Back to Journals » Hepatic Medicine: Evidence and Research » Volume 12
Management Strategies for Patients with Hilar Cholangiocarcinomas: Challenges and Solutions
Authors Mehrotra S, Lalwani S, Nundy S
Received 12 July 2019
Accepted for publication 9 January 2020
Published 23 January 2020 Volume 2020:12 Pages 1—13
DOI https://doi.org/10.2147/HMER.S223022
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Gerry Lake-Bakaar
Siddharth Mehrotra, Shailendra Lalwani, Samiran Nundy
Department of Surgical Gastroenterology and Liver Transplantation, Sir Ganga Ram Hospital, New Delhi, India
Correspondence: Samiran Nundy
Email [email protected]
Abstract: Advances in imaging, pathology and therapy have resulted in major improvements in the management of cholangiocarcinomas; the mortality has come down and with it there has been an improved 5-year survival. Surgical resection remains the treatment of choice and reports from high volume centres have shown an increase in resectability rates, R0 resection, a decrease in mortality and an improvement in 5-year survival; however, the operative morbidity remains high, pointing towards the complexity of the management of these difficult lesions. Complete excision is also often limited by the locally advanced nature of the disease at the time of diagnosis and a proportion of patients who were earlier deemed resectable on imaging are found to have unresectable disease at the time of operation. Neoadjuvant therapy has had only a limited impact on survival. Liver transplantation is also an option in a few patients following strict criteria for selection. Since the large majority of patients are only diagnosed at the late stages of the disease palliation (endoscopic or surgical) is an important part of treatment. Portal vein embolisation and pre-operative biliary drainage have had a major impact on outcomes. Major liver resection with caudate lobe removal remains the standard operation and procedures like routine vascular resection and liver transplant should only be carried out in experienced centres. Improvements in both neo as well as adjuvant therapy may lead to a standardized protocol in the future, as well as an improvement in survival.
Keywords: cholangiocarcinoma, preoperative preparation, surgery
Introduction
Cholangiocarcinoma (CC) is a rare malignancy originating from the biliary epithelium. Klatskin’s article in 1965 described the classical picture but also highlighted the high perioperative mortality (92%) in these patients.1 Advances in imaging, pathology and therapeutic management have made major strides and hence, the mortality has come down and with it there has been an improved 5-year survival. Surgical resection is the treatment of choice but limited by the locally advanced nature of disease at the time of diagnosis. A proportion of patients who were deemed resectable on imaging are also found to have unresectable disease at the time of surgery. Neoadjuvant therapy has had a limited impact. Liver transplantation is also an option in a few patients following strict criteria for selection. Palliation (endoscopic or surgical) is an important part of the treatment since the large majority of patients are only diagnosed at the late stages of the disease.
Epidemiology
Cholangiocarcinoma is anatomically divided into intrahepatic (IHCC) and extrahepatic CC which not only need different management but differ in incidence as well. The highest incidence has been reported from Asian countries like South Korea, China and Thailand (5.6 to 22.9/100,000).2 Europe and the western world have usually reported low incidence rates of 1–-2/100,000. Surveillance, Epidemiology and End Results (SEER)based US data from 1973 to 2012 reported an increase in the incidence of intrahepatic CC from 0.44 to 1.18 cases per 100,000, while the incidence of extrahepatic CC increased modestly from 0.95 to 1.02 per 100,000 during this 40-year period.3 Also reported was a fall in the incidence of carcinoma from an unknown primary during the same period.4 However, the 7th International Classification of Diseases (ICD) coding for CC has been altered to include more tumours to be intrahepatic cholangiocarcinomas rather than liver cancers which may reflect this change in incidence, and recent reports have also supported the view that at least some of the reported increase in IHCC may be an artefact of changes in the World Health Organization’s International Classification of Diseases for Oncology. Improved radiological techniques and better cancer registries may also account for the increase. CC accounts for 3% of gastrointestinal malignancies, but IHCC is the second most common primary liver tumour. The disease is most commonly diagnosed worldwide in the fifth decade of life; however, in the western world, the disease presents later during the seventh decade. It rarely occurs before the age of 40 with the exception of patients with primary sclerosing cholangitis (PSC) in whom it presents earlier. Men are more commonly affected than females with a ratio of 1.5:1.
Risk Factors
Although there are a number of risk factors commonly associated with CCs, the majority of patients who are diagnosed with CC do not have any identifiable ones.
These risk factors include:
● Parasitic Infections
Infections with parasites have been long associated with CC especially in eastern countries. Clonorchis sinensis and Opisthorcis viverrini obtained from the ingestion of raw, undercooked or pickled fish are the two most common liver flukes associated with CC. These organisms may remain in the gall bladder and bile ducts for years and cause repeated episodes of inflammation, cholangitis and fibrosis. The fibrosis does not reverse even after treatment with antihelminthics. Shin et al in a meta-analysis of case-controlled studies showed a correlation between Clonorchis sinensis and CC in Korea and found that the overall odds ratio for CC due to C. sinensis infection was 4.7 (95% CI: 2.2–9.8).5
● Primary Sclerosing Cholangitis (PSC)
PSC is an autoimmune disease of the biliary epithelium and presents as single or multiple strictures of both the intra- and extrahepatic bile ducts. It is rare but is one of the commonly identified risk factors for CC in the West. The lifetime risk of CC in a PSC patient ranges from 6% to 36%; however, only 10% of CC are attributed to PSC. Chronic inflammation, stasis and mutations are responsible for the carcinogenesis. Chapman et al reported that most cases of CC in PSC are identified during the first year of diagnosis6 and hence the age of diagnosis of CC in PSC patients is the fourth decade as compared to the seventh in other patients. The influence of a concomitant presence of IBD is controversial with Claseene et al reporting a significantly higher incidence of CC in patients with both PSC with IBD7 while Chalasani did not show a significant correlation.8
● Choledochal Cysts
Choledochal cysts have been traditionally associated with an increased risk of CC of 6–30%. The risk increases after the second decade of life, and malignancy is diagnosed by a mean age of 32 years, though recent studies quote higher age groups. However, most reports have few patients or the data collection has not been adequate to make accurate predictions. Lee et al in a multicentre retrospective study identified biliary tract malignancy in 10% of the patients undergoing excision for choledochal cysts of which half were CC.9 A meta-analysis of 18 studies also reported an 11 times risk of malignancy especially in the type I and type IV varieties.10 The risk of biliary tract malignancy does not disappear with excision. Taku Ohashi in a long-term follow-up reported cumulative incidences of biliary tract cancer at 15, 20, and 25 years after cyst excision at 1.6%, 3.9%, and 11.3%, respectively.11
● Hepatitis B and C
Hepatitis and cirrhosis have been proven in recent times to be risk factors for CC. Hepatitis B has been shown to be associated with CC more in eastern countries whereas in the west it has been Hepatitis C. A Japanese study has shown a seroprevalence of both the hepatitis viruses in IHCC patients. A meta-analysis showed a statistically significant increased risk of IHCC in hepatitis B patients.12 Most of these studies have shown a relatively higher risk of IHCC than hilar CC. Hepatitis C has also been shown in a systemic review to increase the risk of CC, especially IHCC, and a pooled risk higher in the west compared to the east.13 Sorenson et al reported cirrhotic patients to be at a high risk of CC though the risk is less than HCC.14
● Hepatolithiasis
Hepatolithiasis has been known to cause CC and the incidence reported from Asian countries varies from 2.4% to 16%.15 This condition is much less common in the West, but the incidence of CC arising in patients with hepatolithiasis is the same. CC in the presence of hepatolithiasis carries a worse prognosis than when it is absent and CC is known to occur more often in patients with liver atrophy, truncated portal flow and patients with hepatobiliary-enteric anastomosis.16
● Miscellaneous
Many other diseases have shown a correlation with CC. The metabolic syndrome with obesity and diabetes has shown a relationship but more data are required before making firm conclusions. Environmental toxins and chemicals have also been implicated as causes for CC. Thorotrast, a former radiological agent, was associated with a very high risk of CC and hence banned in 1960. Environmental toxins such as vinyl chloride, dichloromethane and/or1,2-dichloropropane dioxin have also been postulated as causes of CC.
