Back to Journals » Vascular Health and Risk Management » Volume 10
Limitations of real-world treatment with atorvastatin monotherapy for lowering LDL-C in high-risk cardiovascular patients in the US
Authors Marrett E, Zhao C, Zhang NJ, Zhang Q, Ramey D, Tomassini J, Tershakovec A, Neff D
Received 24 September 2013
Accepted for publication 24 January 2014
Published 25 April 2014 Volume 2014:10 Pages 237—246
DOI https://doi.org/10.2147/VHRM.S54886
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Elizabeth Marrett,1 Changgeng Zhao,1 Ning Jackie Zhang,2 Qiaoyi Zhang,1 Dena R Ramey,1 Joanne E Tomassini,1 Andrew M Tershakovec,1 David R Neff1
1Merck & Co, Inc., Whitehouse Station, NJ, USA; 2College of Health and Public Affairs, University of Central Florida, Orlando, FL, USA
Background: Guidelines endorse statin therapy for lowering low-density lipoprotein cholesterol (LDL-C) to recommended levels, in patients with cardiovascular disease (CVD) risk, if needed, after lifestyle changes. Atorvastatin is a common statin with greater LDL-C lowering efficacy than most other statins; its availability in generic form will likely increase its use. This study assessed attainment of guideline-recommended LDL-C levels in high-risk CVD patients treated with atorvastatin monotherapy.
Methods: Analyses of two retrospective US cohorts of patients who received a prescription for atorvastatin monotherapy between January 1, 2008 and December 31, 2010 (index date defined as first prescription date) in the GE Centricity Electronic Medical Record (EMR) (N=10,693) and Humana Medicare (N=16,798) databases. Eligible patients were ≥18 years, diagnosed with coronary heart disease or atherosclerotic vascular disease, with ≥1 LDL-C measurement between 3 months and 1 year postindex date, and continuously enrolled for 1 year prior to and following the index date.
Results: Of the eligible patients, 21.8%, 29.6%, 29.9%, and 18.7% (GE Centricity EMR) and 25.4%, 32.9%, 27.8%, and 14.0% (Humana Medicare) received 10, 20, 40, and 80 mg doses of atorvastatin, respectively. The mean ± standard deviation (SD) follow-up LDL-C levels were 2.1±0.8 mmol/L (83±30 mg/dL) and 2.3±0.8 mmol/L (88±31 mg/dL) for the GE Centricity EMR and Humana Medicare cohorts, respectively. Regardless of dose, only 28.3%-34.8% of patients had LDL-C levels <1.8 mmol/L (<70 mg/dL), and 72.0%-78.0% achieved LDL-C <2.6 mmol/L (<100 mg/dL) in both cohorts. As many as 41% and 13% of patients had LDL-C levels ≥0.5 mmol/L (≥20 mg/dL) above LDL-C 1.8 mmol/L (70 mg/dL) and 2.6 mmol/L (100 mg/dL), respectively, in both cohorts; these percentages were generally similar across atorvastatin doses.
Conclusion: In this real-world US setting, a large number of high-risk CVD patients did not attain guideline-recommended LDL-C levels with atorvastatin monotherapy. More than 65% of the patients had LDL-C levels >1.8 mmol/L (>70 mg/dL), and of these, 30%-40% had LDL-C levels ≥0.5 mmol/L (≥20 mg/dL) above this, regardless of dose. This suggests that more effective lipid-lowering strategies, such as statin uptitration, switching to a higher efficacy statin, and/or combination therapy, may be required to achieve optimal LDL-C lowering in high-risk patients.
