Back to Journals » International Journal of Women's Health » Volume 6
Management of hereditary angioedema in pregnant women: a review
Authors Caballero T, Canabal J, Rivero-Paparoni D, Cabañas R
Received 3 February 2014
Accepted for publication 20 June 2014
Published 9 September 2014 Volume 2014:6 Pages 839—848
DOI https://doi.org/10.2147/IJWH.S46460
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Teresa Caballero,1,2 Julio Canabal,1 Daniela Rivero-Paparoni,1 Rosario Cabañas1
1Hospital La Paz Institute for Health Research, (IdiPaz) 2Biomedical Research Network on Rare Diseases-U754 (CIBERER), Madrid, Spain
Abstract: Three types of hereditary angioedema (HAE) have been described: two are due to C1 inhibitor (C1-INH) deficiency (C1-INH-HAE types I and II) and one is characterized by normal C1-INH (nC1-INH-HAE). The management of pregnancy in patients with HAE is often a clinical challenge owing to potential worsening of the disease in relation to the physiological increase in estrogens and the limited treatment options. This review addresses the potential influence of pregnancy on the clinical severity of hereditary angioedema and the management of this disease during pregnancy with currently available treatments.
Keywords: hereditary angioedema, pregnancy, female, treatment, C1 inhibitor concentrate, tranexamic acid
Introduction
Hereditary angioedema (HAE) is a rare disorder characterized by recurrent, localized, and self-limiting acute edema episodes in different locations.1 A consensus document on the classification of angioedema without wheals has recently been published.2 HAE can be caused by a genetic deficiency of plasma C1 inhibitor (C1-INH; C1-INH-HAE), owing to mutations in the SERPING1 gene, which results in either diminished levels of the C1-INH protein (C1-INH-HAE type I) or dysfunctional forms of the same protein (C1-INH-HAE type II).1,2There is a third type of HAE, previously called type III HAE, in which no C1-INH deficiency is present (nC1-INH-HAE).3,4 Some families with nC1-INH-HAE have mutations in the F12 gene, which encodes coagulation protein factor XII (FXII-HAE), whereas no mutation has been found in others (U-HAE).1–3 Both C1-INH-HAE and nC1-INH-HAE are characterized by an activation of the contact system, which results in an increase in bradykinin formation and subsequently an increase in endothelial permeability, with vascular leakage and angioedema.1
C1-INH-HAE is a rare disease with a prevalence initially estimated to be between 1:10,000 and 1:50,000 inhabitants.5Some national registries have published minimal prevalence, ranging from 1.09 to 1.41 per 100,000 inhabitants.6,7 There have been no studies on the prevalence of nC1-INH-HAE.
Angioedema usually develops in the subcutaneous tissues of the limbs, face, trunk, or genitalia (peripheral angioedema), but also commonly affects the submucosal tissues of the gastrointestinal and respiratory tracts.8 This type of angioedema, which is elicited by local release of bradykinin, does not respond to conventional therapy with antihistamines, corticosteroids or adrenaline.9
Special issues in the management of C1-INH-HAE in female patients, including pregnancy, have been addressed in an international consensus document,10 but data are more limited regarding nC1-INH-HAE. Although both C1-INH-HAE and nC1-INH-HAE are autosomal dominant inherited diseases, and there should be no differences in sex distribution, a female preponderance of patients has been observed in most clinical series.6,11–18 One of the most plausible explanations for the female predominance of C1-INH-HAE patients might be that women have a more severe expression of the disease due to the significant association between estrogen exposure and disease severity, with a higher diagnosis rate and a higher attendance rate at health care services.10,18 Furthermore, the sex prevalence in nC1-INH-HAE shows a higher female prevalence than in C1-INH-HAE, due to a much lower penetrance of the disease in males (>90% of asymptomatic carriers in males versus 40% in women in FXII-HAE).2,17,19–21
The clinical course of C1-INH-HAE and nC1-INH-HAE can worsen during pregnancy due to the physiological increase in estrogens.1,3,4,8,10 This fact, together with limitations regarding the use of certain drugs during pregnancy, makes the management of HAE a challenge for physicians. This review addresses the issue of management of HAE during pregnancy in an attempt to upgrade the health care of patients with C1-INH-HAE and nC1-INH-HAE and decrease the burden HAE will cause for patients and their families in the future.
Bibliographic search
An in-depth search of the available English language literature was performed on PubMed and EMBASE using the following key words: hereditary angioedema, HAE, type III HAE, C1-inhibitor, and C1 inhibitor deficiency. Additional sources were selected from the reference lists of published articles. Searches were last updated on December 31, 2013. All publications consulted were reviewed for data regarding the management of pregnancy and related issues in HAE.
