Back to Journals » ImmunoTargets and Therapy » Volume 4
Management of patients with non-Hodgkin’s lymphoma: focus on adoptive T-cell therapy
Authors Perna S, Huye L, Savoldo B
Received 9 May 2014
Accepted for publication 16 July 2014
Published 19 March 2015 Volume 2015:4 Pages 55—63
DOI https://doi.org/10.2147/ITT.S31389
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Serena Kimi Perna,1 Leslie E Huye,1,† Barbara Savoldo1,2
1Center for Cell and Gene Therapy, Baylor College of Medicine, Houston Methodist Hospital, Houston, TX, 2Department of Pediatrics, Texas Children's Hospital, Houston, TX, USA
†Leslie E Huye passed away on January 1st, 2015
Abstract: Non-Hodgkin's lymphoma (NHL) represents a heterogeneous group of malignancies with high diversity in terms of biology, clinical responses, and prognosis. Standard therapy regimens produce a 5-year relative survival rate of only 69%, with the critical need to increase the treatment-success rate of this patient population presenting at diagnosis with a median age of 66 years and many comorbidities. The evidence that an impaired immune system favors the development of NHL has opened the stage for new therapeutics, and specifically for the adoptive transfer of ex vivo-expanded antigen-specific T-cells. In this review, we discuss how T-cells specific for viral-associated antigens, nonviral-associated antigens expressed by the tumor, T-cells redirected through the expression of chimeric antigen receptors, and transgenic T-cell receptors against tumor cells have been developed and used in clinical trials for the treatment of patients with NHLs.
Keywords: adoptive immunotherapy, cytotoxic T lymphocytes (CTLs), chimeric antigen receptor (CAR), transgenic T-cell receptors
Background
Non-Hodgkin’s lymphomas (NHLs) comprise a heterogeneous group of malignancies of the lymphoid tissues. In the US, the great majority (80%–85%) present with a B-cell phenotype, while 15%–20% of NHLs are derived from T-cell subtypes and rarely from natural killer (NK) cells. Because of the high heterogeneity of diseases included in the NHL category, the clinical presentation varies greatly according to the subtype and area of involvement, ranging from indolent to highly aggressive forms.
In 2013, 69,740 new cases of NHL were estimated, with 19,020 deaths, making NHL the seventh-leading type of cancer in the US and accounting for 4% of new cases and 3% of cancer-related deaths. The incidence of NHL is currently rising, with a 3.7% yearly percentage increase recorded between 1975 and 1991 and a 0.3% yearly increase from 1992 to 2007. This has been at least in part attributed to the strict relationship between the occurrence of NHL and a competent immune system, with diseases or treatments that weaken the immune system often favoring the development of NHL. Indeed, patients affected with human immunodeficiency virus (HIV), inherited immunodeficiency syndromes, or autoimmune diseases, or treated with high-dose chemotherapy, as well as recipients of organ-transplant or hematopoietic stem cells, have a higher risk of developing NHL. Based on similar mechanisms, infections also play an important role in the development of some lymphomas by impairing multiple immune functions or inducing chronic inflammatory responses. Specifically, Epstein–Barr virus (EBV) is strongly associated with the occurrence of Burkitt’s lymphoma, nasal NK-T lymphomas, and posttransplant lymphoproliferative diseases (PTLDs), Helicobacter pylori is a risk factor for gastric mucosal associated lymphomas, hepatitis C virus is associated with splenic marginal-zone lymphomas, Borrelia burgdorferi with cutaneous mucosa-associated lymphoid-tissue lymphomas, and Chlamydophila psittaci with ocular adrenal lymphomas.1
All these conditions are associated, to different extents, with a defect of the B-cell, T-cell, or NK-cell compartments that play a central role in patrolling the body and preventing the proliferation of transformed cell clones. For lymphomas associated with conditions that directly affect the immune system (HIV, primary immunodeficiency, chemotherapy), a failure in immunosurveillance leads to the development of NHL, while in the case of infection-associated lymphomas, dysfunctional immunosurveillance needs to be associated with chronic antigen exposure and the presence of oncogenic viruses. Obviously, NHL can also develop in previously healthy individuals. In this case, genetics and defective deoxyribonucleic acid-damage responses play a relevant role in the pathogenesis of the diseases.2
The median age at diagnosis for NHL is 66 years, with more than 9% of patients over the age of 85 years. Importantly, the patient’s comorbidities related to this older age often restrict the applicability of standard chemotherapy regimens.3 The response rates of patients with NHL to conventional chemotherapy are generally greater than 50%. However, despite the numerous drugs and combinations available, a significant fraction of NHL patients eventually relapse due to incomplete eradication of tumor cells. Numerous regimens have been studied as options for salvage therapy and for aggressive NHL, but despite the inclusion of high-dose chemotherapy and autologous stem cell transplant (auto-SCT), only 50% of patients survive in the long term. Outcomes are even worse in patients with chemotherapy-resistant disease and for those ineligible for transplant because of age or comorbidities, with an expected survival of less than 1 year. Allogeneic SCT offers lower relapse rates compared to auto-SCT, but the myeloablative pretransplant regimens are associated with high treatment-related mortality, which precludes its use in many patients.4
Therefore, important challenges remain for the management of patients that fail complete tumor eradication postchemotherapies and/or are ineligible for transplant. Specifically, for older or frailer patients, new less toxic strategies need to be developed and explored to overcome treatment failure.5
NHL and immunotherapy
Immunotherapy-based approaches have significantly impacted the outcome of NHL. These therapies range from monoclonal antibodies (mAbs), Ab–drug conjugates, radioimmunotherapy, and small-molecule inhibitors targeting cell survival and growth pathways.