Symptoms and Signs
The presentation of cholangiocarcinomas differs with their site.
Intrahepatic Cholangiocarcinomas
The most common presentation of IHCC is of incidental discovery. Twenty to forty percent of the patients are diagnosed while undergoing imaging for some other disease or symptom. The symptoms, if present, are nonspecific like weight loss, anorexia and usually denote advanced disease. A study from the Memorial Sloan Kettering Cancer Center (MSKCC) in New York reported higher resectability rates in asymptomatic (58%) than symptomatic (25%)17 patients.
Extrahepatic Cholangiocarcinomas
These patients usually present with symptoms, and painless jaundice is the most common with other itching, clay coloured stools and dark urine. Cholangitis may be the presenting feature in 10% of the patients. Hepatomegaly is usually present and may be confined to one lobe depending on the site of the lesion and concomitant vascular involvement. Advanced disease presents with features of anorexia, malaise and significant weight loss. In PSC patients these signs may be the initial marker for the presence of CC.
Diagnosis
Laboratory Tests
The liver function tests are usually deranged and suggest obstructive jaundice with raised serum bilirubin and alkaline phosphatase levels in extrahepatic CC. In IHCC the liver function tests may be normal or suggest an obstructive pattern if the lesion is a large mass causing obstruction near the hepatic hilum. Cholangitis may present with altered leukocyte counts and serum creatinine levels. These should always be sought and treated early.
The tumour marker Ca 19-9 is expressed on the cell surfaces of mainly the biliary and pancreatic ducts. It is elevated in CC and at a cut off value of 37IU/mL, the sensitivity is 73% while the specificity is only 63%. However, if we keep a cutoff of 300IU/mL, the specificity increases but the sensitivity decreases. It must be remembered that increased CA 19-9 concentrations up to 100,000 IU/mL have also been reported in patients with bile duct stenosis due to biliary stones,18 which normalized on removal of the obstruction. This is attributed to its increased production and decreased clearance. The estimated sensitivity of CA 19-9 in predicting CC in the context of primary sclerosing cholangitis is 38–89%, and specificity 50–98%. A recent meta-analysis reported an overall pooled sensitivity of 0.72 (0.70–0.75) and specificity of 0.84 (0.82–0.85) for Ca19-9 in CC.19 The trend is important in cholestatic diseases with permanently raised Ca19-9 levels to differentiate the onset of CC. In non-PSC patients, a Ca19-9 value of <100U/L has a negative predictive value of 92%. In PSC patients, a Ca19-9 value <129 has a high negative predictive value of 99% and a positive predictive value of 57%. Values more than 129U/L have also been found in one-third of patients with PSC with no evidence of CC on long-term follow-up.20 Up to 7% patients are Lewis antigen negative and may not secrete Ca19-9.21
CEA is another tumour marker for CC but is elevated in only one third of patients. One study reported CEA to be a better prognostic marker than Ca 19-9,22 but as the tumour marker is also elevated in many other conditions its specificity is low. Other markers like IL-6, preoperative platelet/lymphocyte ratio (PLR), abnormal expression of mucin 5AC (MUC5AC) and even concomitant use of multiple markers have been used but need to be validated in more studies to be applicable in large populations.
Imaging
Ultrasound
This is usually used as the first screening test for patients with jaundice. It may show biliary dilatation and help to identify the site of blockage. The examination may be more specific in IHCC by showing a hypoechoic hepatic mass with minimal vascularity on Doppler ultrasonography. Satellite nodules and retraction of the capsule may also be seen.
Cross-Sectional Imaging
A triple-phase CT scan is the imaging procedure of choice in CC and accurately identifies the lesion in 94–100% patients. It also accurately defines the radial extent of soft tissue involvement, vascular encroachment, lobar atrophy and direct liver invasion which are important for determining resectability (Figure 1). The main disadvantage of CT scanning is its inability to accurately measure the longitudinal extent of biliary tract involvement (accuracy 81% vs 100%).23 With the help of 3 D imaging and reconstruction the tumour and liver volumes can be accurately measured to help decide whether to proceed to surgical resection or perform preoperative optimization. Lymph nodes can also be assessed with CT though the sensitivity for this is low. Distant metastases can also be identified. CT should be done before stenting as these cause image artefacts and reduce its accuracy.
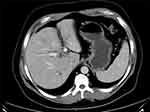 |
Figure 1 CT triphasic image of hilar cholangiocarcinoma adjacent to portal vessels (vascular involvement). |
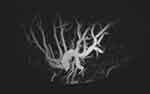 |
Figure 2 MR image of hilar cholangiocarcinoma showing separation of bile ducts (high hilar lesion). |
On non-contrast scans an IHCC appears as a hypodense mass while in the arterial phase there is peripheral rim enhancement with irregular and infiltrative margins; however, compared to HCC, there is no portal venous washout, but there may be delayed portal enhancement. On MRI with MRCPCC appears as hypointense on T1, and hyperintense on T2-weighted images. In addition, rim hyperenhancement in the arterial phase is consistent with IHCC.
MRI with MRCP has the advantage (Figure 2) of more accurately detecting the extent of biliary involvement and, with recent advances, its prediction of vascular invasion and resectability is comparable to CT.24 However, in a large number of centres, MR is a complimentary test to CT rather than an alternative. The ideal time to do an MRCP is also before stent insertion.
Hilar CCs are not FDG avid and hence the routine use of PET scans is not advised. Kim et al reported a sensitivity, specificity and accuracy of 84%, 79% and 83% of PET in detecting the primary tumour but many other studies have shown a lower sensitivity and specificity.25,26 False-negative results may be achieved in periductal fibrosis, infiltrative growths and lesions with a high mucin content. Detection of lymph nodes and distant metastases has a higher sensitivity compared to conventional imaging. It may sometimes help in differentiating malignant from benign lesions.
Endoscopy
Biliary brushing has occasionally been used as a tool for diagnosis but because of the fibrotic nature of the tumour its sensitivity is low (~40%). Newer modalities like fluorescence in situ hybridization
(FISH) targeting the chromosome 3,7,17 increases the sensitivity of brush biopsy with polysomy having the highest specificity. These tests however are not commonly available at all centres and cost is a concern. Endoscopic Ultrasound (EUS) has a low sensitivity but high specificity (60% and 100%) for diagnosis. The sensitivity is higher for distal CCs whereas in proximal tumours ERCP scores over EUS. The advantages are that the positive predictive value is high and lymph node sampling can be done which may help avoid more invasive procedures and plan management. The disadvantages are few but there is a reported incidence of tumour seeding and a decreased sensitivity in the presence of stents.
