Back to Journals » Pharmacogenomics and Personalized Medicine » Volume 11
Influence of ABCB1 and CYP3A5 gene polymorphisms on pharmacokinetics of apixaban in patients with atrial fibrillation and acute stroke
Authors Kryukov AV , Sychev DA, Andreev DA , Ryzhikova KA , Grishina EA, Ryabova AV , Loskutnikov MA, Smirnov VV, Konova OD, Matsneva IA , Bochkov PO
Received 15 November 2017
Accepted for publication 1 February 2018
Published 22 March 2018 Volume 2018:11 Pages 43—49
DOI https://doi.org/10.2147/PGPM.S157111
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Martin H Bluth
Alexander Valerevich Kryukov,1 Dmitry Alekseevich Sychev,1 Denis Anatolevich Andreev,2 Kristina Anatolievna Ryzhikova,1 Elena Anatolievna Grishina,1 Anastasia Vladislavovna Ryabova,1 Mark Alekseevich Loskutnikov,3 Valeriy Valerevich Smirnov,4 Olga Dmitrievna Konova,1 Irina Andreevna Matsneva,2 Pavel Olegovich Bochkov1
1Russian Medical Academy of Continuous Professional Education, Moscow, Russia; 2Department of General Medicine, Sechenov First Moscow State Medical University, Moscow, Russia; 3L.A. Vorohobov City Clinical Hospital, Moscow, Russia; 4NRC Institute of Immunology FMBA of Russia, Moscow, Russia
Introduction: Difficulties in non-vitamin K anticoagulant (NOAC) administration in acute stroke can be associated with changes in pharmacokinetic parameters of NOAC such as biotransformation, distribution, and excretion. Therefore, obtaining data on pharmacokinetics of NOAC and factors that affect it may help develop algorithms for personalized use of this drug class in patients with acute cardioembolic stroke.
Patients and methods: Pharmacokinetics of apixaban in patients with acute stroke was studied earlier by Kryukov et al. The present study enrolled 17 patients with cardioembolic stroke, who received 5 mg of apixaban. In order to evaluate the pharmacokinetic parameters of apixaban, venous blood samples were collected before taking 5 mg of apixaban (point 0) and 1, 2, 3, 4, 10, and 12 hours after drug intake. Blood samples were centrifuged at 3000 rpm for 15 minutes. Separate plasma was aliquoted in Eppendorf tubes and frozen at -70°C until analysis. High-performance liquid chromatography mass spectrometry analysis was used to determine apixaban plasma concentration. Genotyping was performed by real-time polymerase chain reaction. CYP3A isoenzyme group activity was evaluated by determining urinary concentration of endogenous substrate of the enzyme and its metabolite (6-β-hydroxycortisol to cortisol ratio). Statistical analysis was performed using SPSS Statistics version 20.0. The protocol of this study was reviewed and approved by the ethics committee; patients or their representatives signed an informed consent.
Results: ABCB1 (rs1045642 and rs4148738) gene polymorphisms do not affect the pharmacokinetics of apixaban as well as CYP3A5 (rs776746) gene polymorphisms. Apixaban pharmacokinetics in groups with different genotypes did not differ statistically significantly. Correlation analysis showed no statistically significant relationship between pharmacokinetic parameters of apixaban and the metabolic activity of CYP3A.
Conclusion: Questions such as depending on genotyping results for apixaban dosing and implementation of express genotyping in clinical practice remain open for NOACs. Large population studies are required to clarify the clinical significance of genotyping for this drug class.
Keywords: cardioembolic stroke, atrial fibrillation, non-vitamin K anticoagulants, apixaban, pharmacokinetics, pharmacogenetics
Rivaroxaban, apixaban, and edoxaban are oral anticoagulant drugs, which act as direct factor Xa inhibitors.