Rituximab (the chimeric anti-CD20 Ab) is the pivotal example of mAb therapy for NHL. Thanks to its dramatic impact on the overall survival and response rate as front-line therapy, this drug is now part of the standard of care for patients with B-cell lymphomas.6 In the rituximab era, limited data are available on the efficacy of salvage therapy for relapsed/refractory NHL, and the role of rituximab in salvage regimens, when already included in primary therapy, remains unclear.7,8
In some cases, mAbs are conjugated with cytotoxic agents to enhance the therapeutic efficacy of the original Ab and ensure limited side effects.9 An example of Ab–drug conjugate therapy is represented by brentuximab vedotin, (SGN-35), an mAb targeting CD30 used successfully in the last few years for NHLs, such as anaplastic large-cell lymphoma or peripheral T-cell lymphomas, that express the CD30 molecule. This treatment has produced a 41% complete remission (CR) rate in relapsed patients, although the median duration of response has been often limited.10
Alternatively, mAbs can be chemically conjugated to radioactive isotopes for tumor targeting and delivery, or fragments of two mAbs can be coupled to provide increased tumor-targeting specificity through binding of two tumor-specific antigens and enhanced cytotoxic efficacy by engagement of multiple effector mechanisms. The best example of this bispecific Ab technology is blinatumomab, which couples CD19 (a B-cell marker) and CD3 (a T-cell engager) for recruitment of T-cell activity against B-cell malignancies. The first Phase I study of 62 NHL patients demonstrated an overall response rate of 82% across NHL subtypes, maintained up to 3 years for 60% of responders. Unfortunately, severe side effects (encephalopathy, tremor, and aphasia) require discontinuation of therapy in about 20% of patients.11
Adoptive T-cell therapy for NHL
In parallel with the aforementioned approaches, strategies relying on restoring immune cell activities have been developed for the cure of NHL. Specifically, adoptive immunotherapy offers minimal toxicity but significant potential to control NHL disease. In-depth knowledge on the specific characteristics of current T-cell-based approaches available in the clinic and on the advantages and weaknesses of each specific strategy can aid in the management of NHL patients postchemotherapy.
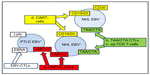
T-cell-based approaches currently available to treat NHL are (Figure 1 and Table 1):
- EBV-specific T-cells
- tumor-associated antigen (TAA)-specific T-cells
- transgenic T-cell receptor (TCR)-engineered T-cells
- chimeric antigen receptor (CAR)-engineered T-cells.
  | Table 1 Reported clinical trials for NHL patients |
Epstein–Barr virus-specific T-cells for the treatment of NHL
Infections are known to play an important role in the development of some lymphomas by impairing multiple immune functions or inducing chronic inflammatory responses. One of the most common infectious agents associated with NHL is EBV. This is a human γ1-herpesvirus with oncogenic potential responsible for causing a self-limiting disease known as infectious mononucleosis when acquired during adolescence or an asymptomatic infection if acquired early in life. Infection of B cells is accompanied by the expression of the highly immunogenic EBV-associated antigens (latent EBNAs and lytic antigens). Cytotoxic T-cells (CTLs) targeting these antigens limit the uncontrolled expansion of the infected B cells. However, the virus can be carried lifelong in the memory B-cell compartment in a latent state by expressing only few poorly immunogenic antigens (EBNA-1 and LMP1 and LMP2). If T-cells are deficient in numbers or functionality, such as post hematopoietic stem cell or solid-organ transplantation, respectively, EBV reactivates and promotes the unrestricted proliferation of infected cells, leading to the development of a group of diseases collectively referred as PTLDs.12
As these tumors express several of the same highly immunogenic antigens commonly present in EBV-infected B cells, the restoration of CTLs targeting these antigens was anticipated as a potential cure. The approach was first explored at the Memorial Sloan Kettering Cancer Center following the transfer of unmanipulated T lymphocytes from EBV-seropositive donors. Although effective, this strategy was hampered by the high risk of graft-versus-host disease.13 A few years later, Rooney et al refined the approach by specifically expanding T-cells targeting only EBV-associated antigens (EBV-specific CTLs), and showed that the infusion of EBV-specific CTLs in patients with PTLD was not only effective but also free from side effects.14 This pioneering work provided the rationale for extending the use of EBV-specific CTLs to many more EBV-associated malignancies.