Keywords: statin therapy, managed-care, lipid-lowering therapy
Introduction
Lowering of low-density lipoprotein cholesterol (LDL-C) levels by statin therapy has been shown to be highly related to cardiovascular disease (CVD) risk reduction in a meta-analysis of 26 randomized-controlled statin clinical studies.1 As such, statin therapy is endorsed by various guidelines as the first-line pharmacotherapeutic approach for reducing LDL-C levels and CVD risk when lifestyle changes, including diet (eg, reduced saturated fats and cholesterol, plant stanol/sterols, and increased fiber), weight control, and exercise do not adequately control LDL-C levels.2–5
Earlier guidelines targeted LDL-C <2.6 mmol/L (<100 mg/dL) as the optimal goal for primary prevention, in patients at high CVD risk with multiple coronary heart disease (CHD) risk factors.6–8 An optional treatment target of LDL-C <1.8 mmol/L (<70 mg/dL) was further recommended in 2004, based on clinical trial evidence showing that LDL-C 2.6 mmol/L (100 mg/dL) did not appear to be a threshold below which no further CV risk reduction could be achieved.4 The latter target is also considered reasonable for high-risk patients with atherosclerotic vascular disease, and similar guidelines have also been adopted internationally.2,9,10 Although LDL-C <1.8 mmol/L (<70 mg/dL) is recommended as a therapeutic option for all high CVD-risk patients, those in the category of very high risk (ie, patients with pre-existing CHD, plus major multiple risk factors [eg, diabetes], severe and poorly controlled risk factors, multiple risk factors of the metabolic syndrome, or acute coronary syndrome) should be most highly considered for this target.4,11–13 It should also be noted that recent evidence-based guidelines have recommended treatment of moderate-risk patients with moderate-intensity statins to reduce LDL-C levels from baseline by 30%–<50% and of high-risk patients with high-intensity statins to reduce LDL-C levels by ≥50%, rather than to reach specific LDL-C targets.14
While lowering of LDL-C to guideline-recommended levels has improved over time due to an increased awareness and use of statin therapy in clinical practice, many higher-risk patients still do not attain recommended LDL-C levels when prescribed statin monotherapy. Although ~2/3 of high-risk patients attain LDL-C levels <2.6 mmol/L (<100 mg/dL), only ~1/3 achieve levels <1.8 mmol/L (<70 mg/dL).15–17 Several observational studies and national surveys have indicated that various barriers, including suboptimal lipid-lowering therapy, noncompliance, tolerability, and cost factors, have contributed to this treatment gap.18–22 Cholesterol guidelines have indicated that more effective lipid-lowering therapy, including uptitration to higher statin doses, switching to higher potency statins, and/or combination statin therapy with other lipid-lowering agents, may be necessary to further improve LDL-C levels in these patients.2,4,5
Atorvastatin is a common statin used in clinical practice that has been shown to effectively reduce LDL-C and CVD risk, in randomized, controlled clinical trials.23 Even though atorvastatin is more potent in terms of LDL-C lowering efficacy than most other statins, it has been less widely used in comparison with generic statins, particularly simvastatin, likely attributable to cost and other factors.24,25 However, the availability of generic atorvastatin will likely increase its use; thus it is of interest to examine LDL-C-lowering in high-risk patients treated with this statin in a real-world setting. This analysis assessed the attainment of LDL-C levels of <1.8 mmol/L (<70 mg/dL) and <2.6 mmol/L (<100 mg/dL) in two retrospective cohort studies of high-risk CVD patients treated with atorvastatin in the US, as well as the distance from these specified cut points in those patients who did not reach these levels.
Study methods
Data sources
Patient records from the GE Centricity Electronic Medical Record (EMR) database (GE Healthcare, Little Chalfont, UK) and administrative claims from Humana Medicare (Humana Inc., Louisville, KY, USA) were assessed between the dates of January 1, 2007 and December 31, 2011. The GE Centricity EMR database captures patient-level clinical data, including drug prescription and laboratory results, from over 25,000 physicians (~66% primary care), representing ~30 million US patients. We also analyzed administrative claims data from the Humana Medicare database. Humana Medicare claims data represent 8.1 million covered lives in 50 states, Washington DC, and Puerto Rico, among whom 4.3 million are Medicare members and 1.9 million are Advantage prescription drug plan beneficiaries. For the purpose of the present analysis, pharmacy data were merged with medical, lab, and patient data for Humana Medicare beneficiaries, using unique patient IDs. In both analyses, data were de-identified, and patient privacy was protected.
