Back to Journals » Clinical and Experimental Gastroenterology » Volume 8
Low serum levels of short-chain fatty acids after lactulose ingestion may indicate impaired colonic fermentation in patients with irritable bowel syndrome
Authors Undseth R, Jakobsdottir G, Nyman M, Berstad A, Valeur J
Received 9 August 2015
Accepted for publication 12 October 2015
Published 27 November 2015 Volume 2015:8 Pages 303—308
DOI https://doi.org/10.2147/CEG.S94084
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Andreas M. Kaiser
Ragnhild Undseth,1 Greta Jakobsdottir,2 Margareta Nyman,2 Arnold Berstad,3 Jørgen Valeur3
1Department of Radiology, Lovisenberg Diaconal Hospital, Oslo, Norway; 2Food for Health Science Centre, Lund University, Lund, Sweden; 3Unger-Vetlesen Institute, Lovisenberg Diaconal Hospital, Oslo, Norway
Background: Ingestion of low-digestible carbohydrates triggers symptoms in patients with irritable bowel syndrome (IBS). These carbohydrates become substrates for microbial fermentation in the colon, yielding short-chain fatty acids (SCFAs) that are readily absorbed. Aiming to compare colonic fermentation in patients with IBS and healthy controls, we analyzed the concentrations of SCFA in serum at fasting and 90 minutes following ingestion of an unabsorbable, but fermentable carbohydrate, lactulose.
Methods: Patients with IBS according to Rome III criteria (n=22) and healthy controls (n=20) ingested 10 g lactulose dissolved in water. Symptoms were graded by questionnaires and SCFA were analyzed using hollow fiber-supported liquid membrane extraction coupled with gas chromatography.
Results: Lactulose induced more symptoms in patients with IBS than in healthy controls (P=0.0001). Fasting serum levels of SCFA did not differ between patients with IBS and controls. However, the postprandial levels of total SCFA (P=0.0002), acetic acid (P=0.005), propionic acid (P=0.0001), and butyric acid (P=0.01) were significantly lower in patients with IBS compared with healthy controls. There was no correlation between the levels of serum SCFA and symptom severity.
Conclusion: Low-serum levels of SCFA after lactulose ingestion may indicate impaired colonic fermentation in patients with IBS. Conceivably, this disturbance is related to symptom generation, but the mechanism is not clear.
Keywords: fermentation, FODMAP, irritable bowel syndrome, microbiota, short-chain fatty acids
Introduction
The pathophysiology of irritable bowel syndrome (IBS) seems to involve disturbances at many levels of the so-called gut-brain axis.1 Recent research has emphasized the role of food in abdominal symptom generation,2 in particular low-digestible carbohydrates.3 Our group has previously shown that the ingestion of the unabsorbable, but fermentable disaccharide lactulose provokes more symptoms in patients with IBS than in healthy individuals.4,5 Intriguingly, the symptoms provoked by lactulose were similar to the patients’ habitual complaints.4 Fermentable carbohydrates that escape digestion and absorption within the small intestine are metabolized by colonic microbiota. Hunter6 has previously suggested that the disturbances of colonic fermentation play a significant role in IBS symptom generation, and has proposed the term “enterometabolic disorder” to describe such alterations. Colonic fermentation yields short-chain fatty acids (SCFAs; mainly acetic, propionic, and butyric acids) and gases (mainly CO2, H2, and CH4). Hence, the efficacy of the fermentation may be evaluated by SCFA analysis. Due to regional differences regarding the availability of fermentable substrates and abundance of saccharolytic microbes, fermentation of low-digestible carbohydrates takes place mainly in the cecum and the proximal colon, rather than in the distal parts.7 A correlation has previously been demonstrated between cecal levels of SCFA and portal and aortic blood levels of SCFA in rats,8 and assessment of SCFA in peripheral venous blood may be considered as an indirect measure of colonic fermentation.9
The aim of the present study was to compare serum SCFA levels in patients with IBS and healthy controls before and after the ingestion of lactulose. Since a previous study has shown that the mean orocecal transit time of a 10 g lactulose solution is approximately 90 minutes,10 and that the onset of symptoms after a meal often occurs within 90 minutes,11 we chose this time point for evaluating the lactulose-stimulated response.