As the etiology of approximately 30%–40% of all NHL is recognized to be associated with EBV,15 it was important to apply the concept of EBV-CTL adoptive immunotherapy for NHL, though to be applicable for NHL, the process for the generation of the EBV-specific CTLs had to be slightly modified. First, unlike PTLD, which expresses the highly immunogenic EBV antigens, NHL almost exclusively expresses the less immunogenic LMP1 and LMP2 antigens. T-cell precursors targeting these weak antigens are usually very limited in the peripheral blood of individuals and patients, requiring the development of more complex protocols for CTL production.
The relevance of LMP2 as a target antigen has been underlined by the high rate of clinical responses in patients infused with autologous EBV-specific CTLs enriched in LMP2-specific precursors. Of the eight NHL patients treated, none experienced immediate or long-term toxicities, four patients remained in CR, two patients treated with disease achieved durable CR (for 9 and 10 months, respectively), and another one had a very good partial response, according to the Response Evaluation Criteria in Solid Tumors.16 To further improve the efficacy of the approach, EBV-specific CTLs were enriched for both LMP1 and LMP2 T-cell precursors, and in a comparative study, 33 patients were infused with CTLs targeting both LMP1 and LMP2 antigens. At a median follow up of 3.1 years, none of the 29 patients treated in CR experienced disease relapse. The 2-year event-free survival (EFS) was 82%, and deaths occurred from nonrelapse causes. No differences were found in EFS in patients infused with LMP2 versus LMP1/LMP2 CTLs. Of the 21 patients treated with clinical evidence of disease, eleven reached CR early after CTL infusion, while one achieved CR only after additional CTL infusions. The overall 2-year EFS for patients treated with resistant/recurrent disease was 50%, and a trend for a better response was seen when CTLs were targeting both LMP1 and LMP2 antigens.15 Lack of toxicity and the reasonably effective profile of LMP-specific CTLs render this approach quite appealing for application in patients who due to their age or presence of comorbidities had not completed or received full treatment and had failed or relapsed in their disease. Some technical challenges, however, remain to render this therapeutic broadly applicable. For instance, the procedure is currently very time-consuming, since more than 2 months are necessary for the preparation of EBV lymphoblastoid cells and then production of virus-specific CTLs. In addition, since the majority of NHL patients are treated with rituximab, which ablates circulating B cells for several months, the generation of EBV lymphoblastoid cells is frequently difficult to achieve. Nevertheless, the use of artificial antigen-presenting cells (APCs) and peptide libraries as a source of specific antigens (EBNA-1, LMP1/2, and BARF-1) may significantly simplify the manufacturing procedures.17 One NIH-registered clinical trial based on this improved manufacturing approach is ongoing in our institution (NCT01555892).
