Back to Journals » Clinical Ophthalmology » Volume 16
Liposomal Ozonated Oil in Ocular Infections: A Review of Preclinical and Clinical Studies, Focusing on Its Antiseptic and Regenerative Properties
Authors Pérez-Santonja JJ , Güell JL, Gris O, Vázquez Dorrego XM, Pellicer E , Benítez-Del-Castillo JM
Received 2 February 2022
Accepted for publication 20 May 2022
Published 14 June 2022 Volume 2022:16 Pages 1953—1962
DOI https://doi.org/10.2147/OPTH.S360929
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Juan J Pérez-Santonja,1 José Luis Güell,2 Oscar Gris,2 Xose Manuel Vázquez Dorrego,3 Esther Pellicer,4 Jose Manuel Benítez-Del-Castillo5– 7
1Department of Ophthalmology, Alicante University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Alicante, Spain; 2Department of Cornea and Refractive Surgery, Institut de Microcirurgia Ocular (IMO), Barcelona, Spain; 3Department of Ophthalmology, Hospital Municipal de Badalona, Badalona (Barcelona), Spain; 4EPT Servicios Científicos, Barcelona, Spain; 5Department of Ophthalmology, Hospital Clínico San Carlos de Madrid, Madrid, Spain; 6Universidad Complutense de Madrid, Instituto de Investigaciones Oftalmológicas Ramón Castroviejo, Madrid, Spain; 7Clínica Rementería, Madrid, Spain
Correspondence: Jose Manuel Benítez-Del-Castillo, Hospital Clinico San Carlos, C/ Martin Lagos s/n, Madrid, 28040, Spain, Tel +34 913303000, Fax +34 913941359, Email [email protected]
Abstract: Ozonated oil has shown antimicrobial, anti-inflammatory and regenerative properties that make it useful in the prevention of infectious diseases and also as an adjuvant in wound-healing management. This review brings together most aspects of the use of liposomal ozonated oil for ocular infections and regeneration of the ocular surface. A search was performed in PubMed, Medline, Web of Science and Cochrane Library for studies published by June 2021. Search terms were combined to sort out papers on the properties and use of ozonated oil in ocular infections. A total of 25 publications were selected for this review on the composition, mechanism of action, restorative action, and preclinical and clinical studies of liposomal ozonated oil focused on ocular infections. In patients with complicated corneal pathology, liposomal ozonated oil has been found to restore corneal ulcers and improve keratitis. In patients with ocular pathologies involving inflammation and infections, liposomal ozonated oil has been found to improve and almost completely restore the signs of vernal, granulomatous and even adenoviral conjunctivitis. Liposomal ozonated oil has also been found to be effective in reducing ocular microbial flora. In conclusion, liposomal ozonated oil has an antiseptic and regenerative effect on corneoconjunctival tissues. It has demonstrated efficacy and safety profile for its use in ocular infections and can be considered as a suitable supportive strategy both alone and combined with other antimicrobial agents.
Keywords: antifungal, antimicrobial, antiviral, liposomal ozonated oil, restorative action
Introduction
Liposomal ozonated oil is a novel ocular antiseptic. Since the 1950s, when povidone-iodine and chlorhexidine were introduced, no other topical antiseptics for ocular surface use had been described. Povidone-iodine has been used as an effective broad-spectrum antiseptic and disinfectant since its inception in 1955, and aqueous chlorhexidine has been described as a possibly better tolerated antiseptic for ophthalmic procedures than povidone-iodine.1
Liposomal-encapsulated ozonated oil is the first ozone-based ocular antiseptic intended to support the prevention and treatment of ocular infections.2–8
Ozone is a powerful oxidant with known bactericidal, antiviral and antifungal properties as well as anti-inflammatory activity and tissue-repair promoting properties, which have been widely used in several pathologies including wound healing in skin diseases, periodontics, musculoskeletal disorders, diabetic ulcers, and eye infections and injuries among others.2,7,8 Ozonated oil maintains the same properties as gaseous ozone. It can be obtained by a reaction between gaseous ozone and double carbon–carbon bonds of unsaturated fatty acids.8,9 With the aim of improving the tolerance of ozonated oil by the ocular surface, it is encapsulated in liposomes and included in a solution of hypromellose.3
The structure of the eye is divided into the anterior and posterior segments, and the different structures provide natural barriers such as the tears, the cornea, the blood-aqueous barrier and the blood-retinal barrier. The most common eye disorders diagnosed in both animals and humans are inflammatory anterior segment diseases, such as conjunctivitis, keratoconjunctivitis, and corneal ulcers.9