Diagnosis of HAE during pregnancy
C1-INH-HAE rarely presents for the first time during pregnancy.10,22,23 However, it is not so rare that symptoms appear for the first time during pregnancy in patients with nC1-INH-HAE.3,19 Early and accurate diagnosis of both diseases is important for both mother and fetus and can be attained by measuring blood complement levels (C4, antigenic C1 inhibitor, and functional C1 inhibitor) and/or by carrying out genetic testing.1,2,4,10,24
Serum C1-INH testing during pregnancy should be carefully interpreted,10 as transient low C1-INH levels with normalization after delivery have been found in normal pregnant women.25–28 A drop in plasma C1-INH levels during pregnancy could be related to an increase in plasma volume.26 Moreover, lower levels of functional plasma C1-INH have also been found in some women with nC1-INH-HAE during pregnancy.3,20,29 In order to avoid misdiagnosis, follow-up tests on patients who have a low blood C1-INH level or function during pregnancy should be run a few months after delivery to confirm a C1-INH-HAE diagnosis.10
Prenatal diagnosis and preimplantation genetic diagnosis
Prenatal diagnosis for C1-INH-HAE and nC1-INH-HAE during pregnancy is rarely requested. The presence of the mutation responsible for the disease in an affected parent must be detected before a fetus is tested for it. Genetic testing for the specific mutation is performed with cells taken from either a chorion villus sample or an amniotic fluid sample.10 The risk of a miscarriage from either procedure carried out by experienced professionals is between 0.5% and 1%.10,30 In cases where the disease-causing mutation is found in the fetus, and where applicable laws and practices allow it, the option of a therapeutic abortion could be made available.10
Preimplantation genetic diagnosis, a technique used to identify genetic defects in embryos prior to implantation and pregnancy, can detect embryos without C1-INH-HAE or FXII-HAE. However, preimplantation genetic diagnosis is expensive and has a low success rate for pregnancy. Moreover, women must be given hormone therapy, which is a risk factor for an increase in HAE severity.10,31 A successful preimplantation genetic diagnosis in C1-INH-HAE has been published.32
Postnatal diagnosis
The C1-INH concentration in umbilical blood of healthy neonates is usually lower than that of the normal adult value (approximately two thirds)33 and the complement system reaches an adult maturity level by the age of 6–36 months.34 C1-INH levels and function can show false-positive and false-negative results in infants under 12 months and thus C1-INH test findings should be confirmed after 1 year of age.35,36 Given the diagnostic uncertainty of biochemical tests in young children, genetic testing might be a useful option for determining whether a child has inherited C1-INH-HAE. Genetic testing is necessary for diagnosing FXII-HAE in cases with clinical symptoms and identifying asymptomatic carriers. However, it must first be known that the family carries the disease-causing mutation. Genetic testing of asymptomatic children with C1-INH-HAE or FXII-HAE is especially beneficial because diagnosis can help to ensure that the correct treatment is provided in the event of an attack,10,37,38 as well as in cases where emergency medical or surgical procedures are necessary. It is noteworthy that there is no diagnostic test for nC1-INH-HAE without mutation in the F12 gene (U-HAE).
Genetic testing
Genetic testing is not always necessary for diagnosis of C1-INH-HAE; however it could aid in the diagnosis of cases in which C1-INH measurements are inconclusive, as may occur with newborns or in cases with C1-INH deficiency in the absence of family history. Genetic testing may be used to detect the presence of C1-INH-HAE in “de novo mutations” or to rule out acquired angioedema due to C1-INH deficiency. Moreover, genetic testing is the only way to confirm a diagnosis of FXII-HAE. It could also be helpful in the identification of family members at risk of C1-INH-HAE or FXII-HAE (presymptomatic testing). The disease-causing mutation in a particular family should be identified in advance in cases where prenatal diagnosis, preimplantation genetic diagnosis, or presymptomatic testing is requested. It is noteworthy that the mutation responsible for C1-INH deficiency is only identified in approximately 90% of patients with C1-INH-HAE39,40 and in a lower percentage of patients with nC1-INH-HAE.4
Genetic counseling
Patients and families with C1-INH-HAE and nC1-INH-HAE should have access to genetic counseling and be provided with a pedigree chart, information on the type of inheritance (autosomal dominant), diagnostic screening of affected relatives, and information on prenatal diagnosis or preimplantation genetic diagnosis according to national laws and practice.10 C1-INH-HAE is a treatable disease with a highly variable phenotype, and prenatal diagnosis is therefore seldom requested.10 Nevertheless, there is no approved therapy for nC1-INH-HAE, and information on the efficacy of drugs available for C1-INH-HAE and nC1-INH-HAE is scattered. The low number of requests for prenatal diagnosis of nC1-INH-HAE might be attributed to the fact that it is a newly described disease, together with low penetrance.
Patients should also be informed of limitations of HAE treatment during pregnancy and lactation, possible worsening of the disease, and available treatments for HAE during pregnancy and patient accessibility to them. It is also important that patients be warned not to receive certain HAE treatments, such as attenuated androgens, prior to conception.10
Management
During pregnancy
Close follow-up during pregnancy, labor, delivery, and breast-feeding is recommended since these events can affect the course of the disease in different ways.10 Pregnancy may affect the course of C1-INH angioedema attacks by worsening, improving, or having no impact at all,10,41 but a higher percentage of pregnancies experienced an increase in C1-INH-HAE attack rates in the largest published series, with 227, 118, and 35 pregnancies respectively.41–43 The influence of pregnancy on C1-INH-HAE severity may even change in different pregnancies in the same woman.9,11,42 Thus, in the series reported by Czaller et al including 41 women, the C1-INH-HAE course was similar in 21 patients (78% of those with several pregnancies) and different in six.42 The effects on C1-INH-HAE symptoms were also reported as similar for each subsequent pregnancy in most women by Bouillet et al (data on 107 women from eight countries who had experienced 227 pregnancies).41
Regarding the pregnancy trimester when C1-INH-HAE symptoms are more severe, the published data show some discrepancies. On the one hand, more severe clinical symptoms have been described during the first trimester in some series,42,44,45 but on the other, increased attack rates in the second and third trimesters have also been reported.43,46 The influence of pregnancy on the course of the disease has been analyzed according to different parameters in a variety of published studies (attack rates, severity of attacks, and number of pregnancies with attacks in each trimester). The differences in the analyzed parameters among studies could explain the different conclusions reached. Martínez-Saguer et al found that the highest mean number of attacks happened during the two last trimesters,43 whereas Czaller et al reported a higher number of pregnancies with angioedematous attacks during the first trimester (49% of pregnancies) than in the second (37%) and third (37%) trimesters.42 However, a higher mean number of attacks was found in the third trimester (6.59) followed by the first (3.78) and then the second (2.29) in the same series.42 As for attack severity, the results were very similar in the first (0.585) and third (0.561) trimester.42 Bouillet et al observed no improvement in the last two trimesters.41
Factors predicting the course of C1-INH-HAE during pregnancy have been analyzed. First, there is a connection between experiencing angioedema attacks at a young age and worsening during pregnancy, with an increase in the frequency and severity of attacks.12,42 Second, women who referred to menstruation as an exacerbating factor improved in the last trimester.42 Third, those who indicated that mechanical trauma triggered their angioedema attacks even before pregnancy had more C1-INH-HAE attacks during all three trimesters.42 Last, being pregnant with a C1-INH-HAE fetus has been found to be associated with a higher frequency of angioedema episodes during the last trimester.42 In contrast, Martínez-Saguer et al found no relationship between attack rate during pregnancy and whether the fetus had C1-INH-HAE,43 whereas they did find that women whose newborns had C1-INH-HAE tended to have a lower level of C1-INH activity during pregnancy.43
