Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Inhaled corticosteroids, blood eosinophils, and FEV1 decline in patients with COPD in a large UK primary health care setting
Authors Whittaker HR, Müllerova H , Jarvis D , Barnes NC , Jones PW , Compton CH, Kiddle SJ , Quint JK
Received 9 January 2019
Accepted for publication 1 April 2019
Published 23 May 2019 Volume 2019:14 Pages 1063—1073
DOI https://doi.org/10.2147/COPD.S200919
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Hannah R Whittaker,1 Hana Müllerova,2 Deborah Jarvis,1 Neil C Barnes,2 Paul W Jones,2 Chris H Compton,2 Steven J Kiddle,3 Jennifer K Quint1
1National Heart and Lung Institute, Imperial College London, London, UK; 2Respiratory Epidemiology, GlaxoSmithKline R&D, Uxbridge, UK; 3MRC Biostatistics Unit, University of Cambridge, Cambridge, UK
Background: Inhaled corticosteroid (ICS)-containing medications slow rate of decline of FEV1. Blood eosinophil (EOS) levels are associated with the degree of exacerbation reduction with ICS.
Purpose: We investigated whether FEV1 decline differs between patients with and without ICS, stratified by blood EOS level.
Patients and methods: The UK Clinical Practice Research Datalink (primary care records) and Hospital Episode Statistics (hospital records) were used to identify COPD patients aged 35 years or older, who were current or ex-smokers with ≥2 FEV1 measurements ≥6 months apart. Prevalent ICS use and the nearest EOS count to start of follow-up were identified. Patients were classified at baseline as higher stratum EOS (≥150 cell/μL) on ICS; higher stratum EOS not on ICS; lower stratum EOS (<150 cells/μL) on ICS; and lower stratum EOS not on ICS. In addition, an incident ICS cohort was used to investigate the rate of FEV1 change by EOS and incident ICS use. Mixed-effects linear regression was used to compare rates of FEV1 change in mL/year.
Results: A total of 26,675 COPD patients met our inclusion criteria (median age 69, 46% female). The median duration of follow up was 4.2 years. The rate of FEV1 change in prevalent ICS users was slower than non-ICS users (−12.6 mL/year vs −21.1 mL/year; P =0.001). The rate of FEV1 change was not significantly different when stratified by EOS level. The rate of FEV1 change in incident ICS users increased (+4.2 mL/year) vs −21.2 mL/year loss in non-ICS users; P<0.001. In patients with high EOS, incident ICS patients showed an increase in FEV1 (+12 mL/year) compared to non-ICS users whose FEV1 decreased (−20.8 mL/year); P<0.001. No statistical difference was seen in low EOS patients. Incident ICS use is associated with an improvement in FEV1 change, however, over time this association is lost.
Conclusion: Regardless of blood EOS level, prevalent ICS use is associated with slower rates of FEV1 decline in COPD.
Keywords: COPD, lung function, eosinophil, inhaled corticosteroids
Introduction
In 2016, COPD ranked the ninth leading cause of death worldwide and it is projected to increase to the third leading cause of death and the seventh leading cause of disability-adjusted life years (DALYs) lost worldwide by 2030.1,2 One of the hallmarks of COPD is decline in lung function, which is thought to be more rapid compared to the decline in the general population.3
Initial treatment of COPD usually comprises long-acting bronchodilators, ie, long-acting beta2-agonists (LABA) and long-acting muscarinic antagonists (LAMA), which increase and maintain lung function, improve health-related quality of life, and reduce the risk of exacerbations of COPD (AECOPD).4 Inhaled corticosteroids (ICS) are additionally recommended by NICE in combination with LABA or LAMA/LABA for people with frequent AECOPD or who remain breathless. Studies to date have shown that ICS reduce the rate of moderate and severe AECOPD, reduce the rate of hospitalization, and decrease the rate of decline of forced expiratory volume in 1 s (FEV1) over time.5,6 However, studies also suggest that ICS increase the risk of pneumonia and increase the risk of upper respiratory tract infections, suggesting that more careful phenotyping of patients most likely to benefit from ICS is required.7,8
Blood eosinophils have been considered a potential biomarker in COPD in relation to ICS. Studies have shown that ICS are more effective in COPD patients with higher blood eosinophil counts rather than lower in terms of reducing AECOPD risk.9–11 A few studies have explored the relationship between ICS and blood eosinophil count in terms of FEV1 decline, but the majority have consisted of randomized control trials with strict inclusion criteria, and short-term follow-up of roughly less than 3 years limiting their external validity to the wider COPD population.9,10,12
Using Clinical Practice Research Datalink (CPRD), a primary care research database, we investigated whether the rate of FEV1 decline in COPD patients differed between patients on ICS and patients not on ICS, stratified by blood eosinophil level.