Ocular infections are caused by a large number of microorganisms, involving bacteria, virus, fungi and yeast. Bacteria such as Staphylococcus aureus, coagulase-negative staphylococci and Pseudomonas aeruginosa are among the most frequent eye pathogens causing acute and chronic infections.3 The most common viral infections in the eye are in the form of conjunctivitis and keratitis, mainly due to adenovirus and occasionally to herpes simplex or herpes zoster virus.2,10 Virus replication in the corneal epithelium leads to epithelial keratitis causing opacities, which can also affect the anterior part of the stroma due to the accumulation of lymphocytes, macrophages, monocytes and fibroblasts in response to the infection.10 Ocular fungal infections related to fungi, such as Aspergillus and Fusarium, and yeast, particularly Candida, can affect any part of the ocular surface and can extend to the interior ocular structures causing endophthalmitis.8 Fungal infections on the ocular surface may affect the cornea causing fungal keratitis, conjunctiva causing conjunctivitis, lacrimal glands causing dacryoadenitis, and eyelids causing blepharitis.8
Usually, infectious and traumatic ocular diseases are treated with different therapies to provide treatment and/or protection against infection and promote re-epithelialization. Nowadays, the excessive use of antibiotics has led to the appearance of multidrug-resistant bacterial strains,11 and there is a need for new antimicrobial formulations with novel mechanisms of action to overcome this major health problem.
The present review is intended to bring the most relevant general aspects and the main preclinical and clinical results related to the use of ozonated oil and liposomal ozonated oil in ocular infections and regeneration of ocular surface, and to bring to light the benefits of its use over other antiseptics, such as povidone-iodine and chlorhexidine.
Methods
Literary sources included PubMed, Medline, Web of Science, and Cochrane Library for the studies published until June 2021. The search was carried out in English and Spanish languages, and the following search terms were used: “ozonated oil”, “ozonized oil”, “liposome-vehiculated”, “liposomal ozonated oil”, “liposomal oil”, “ocular infection”, “ocular surface”, “ozonides”, “ozone”, “sunflower oil”, “antimicrobial”, “antimycotic” and “antiseptic”. Search terms were combined to sort out papers on the properties and use of ozonated oil in ocular infections.
After analyzing the content of the papers found, 25 publications were selected.
Results
Composition
Ozonated oil is produced by a chemical reaction of gaseous ozone and double carbon-carbon bonds of unsaturated fatty acids that leads to intermedium compounds, called ozonides (Figure 1), which are more stable with fatty acids.8,12 However, as ozonated oil is highly irritating to the corneal tissue, ozonated sunflower seed oil needs to be encapsulated in liposomes prepared from soy phospholipids (Figure 2).8,13 The mucilage hydroxypropyl methylcellulose (hypromellose) is added to improve tolerability.8,13 Low concentrations of boric acid, sodium tetraborate, sodium edetate and polyhexamethylenebiguanide (PHMB) are also added as tampers (they do not act as preservatives). Liposome encapsulation is not only addressed to reduce the undesirable effects of ozonated oils but also to modulate their release, to improve physical stability protecting from oxidative reactions with the environment, to decrease volatility, and even to improve compliance.13
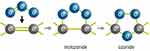 |
Figure 1 Reaction of gaseous ozone and double carbon-carbon bonds of unsaturated fatty acids to form ozonides. |
 |
Figure 2 Ozonated oil encapsulated in liposomes. |
Mechanism of Action
Liposomes bind to the pathogen surface, breaking it with ozonolysis and releasing the ozonides (Figure 3).2,8,13 The ozonides penetrate into the pathogen and become hydrolyzed, forming oxygenated compounds such as lipidic peroxides and reactive oxygen species (ROS).8 These oxygenated compounds alter the pathogen structure acting on proteins, lipids and complex molecules such as enzymes and DNA/RNA, leading to the destruction of the pathogen (Figure 3).8,9,12,14,15
Thus, liposomal ozonated oil has a double mechanism of action: direct oxidation of the pathogen surface, and interaction with the pathogen macromolecules, such as nucleic acids, membrane proteins and enzymes.
