Back to Journals » International Journal of Women's Health » Volume 12
Laparoscopic Myomectomy – Converting to Laparotomy for a Suspicious Intraoperative Appearance with Subsequent Benign Histology – a Pre- and Intra-Operative Dilemma
Authors Wali S , Porter-Hope C , Amin TN , Miskry T
Received 23 April 2020
Accepted for publication 4 August 2020
Published 17 September 2020 Volume 2020:12 Pages 739—744
DOI https://doi.org/10.2147/IJWH.S257136
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Elie Al-Chaer
Sarah Wali,1 Charlotte Porter-Hope,2 Tejal N Amin,1 Tariq Miskry1
1Department of Obstetrics and Gynaecology, St Mary’s Hospital, Paddington, London, W2 1NY, UK; 2Imperial College Medical School, London, UK
Correspondence: Sarah Wali Email [email protected]
Background: Although the most common uterine tumour is leiomyoma, the differential diagnoses also include the rarer adenomyoma and leiomyosarcoma. A lack of clear reliable clinical and radiological features makes the triage of uterine masses to the appropriate surgical procedure difficult. In the case of suspicious appearance of a presumed leiomyoma, an open surgical approach is recommended and morcellation is avoided.
Case: We present a case of a woman undergoing an elective laparoscopic myomectomy for a fibroid that appeared benign on ultrasound but had suspicious features intraoperatively. The operation was converted to a laparotomy to avoid the risk of morcellation-related seeding in the event that the histology was malignant. The histology was subsequently a benign exophytic adenomyoma.
Conclusion: The assessment of the nature of fibroids can be difficult both pre- and intraoperatively, and the need for difficult decision making to convert to open surgery during a laparoscopic myomectomy but with subsequent benign histology should be part of patient counselling.
Keywords: adenomyoma, communication, differential diagnosis, converting to laparotomy, morcellation risks, histology
Precis
In the case of suspicious intraoperative appearance of a presumed leiomyoma during laparoscopy, no morcellation technique should be used and conversion to laparotomy is recommended.
Introduction
Although the most common uterine growth is leiomyoma1 (a benign smooth muscle tumour), the differential diagnoses should also include the rarer adenomyoma (a benign but complex focal tumour of heterotopic endometrial glands and stroma in the myometrium with adjacent myometrial hyperplasia),2 and leiomyosarcoma (a malignant smooth muscle tumour). All three can be either asymptomatic or present with similar symptomatology including abnormal or heavy uterine bleeding, dysmenorrhea, pelvic pain and pressure thus making the use of clinical features unreliable in distinguishing benign from malignant uterine neoplasms.1 The accuracy of radiographic predictors (ultrasound and MRI) of mesenchymal uterine malignancy is limited thus also making imaging unreliable in differentiating malignant from benign uterine masses.3 The lack of clear and reliable clinical and radiological features makes the triage of uterine masses to the appropriate surgical procedure difficult.4 Although laparoscopic excision may be suitable for leiomyomas, hysterectomy is the preferred surgery for leiomyosarcoma. The management of adenomyomas is patient-tailored taking into consideration her fertility desires, symptoms, severity of disease and patient preference for definitive or conservative management.5
On finding oneself in a difficult clinical situation, the General Medical Council (GMC) good medical practice states that the doctor must use their judgement in applying clinical, professional, ethical and legal principles to the situation and must be prepared to explain and justify their decisions and actions.6 In addiction, the doctor must work in partnership with patients and be satisfied that they have consent before they provide any treatment.7
The Case
A 37-year-old nulligravid Caucasian woman presented with menorrhagia and pressure symptoms secondary to a pre-existing diagnosis of fibroids made during a routine gynaecology appointment in Spain. Her pressure symptoms included urinary frequency, constipation and pain, often resulting in syncopal episodes. There were no symptoms of endometriosis (dyschezia, dyspareunia, cyclical pain) or signs to indicate sinister pathology. She had no other past medical history and no family history of concern. On examination, she was of a normal BMI and had a 14-week size uterus on abdominal palpation with associated mild right iliac fossa tenderness. Transvaginal ultrasound (TVS) revealed a 12cm anterior fundal subserosal leiomyoma with no suspicious features (Figure 1). Management options including expectant, medical, embolization and surgical were all offered and discussed. The woman opted for an elective laparoscopic myomectomy with power morcellation.
