Back to Journals » International Journal of General Medicine » Volume 7
Incidence rate of non-Hodgkin’s lymphomas among males in Saudi Arabia: an observational descriptive epidemiological analysis of data from the Saudi Cancer Registry, 2001–2008
Authors Alghamdi I , Hussain I, Alghamdi M, Dohal A, Alghamdi M , El-Sheemy M
Received 6 March 2014
Accepted for publication 5 April 2014
Published 25 June 2014 Volume 2014:7 Pages 311—317
DOI https://doi.org/10.2147/IJGM.S63645
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Ibrahim G Alghamdi,1,2 Issam I Hussain,1 Mohamed S Alghamdi,3 Ahlam A Dohal,4 Mansour M Alghamdi,4 Mohammed A El-Sheemy5
1School of Life Sciences, University of Lincoln, Lincoln, UK; 2Albaha University, Al Baha city, Saudi Arabia; 3General Directorate of Health Affairs, Ministry of Health, Al Baha, 4King Fahad Specialist Hospital, Dammam, Saudi Arabia; 5Research and Development, Lincoln Hospital, United Lincolnshire Hospitals NHSTrust, Lincoln, UK
Background: This study describes epidemiological data of non-Hodgkin’s lymphoma (NHL) diagnosed from 2001 to 2008 among Saudi men.
Materials and methods: Retrospective data from all NHL cancer cases among Saudi men recorded in the Saudi Cancer Registry (SCR) between January 2001 and December 2008 were used. Descriptive statistics, analysis of variance, Poisson regression, and simple linear regression were also used.
Results: In total, 2,555 new cases of NHL were recorded between January 2001 and December 2008. The region of Riyadh, Saudi Arabia had the highest overall age-standardized incidence rate (ASIR) at 7.8, followed by the Eastern region at 6.8, and Makkah at 6.1 per 100,000 men; however, Jazan, Hail, and Baha had the lowest average ASIRs at 2.5, 3.7, and 3.9 per 100,000 men, respectively. The incidence-rate ratio for the number of NHL cases was significantly higher in Riyadh (4.68, 95% confidence interval [CI] 4.11–5.32), followed by Makkah (4.47, 95% CI 3.94–5.07), and the Eastern region of Saudi Arabia (3.27, 95% CI 2.90–3.69) than that in the reference region of Jazan. Jouf had the highest changes in the ASIRs of NHL among Saudi men from 2001 and 2008 (5.0 per 100,000 men).
Conclusion: A significant increase in the crude incidence rate and ASIR for NHL in Saudi Arabia between 2001 and 2008 was found. Riyadh, the Eastern region, and Makkah had the highest overall ASIR in Saudi Arabia. Jazan, Hail, and Baha had the lowest rates. Additionally, Riyadh, Makkah, and the Eastern region had the highest incidence-rate ratio for the number of NHL cases. Finally, Jouf had the highest changes in crude incidence rate and ASIR from 2001 to 2008. Further analytical studies are needed to determine the potential risk factors of NHL among Saudi men.