Most of these imaging and endoscopy modalities are complementary and do not replace one another and patients will generally need a combination of tests before coming to a conclusion. Even after multiple tests, approximately 10% of the patients undergoing surgery for CC will be found to have a benign disease on histopathology.
Management
Staging
Bismuth and Corlette first described the staging of hilar CC based on the level of biliary system involvement. This staging has been modified a number of times and has influenced management as described by Bismuth in 199227(Table 1). However, the classification was limited in assessing vascular involvement and atrophy, hence it did not predict resectability.
 |
Table 1 Comparative Outcomes for Surgery in Hilar Cholangiocarcinoma |
Jarnagin from the Memorial Sloan Kettering Cancer Center (MSKCC) in New York used a modified staging system incorporating local and tumour related factors such as vascular involvement and atrophy and showed it to be a better predictor of resectability, Ro resection and predictor of survival.28 The modified T stage includes (i) biliary tract involvement including the hilum and bilateral ducts (ii) portal vein involvement including contralateral infiltration and the (iii) presence of lobar atrophy. This staging system has since been modified and is now used worldwide, although resectability is still difficult to predict. Two new classifications have been proposed; Blechacz and Sanchez from the Eastern Cooperative Oncology Group proposed a classification in 2009 incorporating tumour size, metastasis and ECOG performance.29 Deoliveira et al in 2011 also described a staging system including tumour size, degree of vascular involvement and lymph node metastasis.30 However, most of these staging systems are complicated and predicting resectability and prognosis is still difficult and the MSKCC staging is still commonly used.
The TNM classification is done after the specimen has been resected and this staging system shows a good correlation with survival. Till the sixth AJCC classification the IHCCs were treated as primary liver cancers; however, with the seventh AJCC IHCC and distal CC were placed as separate entities with their respective staging systems. The system incorporates the extent of tumour spread with biliary and vascular involvement, lymph nodes and distant metastasis. However, it can only be used after operation and not for predicting resectability.
Pre-Operative Management
Preoperative Biliary Drainage
Before the turn of century surgery for hilar CC was associated with high mortality rates and liver failure post-operatively was one of the most common causes. Many centres in Asia started using pre-operative biliary drainage to decrease the risk of postoperative liver failure due to cholestasis and aid in liver regeneration. A large series published by Nagino et al in 2013 showed that preoperative biliary drainage was used in 84% of their patients (all 414 patients with jaundice and 69 patients with biliary dilatation). They operated when the serum bilirubin level was less than 2 mg/dl. Lee et al published their data in 2010 and reported using preoperative drainage in 94-% of the patients and related their good outcomes to this measure. However, there are studies from the western world which are against the routine use of preoperative drainage mainly because of the risk of introducing infection and possibility of tumour seeding. Kennedy et al in 2009 showed that patients with a future liver remnant (FLR) of less than 30% showed a benefit with pre-operative biliary drainage as compared to those with no drainage; while in the group with a larger FLR, preoperative biliary drainage was associated with a higher mortality and patients with no preoperative drainage did not have liver failure. Hence, they suggested the selective use of biliary drainage in patients with FLRs less than 30%.31 Wiggers et al also showed a higher mortality (12% vs 0%) and risk of cholangitis (20% vs 8%) in patients with high FLRs.32 Hence, the use of biliary drainage and level of serum bilirubin before surgery are still based on institutional protocols. However, there are certain indications where biliary drainage is nearly always necessary like in patients with cholangitis, those who have had pre-operative chemotherapy and jaundice associated with renal failure and severe malnutrition.
Endoscopic drainage was preferred initially but was at times technically difficult in high hilar tumours and associated with recurrent cholangitis because of the duodenal communication. Percutaneous transhepatic biliary drainage (PTBD) then became more commonly used before major hepatectomy for high hilar blocks. The main advantages were the fewer procedures necessary and risk of cholangitis compared to ERCP.33 The success of PTBD in high hilar blocks contributed to its greater use and also its application for localization of the ducts at operation. However, PTBD is not without complications. Vascular injury has been noted to occur in a number of patients. Cholangitis is also encountered though in fewer patients compared to ERCP.34 The number of stents inserted is also correlated with higher levels of biliary obstruction and prognosis with some patients requiring 2–3 stents and yet achieving lowered serum bilirubin levels. Tumour tract seeding has been reported after PTBD though this has now decreased to less than 5%. Nagino et al also mentioned tumor tract seeding as being one of their main reason for changing to endoscopic nasobiliary drainage (ENBD). Japanese surgeons have started using ENBD more frequently over the last decade as they are able to refeed the aspirated bile and repeatedly flush the ENBD if it gets blocked. Nagino et al have reported ENBD as the procedure of choice and PTBD to be used only when ENBD is not feasible. However, Aaukura et al have reported difficulty with ENBD in type IV cholangiocarcinomas.35 Drainage of both the lobes may be difficult with ENBD and more easily accomplished with PTBD. The use of ENBD has largely been confined to Japan and few other centres and PTBD still remains to be the procedure of choice till more prospective studies from multiple centres show distinct advantages.
Selective drainage of the future remnant liver has been accepted as the standard procedure over the years. Nagino et al described how they initially drained all the obstructed ducts and later only selected ducts of future remnant. Their indications for drainage of all ducts were segmental cholangitis, insufficient biliary drainage or guide to surgery in high hilar blocks. Even Cho et al in their large experience have used selective drainage of the future remnant. Hence, segmental drainage is preferred over total drainage though in Japan ENBD is preferred where usually one side is drained but with advances catheter draining of both sides may be achieved.
The timing of surgery or duration of pre-operative drainage is not defined. Most institutions define it on the basis of the level of serum bilirubin which also shows variance. A meta-analysis of 13 studies quoted a serum total bilirubin less than 5 mg/dl as the end point while others quoting median bilirubin less than 5. The duration of PTBD in their study was 30.8 (± 27.3) days.
Portal Vein Embolization
Portal vein embolization (PVE) is also being used more commonly over the years to increase the size of the FLR. PVE has been suggested for patients with normal livers and an FLR < 25%, post chemotherapy steatotic livers with FLR < 30% and chronic disease with FLR < 40%. Most of these studies have been done in patients with colorectal liver metastases but have been used for hilar CC as well. Song et al used PVE when preoperative the FLR was <20% and used a time frame of 3 weeks to review and perform the hepatic resection if the FLR was >20%. Park et al described the use of PVE in 18% of the patients with FLR < 25%. Nagino et al reported the use of PVE in 45% of patients and their criterion for choosing was an FLR < 40% and surgery was planned 3 weeks later after assessing the FLR. The increased FLR helps to protect against post-operative liver failure and has made major hepatectomies like trisectionectomy successful. The main aim is to increase the FLR and decrease the risk of post-operative liver failure. The majority of centres use PVE before right trisectionectomy or even right hepatectomy in cholestatic livers; however, there are centres who would not use the same for left hepatectomy or even left trisectionectomy as the right posterior lobe would contribute to approximately 33% of the liver volume compared to the left lateral segment constituting only approximately 20%.36
There is still a debate over the interval of liver resection after PVE but most studies have shown the maximum growth occurs after 2–3 weeks following which patients can be taken up for surgery. Two studies have highlighted that the kinetic growth rate is a better predictor of PVE outcomes than the standardized future liver remnant (sFLR) volume and degree of hypertrophy. Leung et al showed that no patient with a growth rate >2.66% per week suffered from post-operative liver failure.37 Shindou et al compared kinetic growth rates of less than 2% per week vs ≥2% per week and found it to correlate with rates of hepatic insufficiency (21.6% vs 0%, p = 0.0001) and liver-related 90-day mortality (8.1% vs 0%, P=0.04).38 Systematic reviews have also been done to suggest efficacy. A number of other innovations have taken place since the inception of PVE in 1990 like embolization of seg4 branches also in right trisectionectomy to increase the efficacy and growth and puncturing of the contralateral side to be removed. All these innovations have improved efficacy and at the same time helped to reduce complications.