Study design and patient eligibility
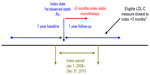
Included in the studies were patients at high CVD risk who received a prescription for atorvastatin monotherapy (the date of the first prescription was defined as the index date) between January 1, 2008 and December 31, 2010 (index period). Patients were required to be ≥18 years of age at the index date and to have at least one recorded LDL-C measure ≥3 months and ≤12 months postindex date. For the Medicare analysis, there was an additional requirement for one atorvastatin prescription within 45 days of the LDL-C measure (not feasible in the data from the GE Centricity EMR, due to data limitations). In the case where multiple eligible LDL-C measures occurred, the measure closest to index date plus 3 months was selected for the analysis (Figure 1). The atorvastatin daily dose was assessed based on the last recorded prescription prior to the LDL-C measure.
Patients were required to be continuously enrolled in the respective system for 1 year prior to the index date (baseline period), and for 1 year following the index date (follow-up period) in the Humana Medicare analyses. Having at least one office visit in both the baseline and follow-up periods was used as a proxy for continuous enrollment over the study period, in the GE Centricity EMR analyses. High-risk patients were identified as those with evidence of CHD or atherosclerotic vascular disease (AVD), including ischemic heart disease, ischemic stroke, peripheral arterial disease, or atherosclerosis. The qualifying disease was identified using the International Classification of Diseases (ninth revision), Clinical Modification (ICD-9-CM) diagnosis/procedure or Current Procedural Terminology (CPT®) procedure codes during the baseline period and up until the follow-up LDL-C measure (Table S1). Excluded were patients who received any lipid-modifying treatment regimen, other than atorvastatin monotherapy, between the index date and the qualified LDL-C measure.
Outcomes of interest
Various guidelines recommend treatment of LDL-C to levels <2.6 mmol/L (<100 mg/dL) for all patients with CHD and/or AVD but also suggest treatment to LDL-C <1.8 mmol/L (<70 mg/dL) as reasonable or optional in such patients.2,4,5 The aim of this analysis was to assess mean LDL-C levels and the proportions of patients with levels <1.8 mmol/L (<70 mg/dL) and <2.6 mmol/L (<100 mg/dL), in high-risk patients with CHD and/or AVD who were treated with atorvastatin monotherapy. In this analysis, all patients were assessed against each specified LDL-C cut point, by atorvastatin dose and across doses. In addition, for patients above the specified LDL-C cut-points, we also assessed the proportions who were ≤0.3, 0.3−0.5, or >0.5 mmol/L (≤10, 11−20, or >20 mg/dL) away from the cut point overall, across atorvastatin doses and by daily atorvastatin dose.
Statistical analysis
The GE Centricity EMR and Humana Medicare analyses were purely descriptive, and there was no hypothesis testing or statistical comparison either within or between the two studies. They are presented side-by-side only to provide a broad view of atorvastatin monotherapy usage and associated LDL-C levels in two US populations.
Descriptive statistics were used to assess baseline patient characteristics, atorvastatin dose, and LDL-C levels. For continuous variables, means and standard deviations were calculated. Categorical variables were expressed as frequencies and proportions, with 95% confidence intervals calculated for the outcome measures of interest. Statistical analyses were performed using SAS versions 9.1 and 9.2 (SAS Institute, Inc., Cary, NC, USA) for the analysis of the GE Centricity EMR and the Humana Medicare databases, respectively.
Results
Patient characteristics
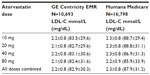
Of 438,403 patients on atorvastatin therapy in the GE Centricity EMR database during the index period, a total of 10,693 patients satisfied the inclusion criteria (Figure 2). Among these patients, the mean age was 68.0 years, and the majority were male (58.7%). The mean baseline LDL-C level was 2.1 mmol/L (82.2 mg/dL), with 34.1% of patients receiving some type of lipid-lowering medication in the 12 months prior to the index date (Table 1). CHD was the most common qualifying diagnosis (73.7%), followed by atherosclerosis (21.9%). In addition, 20.3% of patients had diabetes. The most commonly prescribed doses of atorvastatin were 20 (29.7%) and 40 (29.9%) mg; atorvastatin 80 mg was less-often prescribed (18.7%).
