Back to Journals » International Journal of General Medicine » Volume 14
Isolated Neutropenia/Benign Ethnic Neutropenia: A Common Clinical and Laboratory Finding in Southern and Western Saudi Arabia
Authors Awan ZA, Al Amoudi SM, Saboor M, Alkhaldy HY
Received 6 January 2021
Accepted for publication 28 January 2021
Published 15 February 2021 Volume 2021:14 Pages 451—457
DOI https://doi.org/10.2147/IJGM.S300690
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Zuhier A Awan,1,2 Saeed M Al Amoudi,2 Muhammad Saboor,3,4 Husain Y Alkhaldy5,6
1Department of Clinical Biochemistry, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia; 2Department of Clinical Pathology, Al-Borg Medical Laboratories, Jeddah, Saudi Arabia; 3Department of Medical Laboratory Technology, Faculty of Applied Medical Sciences, Jazan University, Gazan, Saudi Arabia; 4Medical Research Centre (MRC), Jazan University, Jazan, Saudi Arabia; 5Department of Internal Medicine, College of Medicine, King Khalid University, Abha, Saudi Arabia; 6Research Center for Advanced Materials Science, King Khalid University, Abha, Saudi Arabia
Correspondence: Husain Y Alkhaldy
Department of Internal Medicine, College of Medicine, Research Center for Advanced Materials Science, King Khalid University, P.O.Box 9004, Abha, 61413, Saudi Arabia
Tel +966599915567
Email [email protected]
Objective: Isolated mild neutropenia is a common clinical problem in some ethnicities including Arabs and Middle Eastern population. The current study aims to authenticate the prevalence of isolated neutropenia in Southern and Southwestern Saudi Arabia, explore the effect of altitude or regional differences and to suggest a new reference range for neutrophil count.
Methods: In this retrospective cross-sectional study, laboratory results of a commercial laboratory were screened over a period of 5 years (2016– 2020) in seven different cities of different altitudes in South and southwestern Saudi Arabia. Participants’ laboratory investigations were reviewed and excluded for any abnormal complete blood count, renal profile, liver profile, lipid profile, thyroid function test, fasting blood glucose, or HbA1c findings. Descriptive analysis and 95th percentile range were calculated using standard statistical methods.
Results: A total of 91,880 complete blood count results were included in the final analysis. Isolated neutropenia was common laboratory finding, with a prevalence ranging from 11% to 23%. The 2.5th percentile of the neutrophil count was lower than currently utilized 1.5× 109/L in all studied seven cities.
Conclusion: Mild to moderate neutropenia is common in Southern and Southwestern Saudi Arabia. Benign ethnic neutropenia (BEN) likely explains this high prevalence. Since BEN has no clinical significance, the reference range for normal neutrophil counts needs to be adjusted to reflect the effect of BEN.
Keywords: isolated neutropenia, benign ethnic neutropenia, reference intervals, Saudi Arabia
Introduction
Neutrophils are the most abundant leukocytes in the peripheral blood. They play a pivotal role in the innate immune system and are key regulators of the adaptive immune system.1,2 The absolute neutrophil count (ANC) normally ranges from 1.8 to 7.5 × 109/L.3 Marked reduction in the peripheral neutrophil count is regarded as a serious clinical problem that needs to be addressed. Peripheral neutrophils constitute only a minute fraction of the total body neutrophils that are mainly in the bone marrow. The number of neutrophils in the peripheral circulation may be affected by multiple factors, including stress, smoking, pregnancy, and drugs. Altitude may also be associated with lower neutrophil counts.4,5 Reduced ANC is defined as a count less than 1.5 × 109/L and is known as neutropenia. In severe cases, neutropenia predisposes an individual to infections or even life-threatening consequences. Neutropenia is categorized into three subtypes based on the ANC: mild (1.0–1.5 × 109/L), moderate (0.5–1.0 × 109/L), and severe (<0.5 × 109/L).