In bacteria and fungi, it produces lysis with blockage of respiration and replication. The mechanism of the interaction of ozone with gram-positive and gram-negative bacteria is based on the disruption of envelope integrity through peroxidation of phospholipids, lipoproteins, and peptidoglycan. This leads to enzyme inactivation, gene inhibition, and degradation of genetic materials, interrupting the normal cell activity of bacteria.2
In fungi, ozone produces damage to the cell surface by oxidative inactivation of disulfide bonds, enters the cytoplasm and disrupts vital cellular functions.2
In virus, it blocks viral tissue diffusion and viral replication, causing destruction of the capsid and the envelope, and having a direct interaction with the nucleic acids (DNA/RNA).8,15
Restorative Action
Liposomal ozonated oil does not negatively affect corneal and conjunctival epithelial cells. Ozone is a potent oxidant in human cells at high concentrations but, administered at controlled doses, it promotes cellular adaptation to oxidative stress by activating antioxidant and anti-inflammatory endogen mechanisms (Figure 4).2,16,17
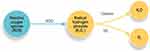 |
Figure 4 Ozone promotes cellular adaptation to oxidative stress activating antioxidant endogen mechanisms. Abbreviation: SOD, superoxide dismutase. |
Ozone is a molecule naturally present in the immune system, both in innate (neutrophils) and acquired (antibody-mediated) immunity. When ozone is combined with organic compounds, it has an immunoeffector activity. It not only attacks and destroys pathogens but also has signaling functions that amplify the immune response through NF-kβ (nuclear factor kappa B), TNFα (tumor necrosis factor alpha) and IL-6 (interleukin 6).14
When applied to the corneoconjunctival tissues, ozone activates immunity by means of an increase in interferon, cytokines and antioxidant enzymes, such as dismutase superoxide, catalase and glutathione peroxidase.2,16,17 It also regulates the levels of nitric oxide, Nf-kβ and TNFα, and finally has shown to promote the synthesis of collagen and stimulation of the production of growth factors such as PDGF (platelet-derived growth factor) and TGFβ (transforming growth factor-beta).2,17,18 The effect on the corneoconjunctival tissues includes three phases: first, direct antimicrobial activity; second, activation of local antioxidant defenses with release of growth factors with proliferative effect; and third, promotion of wound healing with remodeling effect.2,4,16
Liposomal ozonated oil has shown regenerative and anti-inflammatory properties when applied on the ocular surface. At therapeutic doses, it does not show toxicity in the epithelial cells, and in an in vitro study on human corneal epithelium showed no toxicity: after three days of treatment, cellular growth was promoted compared to the control.3
Regarding antiseptics such as chlorhexidine and povidone-iodine, which exhibit broad-spectrum bactericidal effects, they may help in the wound healing process indirectly by maintaining a low recount of microorganisms.19 Although povidone-iodine and chlorhexidine have broad-spectrum germicidal effects, studies have demonstrated their cytotoxic and genotoxic effects in fibroblasts and osteoblasts in vitro which could delay the repair process.19
In summary, liposomal ozonated oil has an antiseptic and reparative effect on corneoconjunctival tissues, which might be of great benefit compared to other antiseptics.2,4,16
Preclinical Efficacy Studies
In vitro Bactericidal Activity
Bactericidal effect of liposomal ozonated oil was demonstrated in vitro against Staphylococcus aureus and Pseudomonas aeruginosa, some of the most frequent eye pathogens causing acute and chronic infections of the ocular surface. Antimicrobial action was found to be dose-dependent.3
Bactericidal action of ozonated oil was further investigated and compared with 10% povidone-iodine and 0.2% chlorhexidine, through a disk diffusion test against Staphylococcus aureus and Porphyromonas gingivalis.20 Growth inhibition was found to be significantly greater with ozonated oil for both tested strains.
Ozonated sunflower oil showed a valuable antimicrobial activity against mycobacteria, staphylococci, streptococci, enterococci, Pseudomonas and Escherichia coli with minimal inhibitory concentrations (MICs) between 1.18 and 9.5 mg/mL.5 Activity was shown against multi-drug resistant bacteria that are of great concern, such as staphylococci, streptococci and enterococci.
