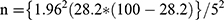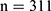Back to Journals » The Application of Clinical Genetics » Volume 13
Incidence of HNF1A and GCK MODY Variants in a South African Population
Authors Matsha TE , Raghubeer S , Tshivhase AM, Davids SFG, Hon GM, Bjørkhaug L, Erasmus RT
Received 15 September 2020
Accepted for publication 6 November 2020
Published 14 December 2020 Volume 2020:13 Pages 209—219
DOI https://doi.org/10.2147/TACG.S281872
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Martin Maurer
Tandi E Matsha,1,* Shanel Raghubeer,1,* Abegail M Tshivhase,1 Saarah FG Davids,1 Gloudina M Hon,1 Lise Bjørkhaug,2 Rajiv T Erasmus1
1SAMRC/Cardiometabolic Health Research Unit, Department of Biomedical Sciences, Faculty of Health & Wellness Sciences, Cape Peninsula University of Technology, Bellville Campus, Cape Town 7530, South Africa; 2Department of Safety, Chemistry, and Biomedical Laboratory Sciences, Western Norway University of Applied Sciences, Bergen, Norway
*These authors contributed equally to this work
Correspondence: Shanel Raghubeer
SAMRC/CPUT/Cardiometabolic Health Research Unit, Department of Biomedical Sciences, Faculty of Health & Wellness Sciences, Cape Peninsula University of Technology, Bellville Campus, Cape Town 7530, South Africa
Tel +27 21 959 6015
Email [email protected]
Background and Aim: Maturity-onset diabetes of the young (MODY) is the result of single gene variants. To date, fourteen different MODY subtypes have been described. Variants in genes coding for glucokinase (GCK, MODY2) and hepatic nuclear factor 1 alpha (HNF1A, MODY3) are most frequently encountered. MODY patients are often misdiagnosed with type 1 or type 2 diabetes, resulting in incorrect treatment protocols. At the time of reporting, no data are available on MODY prevalence in populations from Africa. Our study aimed to investigate and report on the incidence of MODY-related variants, specifically HNF1A variants, in a population from the Western Cape.
Methods: Study participants were recruited (1643 in total, 407 males, 1236 females) and underwent anthropometric tests. Thereafter, blood was collected, and real-time PCR was used to screen for specific variants in HNF1A and GCK genes.
Results: Ninety-seven individuals (5.9%) were identified with a specific HNF1A gene polymorphism (rs1169288) and twelve (0.9%) with a GCK polymorphism (rs4607517).
Conclusion: In total, 6.6% of the study population expressed MODY variants. To our knowledge, we are the first to report on MODY incidence in Africa. This research provides the basis for MODY incidence studies in South Africa, as well as data on non-Caucasian populations.
Keywords: MODY, HNF1A, GCK, diabetes mellitus, monogenic diabetes, South Africa
Introduction
Maturity onset diabetes of the young (MODY) is a monogenic form of diabetes, caused by variation in a single gene, and type 1 (T1DM) and type 2 diabetes mellitus (T2DM) are polygenic in nature. While T1DM is caused by an autoimmune-mediated destruction of pancreatic β-cells in the islets of pancreatic glands, a complex interaction of multiple genes and environmental factors are thought to be drivers of T2DM.1 MODY is associated with pathogenic variants in one of fourteen different genes, resulting in β-cell dysfunction, which causes a defect in insulin secretion in response to glucose stimulation.2 Pathogenic variants in these genes (HNF4A, GCK, HNF1A, PDX1, HNF1B, NEURO1, KLF11, CEL, PAX4, INS, BLK, ABCC8, KCNJ11, and APPL1) determine the type of MODY (MODY1–14), age of onset, treatment responses, and associated extra-pancreatic complications, such as microvascular disorders.1
MODY is characterised by an early age of onset (before 35 years), a strong familial history of diabetes, and often, a lack of insulin resistance.3 Further, patients diagnosed with MODY often present with normal body mass indexes (BMI).3 The illness is inherited in an autosomal dominant manner, where 63% of carriers develop diabetes before 25 years of age, and 96% develop diabetes before 55 years of age.4 MODY is thought to account for roughly 1–3% of all diabetes mellitus (DM) cases,5 where the most prevalent forms involve pathogenic variants in hepatocyte nuclear factor (HNF1A, HNF1B, HNF4A) and glucokinase (GCK) genes, accounting for ~99% of all MODY cases.6 However, prevalence estimates vary by ethnicity, hence the need for more comprehensive prevalence studies, especially in countries where genetic testing is often overlooked.