Keywords: cancer epidemiology, non-Hodgkin’s lymphomas, incidence rate, age standardized incidence rate, Saudi Cancer Registry
Introduction
Non-Hodgkin’s lymphoma (NHL) has been recognized as a cancer that develops extensively in the lymphatic tissues (the immune system), and can be spread to other parts of the body through the bloodstream, making secondary tumours.1,2 Patients can acquire aggressive NHL (fast-growing) or indolent (slow-growing) lymphoma that can be formed from B cells or T cells.3 According to research conducted in the UK in 2008, of the 27 countries in the European Union, Finland had the highest NHL mortality rates for both men and women, at approximately five and seven cases per 100,000 persons, respectively.4 In addition, the American Cancer Society predicted that 37,600 men would be diagnosed with NHL in the US, with approximately 10,590 subsequent deaths in the total population.5
In 2008, NHL was one of the most prevalent types of cancer in Saudi Arabia, and ranked second in cancer incidence among the male population, with a ratio of 122:100 for men to women.6 The International Agency for Research on Cancer estimated that the age-standardized incidence rate (ASIR) for NHL was 6.5 per 100,000 men in 2012, and the age-standardized mortality rate (ASMR) was 4.3 per 100,000 men.7 Furthermore, the registry of King Faisal Specialist Hospital and Research Centre in Saudi Arabia recorded 5,493 cases (7.6%) of NHL with admission to the hospital from 1975 to 2011.8 In Saudi Arabia, the ASIR of NHL is higher than that in the other Arabian Gulf countries. For example, in 2012, the reported ASIRs for Oman, Qatar, the United Arab Emirates, and Bahrain were 6.3, 5.9, 5.8, and 4.7 per 100,000 men, respectively.7 In spite of insufficient data on the geographical distribution of NHL among males in Saudi Arabia, we intended to examine the crude incidence rate (CIR) and ASIR of NHL cancer cases by conducting an observational descriptive epidemiological study of the disease while considering spatial/temporal distribution of recorded cases in the Saudi Cancer Registry (SCR) during 2001 to 2008.9
Materials and methods
We conducted a retrospective, descriptive, epidemiological study of all Saudi men with NHL cancer diagnosed between January 2001 and December 2008. The data for this study are managed by the SCR, which is a population-based registry that was established in 1994 through the Ministry of Health in Saudi Arabia. However, no data were available from 1994 to 2000, and the most recent available data set from the SCR was in 2008.
Since 2001, the SCR has been reporting on the pattern of cancer in Saudi Arabia with the primary objective of defining the population-based incidence of the disease. According to these data, comprehensive reports for 13 administrative regions from 2001 to 2008 currently exist that report the percentage of cases, CIR, and ASIR, adjusted for the provinces of Saudi Arabia, age of cases, and the year of diagnosis. Data from all of these reports were gathered from the SCR with the aim of critically analyzing the descriptive epidemiology of NHL among Saudi Arabian men.
For all data analyses, we used SPSS version 20.0 (IBM, Armonk, NY, USA). The descriptive analysis was performed by calculating the overall percentage, CIR, and ASIR after adjusting for age-group, region, and year of diagnosis. The percentage was calculated by dividing the total number of incident NHL cases by the total number of male cancer cases. Furthermore, analysis of variance (ANOVA) was performed to determine if the incidence of NHL varied significantly among the regions and years of diagnosis.10 In addition, linear regression models were applied to investigate the strength of the association between the independent and dependent variable to predict the annual CIR and ASIR for NHL among Saudi men.11 Poisson regression models were also conducted to calculate the incidence rate ratio (IRR) for the number of NHL cases in each region of Saudi Arabia.
The overall ASIRs are reported as per 100,000 men, and were calculated for all of the SCR datasets based on the Segi world standard12,13 for intermediate-age populations (Table 1). The Segi world standard can be used as a direct method of standardization to calculate the ASIR of NHL cancer. It allows for control of the confounding effects of age when different populations are compared across age-groups.12,13
Results
A total of 2,555 cases of NHL were recorded in the SCR between January 2001 and December 2008. The frequency of cases with NHL increased slightly from 2001 to 2008. We identified 230 cases in 2001 (8%, 95% confidence interval [CI] 6.0%–10.0%) and 332 by 2004 (9.5%, 95% CI 7.5–11.5%), representing a 1.5% increase. This was the highest percentage stated by the SCR (2004), but not statistically significant (P>0.05). From 2005 to 2008, the number of NHL cases remained slightly elevated, and fluctuated between 9.3% and 8.7%, respectively (Figure 1A and B).