Furthermore, the killing rate of ozonated oil for Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA) was of almost 100% within 5 minutes and 15 minutes of exposure, respectively.21
Two recent studies evaluated antibacterial activity and the effect on biofilm formation of liposomal ozonated oil 0.5% (concentrations tested between 250 and 0.25 μg/mL) on antibiotic resistant S. aureus and P. aeruginosa.22,23 Antimicrobial efficacy against these bacterial strains was found.22 Liposomal ozonated oil both inhibits P. aeruginosa and S. aureus biofilm formation and removes P. aeruginosa and S. aureus preformed biofilm on different supports, such as plastic plates and contact lens.23
In vitro Fungicidal Activity
Fungicidal effect of liposomal ozonated oil was tested against Candida albicans, Candida glabrata, Candida krusei, and Candida orthopsilosis by microdilution method, and all Candida isolates were found susceptible with minimum inhibitory concentration (MIC) values ranging from 0.195% to 6.25% concentrations of ozonated oil diluted in 0.9% saline; up to 70% of cells were inactivated after 1 hour of exposure.8
In vitro Antiviral Activity
Antiviral effect was evaluated on different species (herpes simplex virus type-1, Indiana vesicular stomatitis virus, vaccinia virus, adenovirus type-2, and a strain of influenza A virus) in cell cultures. In this study, the level of virus inactivation by ozone suggested a damage and destruction of the lipid viral envelope and protein capsid.6
Bactericidal Action in Animals
Liposomal ozonated oil has demonstrated efficacy in recurrent conjunctivitis and corneal ulcers in animals.9 The efficacy of liposomal ozonated oil was evaluated in different cases of external ophthalmic infectious pathology in animals by MSRA and Pseudomonas aeruginosa, and it was found to show a valid profile (with a dosage of 1–2 drops 3–4 times a day) for the treatment of ocular infectious pathologies, such as conjunctivitis, keratitis, keratoconjunctivitis and corneal ulcers.9,24
The effectiveness of liposomal ozonated oil was compared to ofloxacin and povidone-iodine in conjunctival sac and periocular skin in dogs,25 and results showed that liposomal ozonated oil 0.5% produced a reduction of the bacterial load in the ocular surface comparable to povidone-iodine 5% (10.0 vs 8.0 CFU/mL, respectively, for total microbial count) and superior to ofloxacin (10.0 vs 80 CFU/mL, respectively, for total microbial count) (Table 1). There was a significant decrease in bacterial concentration for the total microbial count and coagulase-positive staphylococci for both liposomal ozone dispersion (p < 0.005) and povidone-iodine (p < 0.05) but not for ofloxacin (p > 0.1).
 |
Table 1 Preoperative Topical Ozonated Oil Reduces Bacterial Colonization in Conjunctival Sac and Periocular Skin |
Clinical Efficacy Studies
A series of three cases were published to present the effect of liposomal ozonated oil in patients with complicated corneal pathology, showing resolution of corneal ulcers and improvement of keratitis within 10 days of treatment.26
Liposomal ozonated oil has also been demonstrated to be a valid and suitable supportive therapy for external ocular pathologies involving inflammation and infections.27 A patient with vernal conjunctivitis, who did not show any improvement after one week with oral prednisone and topical corticosteroid plus chloramphenicol, was found to improve his condition after the first week of adding a liposomal ozonated oil compound with partial resolution after 2 months. A patient with granulomatous conjunctivitis, who did not show any improvement after one week with topical and systemic antibiotics, was found to have improved his condition after the first week of adding a liposomal ozonated oil compound with almost complete resolution after 1 month.
A recent publication reported three cases of corneal inflammation successfully resolved after using topical liposomal ozonated oil.26 One patient with dendritic epithelial keratitis receiving systemic antiviral treatment and liposomal ozonated oil 3 times a day shortened the healing time and reduced the clinical symptoms to one week, and one month after receiving only liposomal ozonated oil he showed complete recovery of sight, without signs of corneal opacities and complete regression of the symptoms. Another patient with corneal trauma, caused by olive leaves receiving liposomal ozonated oil eye drops 6 times a day plus artificial tears 3 times a day, showed complete healing of the corneal epithelium after five days, with complete recovery of symptoms. A third patient with post-herpetic neurotrophic corneal ulcer receiving antibiotic therapy, atropine, systemic antiviral therapy and liposomal ozonated oil eye drops, showed an initial improvement after 3 days, and after 10 days with only liposomal ozonated oil, restoration of epithelial integrity was observed, with resolution of the abscess area and ulcer.
