenter Boston Type 1 Keratoprosthesis Study. Ophthalmology. 2006;113:1779, e1–e7. doi:10.1016/j.ophtha.2006.05.015
122. Hille K, Grabner G, Liu C, et al. Standards for modified osteoodontokeratoprosthesis (OOKP) surgery according to Strampelli and Falcinelli: the Rome-Vienna Protocol. Cornea. 2005;24:895–908. doi:10.1097/01.ico.0000157401.81408.62
123. Gomaa A, Comyn O, Liu C. Keratoprostheses in clinical practice - a review. Clin Exp Ophthalmol. 2010;38:211–224. doi:10.1111/j.1442-9071.2010.02231.x
124. De La Paz MF, De Toledo JA, Charoenrook V, et al. Impact of clinical factors on the long-term functional and anatomic outcomes of osteo-odonto-keratoprosthesis and tibial bone keratoprosthesis. Am J Ophthalmol. 2011;151:829–839.
125. Hirst LW, Summers PM, Griffiths D, Bancroft J, Lillicrap GR. Controlled trial of hyperbaric oxygen treatment for alkali corneal burn in the rabbit. Clin Exp Ophthalmol. 2004;32(1):67e70. doi:10.1046/j.1442-9071.2004.00761.x
126. Sharifipour F, Zamani M, Idani E, Hemmati AA. Oxygen therapy for severe corneal alkali burn in rabbits. Cornea. 2007;26(9):1107e10. doi:10.1097/ICO.0b013e31813349d2
127. Sharifipour F, Baradaran-Rafii A, Idani E, Zamani M, Jabbarpoor Bonyadi MH. Oxygen therapy for acute ocular chemical or thermal burns: a pilot study. Am J Ophthalmol. 2011;151(5):823e8. doi:10.1016/j.ajo.2010.11.005
128. Duchesne B, Tahi H, Galand A. Use of human fibrin glue and amniotic membrane transplant in corneal perforation. Cornea. 2001;20(2):230e2. doi:10.1097/00003226-200103000-00027
129. Sii F, Lee GA. Fibrin glue in the management of corneal melt. Clin Exp Ophthalmol. 2005;33(5):532e4. doi:10.1111/j.1442-9071.2005.01076.x
130. Nishida T, Nakamura M, Mishima H, Otori T. Differential modes of action of fibronectin and epidermal growth factor on rabbit corneal epithelial migration. J Cell Physiol. 1990;145(3):549e54. doi:10.1002/jcp.1041450323
131. Tang X, Wang Z, Dong N. Tissue adhesive combined with amniotic membrane adhering in the treatment of ocular burns. Yan Ke Xue Bao. 2005;21(2):74e8.
132. Lim P, Fuchsluger TA, Jurkunas UV. Limbal stem cell deficiency and corneal neovascularization. Semin Ophthalmol. 2009;24:139–148. doi:10.1080/08820530902801478
133. Wang Y, Yin H, Chen P, Xie L, Wang Y. Inhibitory effect of canstatin in alkali burn-induced corneal neovascularization. Ophthalmic Res. 2011;46(2):66e72. doi:10.1159/000322804
134. Cheng HC, Yeh SI, Tsao YP, Kuo PC. Subconjunctival injection of recombinant AAV angiostatin ameliorates alkali burn induced corneal angiogenesis. Mol Vis. 2007;13:2344e52.
135. Ahmed A, Berati H, Nalan A, Aylin S. Effect of bevacizumab on corneal neovascularization in experimental rabbit model. Clin Exp Ophthalmol. 2009;37(7):730e6. doi:10.1111/j.1442-9071.2009.02112.x
136. Hosseini H, Nejabat M, Mehryar M, Yazdchi T, Sedaghat A, Noori F. Bevacizumab inhibits corneal neovascularization in an alkali burn induced model of corneal angiogenesis. Clin Exp Ophthalmol. 2007;35(8):745e8. doi:10.1111/j.1442-9071.2007.01572.x
137. Dursun A, Arici MK, Dursun F, et al. Comparison of the effects of bevacizumab and ranibizumab injection on corneal angiogenesis in an alkali burn induced model. Int J Ophthalmol. 2012;5:448–451. doi:10.3980/j.issn.2222-3959.2012.04.08
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