HNF1A (MODY3 [MIM: 600496]) pathogenic variants affect mitochondrial function in pancreatic β-cells, often causing progressive dysfunction of β-cells and insulin secretory defects.7 Additional clinical features in MODY3 patients include glycosuria and decreased renal absorption of glucose.8 Whilst the general consensus has been that heterozygous highly penetrant loss-of-function mutations in HNF1A give rise to MODY3, this genotype-phenotype correlation is true for a subset of HNF1A variant carriers, as it represents one end of a broad spectrum of HNF1A variant effects.9 GCK (MODY2) mutations prevent an appropriate response to rising glucose levels, thereby disrupting the normal secretion of insulin and increasing the glucose threshold. This translates to an increased fasting glucose level, delaying insulin secretion.7 While pathogenic variants in the HNF1A and GCK genes each account for ~20–70% of all MODY cases, pathogenic variants in HNF4A and HNF1B account for ~5%.6
MODY is very difficult to diagnose for several reasons, namely, the low prevalence in a population, shared symptoms with DM, and limited awareness of MODY.10 Further, genetic testing is not common practice in most countries, particularly low-middle income countries. Correct diagnosis could considerably alter treatment protocols and patient quality of life. Treatment could be amended to glucose lowering medication in the case of MODY1 (HNF4A) or MODY3 (HNF1A) diagnoses,11 while withdrawal of medication would be appropriate for patients diagnosed with MODY2 (GCK).12
As most genetic studies have been conducted in Caucasian populations, it is vital to determine the specific prevalence of MODY in populations of varied ethnicity.13 The goal of this study was to report on MODY incidence in a mixed-ancestry population of the Western Cape, South Africa, by prevalence of MODY gene variants previously associated with MODY or T2D risk.14 To the best of our knowledge, we are the first research group to report on MODY population prevalence in Africa. We further describe clinical characteristics of South African individuals carrying MODY variants.
Methods
Ethics
The current cross-sectional study forms part of the Vascular and Metabolic Health (VMH) study registered at the Cape Peninsula University of Technology, Bellville South, Cape Town, South Africa. Ethical approval for the study was obtained from the Research Ethics Committees of the Cape Peninsula University of Technology (CPUT) and Stellenbosch University (respectively, NHREC: REC - 230 408–014, CPUT/HW-REC 2015/H01 and N14/01/003). Further ethics approval was specifically granted for MODY study analysis (CPUT/HW-REC 2015/H01). The study was carried out in accordance with the Declaration of Helsinki. Prior to the commencement of this study, all participants signed a consent form after the study principles had been fully explained in the language of their choice. Thereafter, they were free to ask any questions pertaining to the study and their involvement.
Study Setting
This cross-sectional, descriptive study enrolled participants who reside in Bellville South, located in the Northern Suburbs of Cape Town, Western Cape, South Africa. This population largely consists of mixed-ancestry individuals. According to South African census data of 2011, the population is comprised of 76.0% mixed ancestry, 18.5% Black, 1.0% Asian, 0.5% Caucasian, and 4.0% individuals from other ethnicities.15 The aim of this study was to determine MODY variants in a South African community. MODY has been estimated to account for 1–5% of all diabetes mellitus cases world-wide. However, the incidence of MODY is unknown in the South African population. A study conducted by Erasmus et al reported on the increase in diabetes incidence in this population, thus providing the ideal setting to investigate MODY prevalence.16 Therefore, the prevalence of diabetes in the South African population, which has been reported as 28% by Erasmus et al (2012), was used for the sample size calculation. The formula used to calculate the sample size for this study is as follows:
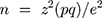 . Where: n = the sample size, z = standard error associated with the chosen level of confidence (1.96), p = estimated population percentage, q = 100-p, e = acceptable sample error (5%).