Patients and methods
Study participants
Patients with IBS were included consecutively from the outpatient clinic of one of the authors at Lovisenberg Diaconal Hospital (Oslo, Norway). The individuals were diagnosed with IBS according to the Rome III criteria,12 and divided into subgroups according to phenotypes based on the Rome III questionnaire for functional bowel disorders: diarrhea predominant IBS, constipation predominant IBS, and IBS with equally diarrhea and constipation. Patients with organic diseases were excluded. Healthy controls were recruited among the hospital staff at Lovisenberg Diaconal Hospital. They were included if they considered themselves to be healthy, and were not otherwise medically examined. However, they were excluded if they met the Rome III criteria for IBS based on our questionnaires. All participants gave written, informed consent, and the study was carried out according to the Declaration of Helsinki and approved by the Regional Committee for Medical Research Ethics (REK Sør-øst; reference number 2011/2474).
Lactulose challenge procedure
The participants met (individually) at the outpatient clinic at 08:00 in the morning after an overnight fast. Venous blood samples, from the antecubital vein of the non-dominant arm, were collected and the subjects filled in a questionnaire to quantify their habitual IBS symptoms. The participants then underwent a lactulose provocation test, as described previously.4 Briefly, the subjects ingested 10 g of lactulose dissolved in 200 mL of tap water. Ninety minutes after the intake of the lactulose solution, another blood sample was collected, and the subjects filled in a questionnaire to grade their lactulose-induced symptoms. The participants were not allowed to drink, eat, or smoke during the test, but their activities were otherwise not restricted.
Symptom questionnaires
Habitual symptoms were assessed using the IBS severity scoring system.13 The maximum achievable score of this system is 500 points, and the questionnaire allows grading of symptom severity as follows: mild (75–175 points), moderate (175–300 points), and severe (>300 points). Increase in symptoms following the lactulose ingestion, as compared to baseline, was graded from 0 to 3 for abdominal borborygmi, bloating, and pain, as well as a feeling of sickness and freezing, giving a maximum achievable sum score of 15 points.
Analysis of blood samples
After collection, the samples were centrifuged and the serum was immediately placed in a freezer at −20°C until analysis. Serum was thawed at room temperature and SCFA (acetic, propionic, butyric, iso-butyric, valeric, and iso-valeric acids) were analyzed by using a method developed by Zhao et al.14 This method enables the detection of serum concentrations of SCFA down to as little as 0.04 μM, as described by Jakobsdottir et al.9 All samples were analyzed at least in duplicate. In addition, the ratios of individual SCFA concentrations to the total SCFA concentrations were calculated. Based on the work of Tjellström et al15 on fecal samples, we calculated two fermentation indices, as follows: fermentation index A, reflecting the fermentation of carbohydrates and conceivably the pro-inflammatory properties of SCFA, was calculated as acetic acid concentrations minus propionic and butyric acid concentrations divided by the total concentration of SCFA. Fermentation index B, reflecting the fermentation of proteins and conceivably the anti-inflammatory properties of SCFA was calculated as the sum of iso-butyric and iso-valeric acids.
Statistical analyses
Graphpad Prism version 6 (Graphpad Software Inc., San Diego, CA, USA) was used to analyze the data. Unless otherwise stated, the data are expressed as means and their SD. Paired t-test was used for comparisons within groups and unpaired t-tests for comparing means between groups. Correlations were assessed with Pearson’s correlation coefficient. To compare fractions, the Fishers exact test was conducted. All tests were two-tailed, and P-values less than 0.05 were set as a limit for statistical significance.
Results
Characteristics of study participants
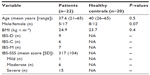
A total of 22 patients with IBS and 20 healthy controls were included in the study. The groups were comparable regarding age, body mass index, and sex (Table 1).
Symptoms following lactulose ingestion
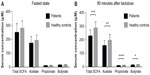
Patients with IBS reported more symptoms in response to lactulose ingestion than healthy controls (Figure 1). Abdominal symptoms (borborygmi, bloating, and pain) were most pronounced, with a mean sum score of 3.2 points (maximum achievable score 9 points), while systemic symptoms (sickness and freezing) had a sum score of 2.2 points (maximum achievable score 6 points).