Methods
Study population
The data
We used general practice (GP) electronic heath records from the Clinical Practice Research Datalink (CPRD) linked Hospital Episode Statistics (HES) data (see supplementary material for further information).
The population
The study population included patients with a validated diagnosis of COPD whose records were deemed to be of research standard from January 1, 2004. Briefly, the validated definition required specific COPD codes.13 COPD patients were included if they were aged 35 years or older, were current or ex-smokers at baseline, had at least one blood eosinophil count at baseline, and had at least two FEV1 measurements at least six months apart over the study period. Patients with a history of asthma were included in the main analysis, but later excluded in a sensitivity analysis due to the potential for misclassification between asthma and COPD.14
Study design
Patients were followed up from their first FEV1 measurements after January 2004, date of COPD diagnosis, date of registration with an eligible practice, and the date at which patients turned 35. Patients were followed until February 2016, date of death, last collection date, date of transfer out of an eligible GP practice, or date of prescribed ICS-containing medication (if they were not on an ICS-containing medication at baseline) whichever was earlier (Figure 1).
 | Figure 1 Study design.Abbreviations: GP, general practice; EOS, eosinophil; ICS, inhaled corticosteroids. |
Variables
Exposures
ICS use was defined as the presence of at least one ICS-containing medication in the year prior to the patient’s index date, determined through recorded ICS-containing prescriptions in the patient’s data. Patients were categorized into those using an ICS-containing medication and those not using an ICS-containing medication at baseline.
Incident ICS use was defined as the presence of at least one ICS-containing medication in the first year after patients’ index date and no prevalent ICS use in the year prior to their index date.
Absolute blood eosinophil counts two years prior to the patient’s index date were identified and counts that were within four weeks of an AECOPD or prescribed oral corticosteroid were not included in order to identify stable eosinophil measurements. The last absolute blood eosinophil count prior to the patient’s index date was used to group patients into high and low blood eosinophil counts using a cut off of 150 cells/µL.15 A recent study on blood eosinophil counts in COPD patients using CPRD found that eosinophil counts are relatively stable over a period of two years.15
Outcome
The outcome of interest was rate of change of FEV1. At least two FEV1 measurements (measured in milliliters) recorded at least 6 months apart were used in order to analyze the rate of change of FEV1. Since 2004 as part of the Quality and Outcomes framework, FEV1 should be measured every 15 months in COPD patients at their GP and quality of spirometry in primary care is of good quality.16,17
Covariates
Covariates included smoking status, gender, age at index date, BMI, Medical Research Council (MRC) dyspnea score, history of gastro-oesophageal reflux disease (GORD), anxiety, depression myocardial infarction (MI), stroke, heart failure, lung cancer, bronchiectasis, heart failure, white blood cell count, neutrophil count, COPD severity, AECOPD frequency in the first year of follow-up, current or history of asthma (see online supplement for further details).
Statistical analysis
Main analysis (prevalent ICS use)
In order to investigate the relationship between blood eosinophil counts and ICS on rate of change in FEV1 patients were grouped into the following based on baseline exposures: 1) high blood eosinophil counts and prevalent ICS use; 2) high blood eosinophil counts and not on ICS; 3) low blood eosinophil counts and prevalent ICS use; 4) low blood eosinophil counts and not on ICS.
Baseline characteristics were described and compared between the four groups of patients. Mixed-effects linear regression was used due to the repeated measures of FEV1 within patients. The model used two reference categories to evaluate the relationship between ICS and blood eosinophil level (Table 1). All covariates were included in the adjusted models.
 | Table 1 Reference and comparison patient groups |
Incident ICS use
We additionally investigated the relationship between incident ICS-containing medication use, blood eosinophil level, and rate of change in FEV1 using an incident ICS-containing medication cohort. This cohort differed as patients were not censored at 1st ICS-containing medication prescription date. Patients not on ICS-containing medication at baseline were included and grouped into incident ICS-containing medication users or non-ICS-containing medication users based on ICS-containing medication prescriptions in their first year of follow-up. Mixed-effects linear regression was used to investigate the relationship between blood eosinophil counts and incident ICS use on the rate of change in FEV1. Reference and comparison groups were the same (Table 1). All covariates were included in the adjusted models.