ALPPS
With the improvements in pre-operative, operative and postoperative measures, liver surgery has now become more safe with mortality rates in major centres reported to be less than 5% and in some to be even less than 1%.39 Hilar CC requires major hepatectomy in most cases, and liver failure was common before portal vein embolization, which stimulates the contralateral liver to hypertrophy, was introduced. However, not all patients achieve a good liver remnant. Associating Liver Partition with Portal vein Ligation for Staged Hepatectomy (ALPPS) was introduced recently to achieve a more rapid and effective hypertrophy of the future liver remnant than portal vein embolisation alone but has had a high mortality and morbidity. However, refinements in technique have decreased mortality as was evident in the first registry report in 2014 but the outcomes have been better in patients with colorectal liver metastasis (8% mortality) than in those with hilar CC (27% mortality).40 Olthof et al in a study compared the outcomes in patients undergoing ALPPS for hilar CC and reported a high mortality (48%) in these patients.41 Hence, PVE is still preferred over ALPPS for increasing the size of future remnant in hilar CC.
Surgery
Surgery is the treatment of choice for hilar CCs. The operation has evolved from simple palliative bile duct resection to major hepatectomies which have now been accompanied by a major decrease in morbidity and mortality rates.
Types of Resection
There has been a controversy regarding major hepatectomy for bile duct resections in patients with hilar CC. In the 1980s, these operations were associated with high morbidity and mortality rates and hence minimal resections even if for palliation were considered to be preferred because major resections were associated with a high mortality. A review article by Boeremain (1990) of 499 operations for hilar CC reported a mortality of 12% and a 5-year survival rate of 13%. The survival was higher in patients undergoing major resection but so was the mortality and hence approximately 43% underwent only local resections.42 By the turn of the century surgery of the liver became more standardized and morbidity and mortality started decreasing.43 Lee et al in 2000 reported an operative mortality after major resection of 6.3% on in hilar CC including a number of major hepatectomies with portal vein resection & pancreaticoduodenectomy (111 vs 17 local resections) with. A free tumour margin was achieved in 78%with major resections as compared to 24% in local resections. They reported a 5-year survival of 24% with major resections and a 4-year survival with local resections.44 Blumgart in a publication in 2000 reported changes over three preceding eras with similar findings of improving free tumour margins and survival with major hepatectomies.45 The mortality decreased but liver failure was a common cause of death. The Nagoya experience published in 2005 further validated the trend with 95% of the patients undergoing major resection and showed more R0 resections and long-term survival. An important trend in these series was the lower mortality rates of less than 5% over the last 5 years. There was a regular use of pre-operative biliary drainage till the serum bilirubin level was less than 2 mg/dl and there were a large number of patients undergoing concomitant portal vein resection with long-term survival.46 They also showed successful use of anatomical right hepatic trisectionectomy with caudate lobectomy for hilar CC.47
With an improvement in mortality and survival, local resection gave way to major resection in most series; however, morbidity and mortality mainly due to liver failure remained. Lee et al in 2010 published their experience on 350 patients with hilar CC from 2001 to 2008. They reported high resectability rates and major hepatectomy being done in 88% of the patients. In their series, they reported the use of biliary decompression in 94% of the patients and portal vein embolization (PVE) in 54% patient before extended hepatic resection. They reported an in-hospital mortality of only 1.7% but morbidity of 43%. They attributed the lower mortality and liver failure rates to the use of pre-operative biliary drainage and PVE. Good long-term survival was shown in patients achieving R0 resection and extended resection as compared to parenchymal preserving resection.48 Even the MSKCC experience published in 2010 stressed the need for R0 resection with major hepatectomy for good survivals.49
Nagino et al published their 34-year experience in 574 patients with hilar CC. They reported an increase in the use of major hepatectomies with local resection as rare procedures. They also reported a decrease in mortality rates from 11% to 1.4% over time and showed how improving radiological techniques, the use of preoperative biliary drainage and PVE along with combined vascular resection were responsible for better surgical outcomes and long-term survival. This landmark article showed how the 5-year survival improved (67% in patients with pM0 N0 R0 over the last 5 years) with many of these factors contributing with more R0 resections despite advanced tumour stages.50
These articles have established the role of major hepatectomies in hilar CC as the treatment of choice (Figure 3). The mortality has decreased however the morbidity still high with Nagino et al reporting a 43% morbidity. However, there are still some controversies associated with surgical management.
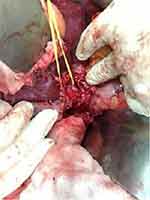 |
Figure 3 Intra-operative image of hilar cholangiocarcinoma showing biliary system (looped). |
Right or Left Hepatectomy
Classically right or left hepatectomy is decided by the disease’s Bismuth stage. Right hepatectomy is performed more for stage IIIa or IV cancers with right predominance while left hepatectomy is done for IIIb or IV with a left predominance. Right lobectomy has certain advantages over the left excision because (a) the left bile duct is longer and easily reconstructed (b) the left portal vein is longer, (c) the right artery runs behind the hilum and has a higher chance of infiltration and (d) the right lobe along with the hilum can be better removed on oncological criteria using the no touch technique as described by Neuhaus.51 However, the disadvantages are a higher tissue mass removed and segment 4 is left which has a high chance of direct infiltration. Hence, the controversy persists. Govil et al tried to circumvent the right artery involvement by doing an artery resection and reconstruction with left lobe resection and showed equivalent outcomes but their study had only a 2-year follow-up and needs further validation. To overcome these, trisectionectomy started becoming more common for oncological clearance.52 Nagino et al in their study showed an increase in the use of trisectionectomy to 30% with a low morbidity and mortality. Hence, more major hepatectomies and reconstruction are being done to achieve R0 resection. Right or left would depend upon the centre’s expertise and local factors like the pattern of tumour spread.
Caudate Lobectomy
The caudate lobe lies embedded between the liver and the inferior vena cava (IVC). Hilar CCsare frequently known to either directly infiltrate the lobe or the caudate duct because of their anatomical relationship and drainage. Mizumoto et al in 1986 identified caudate invasion in 11 patients (direct invasion in 3 and caudate ducts in 8) and suggested the caudate lobe be resected for radical surgery.53 In 1990 Lerut described isolated caudate lobectomy for hilar CC. However, it was Nimura who incorporated caudate lobectomy with major hepatectomy as a routine step for hilar CC as they identified microscopic tumour involvement in nearly all their patients when the caudate lobe was resected. Hence, most centres today routinely resect the caudate lobe along with major hepatectomy; however certain groups do not do so in Bismuth type I and II tumours. Semaya et al reported better R0 resections with right hepatectomy and caudate excision as the right artery was frequently found to have tumour infiltration because of its close proximity to the bile duct.54 Ikeyama further showed that the macroscopic nature of tumour decided the treatment. He suggested major hepatectomy with caudate resection in nodular and infiltrating types while limited hepatic resection could be done for the papillary types of cancer.55 Since the nodular and infiltrating types account for most of these tumours, major hepatectomy with caudate lobe excision is the treatment of choice for most lesions.