Pregnancy can influence the course of the disease in patients with nC1-INH-HAE.3,4,17,47–49 Two families with FXII-HAE in which the affected women experienced severe complications of their disease during pregnancy, including some cases of fetal and neonatal death, have been described.50 Both families had strictly estrogen-dependent swelling with angioedema attacks only during pregnancy or when taking estrogen-containing medications.50
The abdomen is the most frequent localization of angioedema attacks, whether isolated or combined with attacks on different locations.42,43 This might be explained by mechanical trauma due to the progressive stretching of the uterus as a result of fetal growth and movement.42,43 Differential diagnosis of an abdominal attack during pregnancy may be difficult to undertake.42,45,51 An abdominal ultrasound would be very useful. Discovery of free peritoneal fluid and edematous swelling of the intestinal wall are highly suggestive of an abdominal attack of HAE. Clinical improvement and ultrasonographic assessment of regression after treatment with plasma-derived human C1-INH concentrate (pdhC1INH) could confirm the tentative diagnosis.10,42
Labor and delivery
During childbirth, women with C1-INH-HAE/nC1-INH-HAE should be monitored closely in hospital by different specialists (obstetrician, immunologist, anesthesiologist, pediatrician) who can work together with the HAE specialist, thereby providing a multidisciplinary approach.10,42
Despite suspicion that the mechanical trauma involved in vaginal childbirth could trigger angioedema, there is no evidence to support this. Only rarely do vaginal deliveries cause an edematous attack,42 with 6% of women not receiving prophylactic treatment according to the PREHAEAT (Novel methods for predicting preventing and treating attacks in patients with hereditary angioedema) study.41 Those attacks appear immediately after or in the first 48 hours after delivery.9,10,41 Spontaneous vaginal delivery occurs in 80%–90% of births. The number of cesarean sections performed is similar to those in the overall population,41,42,45 and a cesarean delivery rate of 12% was reported by Bouillet et al in the PREHAEAT study.41
After delivery, the prevalence of angioedema of the vulva is higher than that of genital edema before pregnancy.12,42,44,45 Postpartum, some patients suffer a higher rate of edema episodes,23,42,52,53 mainly abdominal attacks.42 Caring for and close monitoring of the mother in the postnatal period is recommended for at least 72 hours after giving birth.10,54,55
Breast-feeding
A temporary increase in the number of episodes, mainly abdominal, has been observed during lactation.42 Prolactin levels have been found to be associated with the number of abdominal episodes suffered by these patients.56 The increased serum prolactin level in susceptible women could be responsible for the temporary increase in angioedema episodes observed in some females postpartum.42 Stopping breast-feeding altogether might decrease the frequency of angioedema episodes by causing the levels of this hormone to drop.
Spontaneous abortion and premature labor
Data on the rate of spontaneous abortion in HAE are scarce and conflicting. Nielsen et al reported that patients with C1-INH-HAE who had symptoms of angioedema had an increased rate of spontaneous miscarriage and early delivery,57 whereas other authors reported no increases in the rate of miscarriage, early delivery, or stillbirths.41–43
Treatment
Available treatments for HAE and pregnancy and their indications
Therapeutic options for patients with HAE are limited during pregnancy. We reviewed all the treatment possibilities for HAE and their tolerability and safety during pregnancy. Most treatments have been studied in C1-INH-HAE, whereas experience in nC1-INH-HAE is limited. Treatment should always be individualized.
Plasma-derived human C1-INH concentrate
pdhC1INH is available from several pharmaceutical companies, and includes Berinert® (CSL-Behring GmbH, Marburg, Germany), Cebitor® (Sanquin, Amsterdam, the Netherlands), and Cinryze® (Viropharma Inc., Exton, PA, USA). pdhC1INH has been proved to be effective in the resolution of acute angioedema episodes as well as for long-term prophylaxis in randomized, placebo-controlled, clinical trials in C1-INH-HAE2,9,54,58,59 and for short-term prophylaxis in a number of open studies and case series in C1-INH-HAE.60–64 However, it has only been tested in isolated cases of nC1-INH-HAE and yielded variable results.3,4,19,20 There are several reports in which pdhC1INH has been proven to be safe and effective during pregnancy and breast-feeding for long-term prophylaxis, short-term prophylaxis, and acute therapy in patients with C1-INH-HAE,10,42,43,65–69 although no controlled studies have been conducted during pregnancy. It is recommended as the first-line therapy during pregnancy and lactation in C1-INH-HAE for all these indications.10,42,43
The long-term prophylactic pdhC1INH dosage and intervals between doses should be determined on a case-by-case basis, taking into account clinical disease severity9,10,70 and can vary from 500 U once a week to 2,000 U twice a week.65,69 A notable case has been published in which a pregnancy was successfully managed in a patient with severe C1-INH-HAE who was first treated with on-demand pdhC1INH and long-term prophylaxis with tranexamic acid and subsequently with both on-demand and long-term prophylaxis with pdhC1INH.71
Antifibrinolytics
Tranexamic acid and epsilon aminocaproic acid are antifibrinolytic agents used in the treatment of angioedema. Antifibrinolytics have been administered for mild edema attacks,51 but there are no controlled studies of their effectiveness in treating acute attacks.10 Long-term prophylaxis with tranexamic acid during pregnancy is considered medically indicated in C1-INH-HAE only when pdhC1INH is unavailable.10 As for patients diagnosed with nC1-INH-HAE, there are no clear therapeutic alternatives for treating acute episodes, although there are reports of isolated cases in which treatment using tranexamic acid and pdhC1INH was successful.4,19,20
Although tranexamic acid crosses the placenta, no mutagenic activity or harmful effects of tranexamic acid on the fetus have been reported10,72 and animal reproduction studies have shown no teratogenic effects.10,73,74 Tranexamic acid dosages similar to those used in patients with C1-INH-HAE have been administered during pregnancy for other diseases, but generally for a shorter period of time and not during the first trimester.10,75 Tranexamic acid has been well tolerated and has not been associated with a prejudicial effect on the delivery of healthy children.10,76 Given the lack of data on the use of tranexamic acid throughout entire pregnancies, its potential risks and benefits must be considered carefully.10 Tranexamic acid is not recommended during breast-feeding as it can pass into breast milk and may harm a nursing baby.10
There are concerns about the possibility of thromboembolism in relation to the suppression of the fibrinolytic system;76 however, controlled clinical studies have not provided any evidence to support them.72 Nevertheless, patients with a personal or family medical history of thromboembolic disease might be at greater risk of venous or arterial thrombosis when taking antifibrinolytics, and an in-depth diagnostic workup, including a hypercoagulability study, may be necessary before initiating tranexamic acid.9
Epsilon aminocaproic acid is a derivative and analog of the amino acid lysine. Its efficacy in the prevention of angioedema attacks was demonstrated in a placebo-controlled, double-blind trial.77 The known side effects are thrombosis and muscle pain, with increased activity of creatine phosphokinase and aldolase, extensive muscle necrosis, asthenia, weakness, abdominal pain, itching, nausea, and dizziness.77 Because of the risk of thrombosis, epsilon aminocaproic acid should be discontinued in patients with prothrombotic states and in all patients prior to surgery.9,58 The standard dose is 1 g every 6–8 hours,9 and can be increased to 12 g/day divided into four doses.8 There are few data on its use during pregnancy or lactation.