Of the 695,639 patients on atorvastatin in the Humana Medicare database during the index period, 16,798 patients met the inclusion criteria (Figure 2). Among these patients, the mean age was 73.7 years, and 52.1% were male. The mean LDL-C at baseline was 2.5 mmol/L (98.2 mg/dL), with 80.8% receiving some type of lipid-lowering therapy in the 12 months prior to the index date (Table 1). Similar to the findings for patients in the GE database, CHD was the most common qualifying diagnosis (69.6%), and atherosclerosis was identified in 23.7% of patients. Overall, 18.8% of this high-risk population also had diabetes. The most commonly prescribed dose of atorvastatin was 20 mg (32.9%), and the least prescribed was 80 mg (14.0%).
LDL-C levels at follow-up
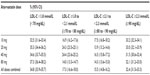
In each study, the mean LDL-C levels in the follow-up period were similar across atorvastatin doses. The mean LDL-C in the GE Centricity EMR analysis ranged from 2.2 mmol/L (83.5 mg/dL) to 2.1 mmol/L (82.4 mg/dL) for patients taking atorvastatin 10 mg and 80 mg, respectively (Table 2). For Humana Medicare patients, mean LDL-C levels ranged from 2.3 mmol/L (88.7 mg/dL) for 10 mg to 2.2 mmol/L (85.9 mg/dL) for 80 mg doses.
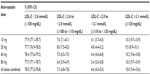
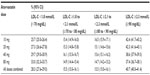
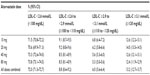
Overall, the proportions of patients with LDL-C levels <1.8 mmol/L (<70 mg/dL) and <2.6 mmol/L (<100 mg/dL) in the GE Centricity EMR analysis were 34.8% and 78.0% respectively (Figure 3). For Humana Medicare patients, the proportions below these LDL-C cut points were smaller, with 28.3% at <1.8 mmol/L (<70 mg/dL) and 72.0% at <2.6 mmol/L (<100 mg/dL). In both studies, the proportion of patients with levels below each LDL-C cut point was similar across atorvastatin doses and ranged from 25.7% (10 mg) to 37.3% (80 mg) for LDL-C <1.8 mmol/L (<70 mg/dL) across both databases (Figure 4). Of note, 32.4% of GE Centricity EMR patients and 40.7% of Humana Medicare patients had LDL-C levels more than 0.5 mmol/L (20 mg/dL) above the 1.8 mmol/L (70 mg/dL) cut point (Figure 3). These percentages were similar regardless of atorvastatin dose in the GE Centricity EMR (33.2%, 33.0%, 31.5%, and 32.1% at 10, 20, 40, and 80 mg, respectively) and Humana Medicare databases (42.4%, 42.3%, 38.6%, and 37.4% at 10, 20, 40, and 80 mg respectively) (Tables S2–S5).
  | Figure 3 Distribution of LDL-C levels around <1.8 mmol/L (<70 mg/dL) and <2.6 mmol/L (<100 mg/dL) cut points. |
Discussion
In this real-world study, a substantial number of high-risk CVD patients did not achieve guideline-recommended LDL-C levels on atorvastatin monotherapy. Although a large proportion of these patients (71%−78%) attained the recommended treatment levels of LDL-C <100 mg/dL, more than 65% did not achieve the optional level of <1.8 mmol/L (<70 mg/dL), even when on therapy at the highest atorvastatin dose of 80 mg. Moreover, 30%−40% of these patients had LDL-C levels more than 0.5 mmol/L (20 mg/dL) above 1.8 mmol/L (70 mg/dL), regardless of atorvastatin dose. Overall, these results indicate a need for more effective LDL-C-lowering therapy in high CVD-risk patients.