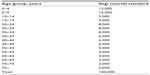
The average number and percentage of NHL cases between 2001 and 2008, adjusted by age-group, was calculated from the SCR (Table 2, Figure 1C and D). The following age-groups were reported: 0–14, 15–29, 30–44, 45–59, 60–74, and older than 75 years. According to the frequency of NHL cases from 2001 to 2008, older men aged 64–74 years had the highest average number of NHL cases (78), representing 25% per year. Those aged 30–44 and 45–59 years had the next-highest average number of cases, with 60, representing 19% of the total number of NHL cases. Additionally, the younger age-groups aged 0–14 and 15–29 years had the lowest number of cases, with less than 44 in total for both groups, representing 11% and 14% of the total number of NHL cases, respectively.
  | Table 2 Non-Hodgkin’s lymphoma case distribution by age-group in male Saudis from 2001 to 2008 |
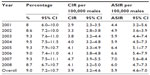
The CIRs were adjusted by the year of diagnosis and calculated for the 2001–2008 data sets. A steady increase of CIRs for NHL was noted as time increased. A CIR of 2.9 (95% CI 2.3–3.5) and 4.1 (95% CI 3.2–5.0) per 100,000 men was estimated in 2001 and 2008, respectively. In 2007, the CIR of 4.7 (95% CI 3.9–5.5) per 100,000 men was significantly higher than that of other years (F7,96=2.34, P<0.05) (Table 3 and Figure 2A). According to the linear regression analysis, the equation to predict the annual increase of CIR for NHL cancer among Saudi men was 2.54 + (0.18× years), and the CIR of NHL increased by a predicted average of 0.18 per 100,000 Saudi men per year.
Table 4 and Figure 2B show data from 2001 to 2008 for the overall CIR per 100,000 men with NHL adjusted for the region of Saudi Arabia. Riyadh had the highest overall CIR for NHL, at 4.6 (95% CI 4.0–5.2) per 100,000 men, followed by Makkah at 4.1 (95% CI 3.3–5.0), and the Eastern region at 3.8 (95% CI 3.1–4.3). Therefore, the results of the ANOVA tests revealed that the incidence rates of NHL were significantly higher for these regions than those of other regions of Saudi Arabia (F12,91=2.891, P<0.001). Furthermore, Jazan and Hail had the lowest average CIRs for NHL among Saudi men, at 2.0 (95% CI 1.4–2.6) and 2.9 (95% CI 1.8–4.0), respectively.
Table 3 and Figure 2C show the ASIR of NHL, adjusted for the year of diagnosis from 2001 to 2008 per 100,000 men. There was a slight increase in the ASIR of NHL diagnosed between 2001 and 2008; the highest ASIR was 7.0 (95% CI 5.6–8.4), recorded in 2007 according to reports released by the SCR. However, the linear regression equation to predict the annual ASIR of NHL cancer among Saudi men was 4.58 + (0.13× years), and the ASIR of NHL increased by a predicted average of 0.13 per 100,000 Saudi men per year. Similarly, Table 4 and Figure 2D indicate that the region of Riyadh had the highest overall ASIR for NHL cancer, at 7.8 (95% CI 6.7–8.9) per 100,000 men, followed by the Eastern region at 6.8 (95% CI 5.8–7.7) and Makkah at 6.1 (95% CI 4.9–7.2). The results of the ANOVA tests indicated that the ASIRs of NHL were significantly higher for these regions than those in other provinces of Saudi Arabia (F12,91=4.532, P<0.001). Alternatively, Jazan, Hail, and Baha had the lowest overall ASIRs for NHL among Saudi men, at 2.5 (95% CI 1.9–3.1), 3.7 (95% CI 2.3–5.2), and 3.9 (95% CI 2.6–5.3), respectively.
The results of the Poisson regression model used to calculate the IRR of NHL cases according to the region of Saudi Arabia are presented in Table 5; the model was statistically significant (likelihood ratio χ213=2823.8, P<0.001). Jazan was used as the reference for all comparisons between other provinces of Saudi Arabia. The IRRs were significantly higher in Riyadh, at 4.68 (95% CI 4.11–5.32), followed by Makkah at 4.47 (95% CI 3.94–5.07) and the Eastern region at 3.27 (95% CI 2.90–3.69). Furthermore, the IRR of NHL cases significantly increased by 1% for every unit increase per year.