Extended Resection
The longitudinal spread of hilar CC in the submucosal layer is estimated to be approximately 0.6 to 2.0 cm while the superficial spread is estimated to be 31–52 mm. It is very difficult for a surgeon to ascertain this spread intra-operatively. Okazaki et al in 2002 found the accuracy, sensitivity, and specificity of intra-operative estimation of the involvement of the proximal duct margin to be only 56.5%, 75.0%, and 46.7%, respectively.56 Mantel et al in 2016 also showed sensitivity and specificity rates of frozen section to be 68% and 97%, respectively.57
On histological examination, positive proximal margins are still present in large numbers after resection. In a retrospective analysis from 2000 to 2009, Lee et al reported invasive carcinoma in 22.8% of the patients and carcinoma in situ (CIS) in 3.7%.58 The Asan group during the same period reported an R1 resection rate of 29.1%. The location of tumour close to the hilar organs and availability of a ductal margin especially on the right side limits how much the resection can be extended. Shingu et al reported that an additional resection of even 5 mm was often difficult and did not improve survival even if it was achieved.59 However, Ribero et al suggested that if a negative margin was attained it was associated with a survival advantage but at the expense of an increased risk of a biliary fistula.60 A multicentre study by Zhang et al in 2018 showed a survival advantage with re-resection to achieve R0 resections with equivalent morbidity and mortality rates.61 These results are at centres which have high volumes and surgeons and pathologists who have a large experience in dealing with hilar CCs and cannot be safely applied in other institutions.
Vascular Resection
The portal vein and hepatic artery are close to the liver hilum and their invasion is not uncommon in hilar CC. Initially, their involvement was thought to make them unresectable, but at the turn of the century, portal vein resection became commonly performed in hilar CCs at high volume centres. Hemming et al in 2011 reported no increase in morbidity and mortality with an increase in survival of patients who achieved R0 resection with portal vein resection.62 At the same time, Nagino et al reported portal vein resection in 36% of the patients. They also reported wedge resection as more commonly done in the 1990s and now most undergo resection of a short segment of the portal vein. The procedure has now become so standardized that some surgeons have suggested that it should be done in nearly all patients in order to achieve an R0 status. However, it should be noted that these results are achieved in high volume centres and morbidity and mortality may increase in centres in which only a few such procedures are performed. The importance of experience was highlighted in a meta-analysis where a subgroup analysis showed patients who underwent portal vein resection after 2007 at high volume centres did not have a difference in postoperative deaths compared to those who did not. However, the study did not show a survival advantage but all these patients had more advanced disease.63 A recent expert consensus statement also advises that portal vein resection in hilar CC should not be done routinely and be undertaken in high volume centres with expertise and after intra-operative evaluation.64
Hepatic artery resection has been undertaken by only a few centres usually combined with portal vein resection. Some studies initially showed poor outcomes with artery resection but Nagino et al in 2013 showed good results. However, a systemic review pointed to higher morbidity and mortality rates with artery resection.65 Hence, the current evidence does not support artery resection and this should be done in a few high volume centres which have the expertise.
Staging Laparoscopy
With the improvements in diagnostic CT/MR, the yield of staging laparoscopy in changing the management in patients with hilar cholangiocarcinoma has decreased to 13–17%.66,67 Song et al and Nagino et al have not described regular use of staging laparoscopy in their studies. However, Jarnagin et al have shown selective use in patients with Blumgart T2 and T3 lesions, or if there is advanced disease on preoperative imaging. The use increased in the subset of patients being considered for liver transplantation where staging laparoscopy has become an essential tool but there is little evidence to prescribe its regular use in patients only undergoing tumour resection.
Lymphadenectomy
The lymph nodes along the portal vein and hepatic artery are always resected and have been shown to influence the prognosis. Some centres have performed extending lymphadenectomy beyond the hepaticoduodenal ligament (HDL) and others have even suggested lymph node clearance till aortic bifurcation although with an increase in accompanying morbidity. Nagino et al showed survival even in patients with involved para-aortic lymph nodes and even they reported routine resection papa-aortic nodes upto 2000 but later only for sampling only. Song et al in a large series also reported lymphadenectomy along the HDL, peri-pancreatic and coeliac axis. The inference from most of these studies is that lymphadenectomy has had no influence on survival but adequate lymphadenectomy is necessary for staging. Recent studies have debated the number of lymph nodes for adequate sampling. A SEER data base analysis reported retrieval of 13 lymph nodes for adequate sampling.68 A systemic analysis identified 7 lymph nodes for adequate staging and even said that 15 lymph nodes are not required as suggested for seventh AJCC staging.69 But these were all retrospective analyses and more studies are needed to yield robust guidelines. Most centres at present perform HDL lymph node retrieval along with nodes along the peri-pancreatic region, coeliac node removal and sampling from the aortic lymph nodes. Sampling is performed to indicate widespread disease and for abandoning a proposed liver transplant procedure.
Outcomes After Resection
The outcomes after surgery for cholangiocarcinoma have improved with standardization and improvement in major liver surgery techniques by the turn of the twentieth century. Most of the articles from the major centres are retrospective covering a decade. A common point in these is the improvement in resectability and decrease in mortality rates overtime (Table 1). However, the morbidity rates are still high largely from bile leaks, sepsis and the small size of the remnant liver. R0 resections and lymph node positivity have been the two most important factors influencing the overall survival. A number of factors have been associated with increased R0 resection like major hepatectomy, vascular resection, use of PVE and preoperative biliary drainage, better frozen section analysis and centre experience. These modifications have increased resectability rates in the curative range from <50% to now approximately 80% and are responsible for the rise in overall long-term survivals. However, these procedures are still complicated and should be done at high volume centres.
Liver Transplantation
Resection has been the mainstay of treatment for CC especially for tumours below the hilum however resectability is often compromised because of the locally advanced nature of the disease. Liver transplantation was tried initially but had poor outcomes with 5-year survival of 30% and high recurrence rates of more than 50%. The Mayo Clinic developed a protocol in 1993 in which they combined neo-adjuvant chemotherapy and radiation with liver transplantation for unresectable patients. Of the 19 patients, 11 underwent surgery and only 1 had recurrence.70 Their follow-up results published in 2005 reported on 38 patients undergoing liver transplantation for hilar CC with a 5-year survival of 82%. The transplanted patients had better survival results and less recurrence rates than those who had resection.71 However, these were a highly selected group of patients (early stage node negative and PSC) who underwent strict protocol adherence and were still operable. Another criticism at the time was that 16 patients did not have tumours at explant though 8 of them had a positive diagnosis preoperatively. The scarcity of donor organs has also discouraged the allocation of livers for CC. The good outcomes reported by the Mayo clinic have now prompted a selected group of patients to undergo strict protocol adherence and MELD exception points were also allotted to these patients.72
Prompted by the Mayo Clinic results many other centres have also started liver transplantation for hilar CCs. Ethun et al published their result from 10 institutions where they showed better 5-year survival (64% vs 18%) for patients undergoing liver transplantation compared to those who had resection for hilar CC. They also showed a better 5-year survival with transplantation in smaller and node-negative hilar CCs.73 However, most of this data is still retrospective and has not come from multiple centres across the globe and till more prospective studies are able to justify transplantation, resection remains the treatment of choice for resectable disease and for selected patients with locally advanced disease who strictly follow the Mayo Clinic protocol can undergo liver transplantation in high volume centres.