Sensitivity and exploratory analyses
Sensitivity analyses included using blood eosinophil cut-offs of 300 and 500 cells/µL, continuous blood eosinophil count, excluding patients with a history of asthma, and restricting the sample to patients with at least 3 FEV1 measurements. Exploratory analyses included stratification by airflow obstruction, AECOPD frequency smoking status, ICS-duration, and type of ICS (see online supplement for further detail).
Power calculation
Sample size calculations were based on the rate of FEV1 change. Table 2 shows the number of patients needed to detect a 2–10 mL/year difference in the rates of change of FEV1 in patients on an ICS and not on an ICS.
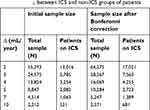 | Table 2 Sample size needed to detect a difference in rates of change of FEV1 between ICS and non-ICS groups of patients |
Results
Baseline characteristics
A total of 26,675 patients were included in this study. Figure 2 illustrates the distribution of patients in each eosinophil/ICS group. Table 3 describes the included population in terms of baseline characteristics, and Table 4 illustrates the proportion of baseline COPD medications used by patients in each ICS use group. The median length of follow-up in this study was 4.2 years (IQR:2.5–6.5), the median number of FEV1 measurements during follow-up was 3 (IQR: 2–5), and the time between FEV1 measurements did not differ between blood eosinophil and ICS group.
 | Table 3 Baseline characteristics for all included patients (N=26,675). Values are n (%) or median (IQR) |
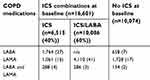 | Table 4 Medication prescribed in the year prior to start of follow-up by baseline ICS use. Values are n (%) |
 | Figure 2 Flow diagram of included patients.Abbreviation: ICS, inhaled corticosteroids. |
Rate of change in FEV1 with prevalent ICS use
The mean adjusted rate of change of FEV1 in those on ICS-containing medication was −12.6 mL/year and −21.1 mL/year in patients not on an ICS-containing medication (P=0.001). Reference classes include women, ex-smokers, no comorbidities, mild airflow obstruction, no AECOPD in first year of follow-up, MRC score of 1, mean BMI (27.1 kg/m2), mean WBC (7,700 cells/µL), and mean neutrophil count (4,600 cells/µL).
Table 5 illustrates the rates of change in FEV1 for each blood eosinophil/ICS group. Irrespective of blood eosinophil level, patients on an ICS-containing medication have slower rates of change in FEV1 compared to those not on an ICS-containing medication.
 | Table 5 Rates of change of FEV1 by blood eosinophil/ICS group in prevalent ICS cohort |
Rate of FEV1 change with incident ICS use
A total of 12,469 patients who were not on an ICS-containing medication at baseline were included in this cohort of which 3,417 newly initiated ICS-containing medication (LABA/ICS 2,604 (76%); ICS 813 (24%)). It is important to note patients prescribed ICS only were on other maintenance therapies including LAMA, SABA, and SAMA. The median number of FEV1 measurements during follow-up was 4 (IQR:3–6) and the time between FEV1 measurements in ICS groups was significantly shorter than the time between FEV1 measurements in non-ICS groups (P<0.001).
The mean adjusted rate of change in FEV1 in patients on newly initiating ICS-containing medication was +4.2 mL/year and −21.2 mL/year in patients not on an ICS-containing medication (P<0.0001). Reference classes include women, ex-smokers, no comorbidities, mild airflow obstruction, no AECOPD in first year of follow-up, MRC score of 1, mean BMI (26.8 kg/m2), mean WBC (8,300 cells/µL), and mean neutrophil count (4,800 cells/µL).