Tumor-associated antigen-specific T-cells for the treatment of NHL
As NHLs are associated with EBV in only approximately 40% of cases, new antigens are needed for extending the benefits of CTL-based therapy to most NHL patients. The majority of well-characterized nonviral TAAs are, however, self-antigens and thus weak stimulators of T-cell immunity. In addition, self-reactive T-cells are typically deleted by thymic selection or frequently rendered unresponsive by tolerance mechanisms. Within the many reported TAAs, cancer testis antigens (CTAs) have attracted significant interest in the community, as they are consistently expressed by multiple tumors but are absent in healthy organs, with the exception of germ-line tissues.18 Although sometimes demanding due to the low frequency of TAA-CTL precursors in the peripheral blood, numerous strategies have been proposed to expand sufficient numbers of TAA-CTLs ex vivo. Advances in the EBV or viral-associated antigen setting have significantly contributed to the TAA-CTL field. Protocols used for the generation of the former have been applied, with modifications, for the generation of the latter. APCs infected with adenoviruses coding the protein of interest or loaded with specific human leukocyte antigen (HLA)-restricted TAA peptide or pepmixes have been used to generate CTA- or TAA-specific CTLs ex vivo.19
Most of the early clinical trials have been performed in patients with melanomas or leukemias and used CD8+ T-cells targeting a single antigen (most frequently MART-1,20 melanoma-associated antigen peptide tyrosinase, or gp10021 for melanoma and WT-122 for leukemia) expressed in the context of specific major histocompatibility complex (MHC) class I molecules, generally HLA-A2 and HLA-B44, and results are rather promising. As NHLs frequently express a number of these CTAs, like SSX2, MAGE-A4, PRAME, and NY-ESO-1, and the TAA survivin, there is increasing interest for extending this therapy to patients with EBV-negative NHL. To expand this application to all patients, including those with less common HLAs, pepmixes loaded to autologous APCs have proven very useful.23 As described earlier for EBV-specific CTLs, the selection and expansion of CTL precursors for TAAs may benefit from improved manufacturing procedures based on the use of artificial APCs and pepmixes as the source of antigens. Recently, our group developed a protocol that uses a combination of several pepmixes to generate CTL specific for multiple TAAs. CTL lines generated from lymphoma patients showed specific responses against a broad array of TAAs, specifically targeting SSX2 in four of eight lines, MAGE-A4 in six of eight lines, survivin in two of eight lines, PRAME in three of three lines, and NY-ESO-1 in three of three lines. The safety, feasibility, and efficacy of this approach are currently being tested in an ongoing clinical trial enrolling patients with either Hodgkin’s lymphoma or NHL.23
Transgenic TCR-engineered T-cells for the treatment of NHL
As the physiological killing of T-cells occurs through the engagement of the TCR, the surface heterodimer between α- and β-chains that mediates recognition of short epitopes presented in the context of the MHC, the transfer of α- and β-TCR chains into T-cells is currently being explored as a means to reliably produce highly functionally avid T-cells in a very short period of time.
Gene-modified T-cells with TCRs cloned from single T-cells screened for high avidity have been used in clinical trials, and shown to be safe and feasible for the treatment of melanoma.24 Although encouraging responses also have been reported in sarcomas, myeloma, and renal cancers through the targeting of NY-ESO-1, MAGE-A3, p53, and carcinoembryonic antigens, the major obstacle to a broad application of this strategy relates to the restricted number of studied HLA molecules (limited to the most common haplotypes), and therefore to the few known epitopes. The expression of MAGE-A and NY-ESO-1 in ~40%–50% of NHL makes the infusion of transgenic TCRs appealing for patients with relapsed NHL.
Still, major technical issues persist for the widespread application of this technology. One example is the mispairing phenomenon. Indeed, the surface expression of the desired TCRs can be hampered by the incorrect pairing of the transgenic chains with the endogenous chains, thus producing not only nonfunctional TCRs but also TCRs with potentially unwanted specificities, including induction of harmful recognition of self-antigens. Second, the isolation of T-cell clones with high functional avidity from which cloning the αβ-TCR chains invariably fails when the antigen expressed by the tumor cells is also present on healthy cells, as thymic selection eliminates these autoreactive clones. In these scenarios, functional avidities have been enhanced empirically by maturing the affinity of the engineered TCRs through phage-display technologies. While quite successful in generating improved antitumor activity, these approaches have recently raised concerns, as unwanted toxicities have occurred. In a recent case, new TCR specificities were generated in T-cells engineered to express a high-affinity enhanced TCR against an HLA-A*01 restricted MAGE-A3 epitope.25,26 The first two patients treated with these engineered T-cells developed cardiac toxicity and died within days of the T-cell infusion. Although heart tissues do not express MAGE-A3, severe myocardial damage and robust proliferation of the transferred T-cells occurred due to the cross-recognition (unexpectedly generated during the ex vivo affinity-enhancement process) by the transgenic TCR of titin, a peptide derived from a striated muscle-specific protein.25,26 In other cases, affinity maturation can result in an increased TCR-threshold level of recognition, which can be dangerous when antigens, such as survivin, are expressed with different threshold between tumors and healthy cells.
Therefore, success with this strategy may be strictly dependent on targeting antigens expressed at different stages of tissue development, of cell activation, or overexpressed by tumor cells but no longer by healthy tissues, or derived from proteins that result from gene mutations or aberrations in tumor cells, making them uniquely expressed by tumor cells but not healthy tissues.
Due to the extensive gene manipulation required for the technology, this option for NHL patients can be offered only in highly specialized facilities with extensive regulatory capacities. Finally, genotoxic risks, although not reported for T-cells, and complexities associated with gene manipulation render this technology still highly experimental.