Cholesterol-lowering management has improved over time due to increased awareness and better use of lipid-lowering therapy. However, several observational studies and national surveys have shown that many high-risk CHD patients still do not meet guideline-recommended LDL-C levels on statin monotherapy, attributed to various reasons, including less than optimal use of lipid-lowering therapy, noncompliance, and cost factors.15–22 The attainment rates for specified LDL-C levels in our study were similar to those previously reported during 1998−2008 in the US, in which 58%−81% of high-risk CHD patients attained LDL-C levels <2.6 mmol/L (<100 mg/dL) and 26%−43% attained LDL-C levels of <1.8 mmol/L (<70 mg/dL).15,25–27
For those high-risk patients who do not achieve recommended LDL-C levels on statin monotherapy, more intensive treatment, by statin uptitration, switching to a higher efficacy statin, and/or combination therapy can be considered.2,4,5 In our study of high-risk CHD and AVD patients, the majority were treated with medium efficacy-level, LDL-C-lowering doses of atorvastatin (20 and 40 mg doses).28 Nonetheless, the proportion of patients who attained LDL-C <1.8 mmol/L (<70 mg/dL) improved little at any atorvastatin dose, in both databases. It should be noted that while our study included patients who were both newly initiated and on existing atorvastatin therapy, as many as 34%−81% of the patients in both databases had been on some type of lipid-lowering therapy during the baseline period, including 22%−70% who were on prior atorvastatin therapy, and this may have accounted for the minimal change observed in LDL-C levels. Atorvastatin uptitration for individual patients was not assessed in this study, but our results are consistent with the percent patient attainment of LDL-C levels <1.8 mmol/L (<70 mg/dL) observed overall with moderate to high doses of statin in other studies.24,25
Several observational and randomized controlled clinical trials have reported that statin uptitration and/or combination therapy with higher LDL-C-lowering efficacy are more effective than moderate-potency therapy for reducing LDL-C and improving goal attainment in high-risk patients.24,25,29–35 However, these studies showed that >50% of these patients still did not achieve the more stringent LDL-C levels of <1.8 mmol/L (<70 mg/dL) and that high-potency statin therapy as well as the combination of statin with ezetimibe or niacin therapies were used less frequently compared with moderate-potency statins in these patients. Lower use of such alternatives was attributed to patient noncompliance/intolerance, physician nonadherence to guidelines, and/or cost factors. A lack of definitive data showing the benefit of combination therapy on cardiovascular outcomes compared with statin monotherapy may limit its use.36–38 Both fibrates and niacin failed to show further reductions of events when combined with statins compared with statins alone.36–38 Furthermore, the coadministration of niacin and fibrates with statins increases the risk of myopathy and rhabdomyolysis. In a recent study, niacin plus the antiflushing agent laropiprant in combination with simvastatin increased the incidence of some types of nonfatal, serious side effects, thus bringing into question the overall safety of niacin.37 An ongoing trial expected to be completed in 2014 will provide additional information on the role of ezetimibe in combination with simvastatin with respect to clinical outcomes.39
Although the reasons for therapy choice were not evaluated in our analysis, taken together, our study and these studies suggest that many high-risk patients would likely benefit from uptitration of their atorvastatin dose, where possible, and/or switching to a higher potency statin (rosuvastatin) or combination therapy. Doubling the statin dose can provide an additional 6% reduction in LDL-C levels,16,28,40 and switching from atorvastatin doses to a higher potency statin (rosuvastatin), can provide further increases in LDL-C-lowering, by as much as 10%, depending on the dose. For example, the LDL-C-lowering efficacy of atorvastatin ranges from 38% to 55% for 10, 20, 40, and 80 mg doses, whereas that of rosuvastatin ranges from 45% to 60% for 5, 10, 20, and 40 mg doses.23,41 In randomized controlled clinical trials, combination statin therapy with ezetimibe has been shown to increase LDL-C lowering by as much as 21.3%−27.0% compared with statin monotherapy, when coadministered in statin-naïve and/or patients on ongoing statin therapy.42–45 In a recent study, ezetimibe added to atorvastatin reduced LDL-C levels significantly more than doubling the atorvastatin dose or switching to (or doubling) higher potency rosuvastatin.46 As noted in these studies, the real-world treatment differences did not achieve the expected results.