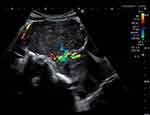 |
Figure 1 Longitudinal view of the uterus with a single 12 cm subserosal fibroid on transabdominal ultrasound scan. Feeder pedicle vessels are demonstrated on Doppler examination. |
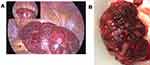
At surgery a diagnostic hysteroscopy was performed to assess the uterine cavity prior to the laparoscopy. A 2cm endometrial polyp was noted (Figure 2) and removed without any complications. The surrounding endometrium appeared normal and there was no intracavity component to her fibroid. Laparoscopic entry was unremarkable with evidence of haemorrhagic fluid within the pelvis secondary to the preceding hysteroscopy. Intraoperatively the uterine corpus, Fallopian tubes and ovaries appeared normal. A 12 cm exophytic mass was noted to be arising from the uterine fundus (Figure 3A) which had an atypical appearance of a leiomyoma. The contour was irregular, nodular, friable and highly vascular with contact bleeding (Figure 3B). These atypical features raised the suspicion of possible malignancy so a systematic exploration of the pelvis and diaphragm was performed. There was evidence of mild endometriosis (seen on the uterovesical fold) and no other obvious macroscopic disease. Peritoneal washings were obtained and the decision to convert to open surgery was made. The intramyometrial aspect of the mass had no discrete margins thus a wide uterine excision was made to avoid leaving abnormal tissue. This approach is known to include a rim of normal tissue and extension of the incision into the uterine cavity. After complete excision of the mass the uterus was repaired in layers with vicryl, achieving satisfactory restoration of anatomy and haemostasis with a total estimated blood loss of 200mls.
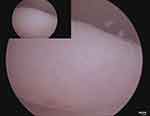 |
Figure 2 Hysteroscopic view of the uterine cavity showing a 2cm endometrial polyp arising from the left posterolateral aspect of the cavity. |
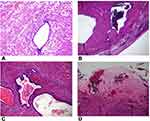
The woman recovered well postoperatively and was discharged from hospital after three days following a full debrief about the intraoperative findings, rational for laparotomy, and plan for follow up with pathology results. Urgent histology revealed the mass was in fact a benign adenomyoma with cystically dilated endometrial glands in smooth muscle stroma and areas of hyaline degeneration (Figure 4). There was no evidence of cytological atypia in the glands and the no architectural complexity to suggest an atypical polypoid adenomyoma. Cytology of peritoneal washings revealed ascitic fluid with no malignant cells. She was reviewed eight weeks postoperatively and had made a full recovery and was satisfied with her surgery with all preoperative symptoms resolving.
Discussion
This case demonstrates two important learning aspects. One is that it can still be difficult to distinguish between different types of uterine tumours despite there being established diagnostic criteria.8 Secondly, we have addressed how clinicians may manage cases when they are faced with unexpected findings at surgery.
Leiomyosarcomas, leiomyomas and adenomyomas may be completely indistinguishable from one another until definitive histological examination.9 Leiomyomas are characteristically greyish white, firm, with a “whorl” cut surface, bulging out of their capsule, with a clear surgical plane and well circumscribed from surrounding tissue. They can be solitary or numerous and vary in size. By contrast, leiomyosarcomas are classically large, solitary masses with a soft consistency and poorly defined margins. Once cut they display a fleshy, tan to grey variegated surface and may be hemorrhagic or necrotic.10 Adenomyomas vary on a spectrum from mostly solid to mostly cystic; thus, their macroscopic appearance varies from a firm consistency with smooth grey-white surfaces that may be indistinguishable from leiomyomas to cystic spaces filled with brown hemorrhagic fluid that are friable and not well circumscribed from adjacent myometrium.11 The intraoperative findings described in this case aroused suspicion of a potential leiomyosarcoma and their identification relies upon surgical experience with ordinary leiomyomas. It is important to highlight that the gross characteristics of a tumour may be suggestive of malignancy but are not diagnostic and must not be the sole reason for proceeding with an unplanned hysterectomy.
Ultrasound features that would rouse suspicion for an adenomyoma rather than a leiomyoma include the presence of cystic areas within the lesion and a circumference of the lesion that merges with the neighboring myometrium.8,12 Although a definite diagnosis can be established only with histopathology, TVS is an effective imaging modality for the preoperative differentiation of adenomyoma from leiomyoma.12 MRI is not routinely used in the initial assessment of women with assumed benign uterine pathology, especially when there are no concerns on TVS or recommendations for further imaging. Furthermore, MRI can be of limited availability and costly.