Attenuated androgens
Attenuated androgens, such as danazol, stanozolol, and oxandrolone, can cross the placental barrier and should be avoided during the entire pregnancy, particularly during the first trimester when they may have direct effects on the fetus, such as virilization and female pseudohermaphroditism.78–83 Notwithstanding, in one case series, seven patients took danazol during the initial weeks of their pregnancies and masculinization was not observed in any of their female neonates.42 In addition, placental insufficiency and fetal growth retardation may occur due to exposure to testosterone during pregnancy.10,84 Consequently, attenuated androgens should be discontinued before pregnancy and at least 1 month prior to conception.9
It is not known whether anabolic steroids are excreted into breast milk. Nevertheless, because of the potential side effects in children, it is recommended that women stop breast-feeding before starting attenuated androgen therapy.10
Virally inactivated fresh frozen plasma
Virally inactivated fresh frozen plasma can be an alternative in countries in which pdhC1INH is not available24,85–87 and in cases where antifibrinolytics are contraindicated or ineffective.10,88,89 Risks and precautions are similar to those in nonpregnant women.10,24,85–87
Virally inactivated fresh frozen plasma, when used for the treatment of acute angioedema attacks, has the potential to lead to worsening of HAE symptoms. In addition to C1-INH, it also supplies substrates (factor XII, prekallikrein, and high molecular weight kininogen) that can produce an increase in bradykinin before the supplied C1-INH has had time to act.10,88,90 Anaphylaxis has been reported in a case in which fresh frozen plasma was administered for an acute C1-INH-HAE attack.85 Physicians must be prepared for such situations. If pdhC1INH is not available and antifibrinolytics are contraindicated or ineffective, virally inactivated fresh frozen plasma could be used for long-term prophylaxis.88,89 Not enough data exist on the use of fresh frozen plasma for long-term prophylaxis during pregnancy.10
Icatibant acetate
Icatibant acetate (Firazyr®; Shire Orphan Therapies GmbH, Zug, Switzerland) is a selective bradykinin B2-receptor antagonist that is effective in the treatment of acute angioedema attacks in adult patients with C1-INH-HAE. Randomized, double-blind, controlled studies have demonstrated its effectiveness.2,9,54,58,91 Beneficial effects on acute attacks in patients with nC1-INH-HAE have also been shown.4 However, no data are available for its use during pregnancy or lactation. For this reason, icatibant acetate is not recommended during pregnancy, except when a life-threatening angioedema attack does not respond to other treatments.9
Ecallantide
Ecallantide (Kalbitor®; Dyax Corporation, Boston, MA, USA) is a kallikrein inhibitor that has been shown to be effective for the treatment of acute attacks in patients older than 16 years.2,9,54,58,91 There are isolated data on its efficacy when used in patients with acute angioedema attacks in nC1-INH-HAE.4 No data are available for the use of ecallantide during pregnancy or lactation.
Recombinant human C1-INH
Recombinant human C1-INH (Ruconest®; Pharming NV, Leiden, the Netherlands), also known as conestat alfa or rhC1INH, is produced in transgenic rabbits in which the human C1NH gene has been inserted.9 rhC1INH is excreted into milk and purified.9 No data are available on its use during pregnancy or lactation.
Management of medical and surgical procedures during pregnancy
Patients with C1-INH-HAE and nC1-INH-HAE are at particular risk of upper airway edema from medical, dental, and surgical procedures,4,9,10,19,54 and should be managed individually with the participation of a multidisciplinary team. The decision to give prophylaxis before a procedure depends on the patient’s personal history and the degree of risk associated with the procedure.54
Short-term prophylaxis should be considered before bronchoscopy, endoscopy, and surgical procedures, especially dental/intraoral surgery, which requires endotracheal intubation and manipulation of the upper airway or pharynx.54
In the case of surgical procedures, there is no specific contraindication to general anesthetic medication.55 However, regional analgesia is preferable because it avoids the need to perform an endotracheal intubation, which can produce laryngeal edema.86 If intubation is absolutely necessary, it is best to use a small endotracheal tube.55
The treatment of choice is pdhC1INH 1–6 hours prior to the procedure (as close to the procedure as possible).10,24 If pdhC1INH is not available, virally inactivated fresh frozen plasma can be administered.92 pdhC1INH should also be available for acute angioedema treatment if necessary.