Decreased production or increased destruction of neutrophils may cause neutropenia.3 Severe congenital neutropenia, as the name suggests, is an inherited condition that leads to life-threatening infections in the first year of life. This condition has also a propensity to progress to acute leukemia.6 Patients whom absolute neutrophil remains low due to decreased production are more prone to develop infections as compared to conditions associated with increased peripheral destruction or margination of neutrophils. Moreover, as first reported in 1941 by Forbes et al, low neutrophil count (<1.5 × 109/L) in certain ethnicities with no predisposition to infections indicates no clinical disadvantage in the affected individuals. This condition is known as benign ethnic neutropenia (BEN).7,8
In ethnicities with reduced circulating neutrophil counts, the bone marrow cellularity, morphology, number of granulocyte colony-forming units, and maturation of myeloid cells remain normal. However, slight reduction in the number of progenitors has been observed in some cases of BEN.7 Additionally, a slight increase in ANC has been reported in individuals with BEN after exercise.9 Subjects with BEN show a circadian variation of the neutrophil count similar to that observed in subjects without BEN.10
BEN is most common in Africans, Afro-Americans, Ethiopian Jews, and Yemenite Jews.8,11–13 We previously showed that a higher prevalence of isolated neutropenia was observed in two cities (Jeddah and Abha) in Saudi Arabia.14 Saudi Arabia, representing the larger part of the Arabian peninsula, is inhabited by mainly Arab tribes of Semitic origin. In this study, we aimed to confirm our previous findings through a large sample size from seven different cities at different altitudes (Jeddah, Taif, Al Baha, Abha, Khamis Mushait, Najran, and Gizan) in Southern and Southwestern Saudi Arabia. We also aimed to explore any potential association of neutropenia prevalence with altitude or any regional differences.
Methods
This study was designed and conducted in accordance with the Declaration of Helsinki principles. The study protocol and study subject consent waiver, due to use of completely de-identified data with no associated risk to the participants, were approved by the ethical and research committee of King Khalid University, Abha, Saudi Arabia. (ECM#2020-0905) – (HAPO-06-B-001). Neither personal identification nor bioinformation of study subjects were collected or disclosed. In this retrospective study, results of a commercial laboratory (Al–Borg Laboratories) were screened over a period of 5 years (2016–2020) in seven different cities in Saudi Arabia. Laboratory investigations included complete blood count (CBC), renal profile, liver profile, lipid profile, thyroid function test, fasting blood glucose measurement, and HbA1c measurement. Screened participants were either customers who do lab tests for health check-ups or follow up of chronic health conditions. Data were collected from the laboratories located in Jeddah (sea level), Taif (1898 meters above sea level), Al Baha (2000 meters), Abha (2270 meters), Khamis Mushait (1700 meters), Najran (1293 meters), and Gizan (sea level). Exclusion lab criteria were applied to exclude any pre-existing health conditions. Subjects selected were all having normal CBC, biochemical, kidney, liver, sugar, lipid and thyroid profiles and thus were assumed healthy and proper to estimate the neutrophil count normal range.
Inclusion and Exclusion Criteria
Male and female patients aged 13–60 years were included. To estimate the white blood cell count (WBC) reference, the following exclusion criteria were applied: any abnormalities, unless mild, in the renal profile (serum blood urea nitrogen, creatinine), liver profile (ALT, AST, total bilirubin, and direct bilirubin), lipid profile (cholesterol, HDL, LDL, and triglyceride), thyroid function test (TSH, T4), and diabetic profile (fasting blood sugar and HbA1c).
For CBC, individuals with hemoglobin (Hb) concentration of 12–19 g/dl, platelet count of 140–450 × 109/L, and WBC count of less than 11.5×109/L were included. For low WBC count, the exclusion was applied only if other blood cell types were abnormal, eg, concurrent anemia or thrombocytopenia. Since BEN is usually mild to moderate, analyses were performed with both inclusion and exclusion of neutrophil counts less than 0.5 × 109/L.
Blood Collection and Laboratory Analysis
To ensure no inter laboratory variations in the obtained results, the different branches follow organizational standard operating protocols of blood sample collection for CBC. Blood samples were collected into liquid K3 EDTA tubes and processed within 2 hours of collection. Sysmex automated analyzers (Sysmex Corporation, Kobe, Japan) were utilized for WBC enumeration and differential count.