Serum levels of SCFA
Levels of SCFA in serum obtained in the fasted state were not significantly different between patients with IBS and healthy controls, for any of the SCFA. The results (in μM) were as follows: total SCFA 249.6±63.9 vs 279.9±53.4 (P=0.1); acetic acid 172.5±48.9 vs 191.1±40.0 (P=0.2); propionic acid 10.8±1.7 vs 11.5±2.3 (P=0.3); and butyric acid 12.0±3.2 vs 16.0±4.2 (P=0.4; Figure 2A). Similar results, with no significant differences, were seen for iso-butyric (13.5±4.8 vs 15.3±5.0; P=0.2), iso-valeric (39.0±5.4 vs 47.4±14.0; P=0.07) and valeric acids (1.2±0.5 vs 1.3±0.6; P=0.4).
The levels of SCFA in serum 90 minutes after lactulose ingestion were significantly lower in patients with IBS than in healthy controls for all the major SCFA: total SCFA 228.0±43.9 vs 285.9±45.1 (P=0.0002); acetic acid 157.2±37.2 vs 195.4±43.7 (P=0.005); propionic acid 10.2±1.2 vs 12.0±1.5 (P=0.0001); and butyric acid 13.3±4.5 vs 16.7±3.2 (P=0.01; Figure 2B). Significant differences were also seen for iso-butyric (35.9±12.8 vs 47.8±11.9; P=0.01), iso-valeric (35.9±12.8 vs 47.4±14.0; P=0.004) and valeric acids (1.3±0.4 vs 1.1±0.2; P=0.08).
No correlation was seen between the levels of SCFA in serum obtained 90 minutes after lactulose ingestion and the degree of symptoms, IBS severity scoring system, age, sex, or body mass index. Additionally, there were no differences between the subgroups with IBS.
Fermentation index A was similar in patients with IBS and healthy controls and did not change following lactulose ingestion. Fermentation index B was similar in patients with IBS and healthy controls in the fasting state. However, a significant decline in index B was seen in patients with IBS after the lactulose ingestion (P=0.01).
Discussion
In the present study, we found that patients with IBS had significantly lower serum levels of SCFA following lactulose ingestion than healthy controls. This novel finding suggests an impairment of colonic fermentation in patients with IBS. This might be surprising since the levels of SCFA in serum reflect the levels of SCFA in cecum and the proximal colon, that is, the actual site for fermentation,8 and prior studies have reported increased (not decreased) levels of SCFA in feces of patients with IBS.16 Lack of prior data concerning SCFA in serum in patients with IBS might be due to the shortage of appropriate analytical methods. However, the present method, recently developed by Zhao et al,14 enables accurate quantification of SCFA, including iso-butyric, iso-valeric, and valeric acids, in serum. It may be speculated that it would have been better to collect several blood samples instead of only one single blood sample after 90 minutes,17 especially since the assessment of orocecal transit time was not included in the present study. However, previous studies evaluating the effects of lactulose in patients with IBS have concluded that 90 minutes after lactulose intake is the average time point at which lactulose reaches the cecum.10 Additionally, this is the average time point for the onset of symptoms after a meal.11
The mechanism by which symptoms are evoked is not known, but lactulose provocation likely overloads the fermentation capacity of the proximal colon. An overload of indigestible carbohydrates in the colon may act osmotically to recruit fluid into the gut lumen. SCFA stimulate the uptake of salt and water,18 and decreased fluid absorption in the proximal colon conceivably contributes to rectal overload and diarrhea. However, we found no correlation between the symptom development in patients with IBS and the serum levels of SCFA. Neither did we find any difference in our principal findings between subgroups of IBS. However, we acknowledge that lack of statistically significant differences might also be due to the small patient material.
We have previously shown that lactulose causes abnormal accumulation of fluid in the small intestine in patients with IBS,19 a phenomenon that might indicate the presence of an underlying motility disorder. When the buildup of fluid in the small intestine enters the cecum, symptoms may be evoked due to poor cecal accommodation,20 particularly if the influx occurs in unregulated flushes, thereby overloading the colonic fermentation and salvaging capacity.21 Logically, microbial fermentation will continue distally throughout the colon as long as substrates remain. Indeed, a recent study may indicate that fermentation, measured as pH, persists throughout the whole colon in patients with IBS.22 Increased levels of SCFA in feces in patients with IBS16 may thus indicate a distal displacement of colonic fermentation. Hence, the levels of SCFA in patients with IBS may be different from healthy subjects throughout the colon, from abnormally low in the proximal part to abnormally high in the distal part.