Management of special obstetric procedures during pregnancy
- Amniocentesis/chorial biopsy: short-term prophylaxis with pdhC1INH is recommended.10
- Medical artificial abortion: the need for short-term prophylaxis in this condition has not been established, but specific acute treatment should be immediately available if necessary.10
- Surgical artificial abortion: short-term prophylaxis with pdhC1INH is recommended to prevent angioedema.42 Alternatively, specific medications for acute treatment should be readily available.10,42
- Delivery and puerperium: there is no evidence as to the best method of delivery in patients with C1-INH-HAE or nC1-INH-HAE. However, there is agreement among experts on the convenience to deliver in hospitals for safety reasons. The rate of cesarean section is similar to the general population, with most births being spontaneous vaginal deliveries.41,42,45 It is noteworthy that although labor and delivery may result in significant mechanical trauma, they seldom induce an edematous attack.10,54–56,65
Thus, routine prophylaxis is not recommended before uncomplicated natural vaginal deliveries, although pdhC1INH should be immediately available in the delivery room.10 Prophylactic treatment has to be considered in patients with additional risk factors for an attack, such as repeated or severe attacks during pregnancy or a medical history of genital edema associated with mechanical trauma.10,11
Although there is no evidence to suggest that the risk of angioedema in vaginal deliveries that require the use of vacuum or forceps is greater than in deliveries where they are not used, most experts agree with the recommendation to administer pdhC1INH as soon as possible.10
Following vaginal delivery, patients with significant perineal swelling or other postpartum complications should be closely monitored for at least 3 days, and treated with pdhC1INH if necessary.10,23,41,53,93,94
- Cesarean section (abdominal delivery): epidural anesthesia is recommended to avoid endotracheal trauma and local airway edema. Short-term prophylaxis with pdhC1INH is recommended prior to the procedure87,95–97 and a treatment dose should be on hand in case of complications.9,10 Emergency procedures should not be delayed if pdhC1INH is not available because pdhC1INH can be administered at any time.10
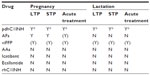
A summary of the different drugs available for the treatment of HAE is shown in Table 1.
Home care
HAE is an unpredictable, stress-provoking, and life-threatening condition that places a huge burden on patients and their families.15,16 Individualized treatment plans created by both physician and patient can increase a patient’s health-related quality of life.54
Once an HAE diagnosis is confirmed, specific treatment should be available at home and every patient should be trained for self-administration with the most suitable specific drug.54 Home therapy has been shown to reduce the severity and duration of attacks, and as a result it reduces morbidity and incapacity.98 Furthermore, home care can improve health-related quality of life for patients, and cut down the cost of care significantly.99,100
pdhC1INH therapy has been shown effective in C1-INH-HAE2,9,42,43,54,58,59,65–69 and there are some data indicating that it may also be effective in nC1-INH-HAE3,19,20,50 and it is the optimal treatment in pregnant and lactating patients with both types of HAE. Therefore, intravenous pdhC1INH home self-administration programs and training should be considered for these patients.43 This is very important, as pregnant women usually suffer from an increase in number and severity of angioedema attacks.3,12,17,19,41–43 Recent surveys reveal that self-administration training programs for intravenous drugs have been established in several HAE centers,101 and it is our hope that the number of HAE centers with training programs will continue to grow.
Author contributions
All authors contributed toward data analysis, drafting the manuscript, revising it critically for important intellectual content, and gave their final approval of the version to be published.
Disclosure
TC has received speaker fees from Shire/Jerini AG and ViroPharma, consultancy fees from Shire/Jerini AG, ViroPharma, Swedish Orphan Biovitrum S.L., and CSL Behring, sponsorship for educational purposes from CSL Behring and Shire, and has participated in clinical trials for Dyax, Pharming, CSL Behring, and Shire/Jerini AG. JC has received sponsorship for educational purposes by Shire HGT and is participating in a clinical trial for Shire. DR-P is participating in a clinical trial for Shire. RC has received sponsorship for educational purposes from CSL Behring, Shire HGT, and Viropharma, and has participated in clinical trials for Pharming and CSL Behring. The authors report no other conflicts of interest in this work.
References
Caballero T, Baeza ML, Cabañas R, et al. Consensus statement on the diagnosis, management, and treatment of angioedema mediated by bradykinin. Part I. Classification, epidemiology, pathophysiology, genetics, clinical symptoms, and diagnosis. J Investig Allergol Clin Immunol. 2011;21(5):333–347. | |
Cicardi M, Aberer W, Banerji A, et al. Classification, diagnosis and approach to treatment in angioedema: consensus report from the Hereditary Angioedema International Working Group. Allergy. 2014;69(5):602–616. | |
Bork K, Wulff K, Hardt J, Witzke G, Staubach P. Hereditary angioedema caused by missense mutations in the factor XII gene: clinical features, trigger factors, and therapy. J Allergy Clin Immunol. 2009;124(1):129–134. | |
Zuraw BL, Bork K, Binkley KE, et al. Hereditary angioedema with normal C1 inhibitor function: consensus of an international expert panel.Allergy Asthma Proc. 2012;33 Suppl 1:S145–S156. | |