Finally, differences among the CIR and ASIR between 2001 and 2008 were calculated. The pattern of NHL among Saudi men according to their region is shown in Table 6. The greatest change in ASIR was observed in the Jouf region, at 5.0 per 100,000 men. However, the CIR and ASIR for all regions of Saudi Arabia fell below the predicted values, based on the linear regression equations listed above.
Discussion
The prevention of NHL is an important issue among the Saudi Arabian population. Epidemiological evidence based on prospective cohort and case-control studies shows that modifiable risk factors, including obesity, lack of physical activity, high fat intake, low fish intake and high consumption of meat, are positively linked with NHL.14 Furthermore, there are nonmodifiable risk factors that may increase the chance of getting NHL. The age of persons older than 60 years is positively associated with NHL. The risk of NHL is higher in men compared to women with unknown causes. In addition, race and ethnicity may affect the occurrence of the NHL. In the US, white Americans are more likely to get NHL compared with African and Asian Americans.
In this study, we reported the CIR and ASIR of NHL among men in Saudi Arabia from 2001 to 2008 to describe the epidemiology of NHL among men according to their region. In addition, this study clarifies the recent trends of NHL in Saudi Arabia and affirms the significance of the disease. In 2012, the ASIRs and ASMRs of NHL cancer per 100,000 men were reported in different countries by the International Agency for Research on Cancer. According to the international comparison of NHL cancer rates with Saudi Arabia in Table 7, the ASIRs of NHL cancer were higher in the US, Australia, Ireland, and the UK, at 14.7, 14.4, 12.5, and 11.9 cases per 100,000 men, respectively, while India, the People’s Republic of China, and Bahrain recorded the lowest rates, at 3, 3.2, and 4.7 cases per 100,000 men, respectively. Furthermore, the highest ASIRs in Arab Gulf countries were estimated in Kuwait, at 7.7 cases followed by Saudi Arabia and Oman at 6.5 and 6.2 per 100,000 men, respectively. Despite the low ASIR of NHL cancer in Saudi Arabia compared to other countries, the ASMR of NHL remained slightly high. For example, the ASMRs of NHL in Australia, Ireland, and the UK were lower than Saudi Arabia, at 3.8, 3.8, and 3.6 cases per 100,000 men, respectively. This means that secondary and tertiary preventions, including early diagnosis, good treatment, and rehabilitation programs, were not adequate for cases in Saudi Arabia. However, the ASMRs in Arab Gulf countries were very similar to each other.
The results of our study indicate that the region of Riyadh, the Eastern region, and Makah have the highest overall ASIRs for NHL among Saudi men. However, the overall ASIRs of NHL for these regions have not increased above the expected levels from 2001 to 2008. Conversely, the regions of Jazan, Hail, and Baha had the lowest overall ASIRs of NHL among men in Saudi Arabia for the same period. However, factors that include socioeconomic status, improvement in technology, and awareness among population may influence the burden of cancer and its incidence rate. The more technologically developed and affluent areas exhibited higher incidence of NHL. This suggests that people in higher socioeconomic areas receive treatment more often, resulting in more cases being diagnosed and reported, than in areas with lower socioeconomic status, where the numbers may be underestimated due to undetected cases. However, we suspect that the false impact of socioeconomic status on the incidence rate of cancer can only be observed in different regions during the short studied period (months), and that with longer surveyed time (years), all cases should eventually be detected, resulting in a more accurate reported incidence rate. The period of time from case diagnosis may be considered a potential bias for studying the real effect of socioeconomic status on cancer. However, it is challenging to study the relationship between socioeconomic status and incidence rate of NHL, because the SCR does not collect data on socioeconomic status for different regions in Saudi Arabia.