Stroke prevention is one of the most important indications for non-vitamin K anticoagulants (NOACs) in patients with atrial fibrillation (AF). The use of anticoagulants and the resumption of their administration after stroke are documented in 2016 European Society of Cardiology Guidelines for the Management of Atrial Fibrillation.1 Anticoagulants are administered depending on the severity of stroke, which is evaluated by the National Institutes of Health Stroke Scale (NIHSS): for patients with a score of <8, anticoagulants are prescribed 3 days after the stroke, with a score of 8–16 after 6 days, and a score of >16 after 12 days in the absence of contraindications.2
Difficulties in NOAC administration in the acute period of stroke can be associated with possible change in the pharmacokinetics of these drugs caused by changes in absorption processes, biotransformation, distribution, and excretion in this category of patients under the influence of various factors.
Thus, obtaining data on the pharmacokinetics of NOACs and the factors that affect it in patients with acute cardioembolic stroke helps develop algorithms for personalizing the choice and dosage of this group of drugs. Pharmacokinetics of apixaban in patients with acute stroke was studied earlier in the study by Kryukov et al.3
Apixaban is predominantly metabolized via CYP3A4/5 isoenzyme and to a lesser extent via isoenzymes CYP1A2, 2C8, 2C9, 2C19, and 2J2. In addition, apixaban is a substrate for transport proteins, P-glycoprotein, and breast cancer resistance protein (BCRP).4
Rivaroxaban is metabolized via isoenzymes CYP3A4 and CYP2J2 and by means of mechanisms independent of the cytochrome system. Rivaroxaban is also a substrate for P-glycoprotein and BCRP carrier proteins.5
Edoxaban is metabolized via isoenzymes CYP3A4 and carboxylesterase 1 (CES1). Edoxaban is a substrate for P-glycoprotein and organic anion transporter protein 1B1 (OATP1B1).6
Nowadays, there is a growing body of literature showing significance of genetic characteristics of patients with respect to metabolism and the distribution of NOACs.7–9
The first aim of the study was to determine the relationship between apixaban pharmacokinetics and ABCB1 gene polymorphisms (rs1045642, rs4148738), encoding P-glycoprotein and CYP3A5 gene polymorphisms (rs776746). The second aim of the study was to evaluate the association between CYP3A (6-β-hydroxycortisol/cortisol) activity and CYP3A5 genotype. Candidate genes were chosen according to the available data on the metabolism of apixaban. The role of other enzymes for apixaban has not been confirmed.
Patients and methods
Research design and participants
The clinical part of the study was conducted at L.A. Vorokhobov State Clinical Hospital, Department of Health, Moscow.
The analysis of apixaban’s pharmacokinetic parameters was carried out at the Research Center of the Russian Medical Academy of Postgraduate Education of the Ministry of Health of the Russian Federation. This study was approved by the ethics committee of the Russian Medical Academy of Postgraduate Education of the Ministry of Health of the Russian Federation. All patients or their representatives signed written informed consent to participate in the study.
All procedures were carried out according to the National legislation (National Standard of the Russian Federation GOST R 52379-2005 “Good Clinical Practice”), Good Clinical Practice (GCP), and international ethical norms (Helsinki Declaration of the World Medical Association, the last changes were made on 64th General Assembly of the WMA, Fortaleza, Brazil, October 2013).
Inclusion criteria were as follows: confirmed diagnoses of acute ischemic stroke and nonvalvular AF (absence of artificial heart valves and hemodynamically significant mitral stenosis). The diagnosis was made by the attending physicians by the clinical picture and the results of the examination (computed tomography [CT] for stroke and electrocardiogram [ECG] for atrial fibrillation).
Exclusion criteria were as follows: confirmed diagnosis of valvular AF (presence of artificial heart valves and hemodynamically significant mitral stenosis), hemorrhagic transformation of cerebral infarction according to CT or magnetic resonance imaging (MRI), impaired swallowing, creatinine clearance of <15 mL/min, age ≥90 years, simultaneous use of NOACs and drug groups such as CYP3A4 and P-glycoprotein inhibitors (ketoconazole, dronedarone, amiodarone, verapamil, diltiazem, naproxen, quinidine, ticagrelor, and clarithromycin), CYP3A4 and P-glycoprotein inductors (rifampicin, carbamazepine, phenobarbital, phenytoin, and St. John’s Wort), pantoprazole, and atenolol. Patients were also excluded in the case of contraindications to apixaban. The study enrolled 17 patients with cardioembolic stroke: 3 males and 14 females, mean age 76.6 ± 9.5 years. Patients had a high recurrent stroke risk; the average risk scale of thromboembolic complications for patients with AF (CHA2DS2-VASc) score was 5.5. Risk of bleeding with anticoagulant treatments was also high; the average HAS-BLED score was 3. Patients did not take anticoagulants before stroke.