A prospective, single-blind, randomized, parallel-groups trial was carried out in 80 patients with adenoviral conjunctivitis.10 Patients were randomized to topical tobramycin 0.3%/dexamethasone 0.1% eye drops plus liposomal ozonated oil eye drops or topical tobramycin 0.3%/dexamethasone 0.1% eye drops four times daily for seven days.10 Results showed that signs of conjunctivitis, such as conjunctival hyperemia, chemosis, and duration of viral infection, were reduced in the group with liposomal ozonated oil. No differences were found in keratitis and corneal infiltrates; however, a higher number of polymerase chain reaction (PCR) negative results were found in the liposomal ozonated oil group (82.3%) than in the control group (59.4%) (Figure 5).
 |
Figure 5 Adenoviral PCR results in patients treated with ozonated oil and control group. Adapted from Cagini C, Mariniello M, Messina M et al. The role of ozonized oil and a combination of tobramycin/dexamethasone eye drops in the treatment of viral conjunctivitis: a randomized clinical trial. Int Ophthalmol. 2020;40(12):3209–3215. Creative Commons license and disclaimer available from: http://creativecommons.org/licenses/by/4.0/legalcode.10 Abbreviations: OZO, ozonated oil group; TS, control group. |
Liposomal ozonated oil has also been demonstrated to be effective in reducing ocular microbial flora. An interventional, non-randomized, paired eyes Phase IV clinical study was designed, and 174 patients undergoing cataract surgery were included.28 Patients scheduled for surgery received treatment with an isotonic ophthalmic solution of 0.5% liposomal ozonated oil in one eye and saline solution in the contralateral eye as control, 2 drops 4 times/day for 3 days. After three days of treatment with liposome ozonated oil, the microbial load was reduced by >90%, while remaining unchanged in the control group (p < 0.001) (Figure 6). The antiseptic effectiveness of liposomal ozonated oil was found even on multi-resistant bacteria without creating microbial resistance. Therefore, this adjuvant home-prophylaxis could enhance the standardized povidone-iodine solution used intraoperatively. Liposomal ozonated oil could be used as an effective preoperative prophylaxis.
 |
Figure 6 Microbial load reduction with ozonated oil versus control group. Data from Spadea L, Zanotto E, Cavallo R et al. Effectiveness of Liposomal Ozonised Oil in Reducing Ocular Microbial Flora in Patients Undergoing Cataract Surgery. J Cataract Refract Surg. 2021; 47(12):1548–1555.28 |
Safety Studies
The safety of the ozonated oils was evidenced in toxicological, histological, mutagenic, genotoxic, and teratogenic tests. Ophthalmic irritability and dermic irritability tests classified ozonated oil as non-irritant. The possibility of sensitization was studied, without being able to detect a sensitizing effect.29
To assess the toxicity of ozonated oil in liposomes, studies carried out in vitro on epithelial keratinocyte cells found no inhibition of cell growth, and after three days of treatment, cell growth was promoted compared to the positive control, with no evidence of any cell toxicity.3 In a more recent study on human corneal and conjunctival epithelial cells, no toxic effects and no metabolic disturbances were found after 1 and 3 hours of contact with liposomal ozonated oil, but it appeared after 24 hours of continuous exposure of the cells to the preparation.22 Therefore, no evidence of toxicity was found with liposomal ozonated oil in the specified concentration range and recommended product posology.
Corneal tolerability of an ozonized solution at a normal and high concentration was tested against 1.25% povidone-iodine and physiological serum in rabbit corneal epithelium.30 Punctate keratitis was detected in 8.3% of eyes treated with the ozonized solution, the same proportion observed with saline, versus 42% observed in the eyes treated with povidone-iodine.
A study comparing liposomal ozonated oil to ofloxacin and povidone-iodine (5% dilution) in dogs25 found that four dogs showed punctate keratitis and a mild degree of corneal oedema immediately after instillation of povidone-iodine, while no side-effects were reported for liposomal ozone dispersion.
The clinical trial in patients with adenoviral conjunctivitis receiving topical tobramycin 0.3%/dexamethasone 0.1% eye drops either with or without liposomal ozonated oil,10 showed that treatment was well tolerated in both groups with none of the patients reporting any treatment-related adverse events.