Chimeric antigen receptor-engineered T-cells for the treatment of NHL
The overall major limitation of all the aforementioned TCR-mediated killing approaches remains the requirement for HLA restriction and for functional antigen-processing machinery in tumor cells. However, to escape immune recognition, tumors invariably interfere with antigen processing or cause downregulation of MHC molecules. This phenomenon has been reported to occur in NHL, and is frequently the cause of relapse. A technical breakthrough was made in 1993, when the antigen-specificity of mAbs targeting a surface molecule essential or preserved in tumor cells was grafted on T-cells to confer lytic activity and prolonged persistence.27 These so-called CAR molecules provide T-cells with the capacity to recognize and kill malignant T-cells in an MHC-independent fashion and to recognize epitopes derived from carbohydrates and glycolipids, thus broadening the array of potential targets. The major advantage of CARs over the corresponding mAbs is represented by the direct activation of ζ-chain signaling with rapid and highly efficient killing and recruitment of many other components of the immune systems. Self-amplification and development of responses mediated by CAR T-cells are finally expected to be sustained in magnitude and prolonged in time, with the possibility to generate memory and thus long-term protection.25
As relapsed NHLs often exhibit low HLA expression, the setting of relapsed NHL disease has highly fueled the application of this approach. CD19, CD20, and CD30 are validated targets for Ab-based therapy of NHL. Numerous efforts have been aimed at the translation of CAR-based therapy toward these molecules for application in NHL. Extensive preclinical work has been done to ensure that CAR-molecule engagement provides appropriate signaling to T-cells. This work has resulted in the incorporation of costimulatory molecules within the CAR for appropriate activation of these cells or in grafting the CAR on antigen-specific T-cells or central memory cells to guarantee long-term persistence.
Thus far, results on 26 patients with NHL receiving CAR-CD19 or CAR-CD20 T-cell therapy in the presence of active disease have been reported in the literature. Because of gene manipulation and the potential for genotoxic risk, this approach is available only to patients that have exhausted other options. Therefore, to date, only patients with multiple relapsed diseases have been enrolled to receive this therapy.
Most of the CARs tested in the clinical setting for NHL incorporate the CD28 endodomain, an early costimulatory signal molecule that is usually supplied by professional APCs. Although effective in improving in vivo expansion of CAR T-cells, persistence and antitumor efficacy in vivo have proven suboptimal, with one CR following auto-SCT, and only a few transient partial responses (27%). As the incorporation of the 4-1BB endodomain (also known as CD137), a member of the TNF-receptor family involved in the late costimulatory signal of T-cells, has produced quite impressive responses in B-cell leukemias, its inclusion instead of or in addition to CD28 may be beneficial for CAR studies targeting NHL. Importantly, the only CRs observed so far in NHL have been in a pilot trial with a CAR that incorporated both the CD28 and 41BB endodomains or in patients heavily pretreated with B-cell-specific chemotherapy regimens prior to CAR-T-cells.28 Further modifications are currently being investigated to achieve optimal expansion and clinical activity of CAR T-cells. Therefore, the results of the many ongoing studies (Table 2), although using CARs generated under different culture conditions and different constructs, will be important to understand the efficacy and the impact of this approach for the treatment of NHL.