A strength of our study is that it provides information on attainment of guideline-recommended LDL-C levels with atorvastatin monotherapy use in a real-world clinical practice setting. However, as with most retrospective, observational studies, there are inherent limitations. We were unable to determine whether LDL-C values were based on direct methods or were estimated based on the Friedewald equation.47 With regard to the cohort selection process, it is possible that the CHD and AVD patients within the databases may not have been fully identified due to a lack of specific information regarding all CHD and AVD risk factors. Although we were able to identify atorvastatin prescriptions written by the physician (in the GE Centricity EMR) and claims for atorvastatin prescriptions filled (in the Humana Medicare database), we were unable to verify patient adherence/compliance to their prescribed medication. Furthermore, because we were unable to confirm if and when atorvastatin prescriptions were filled in the GE Centricity EMR database, we were unable to require as inclusion criteria one atorvastatin prescription fill within 45 days of the follow-up LDL-C measure, in this database. While this study provides an assessment of LDL-C-reduction in atorvastatin users identified who were both newly initiated and on existing therapy during a 3-year time period, LDL-C-reduction was not evaluated separately in these patients groups. Those patients identified as new users and who were not at LDL-C goal may not have yet had the opportunity for further uptitration of lipid-lowering therapy. Inclusion of both new and previous users of lipid-lowering therapy also precluded assessment of LDL-C-reductions from pretreatment lipid levels. Furthermore, the majority of patients in the Humana Medicare analysis were on prior atorvastatin therapy. Although our data suggest that a high percentage of patients were on newly initiated atorvastatin monotherapy in the GE Centricity EMR database, the minimal reduction observed in LDL-C levels suggests that many of these patients may have been on prior lipid-lowering therapy that was not captured in the EMR. We also did not have information on intended patient lipid goals or patient therapy choices, including whether patients underwent atorvastatin uptitration, in either database. We also had no information on side effects or other patient-level factors that may have precluded treatment to lower LDL-C targets.
In conclusion, in this real-world setting, a large percentage of high-risk CVD patients treated with atorvastatin monotherapy were not at LDL-C goal, regardless of dose. These data suggest that more effective lipid-lowering strategies, such as statin uptitration, use of higher potency statins, and/or combination therapy, may be needed to achieve optimal LDL-C-lowering in high-risk patients. It may be prudent for physicians to carefully weigh considerations of safety, tolerability, and anticipated efficacy when making therapeutic decisions, based on individual patient risk. Additionally, improved patient and provider education is still needed to reinforce use of effective therapies and medication adherence.
Acknowledgments
Editorial assistance was provided by Sheila Erespe, Master’s degree in Science, of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Whitehouse Station, NJ, USA. This research was supported by Merck Sharp & Dohme Corp.
Author contributions
All authors jointly developed the manuscript content and were involved in at least one of the following: conception, design, data acquisition, analysis, statistical analysis, interpretation of data, drafting the manuscript, and/or revising the manuscript for important intellectual content. All authors provided final approval of the version to be published.
Disclosure
NJ Zhang received research funding from Merck & Co., Inc. to conduct the Humana Medicare analysis; C Zhao, D Ramey, JE Tomassini, AM Tershakovec, and DR Neff are current employees, and E Marrett and Q Zhang are former employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. and may hold stock/stock options in the company.
References
Baigent C, Blackwell L, Emberson J, et al; Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670–1681. | |