Our Poisson regression model indicates that the IRRs of NHL were significantly higher in Riyadh, followed by Makkah and the Eastern region, than that of Jazan (reference region). However, the most affected age-groups in all of the regions of Saudi Arabia were men older than 60 years.
We also observed changes in the percentage, CIR, and ASIR of NHL among Saudi men according to their region. Jouf had the highest differences in ASIRs of NHL between 2001 and 2008, but the differences fell below the predicted rates for this period. The likely justification for these differences is that Jouf was more affected by NHL over this 8-year period than were the other regions of Saudi Arabia. Despite the shortage of data about the geographic and time distribution of NHL among Saudi men, we sought to uncover the patterns and trends of NHL among men from 2001 to 2008. Therefore, our findings may be useful for improving the public health policy in Saudi Arabia regarding NHL through the discussed epidemiological evidence. This research may help to generate future hypotheses about the potential risk factors of NHL in the highest-affected regions of Saudi Arabia through prospective epidemiological studies, which identify the relationships between exposure and disease.
Conclusion
This descriptive epidemiological analysis of data from the SCR collected from 2001 to 2008 revealed that the CIR and ASIR of NHL cancer among Saudi men show increasing trends. The region of Riyadh, the Eastern region, and Makkah had the highest overall CIRs and ASIRs. Jazan, Hail, and Baha had the lowest. The IRRs of NHL were a concern in certain regions, such as Riyadh, Makkah, and the Eastern region. The greatest differences in the ASIR of NHL among Saudi men from 2001 to 2008 were found in Jouf. Further analytical studies are needed to explore the potential risk factors of NHL among Saudi men.
Disclosure
The authors report no conflicts of interest in this work.
References
Müller AM, Ihorst G, Mertelsmann R, Engelhardt M. Epidemiology of non-Hodgkin’s lymphoma (NHL): trends, geographic distribution, and etiology. Ann Hematol. 2005;84(1):1–12. | |
Hennessy B. Non-Hodgkin lymphoma: an update. Lancet Oncol. 2004;5(6):341–353. | |
Morton L. Lymphoma incidence patterns by WHO subtype in the United States, 1992–2001. Blood. 2006;107(1):265–276. | |
Cancer Research UK. Non-Hodgkin lymphoma mortality statistics. 2014. Available from: http://www.cancerresearchuk.org/cancer-info/cancerstats/types/nhl/mortality/uk-nonhodgkin-lymphoma-mortality-statistics. Accessed May 8, 2013. | |
American Cancer Society. Global Cancer: Facts And Figures. 2nd ed. Atlanta: ACS; 2013. | |
Saudi Cancer Registry. Cancer Incidence Report in Saudi Arabia. Riyadh: Ministry of Health; 2008. | |
Globocan. The global cancer atlas. Available from: http://globocan.iarc.fr/ia/World/atlas.html. Accessed May 8, 2014. | |
King Faisal Specialist Hospital and Research Centre. Tumor Registry Annual Report. Riyadh: Tumor Registry; 2011. | |
Friis RH, Sellers TA. Epidemiology for Public Health Practice. 4th ed. Sudbury (MA): Jones & Bartlett; 2009. | |
McHugh ML. Multiple comparison analysis testing in ANOVA. Biochem Med (Zagreb). 2011;21(3):203–209. | |
Tabatabai MA, Eby WM, Li H, Bae S, Singh KP. TELBS robust linear regression method. Open Access Med Stat. 2012;2012(2):65–84. | |
Ahmad OB, Boschi-Pinto C, Lopez AD, Murray CJL, Lozano R, Inoue M. Age standardization of Rates: A New WHO Standard. Geneva: World Health Organization; 2001. | |
Doll R, Payne PM, Waterhouse JAH. Cancer Incidence in Five Countries. Berlin: Springer-Verlag; 1966. | |
Skibola CF. Obesity, diet and risk of non-Hodgkin lymphoma. Cancer Epidemiol Biomarkers Prev. 2007;16(3):392–395. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.