Adjuvant Treatment
Neoadjuvant
Resection has been the treatment of choice for resectable hilar CC; however, the resectability rate is low and there is a significant group of patients with locally advanced disease. Retrospective studies have shown that neoadjuvant treatment may help in downstaging the tumours and improving resectability but this is not associated with improved survival.74 A systematic review of eight full articles and two abstracts showed some advantage in unresectable patients by converting a few of them to resectability; however, the evidence is not of high quality with no common protocols followed in most of these studies.75 Even in the Mayo Clinic, the locally advanced patients have shown a survival advantage and resectability with their strict protocol which includes neoadjuvant chemotherapy and radiation. Chemotherapy has shown improvement with gemcitabine-based regimes as compared to 5 FU and are being analysed in more Phase II studies. Till more RCTs or prospective studies show better outcomes neo-adjuvant therapy can be used for only locally advanced tumors.
Adjuvant
Recurrence after resection for hilar CC is common. Studies, mostly retrospective, have tried to assess the site and duration for recurrence to occur. The US Extrahepatic Biliary Consortium showed distant metastasis as the predominant site of recurrence. Adjuvant therapy has shown its benefits in a number of reports, but like the neoadjuvant studies, most of the reports are retrospective analyses with different drugs and no defined protocol. A number of randomized trials like ACTICCA-1, PRODIGE-12 and BILCAP are underway to assess the impact of adjuvant treatment on hilar CC. From the literature gathered so far, the conclusions that can be made are that adjuvant treatment is beneficial and more so in patients with margin and lymph nodes positivity. Gemcitabine with cisplatin/oxaliplatin has shown better results than 5FU-based regimes. Radiotherapy alone is not as advantageous as chemotherapy and radiotherapy together.
Conclusions
Surgery still remains the treatment of choice for hilar CC and reports from high volume centres have shown an increase in resectability, R0 resection, a decrease in mortality with an improvement in 5-year survival rates; however, the morbidity remains high pointing towards the complexity of the management of these difficult lesions. PVE and pre-operative biliary drainage have had a major impact on outcomes. Major liver resection with caudate lobe removal remains the standard operation and procedures like routine vascular resection and liver transplant should be carried out in experienced centres only. Improvements in adjuvant therapy may lead to a standardized protocol and improvement in survival.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Klatskin G. Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis. An unusual tumor with distinctive clinical and pathological features. Am J Med. 1965;38:241–256. doi:10.1016/0002-9343(65)90178-6
2. Sripa B, Pairojkul C. Cholangiocarcinoma: lessons from Thailand. Curr Opin Gastroenterol. 2008;24(3):349–356. doi:10.1097/MOG.0b013e3282fbf9b3
3. Saha SK, Zhu AX, Fuchs CS, Brooks GA. Forty-year trends in cholangiocarcinoma incidence in the U.S.: intrahepatic disease on the rise. Oncologist. 2016;21(5):594–599. doi:10.1634/theoncologist.2015-0446
4. Khan SA, Emadossadaty S, Nimzing G, et al. Rising trends in cholangiocarcinoma: is the ICD classification system misleading us? J Hepatol. 2012;56(4):848–854. doi:10.1016/j.jhep.2011.11.015
5. Shin HR, Oh JK, Lim MK, et al. Descriptive epidemiology of cholangiocarcinoma and clonorchiasis in Korea. J Korean Med Sci. 2010;25(7):1011–1016. doi:10.3346/jkms.2010.25.7.1011
6. Chapman MH, Webster GJ, Bannoo S, Johnson GJ, Wittmann J, Pereira SP. Cholangiocarcinoma and dominant strictures in patients with primary sclerosing cholangitis: a 25-year single-centre experience. Eur J Gastroenterol Hepatol. 2012;24(9):1051–1058. doi:10.1097/MEG.0b013e3283554bbf
7. Claessen MM, Vleggaar FP, Tytgat KM, Siersema PD, van Buuren HR. High lifetime risk of cancer in primary sclerosing cholangitis. J Hepatol. 2009;50(1):158–164. doi:10.1016/j.jhep.2008.08.013
8. Chalasani N, Baluyut A, Ismail A, et al. Cholangiocarcinoma in patients with primary sclerosing cholangitis: a multicenter case-control study. Hepatology. 2000;31(1):7–11. doi:10.1002/(ISSN)1527-3350
9. Lee SE, Jang JY, Lee YJ, et al. Choledochal cyst and associated malignant tumors in adults. A multicenter survey in South Korea. Arch Surg. 2011;146(10):1178–1184. doi:10.1001/archsurg.2011.243
10. Hove AT, de Meijer VT, Hulscher JBF, de Kleine RHJ. Meta‐analysis of risk of developing malignancy in congenital choledochal malformation. Br J Surg. 2018;105(5):482–490.
11. Ohashi T, Wakai T, Kubota M, et al. Risk of subsequent biliary malignancy in patients undergoing cyst excision for congenital choledochal cysts. J Gastroenterol Hepatol. 2013;28(2):243–247. doi:10.1111/jgh.2013.28.issue-2
12. Li M, Li J, Li P, et al. Hepatitis B virus infection increases the risk of cholangiocarcinoma: a meta-analysis and systematic review. J Gastroenterol Hepatol. 2012;27(10):1561–1568. doi:10.1111/jgh.2012.27.issue-10
13. Li H, Hu B, Zhou ZQ, et al. Hepatitis C virus infection and the risk of intrahepatic cholangiocarcinoma and extrahepatic cholangiocarcinoma: evidence from a systematic review and meta-analysis of 16 case-control studies. World J Surg Oncol. 2015;13:161. doi:10.1186/s12957-015-0583-9
14. Sorensen HT, Friis S, Olsen JH, et al. Risk of liver and other types of cancer in patients with cirrhosis: a nationwide cohort study in Denmark. Hepatology. 1998;28(4):921–925. doi:10.1002/hep.v28:4
15. Liu CL. Primary biliary stones: diagnosis and management. World J Surg. 1998;22:1162–1166. doi:10.1007/s002689900536
16. Suzuki Y. Predictive factors for cholangiocarcinoma associated with hepatolithiasis determined on the basis of Japanese multicenter study. Hepatol Res. 2012;42:166–170. doi:10.1111/j.1872-034X.2011.00908.x
17. Endo I, Gonen M, Yopp AC, et al. Intrahepatic cholangiocarcinoma: rising frequency, improved survival, and determinants of outcome after resection. Ann Surg. 2008;248(1):84–96. doi:10.1097/SLA.0b013e318176c4d3
18. Basso D, Meggiato T, Fabris C, et al. Extra-hepatic cholestasis determines a reversible increase of glycoprotein tumour markers in benign and malignant disease. Eur J Clin Invest. 1992;22:800–804. doi:10.1111/j.1365-2362.1992.tb01449.x
19. Liang B. Diagnostic accuracy of serum CA19-9 in patients with cholangiocarcinoma: a systematic review and meta-analysis. Med Sci Monit. 2015;21:3555–3563. doi:10.12659/MSM.895040
20. Levy C, Lymp J, Angulo P, Gores GJ, Larusso N, Lindor KD. The value of serum CA 19-9 in predicting cholangiocarcinomas in patients with primary sclerosing cholangitis. Dig Dis Sci. 2005;50:1734–1740. doi:10.1007/s10620-005-2927-8