. Where: n = the sample size, z = standard error associated with the chosen level of confidence (1.96), p = estimated population percentage, q = 100-p, e = acceptable sample error (5%).
The minimum sample size required was 311 participants. The study cohort comprised of individuals who voluntarily participated in the VMH study conducted between 2014 and 2016. A total of 1643 individuals were recruited. All participants were screened for MODY, as misdiagnosis with T1DM or T2DM could have occurred.
Inclusion/Exclusion Criteria
Only participants of mixed-ancestry descent were included. Participants had to be 20 years of age or older, and both males and females were included (both diabetic and non-diabetic). Pregnant women, severely ill individuals, and those who declined to participate were excluded from the study.
Anthropometric Measurements
Anthropometric measurements were recorded for all participants. These included height (cm), weight (kg), and hip (HipC, cm) and waist (WaistC, cm) circumferences. The measurements were performed three times, with the average used for final analysis. In addition, blood pressure (BP) readings were obtained. Blood pressure measurements were performed by a registered nurse according to World Health Organization (WHO) guidelines.17 The Omron M6 Comfort-preformed Cuff Blood Pressure Monitor (Omron, Japan) was used for the measurement. Readings were taken three times with three-minute intervals between each reading. The lowest systolic (SBP) and corresponding diastolic (DBP) readings were used.
Blood Sample Collection and Biochemical Analysis
Blood samples were collected by trained, registered nurses. Participants were requested to fast overnight according to WHO guidelines,17 in order to enable the collection of fasting samples. Samples were collected after the two-hour oral glucose tolerance test (OGTT). Participants who had reported T2DM and/or those using medication had only the fasting blood sample taken. All other participants performed the two-hour OGTT. Fasting and 2hr postprandial blood samples were collected for serological tests, namely serum cotinine, insulin, lipid profile analysis, and measurement of glycated haemoglobin (HbA1c) levels. DNA was extracted using the salt extraction method. The Nanodrop™ One (Thermo Fisher Scientific, Waltham, MA, USA) was used to quantify DNA concentrations, thereafter DNA was stored at −80ºC. Blood samples were transported to PathCare Reference Laboratory (Cape Town, South Africa) for further processing and biochemical analysis. Samples were processed and stored at −80°C for future use.
Biochemical Analysis
Plasma glucose concentrations (mmol/L) were measured using the hexokinase method (Beckman Coulter). Glycated haemoglobin (%) was measured using high performance liquid chromatography (HPLC) (Variant™ II Turbo System, BioRad, Hercules, CA, USA). Insulin (mIU/L) was measured using a paramagnetic particle-based chemiluminescent system. The lipid profile (mmol/L) was analysed using an enzymatic selective protection-endpoint assay (Beckman Coulter) for LDL-cholesterol, an enzymatic immuno-inhibition-endpoint assay (Beckman Coulter) for HDL-cholesterol, and a glycerol phosphate oxidase in the presence of peroxidase (GPO-POD) endpoint assay (Beckman Coulter) for triglycerides.
Definitions and Calculations
Body Mass Index (BMI) was calculated as weight per square meter (kg/m2). According to the Global Report on Diabetes,18 DM is diagnosed if the fasting blood glucose (FBG) measures ≥ 7.0mmol/L or the post-2hr blood glucose measures (2hr BG) ≥ 11.1mmol/L. Impaired fasting glucose (IFG) is diagnosed when the FBG measures between 6.1 and 6.9mmol/L and, if analysed, the post-2hr BG measures < 7.8mmol/L. Impaired glucose tolerance (IGT) is classified as a FBG measured at < 7.0mmol/L and post-2hr BG measured between ≥ 7.8mmol/L and < 11.1mmol/L. Hyperglycaemia includes individuals with T2DM, IFG, and/or IGT.