Opinions on the minimum number of participants included in a pharmacokinetic study differ (US FDA Guidance for Industry: Statistical Approaches to Establishing Bioequivalence, 2001;10 Guidelines for the Evaluation of Bioequivalence of Medicines, 2008;19 and EMEA (Europe, the Middle East and Africa) Guidelines on the Investigation of Bioequivalence, 2010).11 The sample size is determined by the pharmacokinetic design of the study, which traditionally includes a small number of participants. Also, a significant role is played by the severity of the condition of patients enrolled in the study and their age, which together pose technical difficulties in obtaining blood samples.
Renal function measured by the Modification of Diet in Renal Disease (MDRD) was reduced; mean glomerular filtration rate (GFR) was 55 mL/min/1.72 m2, which corresponds to Stage II chronic kidney disease. Hemostasis parameters during hospitalization did not deviate from the reference values (Table 1). To prevent secondary thromboembolic complications, all patients, depending on the severity of the stroke, received an oral anticoagulant apixaban at a dose of 5 mg two times a day. On average, apixaban was prescribed 8 days after the stroke. To determine the pharmacokinetic parameters of the apixaban, blood samples were taken on the first day of drug administration.
The average hospital stay was 14.5 days. A total of 3 patients included in the study experienced a fatal outcome.
In all cases, death was caused due to the consequences of acute cerebrovascular accident.
Methods
Determination of apixaban pharmacokinetic parameters
To determine the concentration of the drug, venous blood was sampled into vacuum tubes with an EDTA-K3 Improvacuter (Guangzhou Improve Medical Instruments Co Ltd, Guangzhou, China) with a capacity of 6 mL. Venous blood sampling was performed before taking 5 mg of apixaban (point 0) and 1, 2, 3, 4, 10, and 12 hours after drug intake. To obtain plasma, blood samples were centrifuged at 3000 rpm for 15 minutes. Separated plasma was aliquoted into Eppendorf tubes and frozen at -70°C until analysis was performed.
Plasma concentration of apixaban was measured by chromatography–mass spectrometric analysis using a high-performance liquid chromatograph Agilent G1978B Multimode Source for 6410 Triple Quade LC/MS (Agilent Technologies, Santa Clara, CA, USA).
Quantitative determination of apixaban and its deuterated analog apixaban-d3 (used as an internal standard) in human plasma samples was performed by high-performance liquid chromatography–tandem mass spectrometry. Sample preparation was carried out by precipitation of plasma proteins with acetonitrile.
Separation of the compounds was carried out using the ACQUITY UPLC BEN C18 (2.1 mm × 50 mm, 1.7 μm; Waters Corporation, Milford, MA, USA) chromatography column in a gradient elution mode at a flow rate of 0.4 mL/min. The retention times were 0.55 minutes for apixaban and 0.56 minutes for apixaban-d3. Total analysis time was 2.0 minutes.
The developed method satisfies acceptance criteria, and thus it can be used for quantitative determination of apixaban in blood plasma samples in a pharmacokinetic study.
Pharmacokinetic parameters (maximum plasma concentration [Cmax], time to maximum plasma concentration [Tmax], area under the curve “concentration–time” to infinity [AUC(0, ∞)], area under the curve “concentration–time” to the last measurement [AUC(0, τ)], and half-life [t1/2]) were determined using the WinNonlin® 5.2 software (Pharsight Corporation, St Louis, MO, USA).