Cicardi M, Agostoni A. Hereditary angioedema. N Engl J Med. 1996;334(25):1666–1667. | |
Roche O, Blanch A, Caballero T, Sastre N, Callejo D, López-Trascasa M. Hereditary angioedema due to C1 inhibitor deficiency: patient registry and approach to the prevalence in Spain. Ann Allergy Asthma Immunol. 2005;94(4):498–503. | |
Bygum A. Hereditary angio-oedema in Denmark: a nationwide survey. Br J Dermatol. 2009;161(5):1153–1158. | |
Agostoni A, Aygören-Pürsün E, Binkley KE, et al. Hereditary and acquired angioedema: problems and progress: proceedings of the third C1 esterase inhibitor deficiency workshop and beyond. J Allergy Clin Immunol. 2004;114 Suppl 3:S51–S131. | |
Caballero T, Baeza ML, Cabañas R, et al. Consensus statement on the diagnosis, management, and treatment of angioedema mediated by bradykinin. Part II. Treatment, follow-up, and special situations. J Investig Allergol Clin Immunol. 2011;21(6):422–441. | |
Caballero T, Farkas H, Bouillet L, et al. International consensus and practical guidelines on the gynecologic and obstetric management of female patients with hereditary angioedema caused by C1 inhibitor deficiency. J Allergy Clin Immunol. 2012;129(2):308–320. | |
Agostoni A, Cicardi M. Hereditary and acquired C1-inhibitor deficiency: biological and clinical characteristics in 235 patients. Medicine (Baltimore). 1992;71(4):206–215. | |
Bork K, Meng G, Staubach P, Hardt J. Hereditary angioedema: new findings concerning symptoms, affected organs, and course. Am J Med. 2006;119(3):267–274. | |
Bork K, Hardt J, Witzke G. Fatal laryngeal attacks and mortality in hereditary angioedema due to C1-INH deficiency. J Allergy Clin Immunol. 2012;130(3):692–697. | |
Bouillet L, Gompel A. Hereditary angioedema in women: specific challenges. Immunol Allergy Clin North Am. 2013;33(4):505–511. | |
Caballero T, Aygören-Pürsün E, Bygum A, et al. The humanistic burden of hereditary angioedema: results from the Burden of Illness Study in Europe. Allergy Asthma Proc. 2014;35(1):47–53. | |
Lumry WR, Castaldo AJ, Vernon MK, et al. The humanistic burden of hereditary angioedema: impact on health-related quality of life, productivity, and depression. Allergy Asthma Proc. 2010;31(5):407–414. | |
Bork K, Gul D, Hardt J, Dewald G. Hereditary angioedema with normal C1 inhibitor: clinical symptoms and course. Am J Med. 2007(11); 120:987–992. | |
Bouillet L, Launay D, Fain O, et al; for French National Reference Center for Hereditary Angioedema (CREAK). Hereditary angioedema with C1 inhibitor deficiency: clinical presentation and quality of life of 193 French patients. Ann Allergy Asthma Immunol. 2013;111(4):290–294. | |
Marcos C, López-Lera A, Varela S, Liñares T, Alvarez-Eire MG, López-Trascasa M. Clinical, biochemical, and genetic characterization of type III hereditary angioedema in 13 Northwest Spanish families. Ann Allergy Asthma Immunol. 2012;109(3):195–200. | |
Vitrat-Hincky V, Gompel A, Dumestre-Perard C, et al. Type III hereditary angio-oedema: clinical and biological features in a French cohort. Allergy. 2010;65(10):1331–1336. | |
Gómez-Traseira C, López-Lera A, Drouet C, et al. Hereditary angioedema caused by the p. Thr309-Lys mutation in the F12 gene: a multifactorial disease. J Allergy Clin Immunol. 2013;6(4):986–989. | |
Hsieh FH, Sheffer AL. Episodic swelling in a pregnant woman from Bangladesh: evaluation and management of angioedema in pregnancy.Allergy Asthma Proc. 2002;23(2):157–161. | |
Cunningham DS, Jensen JT. Hereditary angioneurotic edema in the puerperium. A case report. J Reprod Med. 1991;36(4):312–313. | |
Bowen T, Cicardi M, Farkas H, et al. 2010 international consensus algorithm for the diagnosis, therapy and management of hereditary angioedema. Allergy Asthma Clin Immunol. 2010;6(1):24. | |
Ogston D, Walker J, Campbell DM. C1 inactivator level in pregnancy. Thromb Res. 1981;23(4–5):453–455. | |
Walker JE, Campbell DM, Ogston D. Blood levels of proteinase inhibitors inpregnancy. Br J Obstet Gynaecol. 1982;89(3):208–210. | |
Halbmayer WM, Hopmeier P, Mannhalter C, et al. C1-esterase inhibitor in uncomplicated pregnancy and mild and moderate preeclampsia. Thromb Haemost. 1991;65(2):134–138. | |
Cohen AJ, Laskin C, Tarlo S. C1 esterase inhibitor in pregnancy. J Allergy Clin Immunol. 1992;90(3 Pt 1):412–413. | |
Bouillet L, Ponard D, Rousset H, et al. A case of hereditary angio-oedema type III presenting with C1-inhibitor cleavage and a missense mutation in the F12 gene. Br J Dermatol. 2007;156(5):1063–1065. | |
Sundberg K, Bang J, Smidt-Jensen S, et al. Randomised study of risk of fetal loss related to early amniocentesis versus chorionic villus sampling. Lancet. 1997;350(9079):697–703. | |
Verpoest W, Haentjens P, De Rycke M, et al. Cumulative reproductive outcome after preimplantation genetic diagnosis: a report on 1498 couples. Hum Reprod. 2009;24(11):2951–2959. | |
Bautista-Llácer R, Alberola TM, Vendrell X, Fernández E, Pérez-Alonso M. Case report: first successful application of preimplantation genetic diagnosis for hereditary angiooedema. Reprod Biomed Online. 2010;21(5):658–662. | |
Nielsen EW, Johansen HT, Holt J, Mollnes TE. C1 inhibitor and diagnosis of hereditary angioedema in newborns. Pediatr Res. 1994;35(2):184–187. | |
Davis CA, Vallota EH, Forristal J. Serum complement levels in infancy: age related changes. Pediatr Res. 1979;13(9):1043–1046. | |
Roach B, Kim Y, Jerome E, Michael AF. Influence of age and sex on serum complement components in children. Am J Dis Child. 1981;135(10):918–920. | |
Farkas H, Varga L, Szeplaki G, Visy B, Harmat G, Bowen T. Management of hereditary angioedema in pediatric patients. Pediatrics. 2007;120(3):e713–e722. | |
European Society of Human Genetics. Genetic testing in asymptomatic minors: recommendations of the European Society of Human Genetics.Eur J Hum Genet. 2009;17(6):720–721. | |