In a clinical trial evaluating liposomal ozonated oil in patients undergoing cataract surgery,28 only 3 patients had minimal adverse reactions with trace conjunctival hyperemia, and 7 patients reported discomfort at administration (6 mild and 1 moderate).
Discussion
In this review, 25 papers are summarized on the properties of ozonated oils and the preclinical and clinical studies, which contribute to the further understanding and knowledge of the characteristics and properties of liposomal ozonated oil for treatment of ocular infections.
The overuse of antibiotics in the treatment of infectious diseases, and the consequent appearance of multi-drug resistant bacterial strains, is a growing situation that must be faced in the short time.11 The World Health Organization has classified antibiotic resistance as one of the 3 most important public health threats of the 21st century.31 Antibiotics are widely used in ophthalmic procedures like intravitreal injections, while there is evidence that antibiotic use does not reduce the rate of endophthalmitis.32
This situation is leading us towards the study of other types of antimicrobial agents like antiseptics. Povidone-iodine has a broad antimicrobial activity against gram-positive and gram-negative bacterial isolates and adenoviruses in preclinical studies,33,34 although their limited tolerability with high or frequent doses and efficacy with low/spaced doses restricts their use for external ocular infection treatment.34 Liposomal ozonated oil has been widely studied in several pathologies and has demonstrated beneficial antimicrobial properties, and so far, no bacterial resistance to ozonated oils has been described, making it a good alternative to prevent or to treat external ocular infections both alone or in combination with other antimicrobials. The action of ozonated oil is connected in part to its antimicrobial effect, but also to its ability to promote some growth factors, activating local antioxidant mechanisms and promoting tissue repair, which might be of great advantage compared to povidone-iodine and chlorhexidine.
The results of the studies that have been carried out with liposomal ozonated oil in ocular infections, 6 preclinical studies (4 in vitro3,8,22,23 and 2 in vivo9,24) and 5 clinical studies10,25,26 show its antimicrobial activity as well as the restorative action.
Liposomal ozonated oil is well tolerated in corneoconjunctival tissues. The safety of the liposomal ozonated oil has been assessed in vitro showing no toxic effects on keratinocyte in vivo and in clinical trials showing a good safety profile, tolerability, and biocompatibility on the ocular surface.3,10,15,28
Application of liposomal ozonated oil has shown similar or superior effects to traditional antiseptics and antibiotics, resulting in a broad antimicrobial spectrum and a low rate of adverse events, which is of particular interest in antibiotic resistant bacteria. Furthermore, liposomal ozonated oil may be an interesting supplement to the traditional compounds to have an antioxidant and inflammatory balance in the front part of the eye.
Conclusions
Liposomal ozonated oil has both antiseptic and reparative effects on corneoconjunctival tissues and is well tolerated and biocompatible with the delicate tissue of the ocular surface. It has demonstrated efficacy and safety profiles for its use in ocular infections and can be considered as a new instrument against external ocular infections, both alone and in combination with other antimicrobials.
Future Direction
Further studies with liposomal ozonated oil alone and in combination with the currently used anti-infectious drugs are needed.
Abbreviations
CFU, colony forming units; DNA, deoxyribonucleic acid; IL-6, Interleukin 6; MIC, minimum inhibitory concentration; mL, Milliliters; MRSA, methicillin-resistant Staphylococcus aureus; NF-kβ, nuclear factor kappa B; P., Pseudomonas; PCR, polymerase chain reaction; PDGF, platelet derived growth factor; PHMB, polyhexamethylenebiguanide; RNA, ribonucleic acid; ROS, reactive oxygen species; S., Staphylococcus; TGFβ, transforming growth factor-beta; TNFα, tumor necrosis factor alpha.
Acknowledgments
Supported by Esteve Pharmaceuticals SA, who provided an unrestricted grant. None of the authors have financial disclosures that relate to this manuscript.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
Dr Pérez-Santonja is a consultant for Esteve Pharmaceuticals. Dr Güell is a consultant for Opthec, Meditec, Zeiss, Alcon, Thea Laboratories, Kowa Pharmaceuticals and RxSight. Dr Vázquez is a consultant for Esteve Pharmaceuticals and Visufarma. Dr Benitez-Del-Castillo is a consultant for Allergan Angelini, Esteve Pharmaceuticals, GSK, Lumenis, Novartis, Santen, Sifi, Thea Laboratories and Visufarma. The authors report no other conflicts of interest in this work.