Distal displacement of colonic fermentation may have several clinical consequences.23 For example, SCFA stimulate the enterochromaffin cells in the colorectal mucosa to increase the production of serotonin,24 a neurotransmitter exerting important physiological effects on both sensory and motoric neural transmission. In addition, SCFA from the distal colon bypasses the portal circulation, enabling systemic access and activation of specific SCFA receptors present in both the autonomic and somatic nervous system.25 However, delayed fermentation could be positive since many of the diseases that occur in the gastrointestinal tract are located in the lower part, like colorectal cancer and inflammation, and regarding these conditions, high concentrations of SCFA could be protective.26
A widespread treatment method for IBS is dietary advice, guiding patients to avoid foods high in low-digestible and fermentable short-chain carbohydrates (FODMAP; fermentable oligo-, di- and mono-saccharides, and polyols). The rationale behind this treatment concept is probably to reduce the fermentation burden on the colon.27 However, a low FODMAP diet is probably not a measure to improve the colonic fermenting capacity. Indeed, recent studies suggest that FODMAP restriction may induce unfortunate changes of gut microbiota.28
In the present study, we calculated two fermentation indices according to Tjellström et al:15 Index A, reflecting the fermentation of carbohydrates (high index suggesting a trend toward a pro-inflammatory condition) and index B, indicating the fermentation of proteins (high index suggesting a trend toward an anti-inflammatory condition). According to these indicators, we found that the fermentation of proteins decreases in the postprandial state in patients with IBS compared with healthy controls. This might suggest that the colonic content in patients with IBS has pro-inflammatory properties, conceivably related to subclinical inflammation, as reported in patients with IBS by other groups.29 However, we acknowledge that the two proposed fermentation indices have not yet been formally validated.
Conclusion
We found that the fermentation of lactulose leads to lower serum levels of SCFA in patients with IBS than in healthy controls, suggesting impaired colonic fermentation. When developing new treatments, it might be wise to target the fermentation process itself. Efforts to improve colonic fermentation capacity may be essential for patients with IBS.
Acknowledgment
Our submitted manuscript is a follow-up on our previous article entitled “Abnormal accumulation of intestinal fluid following ingestion of an unabsorbable carbohydrate in patients with irritable bowel syndrome: an MRI study”.19 The abstract of this paper was presented at the 22nd United European Gastroenterology Week (UEGW) in Vienna in 2014 as a poster presentation with interim findings.30 The actual paper, however, has never been published before.
Disclosure
The authors report no conflicts of interest in this work.
References
Camilleri M. Peripheral mechanisms in irritable bowel syndrome. N Engl J Med. 2012;367(17):1626–1635. | |
Chey WD. The role of food in the functional gastrointestinal disorders: introduction to a manuscript series. Am J Gastroenterol. 2013; 108(5):694–697. | |
Staudacher HM, Irving PM, Lomer MC, Whelan K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat Rev Gastroenterol Hepatol. 2014;11(4):256–266. | |
Valeur J, Morken MH, Norin E, Midtvedt T, Berstad A. Carbohydrate intolerance in patients with self-reported food hypersensitivity: comparison of lactulose and glucose. Scand J Gastroenterol. 2009; 44(12):1416–1423. | |
Morken MH, Nysaeter G, Strand EA, Hausken T, Berstad A. Lactulose breath test results in patients with persistent abdominal symptoms following Giardia lamblia infection. Scand J Gastroenterol. 2008;43(2):141–145. | |
Hunter JO. Food allergy–or enterometabolic disorder? Lancet. 1991;338(8765):495–496. | |
Topping DL, Clifton PM. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev. 2001;81(3):1031–1064. | |
Jakobsdottir G, Jadert C, Holm L, Nyman ME. Propionic and butyric acids, formed in the caecum of rats fed highly fermentable dietary fibre, are reflected in portal and aortic serum. Br J Nutr. 2013;110(9):1565–1572. | |