Determination of ABCB1 and CYP3A5 polymorphisms
DNA was extracted from whole blood using CJSC Syntol kits (Moscow, Russian Federation). The carriership of genes ABCB1 (rs1045642) and CYP3A5 (rs776746) was determined by real-time polymerase chain reaction (real-time PCR) using kits SNP-Screen of CJSC Syntol. The program included preliminary denaturation at 95°C, which lasted 3 minutes, 40 cycles of 15 seconds denaturation at 95°C, and then annealing at 60°C for 40 seconds. The carriership of gene ABCB1 (rs4148738) was determined by real-time PCR using commercial kits TaqMan®SNP Genotyping Assays of Thermo Fisher Scientific (Waltham, MA, USA) and Q5® HF DNA Polymerase of New England Biolabs (Ipswich, MA, USA). The program included preliminary denaturation at 95°C, which lasted 2 minutes, 39 cycles of 15 seconds denaturation at 95°C, and then annealing at 56°C for 1 minute. Genotype polymorphisms were detected using Real-Time CFX96 Touch (Bio-Rad Laboratories Inc., Hercules, CA, USA).
Measurement of CYP3A metabolic activity
CYP3A isoenzyme group activity was evaluated by measuring urinary concentration of endogenous substrate of the enzyme and its metabolite (6-β-hydroxycortisol to cortisol ratio). Concentrations of cortisol and 6-β-hydroxycortisol were measured by high-performance liquid chromatography using the Agilent G1978B Multimode Source for 6410 Triple Quade LC/MS (Agilent Technologies, 2008). CYP3A metabolic activity was evaluated by urinary 6-OHC/FC ratio. Low 6-β-hydroxycortisol/cortisol ratios correspond to low CYP3A activity, and high 6-β-hydroxycortisol/cortisol ratios correspond to high activity of CYP3A.
Statistical processing of the results
All statistical analyses were performed using SPSS Statistics 20.0 (IBM Corporation, Armonk, NY, USA). For Cmax, AUC(0, ∞), and AUC(0, τ), geometric mean and coefficient of variation (CV%) were determined. Tmax was described using the median, maximum, and minimum values, for t1/2 the mean (M) and standard deviation (SD) were determined. To assess the differences between the groups, Mann–Whitney and Kruskal–Wallis criteria were used. The results were considered statistically significant at p < 0.05.
Results
Influence of ABCB1 (rs1045642, rs4148738) and CYP3A5 (rs776746) gene polymorphisms on pharmacokinetics of apixaban
Pharmacogenetics testing showed that ABCB1 (rs1045642) genotype distribution was as follows: 5 patients with wild genotype CC, 9 heterozygotes with a CT genotype, and 3 patients with a TT genotype. The distribution of these genotypes was in agreement with the Hardy–Weinberg equilibrium (p=0.76).
Apixaban pharmacokinetics did not differ significantly in groups with different ABCB1 (rs1045642) genotypes (Table 2).
Pharmacogenetics testing showed that ABCB1 (rs4148738) genotype distribution was as follows: 4 patients with wild genotype CC, 7 heterozygotes with a CT genotype, and 6 patients with a TT genotype. The distribution of these genotypes was in agreement with the Hardy–Weinberg equilibrium (p=0.5).
Apixaban pharmacokinetics did not differ significantly in groups with different ABCB1 (rs4148738) genotypes (Table 3).
Pharmacogenetic testing showed that CYP3A5 (rs776746) genotype distribution was as follows: 14 homozygotes with the GG genotype and 3 heterozygotes with the AG genotype. The distribution of these genotypes was in agreement with the Hardy–Weinberg equilibrium (p=0.69).
Apixaban pharmacokinetics did not differ significantly in groups with different CYP3A5 (rs776746) genotypes (Table 4).
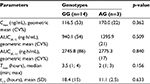
Influence of CYP3A5 (rs776746) genotype on CYP3A metabolic activity
CYP3A metabolic activity was determined in each group of patients with different CYP3A5 (rs776746) genotypes. Differences in CYP3A activity in the groups did not reach statistical significance (Table 5).
  | Table 5 CYP3A metabolic activity depending on CYP3A5 (rs776746) genotypes Abbreviation: SD, standard deviation. |
Pharmacokinetics is more sustained in the AG group according to CV%, but only 3 patients were included in this group. As to activity of CYP3A, the AG group showed a wide range of SDs, and it might be due to high activity of one patient’s enzymes.