A significant difference was seen between patients with higher blood eosinophils on ICS-containing medication and all other groups (Table 6). Whilst FEV1 declined slower in patients with lower eosinophils on an ICS-containing medication, there was no significant difference between patients with ICS and lower blood eosinophils and those with no ICS-containing medication.
 | Table 6 Rates of change of FEV1 by blood eosinophil/ICS group in incident ICS cohort |
Exploratory analyses results
The rate of change of FEV1 between the four groups was not significantly different when stratified by airflow obstruction (Table S1). When stratified by AEOCPD frequency, the rate of change of FEV1 was slower in ICS-containing groups compared to non-ICS-containing medication groups; however, no significant difference was seen (Table S2). The rate of change of FEV1 was significantly slower in ex-smokers with high blood eosinophils whether on ICS or not. Whilst a similar trend was seen in lower blood eosinophil groups, no significant difference was seen between smokers and ex-smokers (Table S3).
Those on ICS-containing medication continuously for 1–3 months prior to start of follow-up had a lower rate of change of FEV1 (−5.5 mL/year) compared to those on ICS continuously for 4–6 months (−15.8 mL/year) and a significantly lower rate compared to 10–12 months (−17.6 mL/year) in the all-patient analysis. Similar differences were seen after stratification by lower blood eosinophils. A significant difference was seen between patients with higher blood eosinophils on ICS-containing medication continuously for 1–3 months and 10–12 months (Table S4). In terms of drug type, no significant difference in the rate of change of FEV1 was seen between patients on any ICS-containing medication (Table S5).
Sensitivity analyses results
When using blood eosinophil cut offs of 300 and 500 cells/µL, we found that in lower blood eosinophil patients, those on an ICS-containing medication had significantly lower rates of change of FEV1 compared to those not on an ICS-containing medication; however, patient numbers were low in these sub-groups and there was inadequate statistical power for a reliable analysis (Table S6). In addition, continuous eosinophil level was not associated with change in FEV1 (Table S7). There were 17,052 patients in the cohort that excluded a history of asthma. The decline in FEV1 across groups varied from −12.3 mL/year to −21.6 mL/year. The pattern of differences between eosinophil/ICS groups was similar to that in the main analysis, but there was no significant difference was seen between groups (Table S8). When restricted to patients with at least 3 FEV1 measurements, similar rates were observed (Table S9).
Discussion
Main findings
This is the first observational study to investigate the relationship between ICS-containing medication and blood eosinophils on the rate of change of FEV1 over an extended period. We have included COPD patients of different degrees of airflow limitation, who may not have met inclusion criteria for some of the randomized clinical trials, in which FEV1 decline has been previously investigated. We found a difference in the rate of decline of FEV1 between patients on prevalent ICS-containing medication at baseline and those not, regardless of blood eosinophil level. In addition, we found an increase in FEV1 in patients with higher blood eosinophils who initiated ICS-containing treatment. These results suggest that whilst receiving an ICS-containing medication slows down the rate of FEV1 decline in COPD patients, new ICS-users with higher blood eosinophils may benefit more when first started on ICS than those with lower blood eosinophils. Overall, however, our results suggest ICS-containing medication use in COPD patients is associated with lower rate of FEV1 decline irrespective of eosinophil level.
Previous literature
Few observational studies have investigated the relationship between the rate of change of FEV1, ICS-containing medication, and blood eosinophils. In contrast to our main finding, a study in non-asthma COPD patients from the Korean COPD Subtype Study (KOCOSS) cohort found no difference in rate of decline in FEV1 between those with higher or lower blood eosinophils (using 200 cells/µL and 600 cells/µL cut-off), whether on ICS/LABA, or not. Findings from KOCOSS showed that FEV1 increased in patients with higher eosinophils, regardless of being on an ICS or not and in those with lower eosinophils (≤200 cells/µL) FEV1 declined faster in ICS users.12 Differences between cohorts may explain discrepancies between findings. Our study included a larger sample of ex- and current smoking COPD patients from primary care in the UK whereas the KOCOSS included a smaller sample of patients from hospitals in Korea.
Furthermore, in contrast to our main finding, a further study on blood eosinophils and FEV1 over time found that higher blood eosinophils >400 cells/µL were associated with greater FEV1 decline in a relatively small general population.18 No difference in the rate of FEV1 decline in high or low blood eosinophil groups was seen in our study; however, we included older patients and used a different definition of high and low eosinophil level.