Statistical Analysis
The participants’ data were de-identified for any personal information before importing to an Excel datasheet. After extraction, data were revised, coded, and transferred into the statistical software IBM SPSS Statistics for Windows, version 22 (IBM Corp., Armonk, N.Y., USA). Normal distribution assessment was applied for all scale parameters. Data for both sex and age were stratified into two groups: young adults (13–40 years old) and middle-aged adults (41–60 years old). Descriptive analyses using the mean and 95% confidence interval, median, range, standard deviation (SD), and percentiles (2.5th, 25th, 75th, and 97.5th) were utilized to assess the normal population range. Student’s t-tests were used to compare continuous variables between different sex and age groups. A p-value <0.05 was considered statistically significant. Since plasma volume is known to be reduced with altitude, we explored any association between neutrophil count and hematocrit considering that lower hematocrit is associated with higher plasma volume and vice versa.
Results
For the present study, 91,880 CBC results were included in the final analysis (supplement 1). Table 1 summarizes the frequency of neutropenia in the studied population, while Table 2 depicts the statistical analysis of the mean, SEM, median, SD, range, and percentiles (2.5th, 25th, 75th, and 97.5th). Comparison between groups based on sex and age group is depicted in Table 3.
 |
Table 1 Frequency of Neutropenia in the Studied Populations |
 |
Table 2 Descriptive Analysis of Distribution of Neutrophils Count (×109/L) in the Studied Populations |
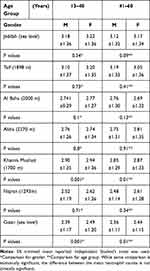 |
Table 3 Mean (±Standard Deviation) of the Neutrophil Count with Respect to Gender and Age Group Distribution |
Mild neutropenia was the most common type of neutropenia among the studied subjects of all cities. The city of Al Baha showed slightly lower frequency in comparison with others, while the highest prevalence was observed in Najran and Gizan (17%). A high prevalence of moderate neutropenia was observed in Abha (5%), Najran (5%), and Gizan (6%). Severe neutropenia was rare among all types, with only 88 cases out of 91,880 (Table 1). Pearson correlation between neutrophil count and hematocrit was not significant.
The lowest value of ANC for the 2.5th percentile was found in Najran (0.83×109/L), while the highest was found in Jeddah (1.05 × 109/L). Additionally, the 2.5th percentile ANC value of the study participants in the remaining cities was between the ANC values found in Jeddah and Najran (0.83–1.05×109/L). The highest prevalence of isolated neutropenia was found in Najran (23% with mild and moderate forms). Gizan had a prevalence of 22%, making it the second most prevalent city for neutropenia, while in Taif, Jeddah, Khamis Mushait, Abha, and Al Baha, the frequency of neutropenia (combined mild and moderate) was found to be 11%, 15%, 16%, 17%, and 18%, respectively. Severe neutropenia was the rarest, with only 88 cases out of 91,880 (0.096%).
Discussion
The present study was conducted primarily to authenticate our previous findings of a high prevalence of isolated neutropenia and explore any geographical association by including other cities of different altitudes and a large dataset of participants. Our aims also include using a larger multi-cities sample to determine a modified reference range suitable to our population. After excluding abnormal tests results, 91,880 CBCs were included for determining the frequency of neutropenia in the study population. The current study validates our previous findings in which the prevalence of neutropenia was found to be 18%.14 The findings of this study are consistent with those of Gari et al12 who also reported a high prevalence (20%) of neutropenia in Jeddah. Several other studies have shown a variable frequency of neutropenia among different populations.11,15–17 Moreover, the findings of this study indicated that the 2.5th percentile of neutrophil count was lower than what is utilized now as lower end of the reference range; 1.5 × 109/L, in all seven cities. This finding is most likely explained by racial effect. We postulate, as explained below, that this prevalence is most likely explained by Benign ethnic neutropenia.
BEN has a positive association with Duffy-null phenotype.18 Atypical chemokine receptor-1 (ACKR1) gene, also known as the Duffy antigen receptor for chemokines (DARC), encodes the Duffy antigens (Fya and Fyb). A single nucleotide polymorphism (SNP) at codon 42 (G125A, rs12075) results in two types of encoded proteins, Fya and Fyb. Fya antigen includes glycine, while Fyb contains aspartic acid.19 The Duffy-null phenotype (Fya–b–), also symbolized as erythroid silent (ES), occurs due to another SNP, rs2814778 at position 46 (– 46 T > C). The Duffy antigen expression is inhibited in individuals with homozygous C/C polymorphisms.20 Individuals with rs2814778 suffer from an abnormal phenotype of neutrophils with enhanced egression into the tissues from the blood.21 Plasmodium vivax enters the red cells through DARC. Duffy-null phenotypes therefore may provide a natural resistance to P. vivax. Consequently, this protective role of Duffy-null phenotypes in malaria endemic regions has undergone positive selection leading to its high prevalence in some ethnicities.22 Although molecular analysis for rs2814778 of Duffy-null phenotypes was not conducted in this study to correlate the high frequency of this phenotype with BEN, the prevalence of Duffy-null phenotypes in Saudi Arabia reported in various studies could be linked with BEN. Duffy-null phenotype (Fya–b–) at a frequency of 62% to 78% has been reported in Saudi Arabia populations.23,24 Therefore, the isolated neutropenia reported in this current study may be Duffy-null phenotype-induced BEN.
