Back to Journals » Clinical Ophthalmology » Volume 15
Keyhole Anterior Lamellar Keratoplasty for Boston Keratoprosthesis Corneal Melt
Authors John T, Tighe S, Sheha H
Received 24 June 2021
Accepted for publication 12 August 2021
Published 2 October 2021 Volume 2021:15 Pages 3993—3996
DOI https://doi.org/10.2147/OPTH.S326306
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Thomas John,1,2,* Sean Tighe,3,4,* Hosam Sheha4,5,*
1Thomas John Vision Institute, Tinley Park, IL, USA; 2Department of Ophthalmology, Loyola University at Chicago, Maywood, IL, USA; 3Department of Biochemistry and Molecular Biology, University of Miami, Miami, FL, USA; 4Department of Ophthalmology, Florida International University Herbert Wertheim College of Medicine & Glaucoma Research Organization, Miami, FL, USA; 5Department of Ophthalmology, Manhattan Eye, Ear, and Throat Hospital, Hofstra Northwell School of Medicine, New York, NY, USA
*These authors contributed equally to this work
Correspondence: Hosam Sheha
Manhattan Eye, Ear, and Throat Hospital, 210 East 64th Street, New York, NY, 10065, USA
Tel +1 917-810-9555
Email [email protected]
Sean Tighe
Department of Ophthalmology, Florida International University, 11200 SW 8th Street, Miami, FL, 33199, USA
Tel +1 813-690-0099
Email [email protected]
Abstract: Corneal melt remains a challenging complication of Boston keratoprosthesis (KPro) and may lead to disastrous consequences such as endophthalmitis, KPro extrusion, and permanent loss of vision. Several procedures have been suggested to repair established melts; however, KPro removal or exchange is often required due to high recurrence. We herein describe a novel surgical technique to repair corneal melt: a lamellar corneal graft is centrally trephined, and a radial cut is extended from the central opening to create a keyhole graft. The graft is draped around the KPro and tightly secured around the central stem. The radial cut is closed with sutures, and the graft is secured into the underlying tissue with fibrin glue and sutures. The graft remains stable with the KPro in place, while the patients maintain a 20/200 vision or better at two years. This technique is a useful alternative for securing KPro and preventing corneal melt recurrence.
Keywords: corneal melt, keratoprosthesis, lamellar keratoplasty
Introduction
Boston keratoprosthesis (KPro) is currently the most popular and widely used artificial cornea worldwide. Typically, it is considered for patients with corneal blindness that are expected to have poor prognosis for conventional corneal transplantation.1 Common indications include failed multiple corneal transplants specially in patients with corneal chemical burns, autoimmune disorders, and congenital anomalies.2 Despite multiple advances in KPro design, surgical technique, and postoperative care, several postoperative challenges remain, such as corneal melt, KPro extrusion, and infection.3 The incidence of KPro corneal melting ranges from 14% to 16%.4–6 Corneal melting usually starts around the KPro stem due to stromal thinning and degradation secondary to persistent inflammation, infection, or mechanical stress.6 Established corneal melting may lead to disastrous consequences such as aqueous leak, hypotony, choroidal effusion, retinal detachment, endophthalmitis, KPro extrusion, permanent loss of vision or ocular structural deformity.7
Several procedures have been suggested to repair established melts including tectonic grafts, however most of the cases often require KPro removal or exchange due to the high rate of recurrence after repair.7 Utine et al reported their unfavorable outcome with the reconstruction of the ocular surface using amniotic membrane and corneal lamellar grafts and recommended opting for repeat KPro.8 Wang et al also observed favorable outcomes of patients undergoing a repeat KPro procedure for a failed KPro, when comparing them with primary KPro patients.9 The recurrence may be triggered or aggravated by the presence of clefts or gaps between the KPro and the carrier graft.10 In this article, we describe a new surgical technique, in which a keyhole anterior lamellar keratoplasty (ALK) is tightly applied around the KPro stem in eyes with extensive corneal melt and partial KPro extrusion. In addition to eliminating the recurrence, the anatomic and visual outcomes were promising.