Similar to our incident ICS-containing medication findings, a post-hoc analysis of the ISOLDE trial found that COPD patients with higher blood eosinophil levels greater than 2% of their total WBC count who were on an ICS had lower rates of FEV1 decline compared to those on a placebo. No difference was seen between patients with lower blood eosinophil counts on an ICS or a placebo.10 However, a post-hoc analysis of two RCTs found that there was a numerically lower mean improvement in trough FEV1 over one year in patients treated with ICS/LABA in with lower eosinophils, compared to those with higher eosinophils, but the confidence intervals between the two groups overlapped widely.9 Those studies, like the majority of RCTs, included a wash-out period before initiating patients on randomized medication and are therefore similar to our incident cohort design.
Limitations
Despite using a highly sensitive algorithm to identify patients with COPD, misdiagnosis of asthma as COPD and vice versa could not be excluded, notably in patients over the age of forty.14,18 Based on findings from previous work on misclassification of asthma and COPD, we included patients with a history of asthma but undertook a sensitivity analysis in which we excluded those with an asthma diagnosis more than 2 years prior to study start.19 After excluding patients with a history of asthma, no significant differences were seen between groups in the rate of decline in FEV1. This is probably due to a smaller sample size and thus underpowered analysis rather than asthma driving the association between ICS-containing medication and rate of change of FEV1, given we adjusted for history of asthma in all other analyses.
Only COPD patients whose GP practices contribute to CPRD were included in our cohort and patients may not be representative of the true UK population of people with COPD. In addition, included patients had to have at least one blood eosinophil measurement at baseline, which introduces selection bias. Blood tests for COPD patients may have been performed due to reasons other than COPD, such as infections. Whilst research suggests that blood eosinophils are stable over relatively short periods of time, the ECLIPSE study showed that 49% of COPD patients had fluctuating eosinophil counts that varied above and below the cut off over 3 years.15,20 The median study duration was 4.2 years and blood eosinophils were identified 2 years prior to study start. Therefore, it is possible that patients’ blood eosinophil measurements did not represent their eosinophil levels throughout the whole study. However, in order to try to obtain a patient’s stable blood eosinophil measurement using the data available, we excluded blood eosinophil counts that were within four weeks of an AECOPD or prescribed oral corticosteroid.
Spirometry is measured in general practice and not standardized in terms of time of day, machine used, and quality of spirometry or pre or post bronchodilation. In addition, we found that the change in FEV1 was highly variable among patients, as seen by wide 95% confidence intervals. This is consistent with previous research, including RCTs, showing that the rate of FEV1 decline is heterogeneous.21,22 Variation in the rate of FEV1 decline was seen when stratified by severity of COPD whereby patients with milder COPD had faster rates of decline compared to those with more severe COPD. This may be because COPD patients with milder disease have more lung function to lose compared to those with severe COPD with less lung function to start with. In addition, rates of FEV1 change in severe to very severe COPD may be influenced by survival bias. Furthermore, shorter time periods between FEV1 measurements in incident ICS users may explain some of the short-term increase in FEV1 in patients with high eosinophils. It is important to note that these results may not reflect changes in FEV1 over long periods of time.
Furthermore, whilst a strength of this study is that it is an observational study and the population is more generalisable to the wider population of COPD patients, it is important to note that the number and frequency of spirometry measurements available in the data may not have been adequate. The median number of spirometry measurements was 3, and the majority of intervals between measurements within patients were not consistent, despite the introduction of QOF.16,17
In addition, missing data were present for the BMI, MRC, neutrophil count, and degree of airflow obstruction, so we performed complete case analysis for our adjusted models and our results may not be generalizable to the wider COPD population.
It is important to note that whilst we found mainly significant differences between the rates of FEV1 in patients on ICS and patients not on ICS the actual differences ranged from 8 mL/year to 33 mL/year. The American Thoracic Society and the European Respiratory Society propose that minimal clinically important differences in FEV1 between treatment groups range from 100 mL to 140 mL.23 Therefore, it could be argued that the differences seen in this study may not be clinically important. Despite this, our estimates are averaged per year and therefore over a longer period of time these differences may become more apparent. In addition, minimal clinically important differences suggested by the American Thoracic Society and the ERS are based off of pharmacological trials and clinically important differences in a more generalizable population of COPD patients may be less.
Lastly, this study is an observational study so we cannot infer causation. It is also important to note that residual confounding may still exist due to the observational nature such as ICS dosage and mortality during follow-up.