As reported for TCR-based approaches and for patients treated with bispecific Abs, some severe toxicities have been reported for CAR-based therapy, in terms of on-target/off-tumor effects and the rapid rise in serum proinflammatory cytokines, respectively. The expression of the CD20 and CD19 target antigens by healthy B cells is responsible for the occurrence of B-cell aplasia.29 While the administration of immunoglobulin can control this side effect in young patients, in older individuals, the prolonged B-cell aplasia when combined with the T-cell dysfunction following heavy chemotherapy regimens may be a fatal combination by predisposing them to life-threatening opportunistic infections. Strategies to spare the normal B-cell compartment while delivering comparable antitumor activity are currently being explored.30 To deal with on-target/off-tumor toxicities of less tumor-selective CARs, such as those targeting molecules like carcinoembryonic antigens or ERBB2, some investigators have proposed transiently expressing CAR through electroporation of T-cells with in vitro-transcribed ribonucleic acid,31 which warrants a self-limiting expression of the CAR. Two patients have been treated with this approach, and although the infusion of these T-cells was safe and induced epitope spreading, the efficacy was very limited, mostly due to reduced persistence of the gene-modified T-cells. Alternatively, groups are exploring the incorporation of a suicide gene into the construct encoding for the CAR to provide a rapid and effective method of controlling adverse events.32 The so-called cytokine-release storm (CRS) is thus far the most common toxicity associated with the administration of CAR T-cells. Although not all patients receiving CAR-T-cells experience CRS, when it occurs, the severity of the clinical presentation is important, and fatalities have occurred.33
Efforts are currently directed to reduce the occurrence of CRS, but increasing evidence suggests that inhibiting T-cell activity too early in order to control CRS may also impair their antitumor activity.34 While encouraging, CAR T-cell immunotherapy appears to be not as successful for NHL as it has been for acute lymphoblastic leukemia (ALL). This experience suggests an important peculiarity for relapsed NHL. While the target antigen is identical, NHL and ALL have different tumor environments, and this may play a significant role in response to therapy. Indeed, numerous evidence suggests that NHL tumors, especially when relapsed, display several immune-escape evasion tools, like upregulation of PDL1,35 infiltration by regulatory T-cells,36 and release of inhibitory molecules37,38 (granzyme B inhibitor or protease inhibitor 9) that are absent in ALL. Furthermore, T-cell activation following antigen engagement is often accompanied by the upregulation of PD1.39 The interaction between PD1 and PDL1 on many tumor cells can deliver a strong inhibitory signal that neutralizes the antitumor activity of residential lymphocytes, as well as of the adoptively transferred T-cells. As the administration of anti-PD1 Abs has proven effective in highly immunogenic diseases in releasing T-cells from the inhibition induced through the PDL1–PD1 axis, the combination with checkpoint inhibitors appears the most promising route to pursue to improve the efficacy of the adoptively transferred T-cells. Lessons learned from these trials will be applicable for all previously described (native or transgenic) TCR-based immunotherapeutic approaches.
Conclusion
High hopes are held for the adoptive transfer of antigen-specific T-cells to complement the standard therapies for patients affected by NHL. Although preliminary, available results from current and ongoing clinical trials support the administration of LMP1/2-specific CTLs to consolidate CRs in patients treated with no evidence of disease and to induce durable responses in those treated with measurable disease. The major benefit of this approach is its safety profile and great tolerability, particularly in more fragile or heavily pretreated patients. Similar results are anticipated when using TAA-CTL adoptive immunotherapy for NHL lacking EBV antigens. Encouraging results are now seen with CAR T-cells. Although data are promising, T-cells infused in NHL have shown limited persistence and expansion, explaining, at least in part, the more limited antitumor activity observed in NHL compared to patients affected by other diseases but treated with comparable T-cells/CTLs in clinical trials. A more in-depth understanding of the biology of T-cell immune responses in NHL patients will certainly shed light into this diversity in clinical responses.
Several strategies have been developed to increase the effectiveness of adoptively transferred T-cells. On one side, investigators are exploring ex vivo strategies to increase the stemness of infused T-cells to improve persistence and proliferation. This can be achieved by reducing the length of the ex vivo culture and/or by manipulating the culture conditions using, eg, specific cytokine cocktails,40 or by selecting specific T-cell subpopulations41 or antigen-specific T-cells.42 In parallel, new costimulatory molecules or combinations of costimulatory pathways continue to be explored to facilitate prolonged CAR-T-cell persistence. On the other hand, in vivo tools are being used to deliver potent proliferative signals. Specifically, in vivo proliferation can be driven by preconditioning regimens that induce profound lymphodepletion and as a consequence the release of significant amounts of homeostatic cytokines. Fine tuning of these approaches is ongoing, as the massive proliferation associated with increased stemness and concentration of homeostatic cytokines has resulted in some cases in fatalities.
Although confounding for the interpretation of the relevance played by each component in the final clinical outcome, the systematic combination of adoptively transferred T-cells with standard therapies or with the new immunomodulatory drugs available in the clinical arena (like checkpoint inhibitors, anti-CTLA4, or anti-PD1 Abs) may provide T-cells with the right environment to fulfill their potential and result in better control of the disease.
Disclosure
Barbara Savoldo is is a researcher in a collaborative research agreement between the Center for Cell and Gene Therapy and Celgene (Summit, NJ, USA) to develop genetically modified T-cells. The authors report no other conflicts of interest in this work.