Intergroup comparison revealed that the ratio of 6β-hydroxy cortisol/cortisol was 2.94 for the group with the GG genotype and 8.87 for the group with the AG genotype, respectively.
Correlation analysis showed no statistically significant relationship between pharmacokinetic parameters of apixaban and metabolic activity of CYP3A.
Discussion
In the study by Dimatteo et al,6 where the influence of ABCB1 (gene encoding P-glycoprotein) rs4148738 SNP on apixaban metabolism was studied, it was shown that apixaban peak equilibrium concentration in patients with wild genotype was significantly lower. However, there was an increased apixaban peak equilibrium concentration in patients with ABCB1 polymophism.7
Ing Lorenzini et al8 described the case of gastrointestinal bleeding associated with rivaroxaban in the patient–carrier of allelic variants of both studied ABCB1 polymorphisms (rs2032582 and rs1045642). In this example, high levels of anti-Xa activity and rivaroxaban concentration were found despite the withdrawal of the drug; t1/2 was 2–3 times higher than that indicated in the manual.
Gouin-Thibault et al12 showed no significant influence of rs2032582 and rs1045642 ABCB1 polymorphisms on rivaroxaban pharmacokinetics: the AUC ratio between carriers of allelic variants and wild genotype was 1.20 (95% CI: 0.96–1.51).
In the study of Vandell et al,9 the effect of allelic variants of the ABCB1 (rs1045642) and SLCO1B1 (rs4149056) genes (genes encoding P-glycoprotein and OATP1B1) on the pharmacokinetics of edoxaban was studied. Edoxaban is a substrate for these transporters. It was found that allelic variants of ABCB1 and SLCO1B1 genes do not affect the edoxaban pharmacokinetics.
However, in SLCO1B1 allelic variant carriers, an increase in the concentration of the edoxaban metabolite was observed. It was not clinically significant since the concentration of the metabolite is 10% of the concentration of the basic substance.
The data obtained in this study on the activity of CYP3A correlate with the literature data. In the study by Chan et al,13 where the pharmacokinetics of midazolam was evaluated in patients with different CYP3A5 genotypes (rs776746), no statistically significant difference was found between the groups.
However, in other studies, the use of CYP3A phenotyping (determination of 6b-hydroxycortisol/cortisol ratio in morning urine) has shown significant changes in enzyme activity depending on characteristics of patient and presence of CYP3A inhibitors.14–18
The limitations of the study include a limited number of polymorphisms included, small sample size, and variability of pharmacokinetic parameters. The most likely cause is the difference in characteristics of patient. The pharmacokinetic parameters of apixaban were obtained in real clinical practice. Peculiarities of pharmacokinetics are due to severity of the underlying disease, the concomitant pathology, and age of the patients; all these are reflected by CV% variability.3 However, this reflects the actual clinical practice and the portrait of the average patient with a stroke; practicing doctors have to deal with this. Steady state concentration was not studied, but it could be more relevant for a pharmacogenetic study. The study reflected mostly an acute situation with its features and problems. Regular monitoring of the anticoagulant effect of apixaban in such patients may be considerable.
Conclusion
In this study, the genetic characteristics of patients with stroke (ABCB1 [gene encoding P-glycoprotein] rs1045642, rs4148738 polymorphisms and CYP3A5 [gene encoding enzyme that metabolizes apixaban] rs776746 polymorphisms) did not affect pharmacokinetics of apixaban.
Questions such as depending on genotyping results for apixaban dosing and implementation of express genotyping in clinical practice remain open for NOACs. Large population studies are required to clarify the clinical significance of genotyping for this drug group.
Acknowledgment
This study was supported by the Russian Science Foundation, project 16-15-00227 “Fundamental and exploratory researches in priority thematic research areas”.
Disclosure
The authors report no conflicts of interest in this work.