Ethical committee review and approval was not required for this surgical report as it is not considered a research study. In addition, the description of the surgical technique does not contain any identifiable patient information.
Surgical Technique
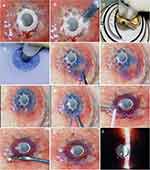
The surgery can be performed under monitored local anesthesia; however, general anesthesia is preferred, especially if corneal melt is associated with excessive aqueous leak and hypotony. Corneal melt is usually detected around the stem of the collar-button design and the anterior surface of the KPro, exposing the titanium backplate (Figure 1A). Hence the goal is to apply a lamellar corneal graft around the KPro stem and completely cover the exposed backplate. Before preparing the donor graft, the space between the front and back plates of KPro should be carefully cleared of all remaining tissue and debris. A curved Crescent knife (Alcon, Inc., Geneva, Switzerland) is used to initially dislodge the remaining tissue (Figure 1B), followed by a balanced salt solution (BSS) irrigation to flush the remaining debris.
Attention is then directed to the donor cornea, mounted into an artificial anterior chamber (Moria, Inc., Georges Besse, France). An automated microkeratome is used to fashion a 9mm lamellar graft (Figure 1C) with an average thickness of 400µm to fit in the cleared space between the KPro plates. The graft size is adjusted to cover any surrounding defect. The graft center is marked by a sterile surgical marker pen, and trypan blue stain (Vision Blue, Dorc, Zuidland, The Netherlands) is used to augment visualization of the edges. A 3.0 mm trephine punch is used to create a central opening to fit tightly around the central stem of the KPro collar button (Figure 1D). A radial cut is then extended from the central opening to the outer edge (Figure 1E) to create a keyhole lamellar graft (Figure 1F). The graft is then draped around the KPro stem (Figure 1G), while the keyhole’s cut edges are approximated using interrupted 10–0 nylon sutures (Figure 1H). This results in a circular, collar of donor lamellar graft that is firmly surrounding the central KPro stem and completely covers the areas of corneal melt. Fibrin glue (Tisseel, Baxter, Deerfield, IL) is then used to attach the lamellar graft to the ocular surface (Figure 1I), and a muscle hook is used to iron the graft to ensure uniform attachment to the underlying tissues (Figure 1J). Also, graft edges are anchored to the underlying tissues using interrupted 10–0 nylon sutures (Figure 1K). Partial or complete peritomy may be performed as needed to facilitate securing the edges of larger grafts at the limbus. The conjunctiva is then secured back using fibrin glue and additional sutures as needed. An intra-operative slit-lamp examination confirms the graft’s uniform attachment to the recipient ocular surface without any debris or air bubbles at the donor-recipient interface (Figure 1L). The patient received routine postoperative care including prednisolone acetate 1% and ofloxacin 0.3% one drop four times daily along with lubrication using artificial tears as needed. Continuous wear of a bandage contact lens is also recommended to protect the ocular surface from desiccation and related complications. Complete healing was noted within two weeks, and the graft remained stable with the KPro in place, while patients maintained a visual acuity of 20/200 or better at two years.
Discussion
The development of new strategies to prevent melting after KPro is crucial to improve the outcomes, especially in eyes with autoimmune disease, as this group of patients is particularly susceptible to such complications.11
Several procedures have been suggested to repair established melts; however, KPro exchange is often required for severe corneal melting with full-thickness melting or aqueous leak. If a new KPro is not available, several investigators have reported the use of tectonic and lamellar grafts as well as the use of amniotic membrane. However these endeavors generally fail probably due to the formation of periprosthetic fistulous tracts from epithelial downgrowth into the anterior chamber.8,9 Lower incidence of melt recurrence and longer survival after repeat KPro may be attributed to the tight apposition of the new graft around the new KPro. However, delaying the melt management until a graft and KPro are obtained can put the eye at greater risk of hypotony, retinal detachment and infection.