Jakobsdottir G, Bjerregaard JH, Skovbjerg H, Nyman M. Fasting serum concentration of short-chain fatty acids in subjects with microscopic colitis and celiac disease: no difference compared with controls, but between genders. Scand J Gastroenterol. 2013;48(6):696–701. | |
Yu D, Cheeseman F, Vanner S. Combined oro-caecal scintigraphy and lactulose hydrogen breath testing demonstrate that breath testing detects oro-caecal transit, not small intestinal bacterial overgrowth in patients with IBS. Gut. 2011;60(3):334–340. | |
Arsie E, Coletta M, Cesana BM, Basilisco G. Symptom-association probability between meal ingestion and abdominal pain in patients with irritable bowel syndrome. Does somatization play a role? Neurogastroenterol Motil. 2015;27:416–422. | |
Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders. Gastroenterology. 2006; 130(5):1480–1491. | |
Francis CY, Morris J, Whorwell PJ. The irritable bowel severity scoring system: a simple method of monitoring irritable bowel syndrome and its progress. Aliment Pharmacol Ther. 1997;11(2):395–402. | |
Zhao G, Liu JF, Nyman M, Jonsson JA. Determination of short-chain fatty acids in serum by hollow fiber supported liquid membrane extraction coupled with gas chromatography. J Chromatogr B Analyt Technol Biomed Life Sci. 2007;846(1–2):202–208. | |
Tjellström B, Hogberg L, Stenhammar L, et al. Effect of exclusive enteral nutrition on gut microflora function in children with Crohn’s disease. Scand J Gastroenterol. 2012;47(12):1454–1459. | |
Tana C, Umesaki Y, Imaoka A, Handa T, Kanazawa M, Fukudo S. Altered profiles of intestinal microbiota and organic acids may be the origin of symptoms in irritable bowel syndrome. Neurogastroenterol Motil. 2010;22(5):512–515. | |
Florent C, Flourie B, Leblond A, Rautureau M, Bernier JJ, Rambaud JC. Influence of chronic lactulose ingestion on the colonic metabolism of lactulose in man (an in vivo study). J Clin Invest. 1985; 75(2):608–613. | |
Binder HJ. Role of colonic short-chain fatty acid transport in diarrhea. Annu Rev Physiol. 2010;72:297–313. | |
Undseth R, Berstad A, Klow NE, Arnljot K, Moi KS, Valeur J. Abnormal accumulation of intestinal fluid following ingestion of an unabsorbable carbohydrate in patients with irritable bowel syndrome: an MRI study. Neurogastroenterol Motil. 2014;26(12):1686–1693. | |
Pritchard SE, Marciani L, Garsed KC, et al. Fasting and postprandial volumes of the undisturbed colon: normal values and changes in diarrhea-predominant irritable bowel syndrome measured using serial MRI. Neurogastroenterol Motil. 2014;26(1):124–130. | |
Read NW. Diarrhoea: the failure of colonic salvage. Lancet. 1982; 2(8296):481–483. | |
Farmer AD, Mohammed SD, Dukes GE, Scott SM, Hobson AR. Caecal pH is a biomarker of excessive colonic fermentation. World J Gastroenterol. 2014;20(17):5000–5007. | |
Bourdu S, Dapoigny M, Chapuy E, et al. Rectal instillation of butyrate provides a novel clinically relevant model of noninflammatory colonic hypersensitivity in rats. Gastroenterology. 2005;128(7):1996–2008. | |
Reigstad CS, Salmonson CE, Rainey JF III, et al. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015;29:1395–1403. | |
Nohr MK, Egerod KL, Christiansen SH, et al. Expression of the short chain fatty acid receptor GPR41/FFAR3 in autonomic and somatic sensory ganglia. Neuroscience. 2015 2;290:126–137. | |
Valeur J, Morken MH, Norin E, Midtvedt T, Berstad A. Intestinal fermentation in patients with self-reported food hypersensitivity: painful, but protective? Clin Exp Gastroenterol. 2010;3:65–70. | |
Ong DK, Mitchell SB, Barrett JS, et al. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J Gastroenterol Hepatol. 2010; 25(8):1366–1373. | |
Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut. 2015;64(1):93–100. | |
Ohman L, Simren M. Pathogenesis of IBS: role of inflammation, immunity and neuroimmune interactions. Nat Rev Gastroenterol Hepatol. 2010;7(3):163–173. | |
Undseth R, Jakobsdottir G, Nyman M, Berstad A, Valeur J. Low serum levels of short-chain fatty acids after lactulose ingestion may indicate impaired microbial fermentation in patients with irritable bowel syndrome. United Eur Gastroenterol J. 2014;2(Suppl 1):A556. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.