References
Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893–2962. | ||
Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893–2962. | ||
Kryukov AV, Sychev DA, Andreev DA, et al. The pharmacokinetics of apixaban in patients with cardioembolic stroke in acute phase. Ration Pharmacother Cardiol. 2016;12(3):253–259. | ||
ELIQUIS® (apixaban) [prescribing information]. Princeton, NJ: Bristol-Myers Squibb Company; 2017. | ||
XARELTO® (rivaroxaban) [prescribing information]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2014. | ||
SAVAYSA (edoxaban) [prescribing information]. Tokyo: Daiichi Sankyo Co., LTD.; 2015. | ||
Dimatteo C, D’Andrea G, Vecchione G, et al. ABCB1 SNP rs4148738 modulation of apixaban interindividual variability. Thromb Res. 2016;145:24–26. | ||
Ing Lorenzini K, Daali Y, Fontana P, Desmeules J, Samer C. Rivaroxaban-induced hemorrhage associated with ABCB1 genetic defect. Front Pharmacol. 2016;7:494. | ||
Vandell AG, Lee J, Shi M, Rubets I, Brown KS, Walker JR. An integrated pharmacokinetic/pharmacogenomic analysis of ABCB1 and SLCO1B1 polymorphisms on edoxaban exposure. Pharmacogenomics J. 2016;18(1):153–159. | ||
US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Guidance for Industry, Statistical Approaches to Establishing Bioequivalence. Silver Spring, MD: US Food and Drug Administration; 2001. Available from: https://www.fda.gov/downloads/Drugs/Guidances/ucm070244. Accessed March 8, 2018. | ||
European Medicines Agency. Guideline on the Investigation of Bioequivalence. London: EMEA; 2010. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2010/01/WC500070039.pdf. Accessed March 8, 2018. | ||
Gouin-Thibault I, Delavenne X, Blanchard A, et al. Inter-individual variability in dabigatran and rivaroxaban exposure: contribution of ABCB1 genetic polymorphisms and interaction with clarithromycin. J Thromb Haemost. 2016;15(2):273–283. | ||
Chan SW, Xiao Y, Hu M, et al. Associations of the CYP3A5*3 and CYP3A4*1G polymorphisms with the pharmacokinetics of oral midazolam and the urinary 6 b-hydroxycortisol/cortisol ratio as markers of CYP3A activity in healthy male Chinese. J Clin Pharm Ther. 2016;41(5):552–558. | ||
Dutreix C, Lorenzo S, Wang Y. Comparison of two endogenous biomarkers of CYP3A4 activity in a drug – drug interaction study between midostaurin and rifampicin. Eur J Clin Pharmacol. 2014;70(8):915–920. | ||
Eldesoky ES, Kamel SI, Farghaly AM, Bakheet MY, Hedaya MA, Siest J-P. Study of the urinary ratio of 6 beta-hydroxycortisol/cortisol as a biomarker of CYP3A4 activity in Egyptian patients with chronic liver diseases. Biomark Insights. 2007;1:157–164. | ||
Kawaguchi A, Ohmori M, Tsuruoka S, et al. Drug interaction between St John’s Wort and quazepam. Br J Clin Pharmacol. 2004;58(4):403–410. | ||
Konishi H, Tanaka K, Minouchi T, Yamaji A. Urinary 6beta-hydroxycortisol/17-hydroxycorticosteroids ratio as a measure of hepatic CYP3A4 capacity after enzyme induction. Ann Clin Biochem. 2004;41(pt 4):335–337. | ||
Zheng W, Jin F, Dunning LA, et al. Epidemiological study of urinary 6beta-hydroxycortisol to cortisol ratios and breast cancer risk. Cancer Epidemiol Biomarkers Prev. 2001;10(3):237–242. | ||
Ministry of Health and Social Development of Russian Federation. Guidelines for the Evaluation of Bioequivalence of Medicines. Moscow: Ministry of Health and Social Development of Russian Federation; 2008. Available from: http://dissolutiontech.ru/assets/files/be-2008.pdf. Accessed March 12, 2018. Russian. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.