Conclusion
In conclusion, we found that in a large primary care cohort of COPD patients, decline in FEV1 was slower in prevalent ICS-containing medication patients, regardless of blood eosinophil level. Incident ICS-containing medication in patients with higher blood eosinophil levels showed more benefit compared to patients with lower blood eosinophils however, over time this difference was lost. Further, long-term observational studies on the use of ICS-containing medications stratified by eosinophil levels are needed. Research into further possible biomarkers and patient characteristics may help define a subgroup of COPD patients who benefit from ICS-containing medications more than others in terms of FEV1 decline.
Abbreviation list
ICS, Inhaled corticosteroids; EOS, eosinophils; FEV1, Forced expiratory volume in 1 s; LAMA, Long-acting muscarinic antagonist; LABA, long active beta agonist; BMI, Body mass index; MI, Myocardial infarction; HF, Heart failure; GORD, Gastro-oesophageal reflux disease; WBC, White blood cell; AECOPD, Exacerbation of COPD; RCT, Randomized control trial.
Ethics approval
The protocol for this research was approved by the Independent Scientific Advisory Committee (ISAC) for MHRA Database Research (protocol number 17_229R) and the approved protocol was made available to the journal and reviewers during peer review. Generic ethical approval for observational research using the CPRD with approval from ISAC has been granted by a Health Research Authority (HRA) Research Ethics Committee (East Midlands – Derby, REC reference number 05/MRE04/87).
Acknowledgments
This manuscript was funded by GlaxoSmithKline; study number 208602. This study has been previously presented as an abstract at the European Respiratory Society Conference in Paris, September 2018. Steven J Kiddle and Jennifer K Quint are joint last authors for this study.
Author contributions
Hannah R Whittaker: data management, data analysis, data interpretation, writing, figures & tables. Hana Müllerova, Steven J Kiddle, Jennifer K Quint: data analysis, data interpretation, writing. Deborah Jarvis, Neil C Barnes, Paul W Jones, Chris H Compton: data interpretation, writing. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
Miss Whittaker reports grants from GlaxoSmithKline, during the conduct of the study; Dr Müllerova was employed by and reported personal fees from GlaxoSmithKline, during the conduct of the study; Dr Jarvis reports grants from European Union and Cystic Fibrosis Trust, outside the submitted work; she also reports grants from GlaxoSmithKline, during the conduct of the study. Dr Barnes is employed, holds shares and reports personal fees from GlaxoSmithKline, outside the submitted work; he also received grants from GlaxoSmithKline, during the conduct of the study. Dr Jones is employed by and reports personal fees from GlaxoSmithKline, during the conduct of the study; Dr Compton is employed, holds shares and reports personal fees from GlaxoSmithKline, during the conduct of the study; Dr Kiddle reports grants from MRC, during the conduct of the study; grants from Roche Diagnostics, Engineering and Physical Sciences Research Council, Biotechnology and Biological Sciences Research Council, National Institute for Health Research, Alzheimer’s Society, Eli Lily, and Jannsen, outside the submitted work; Dr Kiddle was supported by a MRC Career Development Award (MR/PO21573/1). Dr Quint reports grants from GlaxoSmithKline, during the conduct of the study; grants from The Health Foundation, MRC, Wellcome Trust, British Lung Foundation, Insmed, AstraZeneca, Bayer, Boehringer Ingelheim, Chiesi, Teva, and GlaxoSmithKline, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1.
2. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 . GOLD executive summary report. Am J Respir Crit Care Med. 2017;195(5):557–582. doi:10.1164/rccm.201701-0218PP
3. Fletcher C, Peto R. The natural history of chronic airflow obstruction. Br Med J. 1977;1:1645–1648.
4. Rossi A, Khirani S, Cazzola M. Long-acting β2-agonists (LAB) in chronic obstructive pulmonary disease: efficacy and safety. Int J COPD. 2008;3(4):521–529.