Diffuse adenomyosis is common13 however adenomyomas are uncommon14 and exophytic ones are rarely described in the literature.15 The exophytic adenomyoma in this case did not have an intracavity component and was a FIGO type 6 that may have developed by either one or a combination of two theoretical mechanisms: invagination of the basalis endometrium into the myometrium16 or invasion of pelvic endometriosis through the uterine serosa.17 Although hysterectomy is the definitive treatment for adenomyomas, an adenomyomectomy is feasible and can be approached either laparoscopically or via laparotomy, both with their own advantages and disadvantages.15 When a preoperative suspicion of adenomyosis is present, discussions with the patient are had about the difficulties in removing an adenomyoma laparoscopically due to poor dissection planes, friable tissues, bleeding, incomplete excision, breaching the cavity, excising healthy myometrium, and risk of converting to laparotomy.5 Importantly, the laparoscopic approach to fertility sparing fibroid surgery requires the use of power morcellation to remove the specimen through the port site incisions. The use of morcellation has attracted controversy due to cases of seeding of undiagnosed uterine sarcoma with potential detrimental effects on prognosis.18 Even in benign disease, seeding has also been reported resulting in adenomyotic masses and parasitic leiomyomas through the pelvis.19 Alternative specimen retrieval techniques include extending the port incision to form a mini-laparotomy or performing a posterior colpotomy, however with larger specimens some morcellation may still be required. Contained power morcellation techniques using endoscopic bags are available however their effectiveness is unknown, and leakage, although of unknown clinical significance, has been reported when bag integrity is tested after contained morcellation.20 More research and longer term follow-up is needed to further assess the theoretical protection provided by the use of in-bag morcellation in the face of unexpected malignancy.
The risk of unsuspected uterine sarcoma at the time of surgery for presumed benign leiomyoma increases with age and medical guidance bodies, including the Food and Drug Administration, have discouraged the use of morcellators especially in peri- and postmenopausal women who have a higher risk for sarcoma.21 The risk of uterine sarcoma is 1 in 574 for women aged between 25–29 years and increases to 1 in 377 in 35–39 year olds22 including for the woman in our case. Even if in bag morcellation was an available option at the time of the operation the surgeons would not have proceeded laparoscopically in view of the suspicious intraoperative appearance of the tumour. Unplanned conversion to laparotomy is estimated to occur in 1% of laparoscopic myomectomies,23 most commonly for haemorrhage and surgical difficulty caused by large, multiple and difficult to access leiomyomas. It is routine practice to discuss this possibility with the patient preoperatively and to document it on the consent form. It should also be routine practice to discuss and document on the consent form the age-related risk of unsuspected uterine malignancy at the time of surgery for laparoscopic myomectomies where power morcellation will be used for specimen removal.
Conclusion
Due to the difficulty in differentiating benign from malignant lesions macroscopically, the chance of both malignant histology despite non concerning intraoperative features as well as benign histology despite suspicious intraoperative appearance should be explained to ensure patient understanding of the dilemma. A negative result (i.e. not malignancy) after conversion to laparotomy may require even more explanation, communication, and trust between the doctor and patient. Adenomyomas have similar preoperative symptomatology and appearance to leiomyomas but intraoperatively they may present peculiarities involving difficulties in their dissection and manipulation. Although laparoscopic management of adenomyomas is feasible, if there is intraoperative suspicion of a sinister nature of the specimen no morcellation technique should be used and conversion to laparotomy is recommended.24
Teaching Points
- Prior to histological examination, the nature of fibroids can be difficult to predict both preoperatively and intraoperatively.
- In the case of suspicious intraoperative appearance of a presumed leiomyoma during laparoscopy, no morcellation technique should be used and conversion to laparotomy is recommended.
- Thorough consenting of patients preoperatively in addition to postoperative debriefing to explain difficult intraoperative decision making is essential for good medical practice and patient satisfaction in cases of conversion to laparotomy.
Written Informed Consent for Publication of the Case Details Was Obtained
This case report was exempt from the Institutional Review Broad at St Mary’s Hospital because it is a medical/educational activity that does not meet the DHHS definition of “research”.
Acknowledgment
Dr Viola Patrizia for providing histology images from the case and guiding on the histological diagnosis of adenomyomas.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Woźniak A, Woźniak S. Ultrasonography of uterine leiomyomas. Prz Menopauzalny. 2017;16(4):113–117. doi:10.5114/pm.2017.72754
2. Ferenczy A. Pathophysiology of adenomyosis. Hum Reprod Update. 1998;4(4):312–322. doi:10.1093/humupd/4.4.312
3. DeMulder D, Ascher SM. Uterine leiomyosarcoma: can MRI differentiate leiomyosarcoma from benign leiomyoma before treatment? AJR Am J Roentgenol. 2018;211(6):1405–1415. doi:10.2214/AJR.17.19234
4. Liu FW, Galvan-Turner VB, Pfaendler KS, Longoria TC, Bristow RE. A critical assessment of morcellation and its impact on gynecologic surgery and the limitations of the existing literature. Am J Obstet Gynecol. 2015;212(6):717–724. doi:10.1016/j.ajog.2015.01.012
5. Grimbizis GF, Mikos T, Zepiridis L, et al. Laparoscopic excision of uterine adenomyomas. Fertil Steril. 2008;89(4):953–961. doi:10.1016/j.fertnstert.2007.04.063
6. Good medical practice. Available from: https://www.gmc-uk.org/ethical-guidance/ethical-guidance-for-doctors/good-medical-practice.