21. Alvaro D. Serum and bile biomarkers for cholangiocarcinoma. Curr Opin Gastroenterol. 2009;25:279–284. doi:10.1097/MOG.0b013e328325a894
22. Loosen SH, Roderburg C, Kauertz KL, et al. CEA but not CA19-9 is an independent prognostic factor in patients undergoing resection of cholangiocarcinoma. Sci Rep. 2017;7(1):16975. doi:10.1038/s41598-017-17175-7
23. Unno M, Okumoto T, Katayose Y, et al. Preoperative assessment of hilar cholangiocarcinoma by multidetector row computed tomography. J Hepatobiliary Pancreat Surg. 2007;14:434–440. doi:10.1007/s00534-006-1191-4
24. Park HS, Lee JM, Choi JY, et al. Preoperative evaluation of bile duct cancer: MRI combined with MR cholangiopancreatography versus MDCT with direct cholangiography. AJR Am J Roentgenol. 2008;190(2):396–405. doi:10.2214/AJR.07.2310
25. Kim JY, Kim MH, Lee TY, et al. Clinical role of 18F-FDG PET-CT in suspected and potentially operable cholangiocarcinoma: a prospective study compared with conventional imaging. Am J Gastroenterol. 2008;103(5):1145–1151. doi:10.1111/j.1572-0241.2007.01710.x
26. Ruys AT, Bennink RJ, van Westreenen HL, et al. FDG-positron emission tomography/computed tomography and standardized uptake value in the primary diagnosis and staging of hilar cholangiocarcinoma. HPB (Oxford). 2011;13(4):256–262. doi:10.1111/j.1477-2574.2010.00280.x
27. Bismuth H, Nakache R, Diamond T. Management strategies in resection for hilar cholangiocarcinoma. Ann Surg. 1992;215(1):31–38. doi:10.1097/00000658-199201000-00005
28. Jarnagin WR, Fong Y, DeMatteo RP, et al. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234(4):507–517. doi:10.1097/00000658-200110000-00010
29. Blechacz BR, Sanchez W. Gores GJ A conceptual proposal for staging ductal cholangiocarcinoma. Curr Opin Gastroenterol. 2009;25(3):238–239. doi:10.1097/MOG.0b013e3283292383
30. Deoliveira ML, Schulick RD, Nimura Y, et al. New staging system and a registry for perihilar cholangiocarcinoma. Hepatology. 2011;53(4):1363–1371. doi:10.1002/hep.24227
31. Kennedy TJ, Yopp A, Qin Y, et al. Role of preoperative biliary drainage of liver remnant prior to extended liver resection for hilar cholangiocarcinoma. HPB. 2009;11:445–451. doi:10.1111/j.1477-2574.2009.00090.x
32. Wiggers JK, Koerkamp BG, Coelen RJ, et al. Preoperative biliary drainage in perihilar cholangiocarcinoma: identifying patients who require percutaneous drainage after failed endoscopic drainage. Endoscopy. 2015;47:1124–1131. doi:10.1055/s-00000012
33. Kloek JJ, van der Gaag NA, Aziz Y, et al. Endoscopic and percutaneous preoperative biliary drainage in patients with suspected hilar cholangiocarcinoma. J Gastrointest Surg. 2010;14(1):119–125. doi:10.1007/s11605-009-1009-1
34. Kawakami H, Kuwatani M, Onodera M, et al. Endoscopic nasobiliary drainage is the most suitable preoperative biliary drainage method in the management of patients with hilar cholangiocarcinoma. J Gastroenterol. 2011;46(2):242–248. doi:10.1007/s00535-010-0298-1
35. Arakura N, Takayama M, Ozaki Y, et al. Efficacy of preoperative endoscopic nasobiliary drainage for hilar cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 2009;16(9):473–477. doi:10.1007/s00534-009-0076-8
36. Abdalla EK, Denys A, Chevalier P, Nemr RA, Vauthey JN. Total and segmental liver volume variations: implications for liver surgery. Surgery. 2004;135(4):404–410. doi:10.1016/j.surg.2003.08.024
37. Leung U, Simpson AL, Araujo RL, et al. Remnant growth rate after portal vein embolization is a good early predictor of post-hepatectomy liver failure. J Am Coll Surg. 2014;219(4):620–630. doi:10.1016/j.jamcollsurg.2014.04.022
38. Shindoh J, Truty MJ, Aloia TA, et al. Kinetic growth rate after portal vein embolization predicts posthepatectomy outcomes: toward zero liver-related mortality in patients with colorectal liver metastases and small future liver remnant. J Am Coll Surg. 2013;216(2):201–209. doi:10.1016/j.jamcollsurg.2012.10.018
39. Fan ST, Lo CM, Liu CL, et al. Hepatectomy for hepatocellular carcinoma: toward zero hospital deaths. Ann Surg. 1999;229(3):322–330. doi:10.1097/00000658-199903000-00004
40. Schadde E, Ardiles V, Robles-Campos R, et al.; ALPPS Registry Group. Early survival and safety of ALPPS: first report of the International ALPPS registry. Ann Surg. 2014;260(5):829–836. doi:10.1097/SLA.0000000000000947
41. Olthof PB, Coelen RJS, Wiggers JK, et al. High mortality after ALPPS for perihilar cholangiocarcinoma: case-control analysis including the first series from the international ALPPS registry. HPB (Oxford). 2017;19(5):381–387. doi:10.1016/j.hpb.2016.10.008
42. Boerma EJ. Research into the results of resection of hilar bile duct cancer. Surgery. 1990;108(3):572–580.
43. Miyazaki M, Ito H, Nakagawa K, et al. Parenchyma-preserving hepatectomy in the surgical treatment of hilar cholangiocarcinoma. Am Coll Surg. 1999;189(6):575–583. doi:10.1016/S1072-7515(99)00219-7
44. Lee SG, Lee YJ, Park KM, Hwang S, Min PC. One hundred and eleven liver resections for hilar bile duct cancer. J Hepatobiliary Pancreat Surg. 2000;7(2):135–141. doi:10.1007/s005340050167
45. Saldinger PF, Blumgart LH. Resection of hilar cholangiocarcinoma–a European and United States experience. J Hepatobiliary Pancreat Surg. 2000;7(2):111–114. doi:10.1007/s005340050163
46. Nishio H, Nagino M, Nimura Y. Surgical management of hilar cholangiocarcinoma: the Nagoya experience. HPB (Oxford). 2005;7(4):259–262. doi:10.1080/13651820500373010
47. Nagino M, Kamiya J, Arai T, Nishio H, Ebata T, Nimura Y. “Anatomic” right hepatic trisectionectomy (Extended right hepatectomy) with caudate lobectomy for hilar cholangiocarcinoma. Ann Surg. 2006;243:28–32. doi:10.1097/01.sla.0000193604.72436.63
48. Lee SG, Song GW, Hwang S, et al. Surgical treatment of hilar cholangiocarcinoma in the new era: the Asan experience. J Hepatobiliary Pancreat Sci. 2010;17(4):476–489. doi:10.1007/s00534-009-0204-5
49. Rocha FG, Matsuo K, Blumgart LH, Jarnagin WR. Hilar cholangiocarcinoma: the Memorial Sloan-Kettering Cancer Center experience. J Hepatobiliary Pancreat Sci. 2010;17(4):490–496. doi:10.1007/s00534-009-0205-4
50. Nagino M, Ebata T, Yokoyama Y, et al. Evolution of surgical treatment for perihilar cholangiocarcinoma: a single-center 34-year review of 574 consecutive resections. Ann Surg. 2013;258(1):129–140. doi:10.1097/SLA.0b013e3182708b57
51. Neuhaus P, Thelen A, Jonas S, et al. Oncological superiority of hilar en bloc resection for the treatment of hilar cholangiocarcinoma. Ann Surg Oncol. 2011;19(5):1602–1608. doi:10.1245/s10434-011-2077-5
52. Govil S, Bharatan A, Rammohan A, et al. Liver resection for perihilar cholangiocarcinoma - why left is sometimes right. HPB (Oxford). 2016;18(7):575–579. doi:10.1016/j.hpb.2016.05.002
53. Mizumoto R, Kawarada Y, Suzuki H. Surgical treatment of hilar carcinoma of the bile duct. Surg Gynecol Obstet. 1986;162(2):153–158.