Our technique appears effective in securing KPro and reducing corneal melt recurrence. We believe that the exact thickness graft and its firm apposition eliminates the potential dead space around the KPro stem, which is likely a conduit of microorganisms or proteolytic enzymes responsible for graft melting. Tight apposition may also reduce the friction between the carrier graft and the KPro stem, which is expected to occur continuously due to regular eye movements and eye rubbing.12 Therefore, our technique helps decrease the mechanical stress on the carrier graft which is known to increase the expression of MMP, IL-6, and TNF-a, and hence implicated in the progressive corneal thinning.7 Collectively, our technique helps improve the outcomes and reduce devastating complications of KPro, especially for those group of patients with the most desperate need for visual rehabilitation.
What Was Known
- KPro exchange is often required for severe corneal melting with full-thickness melting and aqueous leak.
- Delaying management of the melt until a graft and KPro are obtained can put the eye at greater risk of hypotony, retinal detachment, and infection.
- Several investigators have reported failure of KPro repair using amniotic membrane, tectonic and lamellar corneal grafts.
What This Paper Adds
- This is the first reported use of keyhole anterior lamellar corneal graft to successfully repair corneal melt along with partial KPro extrusion.
- This technique appears effective in securing KPro with a low risk of melt recurrence and longer survival after repair.
- In addition to eliminating the recurrence, the anatomic and visual outcomes were promising.
Acknowledgments
This surgical technique was presented at the American Society of Cataract and Refractive Surgery (ASCRS) Meeting, San Diego, CA, May 2019.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Avadhanam VS, Smith HE, Liu C. Keratoprostheses for corneal blindness: a review of contemporary devices. Clin Ophthalmol. 2015;9:697–720. doi:10.2147/OPTH.S27083
2. Sejpal K, Yu F, Aldave AJ. The Boston keratoprosthesis in the management of corneal limbal stem cell deficiency. Cornea. 2011;30:1187–1194. doi:10.1097/ICO.0b013e3182114467
3. Aldave AJ, Kamal KM, Vo RC, Yu F. The Boston type I keratoprosthesis: improving outcomes and expanding indications. Ophthalmology. 2009;116:640–651. doi:10.1016/j.ophtha.2008.12.058
4. Chew HF, Ayres BD, Hammersmith KM, et al. Boston keratoprosthesis outcomes and complications. Cornea. 2009;28:989–996. doi:10.1097/ICO.0b013e3181a186dc
5. Chan CC, LoVerde L, Qiang J, et al. Incidence, risk factors, and surgical management of Boston type 1 keratoprothesis corneal melts, leaks, and extrusions. Cornea. 2016;35:1049–1056. doi:10.1097/ICO.0000000000000911
6. Robert MC, Dohlman CH. A review of corneal melting after Boston Keratoprosthesis. Semin Ophthalmol. 2014;29:349–357. doi:10.3109/08820538.2014.959186
7. Bouhout S, Robert MC, Deli S, Harissi-Dagher M. Corneal melt after Boston keratoprosthesis: clinical presentation, management, outcomes and risk factor analysis. Ocul Immunol Inflamm. 2018;26:693–699. doi:10.1080/09273948.2016.1269930
8. Utine CA, Tzu JH, Akpek EK. Clinical features and prognosis of Boston type 1 keratoprosthesis-associated corneal melt. Ocul Immunol Inflamm. 2011;19(6):413–418. doi:10.3109/09273948.2011.621580
9. Wang JC, Rudnisky CJ, Belin MW, Ciolino JB. Outcomes of Boston keratoprosthesis type 1 reimplantation: multicentre study results. Can J Ophthalmol. 2018;53(3):284–290. doi:10.1016/j.jcjo.2017.10.021
10. Dudenhoefer EJ, Nouri M, Gipson IK, et al. Histopathology of explanted collar button keratoprostheses: a clinicopathologic correlation. Cornea. 2003;22(5):424–428. doi:10.1097/00003226-200307000-00007
11. Yaghouti F, Nouri M, Abad JC, et al. Keratoprosthesis: preoperative prognostic categories. Cornea. 2001;20:19–23. doi:10.1097/00003226-200101000-00003
12. Balasubramanian S, Pye D, Willcox M. Effects of eye rubbing on the levels of protease, protease activity and cytokines in tears: relevance in keratoconus. Clin Exp Optom. 2013;96(2):214–218. doi:10.1111/cxo.12038
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.