5. Vestbo J, Anderson JA, Brook RD, et al. Fluticasone furoate and vilanterol and survival in chronic obstructive pulmonary disease with heightened cardiovascular risk (SUMMIT): a double-blind randomised controlled trial. Lancet. 2016;387(10030):1817–1826. doi:10.1016/S0140-6736(16)30069-1
6. Vestbo J, Leather D, Diar Bakerly N, et al. Effectiveness of fluticasone furoate-vilanterol for COPD in clinical practice. N Engl J Med. 2016;375(13):1253–1260. doi:10.1056/NEJMoa1608033
7. Suissa S, Patenaude V, Lapi F, Ernst P. Inhaled corticosteroids in COPD and the risk of serious pneumonia. Thorax. 2013;68(11):1029–1036. doi:10.1136/thoraxjnl-2012-202872
8. Yang M, Chen H, Zhang Y, et al. Long-term use of inhaled corticosteroids and risk of upper respiratory tract infection in chronic obstructive pulmonary disease: a meta-analysis. Inhal Toxicol. 2017;29(5):219–226. doi:10.1080/08958378.2017.1346006
9. Pascoe S, Locantore N, Dransfield MT, Barnes NC, Pavord ID. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomised controlled trials. Lancet Respir Med. 2015;3(6):435–442. doi:10.1016/S2213-2600(15)00106-X
10. Barnes NC, Sharma R, Lettis S, Calverley PM. Blood eosinophils as a marker of response to inhaled corticosteroids in COPD. Eur Respir J. 2016;47(5):1374–1382. doi:10.1183/13993003.01370-2015
11. Pavord ID, Lettis S, Locantore N, et al. Blood eosinophils and inhaled corticosteroid/long-acting beta-2 agonist efficacy in COPD. Thorax. 2016;71(2):118–125. doi:10.1136/thoraxjnl-2015-207021
12. Song JH, Lee CH, Kim JW, et al. Clinical implications of blood eosinophil count in patients with non-asthma-COPD overlap syndrome COPD. Int J Chron Obstruct Pulmon Dis. 2017;12:2455–2464. doi:10.2147/COPD.S129321
13. Quint JK, Mullerova H, DiSantostefano RL, et al. Validation of chronic obstructive pulmonary disease recording in the Clinical Practice Research Datalink (CPRD-GOLD). BMJ Open. 2014;4(7):e005540. doi:10.1136/bmjopen-2014-005540
14. Tinkelman DG, Price DB, Nordyke RJ, Halbert RJ. Misdiagnosis of COPD and asthma in primary care patients 40 years of age and over. J Asthma. 2006;43(1):75–80. doi:10.1080/02770900500448738
15. Landis SH, Suruki R, Hilton E, Compton C, Galwey NW. Stability of blood eosinophil count in patients with COPD in the UK clinical practice research datalink. Copd. 2017;14(4):382–388. doi:10.1080/15412555.2017.1313827
16. National Institute for Health and Care Excellence (NICE). Chronic obstructive pulmonary disease in over 16s: diagnosis and management. London: National Institute for Health and Care Excellence Publication; 2018.
17. Rothnie KJ, Chandan JS, Goss HG, Mullerova H, Quint JK. Validity and interpretation of spirometric recordings to diagnose COPD in UK primary care. Int J Chron Obstruct Pulmon Dis. 2017;12:1663–1668. doi:10.2147/COPD.S133891
18. Hancox RJ, Pavord ID, Sears MR. Associations between blood eosinophils and decline in lung function among adults with and without asthma. Eur Respir J. 2018. doi:10.1183/13993003.02536-2017
19. Nissen F, Morales D, Mullerova H, Smeeth L, Quint JK, Douglas I. Misdiagnosis of COPD in asthma patients in the UK electronic health records (Clinical practice research datalink). A59. Epidemiol Airways Chron Lung Dis. 2017;195:A2026–A2026.
20. Singh D, Kolsum U, Brighling CE, Locantore N, Agusti A, Tal-Singer R. Eosinophilic inflammation in COPD: prevalence and clinical characteristics. Eur Respir J. 2014;44:1697–1700. doi:10.1183/09031936.00162414
21. Tashkin DP. Variations in FEV(1) decline over time in chronic obstructive pulmonary disease and its implications. Curr Opin Pulm Med. 2013;19(2):116–124. doi:10.1097/MCP.0b013e32835d8ea4
22. Babu KS, Kastelik JA, Morjaria JB. Inhaled corticosteroids in chronic obstructive pulmonary disease: a pro-con perspective. Br J Clin Pharmacol. 2014;78(2):282–300. doi:10.1111/bcp.12334
23. Jones PW, Beeh KM, Chapman KR, Decramer M, Mahler DA, Wedzicha JA. Minimal clinically important differences in pharmacological trials. Am J Respir Crit Care Med. 2014;189(3):250–255. doi:10.1164/rccm.201310-1863PP. Available from:
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