Arabs are among the ethnicities where higher prevalence of BEN has been reported. A previous study has shown that the prevalence of benign neutropenia was high in Saudi participants as compared with none Saudis.12 This finding suggests that BEN is more common in certain ethnicities. Herein this point also needs to mentioned that consanguineous marriages are common in Saudi Arabia that could be attributed to the high prevalence of BEN in Saudis.25 Thirdly, as Saudi Arabia is in close proximity to the African countries where the prevalence of BEN is up to 50%, the migration of African to this region could be considered a contributing factor in the high prevalence of BEN is Saudi population. We previously noticed a higher prevalence of isolated neutropenia at a high altitude in comparison with sea level.14 In this study, the prevalence of isolated neutropenia in Taif city (1898 m above sea level) was not different from that in the coastal Jeddah city in the current study. Furthermore, in this study, we did not find any association between altitude levels and hematocrit (as a surrogate for decreased plasma volumes). Nonetheless, owing to many confounding factors, including different prevalence of BEN, the effect of altitude on the neutrophil count should not be dismissed without further research.
The ANC, usually above 1.5 × 109/L, is used as an inclusion criterion for clinical trials.26 This can potentially lead to exclusion of otherwise healthy individuals with physiologically lower neutrophil counts.27 The same criterion is also used to grade chemotherapy-associated toxicity, which helps in dose adjustment of crucial treatments. Dose intensity of chemotherapy is important, and dose reduction or delay is associated with increased mortality.28,29 In high-prevalence BEN populations, low baseline ANC should not be considered a factor for altering the treatment strategies in certain clinical conditions. For instance, use of clozapine and deferiprone also induces neutropenia. Administration of these drugs can commence if the ANC is above 0.5 × 109/L, in the absence of any other comorbid condition. In patients with malignancies, if the ANC ranges between 0.5 × 109/L and 1.0 × 109/L, chemotherapy may be administered safely.30
In clinical practice, individuals with ANC <1.5 × 109/L usually undergo unwarranted laboratory and clinical investigations without any significant findings. Although the differential diagnoses of neutropenia are wide, BEN can be determined based on the appropriate clinical scenario. BEN is usually an incidental finding of isolated mild neutropenia with no other hematological abnormalities. It is not associated with increased risks of infections.31 Previous studies have indicated the chronicity of the condition, and follow-up usually indicates the stability and benign nature of the condition.32 Clinical history can also suggest the presence of other causes of neutropenia. For instance, severe congenital neutropenia is associated with severe neutropenia and increased risk of infections. In cyclic neutropenia, recurrent infections, especially oral lesions and low neutrophil counts, at 21-day intervals are common. Meanwhile, in BEN, recurrent infections are infrequent, while ANC usually are only mildly low. Moreover, since molecular analysis for rs2814778 of Duffy-null phenotypes is not available in clinical laboratories, the Duffy phenotype could be utilized as a marker for identifying individuals with BEN.33 In current clinical practice, the diagnosis of BEN depends on the exclusion of other causes of neutropenia.
This study has the advantage of large sample size and the availability of extended lab tests to exclude many health conditions. Nonetheless, it has some limitations. It was a retrospective analysis with few demographics and health-related data. Some micronutrient deficiencies that may affect neutrophil counts such as B12 and folate deficiencies were not assessed.
Conclusion
Isolated neutropenia is common in the southern and southwestern Saudi Arabia. BEN, extensively reported in Middle Eastern countries, is likely to explain many cases of mild isolated neutropenia. However, the effects of other factors, including geography, altitude, and autoimmune diseases, should be explored. In light of the above results, we suggest that the 2.5th percentile of ANC for Saudi Arabians could be set at 1.0 × 109/L. Additionally, ANC of >0.8×109/L, when isolated, chronic and stable with benign medical history, should not be considered alarming in the absence of fever, pyogenic infection, or inflammation.