7. GMC. Good medical practice. Published 2013. Available from: https://www.gmc-uk.org/ethical-guidance/ethical-guidance-for-doctors/good-medical-practice.
8. Van den Bosch T, Dueholm M, Leone FPG, et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015;46(3):284–298. doi:10.1002/uog.14806
9. Stewart E. Uterine Fibroids (Leiomyomas): Differentiating Fibroids from Uterine Sarcomas. Ted W, editor. UpToDate.
10. Oliva E. Practical issues in uterine pathology from banal to bewildering: the remarkable spectrum of smooth muscle neoplasia. Mod Pathol. 2016;29(Suppl 1):S104–S120. doi:10.1038/modpathol.2015.139
11. Schwartz PE, Kelly MG. Malignant transformation of myomas: myth or reality? Obstet Gynecol Clin North Am. 2006;33(1):183–198, xii. doi:10.1016/j.ogc.2005.12.003
12. Botsis D, Kassanos D, Antoniou G, Pyrgiotis E, Karakitsos P, Kalogirou D. Adenomyoma and leiomyoma: differential diagnosis with transvaginal sonography. J Clin Ultrasound. 1998;26(1):21–25. doi:10.1002/(SICI)1097-0096(199801)26:1<21::AID-JCU5>3.0.CO;2-L
13. Naftalin J, Hoo W, Pateman K, Mavrelos D, Holland T, Jurkovic D. How common is adenomyosis? A prospective study of prevalence using transvaginal ultrasound in a gynaecology clinic. Hum Reprod. 2012;27(12):3432–3439. doi:10.1093/humrep/des332
14. Song SE, Sung DJ, Park BJ, Kim MJ, Cho SB, Kim KA. MR imaging features of uterine adenomyomas. Abdom Imaging. 2011;36(4):483–488. doi:10.1007/s00261-010-9640-6
15. Takeda A, Imoto S, Sugiyama C, Nakamura H. Uterine adenomyoma with exophytic subserosal growth: case report of rare manifestation with image diagnosis and laparoscopic-assisted excision. J Minim Invasive Gynecol. 2013;20(5):717–722. doi:10.1016/j.jmig.2013.04.011
16. Garcia L, Isaacson K. Adenomyosis: review of the literature. J Minim Invasive Gynecol. 2011;18(4):428–437. doi:10.1016/j.jmig.2011.04.004
17. Kishi Y, Suginami H, Kuramori R, Yabuta M, Suginami R, Taniguchi F. Four subtypes of adenomyosis assessed by magnetic resonance imaging and their specification. Am J Obstet Gynecol. 2012;207(2):
18. Raspagliesi F, Maltese G, Bogani G, et al. Morcellation worsens survival outcomes in patients with undiagnosed uterine leiomyosarcomas: a retrospective MITO group study. Gynecol Oncol. 2017;144(1):90–95. doi:10.1016/j.ygyno.2016.11.002
19. Van der Meulen JF, Pijnenborg JMA, Boomsma CM, Verberg MFG, Geomini PMAJ, Bongers MY. Parasitic myoma after laparoscopic morcellation: a systematic review of the literature. BJOG. 2016;123(1):69–75. doi:10.1111/1471-0528.13541
20. Tissue Extraction Task Force Members. Electronic address: [email protected]. Morcellation during uterine tissue extraction: an update. J Minim Invasive Gynecol. 2018;25(4):543–550. doi:10.1016/j.jmig.2018.03.010
21. FDA. Laparoscopic power morcellators. Available from: www.fda.gov/medicaldevices/productsandmedicalprocedures/surgeryandlifesupport/ucm584463.htm.
22. Brohl AS, Li L, Andikyan V, et al. Age stratified risk of unexpected uterine sarcoma following surgery for presumed benign leiomyoma. Oncologist. 2015;20(4):433–439. doi:doi:10.1634/theoncologist.2014-0361
23. Bryant-Smith A, Holland T. Laparoscopic myomectomy: a review of alternatives, techniques and controversies. Obstet Gynaecol. 2018;20(4):261–268. doi:doi:10.1111/tog.12520
24. BSGE. BSGE statement on power morcellation - BSGE. Available from: https://www.bsge.org.uk/news/bsge-statement-power-morcellation/.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.