References
Shankland KR, Armitage JO, Hancock BW. Non-Hodgkin lymphoma. Lancet. 2012;380:848–857. | |
Leechawengwongs E, Shearer WT. Lymphoma complicating primary immunodeficiency syndromes. Curr Opin Hematol. 2012;19: 305–312. | |
Zelenetz AD, Wierda WG, Abramson JS, et al. Non-Hodgkin’s lymphomas, version 1.2013. J Natl Compr Canc Netw. 2013;11:257–272. | |
Abramson JS, Zelenetz AD. Recent advances in the treatment of non-Hodgkin’s lymphomas. J Natl Compr Canc Netw. 2013;11:671–675. | |
Gangatharan S, Kuruvilla J. Relapsed and refractory aggressive NHL: time for a change. Transfus Apher Sci. 2013;49:72–79. | |
Harrison AM, Thalji NM, Greenberg AJ, Tapia CJ, Windebank AJ. Rituximab for non-Hodgkin’s lymphoma: a story of rapid success in translation. Clin Transl Sci. 2014;7:82–86. | |
Gisselbrecht C. Is there any role for transplantation in the rituximab era for diffuse large B-cell lymphoma? Hematology Am Soc Hematol Educ Program. 2012;2012:410–416. | |
Rodrigues CA, Patah PA, Novis YA, Hosing C, de Lima M. The role of transplantation in diffuse large B-cell lymphoma: the impact of rituximab plus chemotherapy in first-line and relapsed settings. Curr Hematol Malig Rep. 2011;6:47–57. | |
Chao MP. Treatment challenges in the management of relapsed or refractory non-Hodgkin’s lymphoma – novel and emerging therapies. Cancer Manag Res. 2013;5:251–269. | |
Horwitz SM, Advani RH, Bartlett NL, et al. Objective responses in relapsed T-cell lymphomas with single-agent brentuximab vedotin. Blood. 2014;123:3095–3100. | |
Schlegel P, Lang P, Zugmaier G, et al. Pediatric posttransplant relapsed/refractory B-precursor acute lymphoblastic leukemia shows durable remission by therapy with the T-cell engaging bispecific antibody blinatumomab. Haematologica. 2014;99:1212–1219. | |
Bollard CM, Rooney CM, Heslop HE. T-cell therapy in the treatment of post-transplant lymphoproliferative disease. Nat Rev Clin Oncol. 2012;9:510–519. | |
Papadopoulos EB, Ladanyi M, Emanuel D, et al. Infusions of donor leukocytes to treat Epstein-Barr virus-associated lymphoproliferative disorders after allogeneic bone marrow transplantation. N Engl J Med. 1994;330:1185–1191. | |
Rooney CM, Smith CA, Ng CY, et al. Use of gene-modified virus-specific T lymphocytes to control Epstein-Barr-virus-related lymphoproliferation. Lancet. 1995;345:9–13. | |
Bollard CM, Gottschalk S, Torrano V, et al. Sustained complete responses in patients with lymphoma receiving autologous cytotoxic T lymphocytes targeting Epstein-Barr virus latent membrane proteins. J Clin Oncol. 2014;32:798–808. | |
Bollard CM, Gottschalk S, Leen AM, et al. Complete responses of relapsed lymphoma following genetic modification of tumor-antigen presenting cells and T-lymphocyte transfer. Blood. 2007;110:2838–2845. | |
Ngo MC, Ando J, Leen AM, et al. Complementation of antigen-presenting cells to generate T lymphocytes with broad target specificity. J Immunother. 2014;37:193–203. | |
Nishikawa H, Maeda Y, Ishida T, et al. Cancer/testis antigens are novel targets of immunotherapy for adult T-cell leukemia/lymphoma. Blood. 2012;119:3097–3104. | |
Turtle CJ, Riddell SR. Artificial antigen-presenting cells for use in adoptive immunotherapy. Cancer J. 2010;16:374–381. | |
Butler MO, Friedlander P, Milstein MI, et al. Establishment of antitumor memory in humans using in vitro-educated CD8+ T cells. Sci Transl Med. 2011;3:80ra34. | |
Chapuis AG, Thompson JA, Margolin KA, et al. Transferred melanoma-specific CD8+ T cells persist, mediate tumor regression, and acquire central memory phenotype. Proc Natl Acad Sci USA. 2012;109: 4592–4597. | |
Chapuis AG, Ragnarsson GB, Nguyen HN, et al. Transferred WT1-reactive CD8+ T cells can mediate antileukemic activity and persist in post-transplant patients. Sci Transl Med. 2013;5:174ra27. | |
Gerdemann U, Katari U, Christin AS, et al. Cytotoxic T lymphocytes simultaneously targeting multiple tumor-associated antigens to treat EBV negative lymphoma. Mol Ther. 2011;19:2258–2268. | |
Morgan RA, Dudley ME, Wunderlich JR, et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science. 2006;314:126–129. | |
Maus MV, Grupp SA, Porter DL, June CH. Antibody-modified T cells: CARs take the front seat for hematologic malignancies. Blood. 2014;123:2625–2635. | |