Real-Time Polymerase Chain Reaction (RT-PCR) Single-Nucleotide Polymorphism (SNP) Genotyping
Six different SNPs (rs140491072 (p.Y322C), rs115080759 (p.L389V), rs142318174 (p.G52A), rs137853245 (p.A276D), rs1169288 (p.I27L), and rs4607517; Supplementary Table 1) were analysed using PCR techniques performed in two different laboratories, thereafter results were confirmed by sequencing. Firstly, real-time PCR was carried out using the Applied Biosystems® QuantStudio™ 7 Flex Real-time PCR system (Thermo Fisher Scientific) and TaqMan® Universal PCR Master Mix, according to the manufacturer’s protocol. The initial denaturation occurred at 95°C (10min), followed by 50 cycles of denaturation (95°C, 15s), annealing (60°C, 90s), and extension (60°C, 90s). Thereafter, 1643 genomic DNA samples were sent to Inqaba Biotechnical Industries (Pretoria, South Africa) for SNP genotyping and sequencing. Inqaba Biotechnical Industries employed an in-house protocol, which included an initial locus-specific PCR reaction, followed by single-base extension using mass-modified dideoxynucleotide terminators of an oligonucleotide primer. Reagents and primers (TaqMan® SNP Genotyping Assays) were sourced from Thermo Fisher Scientific.
Statistical Analysis
Data were analysed using Statistica version 13.5 (StatSoft, Southern Africa Analytics) and SPSS v.24 (IBM Corp, 2011). General characteristics of the study participants are summarized as mean ± standard deviation (SD) or median (25th, 75th) percentiles for continuous variables and number (%) for categorical variables. Analysis of variance was used to calculate the mean and standard deviation values. SNPs were assessed using the Hardy-Weinberg Equilibrium (HWE) expectation with a Chi square goodness-of-fit test. Linear regression models were used for the analysis of quantitative traits assuming the dominant, recessive, and additive genetic models. A p-value of < 0.05 was considered statistically significant.
Results
Study Population Characteristics
In total, 1643 individuals participated in the study (407 males, 1236 females). Characteristics of the study population are indicated in Table 1. Females (50.0 ± 15.1 years) were significantly older than males (47.0 ± 15.6 years; p = 0.0008) and exhibited significantly greater glycaemic measurements. Consequently, the incidence of diabetes (males 15% vs females 20%) and pre-diabetes (pre-DM; males 10.8% vs females 15.8%) was significantly greater in females (p = 0.0539 and p = 0.0313, respectively). The BMI (males 24.7 ± 6.6 vs females 30.8 ± 8.0), waist circumference (WaistC, cm; males 86.2 ± 16.8 vs females 95.1 ± 16.9), and hip circumference (HipC, cm; males 94.5 ± 12.5 vs females 107.9 ± 16.4) measurements were significantly greater in females as compared to males (p < 0.0001 for all three measurements; Table 1).
 |
Table 1 Characteristics of the Study Population According to Gender |
Allele and Genotype Distribution Amongst Study Participants
The SNP frequency was tested for Hardy-Weinberg Equilibrium (HWE). Table 2 indicates the genotype distribution and minor allele frequencies within the study population. Ninety-seven individuals (5.9%) presented with the C/C genotype for HNF1A (rs1169288), while 557 individuals (34.3%) presented with the C/A genotype. The allele percentage was calculated at 23.1% for rs1169288 (751/3246; HWE p = 0.1576). Twelve individuals (0.7%) presented with the G/T genotype for HNF1A (rs115080759), with a rare allele percentage of 0.4% (12/3280; HWE p = 0.8818). The GCK SNP (rs4607517) results indicated that 28 individuals (2.1%) presented with the A/G genotype, while 12 presented with A/A (0.9%), indicating a rare allele percentage of 1.9% (52/2722; HWE p < 0.0001; Table 2).