References
1. Ali FS, Jenkins TL, Boparai RS, et al. Aqueous chlorhexidine compared with povidone-iodine as ocular antisepsis before intravitreal injection: a Randomized Clinical Trial. Ophthalmol Retina. 2020;5:788–796. doi:10.1016/j.oret.2020.11.008
2. Martínez-Sánchez G. Ozonized water, background, general use in medicine and preclinic support. Ozone Ther Global J. 2019;9(1):33–60.
3. Cutarelli A, Carlini G, Sarno F, et al. The role of ozone carried by liposomes in the therapy of infectious and skin-regenerating ocular surface. J Biomed Sci Eng. 2019;12(7):347–353. doi:10.4236/jbise.2019.127026
4. Martínez- Sánchez G, Re L, Perez-Davison G, Delaporte RH. Medical uses of ozonized oils, an up-date. Revista Española de Ozonoterapia. 2012;2(1):121–139.
5. Sechi LA, Lezcano I, Nunez N, et al. Antibacterial activity of ozonized sunflower oil (Oleozon). J Appl Microbiol. 2001;90(2):279–284. doi:10.1046/j.1365-2672.2001.01235.x
6. Murray BK, Ohmine S, Tomer DP, et al. Virion disruption by ozone-mediated reactive oxygen species. J Virol Method. 2008;153(1):74–77. doi:10.1016/j.jviromet.2008.06.004
7. Kim HS, Noh SU, Han YW, et al. Therapeutic effects of topical application of ozone on acute cutaneous wound healing. J Korean Med Sci. 2009;24(3):368–374. doi:10.3346/jkms.2009.24.3.368
8. Celenza G, Iorio R, Cracchiolo S, et al. Antimycotic activity of ozonized oil in liposome eye drops against candida spp. Transl Vis Sci Technol. 2020;9(8):4. doi:10.1167/tvst.9.8.4
9. Marchegiani A, Spaterna A. Ozone-based eye drops in anterior segment pathologies: rationale and pre-clinical data. Ozone Ther. 2017;2(1):6743. doi:10.4081/ozone.2017.6743
10. Cagini C, Mariniello M, Messina M, et al. The role of ozonized oil and a combination of tobramycin/dexamethasone eye drops in the treatment of viral conjunctivitis: a randomized clinical trial. Int Ophthalmol. 2020;40(12):3209–3215. doi:10.1007/s10792-020-01503-4
11. Asbell PA, Sanfilippo CM, Pillar CM, DeCory HH, Sahm DF, Morris TW. Antibiotic resistance among ocular pathogens in the United States: five-year results from the Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR) Surveillance Study. JAMA Ophthalmol. 2015;133(12):1445–1454. doi:10.1001/jamaophthalmol.2015.3888
12. Kalinowski J, Heinonen P, Kilpeläinen I, Räsänen M, Gerber RB. Stability of Criegee intermediates formed by ozonolysis of different double bonds. J Phys Chem A. 2015;119(11):2318–2325. doi:10.1021/jp506525g
13. Ugazio E, Tullio V, Binello A, Tagliapietra S, Dosio F. Ozonated oils as antimicrobial systems in topical applications. Their characterization, current applications, and advances in improved delivery techniques. Molecules. 2020;25(2):334. doi:10.3390/molecules25020334
14. Wentworth P, McDunn JE, Wentworth AD, et al. Evidence for antibody-catalyzed ozone formation in bacterial killing and inflammation. Science. 2002;298(5601):2195–2199. doi:10.1126/science.1077642
15. Giannangelo C, Fowkes F, Simpson JA, Charman SA, Creek DJ. Ozonide antimalarial activity in the context of artemisinin-resistant malaria. Trends Parasitol. 2019;35(7):529–543. doi:10.1016/j.pt.2019.05.002
16. Elvis AM, Ekta JS. Ozone therapy: a clinical review. J Nat Sci Biol Med. 2011;2(1):66–70. doi:10.4103/0976-9668.82319
17. Anzolin AP, da Silveira-Kaross NL, Bertol CD. Ozonated oil in wound healing: what has already been proven? Med Gas Res. 2020;10(1):54–59. doi:10.4103/2045-9912.279985
18. Zhang J, Guan M, Xie C, Luo X, Zhang Q, Xue Y. Increased growth factors play a role in wound healing promoted by noninvasive oxygen-ozone therapy in diabetic patients with foot ulcers. Oxid Med Cell Longev. 2014;2014:273475. doi:10.1155/2014/273475
19. da Silveira Teixeira D, de Figueiredo MAZ, Cherubini K, de Oliveira SD, Salum FG. The topical effect of chlorhexidine and povidone-iodine in the repair of oral wounds. A review. Stomatologija. 2019;21(2):35–41.