Weiler CR, van Dellen RG. Genetic test indications and interpretations in patients with hereditary angioedema. Mayo Clin Proc. 2006;81(7):958–972. | |
Kalmar L, Bors A, Farkas H, et al. Mutation screening of the C1 inhibitor gene among Hungarian patients with hereditary angioedema.Hum Mutat. 2003;22(6):498. | |
Pappalardo E, Caccia S, Suffritti C, Tordai A, Zingale LC, Cicardi M. Mutation screening of C1 inhibitor gene in 108 unrelated families with hereditary angioedema: functional and structural correlates. Mol Immunol. 2008;45(13):3536–3544. | |
Bouillet L, Longhurst H, Boccon-Gibod I, et al. Disease expression in women with hereditary angioedema. Am J Obstet Gynecol.2008;199(5):484. e1–e4. | |
Czaller I, Visy B, Csuka D, Füst G, Tóth F, Farkas H. The natural history of hereditary angioedema and the impact of treatment with human C1-inhibitor concentrate during pregnancy: a long-term survey. Eur J Obstet Gynecol Reprod Biol. 2010;152(1):44–49. | |
Martinez-Saguer I, Rusicke E, Aygören-Pürsün E, Heller C, Klingebiel T, Kreuz W. Characterization of acute hereditary angioedema attacks during pregnancy and breast-feeding and their treatment with C1 inhibitor concentrate. Am J Obstet Gynecol.2010;203(2):131. e1–e7. | |
Winnewisser J, Rossi M, Spath P, Burgi H. Type I hereditary angio-oedema, variability of clinical presentation and course within two large kindreds. J Intern Med. 1997;241(1):39–46. | |
Chinniah N, Katelaris CH. Hereditary angioedema and pregnancy. Aust N Z J Obstet Gynaecol. 2009;49(1):2–5. | |
Caliskaner Z, Ozturka S, Guleca M, Dedeb M, Erela F, Karaayvaza M. A successful pregnancy and uncomplicated labor with C1-INH concentrate prophylaxis in a patient with hereditary angioedema. Allergol Immunopathol. 2007;35(3):117–119. | |
Bork K, Barnstedt SE, Koch P, Traupe H. Hereditary angioedema with normal C1 inhibitor activity in women. Lancet. 2000;356(9225):213–217. | |
Binkley KE, Davis A 3rd. Clinical, biochemical and genetic characterization of a novel estrogen-dependent inherited form of angioedema. J Allergy Clin Immunol. 2000;106(3):546–550. | |
Martin L, Raison-Peyron N, Nothen MM, Cichon S, Drouet C. Hereditary angioedema with normal C1 inhibitor gene in a family with affected women and men is associated with the pThr328Lys mutation in the F12 gene. J Allergy Clin Immunol. 2007;120(4):975–977. | |
Picone O, Donnadieu AC, Brivet FG, et al. Obstetrical complications and outcome in two families with hereditary angioedema due to mutation in the F12 gene. Obstet Gynecol Int. 2010;2010:957507. | |
Logan RA, Greaves MW. Hereditary angio-oedema: treatment with C1 esterase inhibitor concentrate. J R Soc Med. 1984;77(12):1046–1048. | |
Ebert A, Pritze W, Weitzel HK. [C-1 esterase inhibitor deficiency as an obstetric problem: a case report]. Zentralbl Gynakol. 1992;114(10):519–522. German. | |
Böckers M, Bork K. [Contraception and pregnancy in hereditary angioedema]. Dtsch Med Wochenschr. 1987;112(13):507–509. German. | |
Craig T, Aygören-Pürsün E, Bork K, et al. WAO guideline for the management of hereditary angioedema.World Allergy Organ J. 2012;5(12):182–199. | |
Nathani F, Sullivan H, Churchill D. Pregnancy and C1 esterase inhibitor deficiency: a successful outcome. Arch Gynecol Obstet. 2006;274(6):381–384. | |
Visy B, Füst G, Varga L, et al. Sex hormones in hereditary angioneurotic oedema. Clin Endocrinol (Oxf). 2004;60(4):508–515. | |
Nielsen EW, Gran JT, Straume B, Mellbye OJ, Johansen HT, Mollnes TE. Hereditary angio-oedema: new clinical observations and autoimmune screening, complement and kalikrein-kinin analyses. J Intern Med. 1996;239(2):119–130. | |
Cicardi M, Bork K, Caballero T, et al. Evidence-based recommendations for the therapeutic management of angioedema owing to hereditary C1 inhibitor deficiency: consensus report of an International Working Group. Allergy. 2012;67(2):147–157. | |
Costantino G, Casazza G, Bossi I, Duca P, Cicardi M. Long-term prophylaxis in hereditary angio-oedema: a systematic review. BMJ Open. 2012;2(4):pii e000524. | |
Bork K, Hardt J, Staubach-Renz P, Witzke G. Risk of laryngeal edema and facial swellings after tooth extraction in patients with hereditary angioedema with and without prophylaxis with C1 inhibitor concentrate: a retrospective study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(1):58–64. | |
Farkas H, Zotter Z, Csuka D, et al. Short-term prophylaxis in hereditary angioedema due to deficiency of the C1-inhibitor – a long-term survey. Allergy. 2012;67(12):1586–1593. | |
Grant JA, White MV, Lee HH, et al. Preprocedural administration of nanofiltered C1 esterase inhibitor to prevent hereditary angioedema attacks. Allergy Asthma Proc. 2012;33(4):348–353. | |
Jurado-Palomo J, Muñoz-Caro JM, López-Serrano MC, et al. Management of dental-stomatologic procedures in patients with hereditary angio-oedema due to C1 inhibitor deficiency (HAE-C1-INH). J Investig Allergol Clin Immunol. 2013;23:81–83. | |
Aygören-Pürsün E, Martinez Saguer I, Kreuz W, Klingebiel T, Schwabe D. Risk of angioedema following invasive or surgical procedures in HAE type I and II – the natural history. Allergy. 2013;68(8):1034–1039. | |
Hermans C. Successful management with C1-inhibitor concentrate of hereditary angioedema attacks during two successive pregnancies: a case report. Arch Gynecol Obstet. 2007(3);276:271–276. | |
Farkas H, Jakab K, Temesszentandrási F, et al. Hereditary angioedema: a decade of human C1-inhibitor concentrate therapy.J Allergy Clin Immunol. 2007;120(4):941–947. | |
Gorman PJ. Hereditary angioedema and pregnancy: a successful outcome using C1 esterase inhibitor concentrate. Can Fam Physician. 2008;54(3):365–366. | |
Baker JW, Craig TJ, Riedl MA, et al. Nanofiltered C1 esterase inhibitor (human) for hereditary angioedema attacks in pregnant women. Allergy Asthma Proc. 2013;34(2):162–169. | |