54. Seyama Y, Kubota K, Sano K, et al. Long-term outcome of extended hemihepatectomy for hilar bile duct cancer with no mortality and high survival rate. Ann Surg. 2003;238(1):73–83. doi:10.1097/01.SLA.0000074960.55004.72
55. Ikeyama T, Nagino M, Oda K, Ebata T, Nishio H, Nimura Y. Surgical approach to bismuth Type I and II hilarcholangiocarcinomas: audit of 54 consecutive cases. Ann Surg. 2007;246(6):1052–1057. doi:10.1097/SLA.0b013e318142d97e
56. Okazaki Y, Horimi T, Kotaka M, Morita S, Takasaki M. Study of the intrahepatic surgical margin of hilar bile duct carcinoma. Hepatogastroenterology. 2002;49(45):625–627.
57. Mantel HT, Westerkamp AC, Sieders E, et al. Intraoperative frozen section analysis of the proximal bile ducts in hilar cholangiocarcinoma is of limited value. Cancer Med. 2016;5(7):1373–1380. doi:10.1002/cam4.693
58. Lee JH, Hwang DW, Lee SY, Park KM, Lee YJ. The proximal margin of resected hilar cholangiocarcinoma: the effect of microscopic positive margin on long-term survival. Am Surg. 2012;78(4):471–477.
59. Shingu Y, Ebata T, Nishio H, Igami T, Shimoyama Y, Nagino M. Clinical value of additional resection of a margin-positive proximal bile duct in hilar cholangiocarcinoma. Surgery. 2010;147(1):49–56. doi:10.1016/j.surg.2009.06.030
60. Ribero D, Amisano M, Lo Tesoriere R, Rosso S, Ferrero A, Capussotti L. Additional resection of an intraoperative margin-positive proximal bile duct improves survival in patients with hilar cholangiocarcinoma. Ann Surg. 2011;254(5):776–781. doi:10.1097/SLA.0b013e3182368f85
61. Zhang XF, Squires MH, Bagante F, et al. The impact of intraoperative re-resection of a positive bile duct margin on clinical outcomes for hilar cholangiocarcinoma. Ann Surg Oncol. 2018;25(5):1140–1149. doi:10.1245/s10434-018-6382-0
62. Hemming AW, Mekeel K, Khanna A, Baquerizo A, Kim RD. Portal vein resection in management of hilar cholangiocarcinoma. J Am Coll Surg. 2011;212(4):604–613. doi:10.1016/j.jamcollsurg.2010.12.028
63. Wu XS, Dong P, Gu J, et al. Combined portal vein resection for hilar cholangiocarcinoma: a meta-analysis of comparative studies. J Gastrointest Surg. 2013;17(6):1107–1115. doi:10.1007/s11605-013-2202-9
64. Mansour JC, Aloia TA, Crane CH, Heimbach JK, Nagino M, Vauthey JN. Hilar Cholangiocarcinoma: expert consensus statement. HPB (Oxford). 2015;17(8):691–699. doi:10.1111/hpb.12450
65. Abbas S, Sandroussi C. Systematic review and meta-analysis of the role of vascular resection in the treatment of hilar cholangiocarcinoma. HPB (Oxford). 2013;15(7):492–503. doi:10.1111/j.1477-2574.2012.00616.x
66. Rotellar F, Pardo F. Laparoscopic staging in hilar cholangiocarcinoma: is it still justified? World J Gastrointest Oncol. 2013;5(7):127–131. doi:10.4251/wjgo.v5.i7.127
67. Ruys AT, Busch OR, Gouma DJ, van Gulik TM. Staging laparoscopy for HilarCholangiocarcinoma: is it still worthwhile? Ann Surg Oncol. 2011;18(9):2647–2653. doi:10.1245/s10434-011-1576-8
68. Mao K, Liu J, Sun J, et al. Patterns and prognostic value of lymph node dissection for resected perihilar cholangiocarcinoma. J Gastroenterol Hepatol. 2016;31(2):417–426. doi:10.1111/jgh.2016.31.issue-2
69. Kambakamba P, Linecker M, Slankamenac K, DeOliveira ML. Lymph node dissection in resectable perihilar cholangiocarcinoma: a systematic review. Am J Surg. 2015;210(4):694–701. doi:10.1016/j.amjsurg.2015.05.015
70. De Vreede I, Steers JL, Burch PA, et al. Prolonged disease-free survival after orthotopic liver transplantation plus adjuvant chemoirradiation for cholangiocarcinoma. Liver Transpl. 2000;6(3):309–316. doi:10.1053/lv.2000.6143
71. Rea DJ, Heimbach JK, Rosen CB, et al. Liver transplantation with neoadjuvant chemoradiation is more effective than resection for hilar cholangiocarcinoma. Ann Surg. 2005;242(3):451–458. doi:10.1097/01.sla.0000179678.13285.fa
72. Gores GJ, Gish RG, Sudan D, Rosen CB. Model for end-stage liver disease (MELD) exception for cholangiocarcinoma or biliary dysplasia. Liver Transpl. 2006;12(12 Suppl 3):S95–S97. doi:10.1002/lt.20965
73. Ethun CG, Lopez-Aguiar AG, Anderson DJ, et al. Transplantation versus resection for hilar cholangiocarcinoma: an argument for shifting treatment paradigms for resectable disease. Ann Surg. 2018;267(5):797–805. doi:10.1097/SLA.0000000000002574
74. Jung JH, Lee HJ, Lee HS, et al. Benefit of neoadjuvant concurrent chemoradiotherapy for locally advanced perihilar cholangiocarcinoma. World J Gastroenterol. 2017;23(18):3301–3308. doi:10.3748/wjg.v23.i18.3301
75. Grendar J, Grendarova P, Sinha R, Dixon E. Neoadjuvant therapy for downstaging of locally advanced hilar cholangiocarcinoma: a systematic review. HPB (Oxford). 2014;16(4):297–303. doi:10.1111/hpb.12150
76. Song SC, Choi DW, Kow AW, et al. Surgical outcomes of 230 resected cholangiocarcinomas in a single centre. ANZ J Surg. 2013;83(4):268–274.
77. Cho MS, Kim SH, Park SW, et al. Surgical outcomes and predicting factors of curative resection in patients with hilar cholangiocarcinoma: 10-year single-institution experience. J Gastrointest Surg. 2012;19(9):1672–1679.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