Catapano AL, Reiner Z, De Backer G, et al; European Society of Cardiology (ESC); European Atherosclerosis Society (EAS). ESC/EAS Guidelines for the management of dyslipidaemias. The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Atherosclerosis. 2011;217(1):3–46. | |
Nash DT, Fillit H. Cardiovascular disease risk factors and cognitive impairment. Am J Cardiol. 2006;97(8):1262−1265. | |
Grundy SM, Cleeman JI, Merz CN, et al; Coordinating Committee of the National Cholesterol Education Program. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol. 2004;44(3):720–732. | |
Smith SC, Allen J, Blair SN, et al; AHA/ACC; National Heart, Lung, and Blood Institute. AHA/ACC guidelines for secondary prevention for patients with coronary and other atherosclerotic vascular disease: 2006 update: endorsed by the National Heart, Lung, and Blood Institute. Circulation. 2006;113(19):2363–2372. | |
National Cholesterol Education Program. Second report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel II). Circulation. 1994;89(3):1333–1445. | |
Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) Final Report. Circulation. 2002;106:3143−3421. | |
Goodman DS, Hulley SB, Clark LT, et al. Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Arch Intern Med. 1988;148(1):36−69. | |
International Atherosclerosis Society. An International Atherosclerosis Society Position Paper: Global Recommendations for the Management of Dyslipidemia. Houston, TX: International Atherosclerosis Society; 2013. Available from: http://www.athero.org/IASPositionPaper.asp. Accessed January 30, 2014. | |
Anderson TJ, Gregoire J, Hegele RA et al. Update of the canadian cardiovascular society guidelines for the diagnosis and treatment of dyslipidemia for the prevention of cardiovascular disease in the adult. Can.J.Cardiol. 2013;29(2):151–167. | |
Standards of medical care in diabetes – 2014. Diabetes Care. 2014; 37(Suppl 1):S14−S80. | |
Brunzell JD, Davidson M, Furberg CD, et al; American Diabetes Association; American College of Cardiology Foundation. Lipoprotein management in patients with cardiometabolic risk: consensus statement from the American Diabetes Association and the American College of Cardiology Foundation. Diabetes Care. 2008;31(4):811−822. | |
Jellinger PS, Smith DA, Mehta AE, et al; AACE Task Force for Management of Dyslipidemia and Prevention of Atherosclerosis. American Association of Clinical Endocrinologists’ guidelines for management of dyslipidemia and prevention of atherosclerosis. Endocr Pract. 2012;18(Suppl 1):S1–S78. | |
Stone NJ, Robinson J, Lichtenstein AH, et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. Epub November 12, 2013. | |
Foody JM, Sajjan SG, Hu XH, et al. Loss of early gains in low-density lipoprotein cholesterol goal attainment among high-risk patients. J Clin Lipidol. 2010;4(2):126–132. | |
Nichols GA, Nag S, Chan W. Intensity of lipid-lowering therapy and low-density lipoprotein cholesterol goal attainment among the elderly before and after the 2004 National Cholesterol Education Program Adult Treatment Panel III update. Am Heart J. 2007;154(3):554–560. | |
Waters DD, Brotons C, Chiang CW, et al; Lipid Treatment Assessment Project 2 Investigators. Lipid treatment assessment project 2: a multinational survey to evaluate the proportion of patients achieving low-density lipoprotein cholesterol goals. Circulation. 2009;120(1):28–34. | |
Cullen MW, Stein JH, Gangnon R, McBride PE, Keevil JG. National improvements in low-density lipoprotein cholesterol management of individuals at high coronary risk: National Health and Nutrition Examination Survey, 1999 to 2002. Am Heart J. 2008;156(2):284–291. | |
Jukema JW, Chiang CW, Ferrières J, et al. Lipid goals among patients with diabetes or metabolic syndrome: Lipid Treatment Assessment Project (L-TAP) 2. Curr Med Res Opin. 2010;26(11):2589–2597. | |
Kotseva K, Wood D, De Backer G, De Bacquer D, Pyörälä K, Keil U; EUROASPIRE Study Group. Cardiovascular prevention guidelines in daily practice: a comparison of EUROASPIRE I, II, and III surveys in eight European countries. Lancet. 2009;373(9667):929–940. | |
Santos RD, Waters DD, Tarasenko L, et al. A comparison of non-HDL and LDL cholesterol goal attainment in a large, multinational patient population: the Lipid Treatment Assessment Project 2. Atherosclerosis. 2012;224(1):150–153. | |
Steinberg BA, Bhatt DL, Mehta S, et al. Nine-year trends in achievement of risk factor goals in the US and European outpatients with cardiovascular disease. Am Heart J. 2008;156(4):719–727. | |