Data Sharing Statement
Data used in this research are available through corresponding author to readers upon request.
Acknowledgment
The authors extend their appreciation to the research center for advanced materials (RCAMS) at King Khalid University, Abha, Saudi Arabia for supporting this work through the research group program under grant number (RCAMS/KKU/0020/20).
Disclosure
All authors declare that there are no conflicts of interest in this work.
References
1. Nauseef WM. Neutrophils, from cradle to grave and beyond [Internet]. Immunol Rev. 2016;273:5–10. doi:10.1111/imr.12463
2. Kobayashi Y. Neutrophil biology: an update [Internet]. EXCLI J. 2015;14:220–227. doi:10.17179/excli2015-102
3. Hoffbrand AVMP. The white cells 1: granulocytes, monocytes and their benign disorders. In: Hoffbrand AVMP, editor. Hoffbrand’s Essential Hematology.
4. Peterson RF, Peterson WG. The differential count at high altitudes. J Lab Clin Med. 1935;20(7):723–726.
5. Siqués P, Brito J, León-Velarde F, et al. Hematological and lipid profile changes in sea-level natives after exposure to 3550-m altitude for 8 months. High Alt Med Biol. 2007;8(4):286–295. doi:10.1089/ham.2007.8405
6. Xia J, Miller CA, Baty J, et al. Somatic mutations and clonal hematopoiesis in congenital neutropenia. Blood. 2018;131:408–416. doi:10.1182/blood-2017-08-801985
7. Atallah-Yunes SA, Ready A, Newburger PE. Benign ethnic neutropenia. Blood Rev. 2019;37:100586. doi:10.1016/j.blre.2019.06.003
8. Lakhotia R, Aggarwal A, Link ME, Rodgers GP, Hsieh MM. Natural history of benign ethnic neutropenia in individuals of African ancestry. Blood Cells Mol Dis. 2019;77:12–16. doi:10.1016/j.bcmd.2019.01.009
9. Phillips D, Rezvani K, Bain BJ. Exercise induced mobilisation of the marginated granulocyte pool in the investigation of ethnic neutropenia. J Clin Pathol. 2000;53(6):448–483. doi:10.1136/jcp.53.6.481
10. Souto Filho JTD, Portugal RD, Nucci M. Effect of circadian variation on neutrophil mobilization to the peripheral blood in benign constitutional neutropenia. Exp Hematol. 2019;69:22–26. doi:10.1016/j.exphem.2018.10.007
11. Denic S, Narchi H, Al Mekaini LA, Al-Hammadi S, Al Jabri ON, Souid AK. Prevalence of neutropenia in children by nationality. BMC Hematol. 2016;16(1):1–7. doi:10.1186/s12878-016-0054-8
12. Gari M, Dakhakhni M, Gari A, et al. Incidence and potential causative factors associated with chronic benign neutropenia in the Kingdom of Saudi Arabia. BMC Proc. 2015;9(2):1–7. doi:10.1186/1753-6561-9-S2-S1
13. Ortiz MV, Meier ER, Hsieh MM. Identification and clinical characterization of children with benign ethnic neutropenia. J Pediatr Hematol Oncol. 2016;38(3):e140–3. doi:10.1097/MPH.0000000000000528
14. Alkhaldy H, Awan ZA, Abouzaid AA, et al. The prevalence of isolated neutropenia at high altitude in Southern Saudi Arabia: does altitude affect leucocyte count? Int J Gen Med. 2020;13:1373–1379. doi:10.2147/IJGM.S284133
15. Wonodi I, Oduguwa T, Amoo I, et al. Benign ethnic neutropenia in a sample of Nigerian healthy controls and clozapine-treated schizophrenia patients with DARC null variant. Schizophr Bull. 2017;43(suppl_1):S67. doi:10.1093/schbul/sbx021.178
16. Goswami R, Kakkar N, John MJ. Lower total leukocyte and neutrophil counts in healthy young Africans from Uganda. Indian J Hematol Blood Transfus. 2018;34(2):278–281. doi:10.1007/s12288-017-0866-0