20. Montevecchi M, Dorigo A, Cricca M, Checchi L. Comparison of the antibacterial activity of an ozonated oil with chlorhexidine digluconate and povidone-iodine. A disk diffusion test. New Microbiol. 2013;36(3):289–302.
21. Song M, Zeng Q, Xiang Y, et al. The antibacterial effect of topical ozone on the treatment of MRSA skin infection. Mol Med Rep. 2018;17(2):2449–2455. doi:10.3892/mmr.2017.8148
22. Paduch R, Urbanik-Sypniewska T, Kutkowska J, et al. Ozone-based eye drops activity on ocular epithelial cells and potential pathogens infecting the front of the eye. Antioxidants. 2021;10:968. doi:10.3390/antiox10060968
23. Zerillo L, Polvere I, Varricchio R, et al. Antibiofilm and repair activity of ozonated oil in liposome. Microb Biotechnol. 2021;15:1422–1433. doi:10.1111/1751-7915.13949
24. Spadea L, Tonti E, Spaterna A, Marchegiani A. Use of ozone-based eye drops: a series of cases in veterinary and human spontaneous ocular pathologies. Case Rep Ophthalmol. 2018;9(2):287–298. doi:10.1159/000488846
25. Marchegiani A, Magagnini M, Cerquetella M, et al. Preoperative topical liposomal ozone dispersion to reduce bacterial colonization in conjunctival sac and periocular skin: preliminary study in dogs. Exp Eye Res. 2019;189:107848. doi:10.1016/j.exer.2019.107848
26. Passidomo F, Pignatelli F, Addabbo G, Costagliola C. Topical liposomal ozonated oil in complicated corneal disease: a report on three clinical cases. Int Med Case Rep J. 2021;14:327–332. doi:10.2147/IMCRJ.S311839
27. Basile AA, Cendali M, Mandelli G, Fioretto G. Anti-inflammatory effect of an eye drops solution based on liposomal ozonated oil in different corneal and anterior segment human diseases. Ozone Ther. 2019;4(2):8377. doi:10.4081/ozone.2019.8377
28. Spadea L, Zanotto E, Cavallo R, et al. Effectiveness of liposomal ozonised oil in reducing ocular microbial flora in patients undergoing cataract surgery. J Cataract Refract Surg. 2021;47(12):1548–1555. doi:10.1097/j.jcrs.0000000000000672
29. Martínez-Sánchez G. Scientific rational for the medical application of ozonized oils, an up-date. Ozone Ther Global J. 2021;11(1):239–272.
30. Kashiwagi K, Saito K, Wang YD, Takahashi H, Ishijima K, Tsukahara S. Safety of ozonated solution as an antiseptic of the ocular surface prior to ophthalmic surgery. Ophthalmologica. 2001;215(5):351–356. doi:10.1159/000050884
31. Munita JM, Arias CA. Mechanisms of antibiotic resistance. Microbiol Spectr. 2016;4:
32. Menchini F, Toneatto G, Miele A, Donati S, Lanzetta P, Virgili G. Antibiotic prophylaxis for preventing endophthalmitis after intravitreal injection: a systematic review. Eye. 2018;32(9):1423–1431. doi:10.1038/s41433-018-0138-8
33. Berkelman RL, Holland BW, Andreson RL. Increased bactericidal activity of dilute preparations of povidone-iodine solutions. J Clin Microbiol. 1982;15:635–639. doi:10.1128/jcm.15.4.635-639.1982
34. Kawana R, Kitamura T, Nakagomi O, et al. Inactivation of human viruses by povidone-iodine in comparison with other antiseptics. Dermatology. 1997;195:29–35. doi:10.1159/000246027
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