Linette GP, Stadtmauer EA, Maus MV, et al. Cardiovascular toxicity and titin cross-reactivity of affinity-enhanced T cells in myeloma and melanoma. Blood. 2013;122:863–871. | |
Eshhar Z, Waks T, Gross G, Schindler DG. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci USA. 1993;90:720–724. | |
Till BG, Jensen MC, Wang J, et al. CD20-specific adoptive immunotherapy for lymphoma using a chimeric antigen receptor with both CD28 and 4-1BB domains: pilot clinical trial results. Blood. 2012;119: 3940–3950. | |
Kochenderfer JN, Wilson WH, Janik JE, et al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood. 2010;116: 4099–4102. | |
Vera J, Savoldo B, Vigouroux S, et al. T lymphocytes redirected against the kappa light chain of human immunoglobulin efficiently kill mature B lymphocyte-derived malignant cells. Blood. 2006;108:3890–3897. | |
Beatty GL, Haas AR, Maus MV, et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol Res. 2014;2:112–120. | |
Budde LE, Berger C, Lin Y, et al. Combining a CD20 chimeric antigen receptor and an inducible caspase 9 suicide switch to improve the efficacy and safety of T cell adoptive immunotherapy for lymphoma. PLoS One. 2013;8:e82742. | |
Lee DW, Gardner R, Porter DL, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124: 188–195. | |
Maude SL, Barrett D, Teachey DT, Grupp SA. Managing cytokine release syndrome associated with novel T cell-engaging therapies. Cancer J. 2014;20:119–122. | |
Rossille D, Gressier M, Damotte D, et al. High level of soluble programmed cell death ligand 1 in blood impacts overall survival in aggressive diffuse large B-cell lymphoma: results from a French multicenter clinical trial. Leukemia. Epub April 15, 2014. | |
Liu XQ, Lu K, Feng LL, et al. Up-regulated expression of indoleamine 2,3-dioxygenase 1 in non-Hodgkin lymphoma correlates with increased regulatory T-cell infiltration. Leuk Lymphoma. 2014;55:405–414. | |
Bossard C, Belhadj K, Reyes F, et al. Expression of the granzyme B inhibitor PI9 predicts outcome in nasal NK/T-cell lymphoma: results of a Western series of 48 patients treated with first-line polychemotherapy within the Groupe d’Etude des Lymphomes de l’Adulte (GELA) trials. Blood. 2007;109:2183–2189. | |
Bladergroen BA, Meijer CJ, ten Berge RL, et al. Expression of the granzyme B inhibitor, protease inhibitor 9, by tumor cells in patients with non-Hodgkin and Hodgkin lymphoma: a novel protective mechanism for tumor cells to circumvent the immune system? Blood. 2002;99: 232–237. | |
Taube JM, Klein AP, Brahmer JR, et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res. Epub April 8, 2014. | |
Xu Y, Zhang M, Ramos CA, et al. Closely related T-memory stem cells correlate with in vivo expansion of CAR.CD19-T cells and are preserved by IL-7 and IL-15. Blood. 2014;123:3750–3759. | |
Wang X, Naranjo A, Brown CE, et al. Phenotypic and functional attributes of lentivirus-modified CD19-specific human CD8+ central memory T cells manufactured at clinical scale. J Immunother. 2012;35: 689–701. | |
Papadopoulou A, Gerdemann U, Katari UL, et al. Activity of broad-spectrum T cells as treatment for AdV, EBV, CMV, BKV, and HHV6 infections after HSCT. Sci Transl Med. 2014;6:242ra83. | |
Savoldo B, Ramos CA, Liu E, et al. CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma patients. J Clin Invest. 2011;121:1822–1826. | |
Kochenderfer JN, Wilson WH, Janik JE, et al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood. 2010;116:4099–4102. | |
Kochenderfer JN, Dudley ME, Feldman SA, et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood. 2012;119:2709–2720. | |
Kochenderfer JN, Dudley ME, Carpenter RO, et al. Donor-derived CD19-targeted T cells cause regression of malignancy persisting after allogeneic hematopoietic stem cell transplantation. Blood. 2013;122: 4129–4139. | |
Till BG, Jensen MC, Wang J, et al. Adoptive immunotherapy for indolent non-Hodgkin lymphoma and mantle cell lymphoma using genetically modified autologous CD20-specific T cells. Blood. 2008;112:2261–2271. | |
Jensen MC, Popplewell L, Cooper LJ, et al. Antitransgene rejection responses contribute to attenuated persistence of adoptively transferred CD20/CD19-specific chimeric antigen receptor redirected T cells in humans. Biol Blood Marrow Transplant. 2010;16:1245–1256. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.