17. Merz LE, Achebe M. When non-whiteness becomes a condition. Blood. 2020.
18. Rappoport N, Simon AJ, Lev A, et al. Correlation between “ACKR1/DARC null” polymorphism and benign neutropenia in Yemenite Jews [Internet]. Br J Haematol. 2015;170:892–895. doi:10.1111/bjh.13345
19. Rappoport N, Simon AJ, Amariglio N, Rechavi G. The Duffy antigen receptor for chemokines, ACKR1,– ‘Jeanne DARC’ of benign neutropenia. Br J Haematol. 2019;184(4):497–507. doi:10.1111/bjh.15730
20. Fragiadaki I, Papadakis S, Sevastaki G, et al. Increased frequency of the single nucleotide polymorphism of the DARC/ACKR1 gene associated with ethnic neutropenia in a cohort of European patients with chronic idiopathic neutropenia. Am J Hematol. 2020;95(7):E163–6. doi:10.1002/ajh.25813
21. Palmblad J, Höglund P. Ethnic benign neutropenia: A phenomenon finds an explanation. Pediatr Blood Cancer. 2018;65(12):e27361. doi:10.1002/pbc.27361
22. Dinardo CL, Kerbauy MN, Santos TC, et al. Duffy null genotype or Fy(a-b-) phenotype are more accurate than self-declared race for diagnosing benign ethnic neutropenia in Brazilian population [Internet]. Int J Lab Hematol. 2017;39:e144–6. doi:10.1111/ijlh.12712
23. Fahad Almsned LA. Frequency of the ABO and Duffy Phenotypes among Saudi Population in Eastern Provence. J Med Sci Clin Res. 2017;05(03):19295–19298. doi:10.18535/jmscr/v5i3.154
24. Owaidah AY, Naffaa NM, Alumran A, Alzahrani F. Phenotype frequencies of major blood group systems (Rh, kell, kidd, duffy, mns, p, lewis, and lutheran) among blood donors in the eastern region of Saudi Arabia. J Blood Med. 2020;11:59–65. doi:10.2147/JBM.S236834
25. El-Hazmi MAF, Al-Swailem AR, Warsy AS, Al-Swailem AM, Sulaimani R, Al-Meshari AA. Consanguinity among the Saudi Arabian population. J Med Genet. 1995;32:623–626. doi:10.1136/jmg.32.8.623
26. Jin S, Pazdur R, Sridhara R. Re-evaluating eligibility criteria for oncology clinical trials: analysis of investigational new drug applications in 2015. J Clin Oncol. 2017;35(33):3745–3752. doi:10.1200/JCO.2017.73.4186
27. Vastola ME, Yang DD, Muralidhar V, et al. Laboratory eligibility criteria as potential barriers to participation by black men in prostate cancer clinical trials [Internet]. JAMA Oncol. 2018;4:413–414. doi:10.1001/jamaoncol.2017.4658
28. Denduluri N, Lyman GH, Wang Y, et al. Chemotherapy dose intensity and overall survival among patients with advanced breast or ovarian cancer. Clin Breast Cancer. 2018;18(5):380–386. doi:10.1016/j.clbc.2018.02.003
29. Crawford J, Denduluri N, Patt D, et al. Relative dose intensity of first-line chemotherapy and overall survival in patients with advanced non-small-cell lung cancer. Support Care Cancer. 2020;28(2):925–932. doi:10.1007/s00520-019-04875-1
30. Hsieh MM, Tisdale JF, Rodgers GP, Young NS, Trimble EL, Little RF. Neutrophil count in African Americans: lowering the target cutoff to initiate or resume chemotherapy? J Clin Oncol. 2010;28:1633–1637. doi:10.1200/JCO.2009.24.3881
31. Legge SE, Christensen RH, Petersen L, et al. The Duffy-null genotype and risk of infection. Hum Mol Genet. 2020;1–9. doi:10.1093/hmg/ddz215
32. Donadieu J, Beaupain B, Fenneteau O, Bellanné-Chantelot C. Congenital neutropenia in the era of genomics: classification, diagnosis, and natural history [Internet]. Br J Haematol. 2017;557–574. doi:10.1111/bjh.14887
33. Barreto MESF, Lipay ME, Santos LD, et al. Duffy phenotyping and FY*B-67T/C genotyping as screening test for benign constitutional neutropenia. Hematol Transfus Cell Ther. 2020. doi:10.1016/j.htct.2020.08.015
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