Chan W, Berlin N, Sussman GL. Management of hereditary angioedema with C1-inhibitor concentrate during two successive pregnancies.Int J Gynaecol Obstet. 2013;120(2):189–190. | |
Pedrosa M, Lobera T, Panizo C, Jurado J, Caballero T. Long-term prophylaxis with C1-inhibitor concentrate in patients with hereditary angioedema. J Invest Allergy Clin Immunol. In press 2014. | |
arkas H, Csuka D, Tóth F, Koszegi L, Varga L. Successful pregnancy outcome after treatment with C1-inhibitor concentrate in a patient with hereditary angioedema and a history of four miscarriages. Eur J Obstet Gynecol Reprod Biol. 2012;165(2):366–367. | |
Dunn CJ, Goa KL. Tranexamic acid: a review of its use in surgery and other indications. Drugs. 1999;57(6):1005–1032. | |
Kullander S, Nilsson IM. Human placental transfer of an antifibrinolytic agent (AMCA). Acta Obstet Gynecol Scand. 1970;49(3):241–242. | |
Walzman M, Bonnar J. Effects of tranexamic acid on the coagulation and fibrinolytic systems in pregnancy complicated by placental bleeding.Arch Toxicol Suppl. 1982;5:214–220. | |
Chi C, Kulkarni A, Lee CA, Kadir RA. The obstetric experience of women with factor XI deficiency. Acta Obstet Gynecol Scand. 2009;88(10):1095–1100. | |
Frank MM, Gelfand JA, Atkinson JP. Hereditary angioedema: the clinical syndrome and its management. Ann Intern Med. 1976;84(5):580–593. | |
Frank MM, Sergent JS, Kane MA, Alling DW. Epsilon aminocaproic acid therapy of hereditary angioneurotic edema: a double-blind study. N Engl J Med. 1972;286(15):808–812. | |
Reschini E, Giustina G, D’Alberton A, Candiani GB. Female pseudohermaphroditism due to maternal androgen administration: 25-year follow-up. Lancet. 1985;1(8439):1226. | |
Duck SC, Katayama KP. Danazol may cause female pseudohermaphroditism. Fertil Steril. 1981;35(2):230–231. | |
Cicardi M, Bergamaschini L, Cugno M, Hack E, Agostoni G, Agostoni A. Long term treatment of hereditary angioedema with attenuated androgens: a survey of a 13 year experience. J Allergy Clin Immunol. 1991;87(4):768–773. | |
Manikkam M, Crespi EJ, Doop DD, et al. Fetal programming: prenatal testosterone excess leads to fetal growth retardation and postnatal catch-up growth in sheep. Endocrinology. 2004;145(2):790–798. | |
Castro-Magana M, Cheruvanky T, Collipp PJ, Ghavami-Maibodi Z, Angulo M, Stewart C. Transient adrenogenital syndrome due to exposure to danazol in utero. Am J Dis Child. 1981;135(11):1032–1034. | |
Schwartz R. Ambiguous genitalia in a term female infant due to exposure to danazol in utero. Am J Dis Child. 1982;136(5):474. | |
Sathishkumar K, Elkins R, Yallampalli U, Yallampalli C. Elevated androgens levels during pregnancy impair fetal growth due to placental insufficiency and programs for adult hypertension in rats. Biol Reprod. 2009;81:250. | |
Boulos AN, Brown R, Hukin A, Williams RM. Danazol prophylaxis for delivery in hereditary angioneurotic oedema. Br J Obstet Gynaecol. 1994;101(12):1094–1095. | |
McGlinchey PG, Golchin K, McCluskey DR. Life-threatening laryngeal oedema in a pregnant woman with hereditary angioedema. Ulster Med J. 2000;69(1):54–57. | |
Hopkinson RB, Sutcliffe AJ. Hereditary angioneurotic oedema. Anaesthesia. 1979;34(2):183–186. | |
Galan HL, Reedy MB, Starr J, Knight AB. Fresh frozen plasma prophylaxis for hereditary angioedema during pregnancy. A case report. J Reprod Med. 1996;41(7):541–544. | |
Prematta M, Gibbs JG, Pratt EL, Stoughton TR, Craig TJ. Fresh frozen plasma for the treatment of hereditary angioedema. Ann Allergy Asthma Immunol. 2007;98(4):383–388. | |
Longhurst HJ. Emergency treatment of acute attacks in hereditary angioedema due to C1 inhibitor deficiency: what is the evidence? Int J Clin Pract. 2005;59(5):594–599. | |
Zuraw BL, Bernstein JA, Lang DM, et al. A focused parameter update: hereditary angioedema, acquired C1 inhibitor deficiency, and angiotensin-converting enzyme inhibitor-associated angioedema. J Allergy Clin Immunol. 2013;131(6):1491–1493. | |
Szema AM, Paz G, Merriam L, Stellaccio F, Jen J. Modern preoperative and intraoperative management of hereditary angioedema. Allergy Asthma Proc. 2009;30(3):338–342. | |
Postnikoff IM, Pritzker KP. Hereditary angioneurotic edema: an unusual case of maternal mortality. J Forensic Sci. 1979;24(2):473–478. | |
Blohme G, Ysander L, Korsan-Bengtsen K, Laurell AB. Hereditary angioneurotic oedema in three families. Symptomatic heterogeneity, complement analysis and therapeutic trials. Acta Med Scand. 1972; 191(3):209–219. | |
Gompels MM, Lock RJ, Abinun M, et al. C1 inhibitor deficiency: consensus document. Clin Exp Immunol. 2005;139(3):379–394. | |
Cox M, Holdcroft A. Hereditary angioneurotic oedema: current management in pregnancy. Anaesthesia. 1995;50(6):547–549. | |
Hardy F, Ngwingtin L, Bazin C, Babinet P. Hereditary angioneurotic edema and pregnancy. J Gynecol Obstet Biol Reprod (Paris). 1990;19:65–68. | |
Longhurst HJ, Farkas H, Craig T, et al. HAE international home therapy consensus document. Allergy Asthma Clin Immunol. 2010;6(1):22. | |
Bygum A, Andersen KE, Mikkelsen CS. Self-administration of intravenous C1-inhibitor therapy for hereditary angioedema and associated quality of life benefits. Eur J Dermatol. 2009;19(2):147–151. | |
Wilson DA, Bork K, Shea EP, Rentz AM, Blaustein MB, Pullman WE. Economic costs associated with acute attacks and long-term management of hereditary angioedema. Ann Allergy Asthma Immunol. 2010;104(4):314–320. | |
Caballero T, Sala-Cunill A, Cancian M, et al. Current status of implementation of self-administration training in various regions of Europe, Canada and the USA in the management of hereditary angioedema. Int Arch Allergy Immunol. 2013;161 Suppl 1: 10–16. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.