Lipitor® (atorvastatin calcium) tablets [package insert]. New York, NY: Pfizer, Inc.; 2012. | |
Karalis DG, Victor B, Ahedor L, Liu L. Use of lipid-lowering medications and the likelihood of achieving optimal LDL-cholesterol goals in coronary artery disease patients. Cholesterol. 2012;2012:861924. | |
Kauffman AB, Olson KL, Youngblood ML, Zadvorny EB, Delate T, Merenich JA. Attainment of low-density lipoprotein cholesterol goals in coronary artery disease. J Clin Lipidol. 2010;4(3):173−180. | |
Kitkungvan D, Lynn Fillipon NM, Dani SS, Downey BC. Low-density lipoprotein cholesterol target achievement in patients at high risk for coronary heart disease. J Clin Lipidol. 2010;4(4):293−297. | |
Nag SS, Daniel GW, Bullano MF, et al. LDL-C goal attainment among patients newly diagnosed with coronary heart disease or diabetes in a commercial HMO. J Manag Care Pharm. 2007;13(8):652–663. | |
Maron DJ, Fazio S, Linton MF. Current perspectives on statins. Circulation. 2000;101(2):207−213. | |
Aronow HD, Hess G, Hill J, Kuznik A, Liu LZ. Switching from atorvastatin to simvastatin in patients at high cardiovascular risk: effects on low-density lipoprotein cholesterol. Am J Ther. 2010;17(2):167–175. | |
Atar D, Carmena R, Clemmensen P, et al. Clinical review: impact of statin substitution policies on patient outcomes. Ann Med. 2009;41(4):242–256. | |
Athyros VG, Tziomalos K, Karagiannis A, Mikhailidis DP. To switch (statins) or not to switch? That is the question. Expert Opin Pharmacother. 2010;11(18):2943–2946. | |
Jones PH, Davidson MH, Stein EA, et al; STELLAR Study Group. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol. 2003;92(2):152–160. | |
Mikhailidis DP, Sibbring GC, Ballantyne CM, Davies GM, Catapano AL. Meta-analysis of the cholesterol-lowering effect of ezetimibe added to ongoing statin therapy. Curr Med Res Opin. 2007;23(8):2009–2026. | |
Tunceli K, Sajjan SG, Ramey DR, et al. Switching from high-efficacy lipid-lowering therapies to simvastatin and low-density lipoprotein cholesterol goal attainment in coronary heart disease/coronary heart disease-equivalent patients. J Clin Lipidol. 2010;4(6):491–500. | |
Willey VJ, Bullano MF, Shoetan NN, Gandhi SK. Therapy modifications and low-density lipoprotein cholesterol goal attainment rates associated with the initiation of generic simvastatin. Curr Med Res Opin. 2010;26(1):121–128. | |
Boden WE, Probstfield JL, Anderson T, et al; AIM-HIGH Investigators. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365(24):2255–2267. | |
hps2-thrive.org [homepage on the Internet]. Welcome to the HPS2-THRIVE trial. University of Oxford Clinical Trial Service Unit (CTSU); 2013 [updated May 2, 2013]. Available from: http://www.hps2-thrive.org/. Accessed January 31, 2013. | |
Accord Study Group; Ginsberg HN, Elam MB, Lovato LC et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010;362(17):1563−1574. | |
Califf RM, Lokhnygina Y, Cannon CP, et al. An update on the IMProved reduction of outcomes: Vytorin Efficacy International Trial (IMPROVE-IT) design. Am Heart J. 2010;159(5):705–709. | |
Roberts WC. The rule of 5 and the rule of 7 in lipid-lowering by statin drugs. Am J Cardiol. 1997;80(1):106–107. | |
Crestor® (rosuvastatin calcium) tablets [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2012. | |
Catapano A, Toth PP, Tomassini JE, Tershakovec AM. The efficacy and safety of ezetimibe co-administered with statin therapy in various patient groups. Clinical Lipidol. 2013;8(1):13−41. | |
Liptruzet® (ezetimibe/atrovastatin) tablets [package insert]. Whitehouse Station, NJ: Merck and Co, Inc.; 2013. | |
Vytorin® (ezetimibe/simvastatin) tablets [product insert]. Whitehouse Station, NJ: Merck and Co, Inc.; 2012. | |
Toth PP, Catapano A, Tomassini JE, Tershakovec AM. Update on the efficacy and safety of combination ezetimibe plus statin therapy. Clinical Lipidol. 2010;5(5):655−684. | |
Bays HE, Averna M, Majul C, et al. Efficacy and safety of ezetimibe added to atorvastatin versus atorvastatin uptitration or switching to rosuvastatin in patients with primary hypercholesterolemia. Am J Cardiol. 2013;112(12):1885–1895. | |
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502. |
Supplementary materials
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.