 |
Table 2 Genotype Distributions and Minor Allele Frequencies |
Participant Characteristics Compared Across HNF1A rs1169288 SNP Genotypes According to Gender
Ninety-seven participants expressed the HNF1A p.I27L SNP (C/C), with 557 expressing A/C and 969 expressing A/A (Table 3). No significant difference was observed between genotypes and the percentage of diabetic, hyperglycaemic, and normoglycaemic participants. No age difference was observed between participants and the three varying genotypes. Similarly, no significant differences were observed between BMI, WaistC, HipC, and glycaemic measurements, as well as cholesterol profiles across the genotypes. Interestingly, C-reactive protein (CRP, mg/L) levels were significantly lower in participants with the C/C genotype (5.62±6.36) compared to participants with the A/A (9.47±17.43) and C/A (7.33±13.64) genotypes (p = 0.0068; Table 3). Regression analysis indicated decreased CRP levels across the models (Table 4), with significantly decreased CRP observed in the dominant and additive models (p = 0.04 and p = 0.017, respectively). Additionally, significantly increased LDL cholesterol levels were observed in the dominant and additive models (p = 0.019 and p = 0.019, respectively; Table 4), while HDL cholesterol appeared to decrease across all three models, with significance observed in the additive model (p = 0.097). The odds ratios were calculated for HNF1A (rs1169288) regarding potential determinants of MODY, such as C-reactive protein, FBG, HbA1C, and lipid profile components (Supplementary Table 2).
 |
Table 3 Participant Characteristics Compared Across HNF1A rs1169288 SNP Alleles |
 |
Table 4 Generalized Linear Regression Models Showing Phenotypes Associated with HNF1A rs1169288 |
Participant Characteristics Compared Across HNF1A rs115080759 SNP Genotypes
Participants expressed the normal T/T (N=1628) genotype and the MODY-associated T/G (N=12) genotype (Table 5). No participants expressed the G/G variation. No significant differences were observed between the genotypes and the percentage of diabetic, hyperglycaemic, and normoglycaemic participants. No significant age difference was observed. Additionally, there were no significant differences between BMI, WaistC, HipC, and lipid profiles across the genotypes. However, significant differences were observed in glycaemic measurements. Participants expressing the T/G genotype exhibited significantly increased FBG levels (7.92±4.99 vs 5.72±2.74; p = 0.0061), increased HbA1c percentages (7.30±2.73 vs 6.18±1.52; p = 0.0116), and an increased glucose to insulin ratio (1.56±1.76 vs 1.00±0.87; p = 0.0284). A significant difference was observed in FBG (p = 0.0098), HbA1c % (p = 0.0127), and the glucose/insulin ratio (p = 0.0213) across genders and SNP genotypes (Table 5).
 |
Table 5 Participant Characteristics Compared Across HNF1A rs115080759 SNP Alleles |
Participant Characteristics Compared Across GCK rs4607517 SNP Genotypes According to Gender
Twelve participants expressed the GCK rs4607517 SNP (A/A), with 28 expressing A/G, and 1321 expressing G/G (Table 6). No significant differences were observed in all participant parameters across SNP genotypes. However, participants expressing the MODY-associated allele (either heterozygous A/G or homozygous A/A) exhibited slightly elevated glycaemic measurements compared to participants expressing the G/G genotype (Table 6), although non-significant. Linear regression analysis indicates significantly increased levels of triglycerides across the three models (p = 0.001 (dominant), p < 0.001 (recessive and additive); Table 7). The odds ratios were calculated for GCK (rs4607517) regarding potential determinants of MODY, such as C-reactive protein, FBG, HbA1C, and lipid profile components (Supplementary Table 3).
 |
Table 6 Participant Characteristics Compared Across GCK rs4607517 SNP Alleles |
 |
Table 7 Generalized Linear Regression Models Showing Phenotypes Associated with GCK rs4607517 |
Discussion
MODY is a rare form of diabetes caused by single-gene defects, and is often misdiagnosed as T1DM or T2DM, thereby compromising treatment protocols.10 Mutations in GCK and HNF1A genes are most common, accounting for approximately 99% of MODY diagnoses, representing 1–5% of all DM cases. Previous research on the South African study population reported a high incidence of diabetes and cardiovascular disease,16,19 providing a suitable setting for an investigation into MODY incidence. To our knowledge, this is the first study to identify individuals carrying pathogenic variants for monogenic diabetes in South Africa and Africa in general. We aimed to determine the presence of MODY by the risk allele frequency of five HNF1A SNPs, one common (MAF > 5%) and four rare (MAF < 1%), of which the four have been found in an African population alone, according to Gnomad, and one GCK SNP, and their association with cardiometabolic traits in a general South African population characterised by a high DM prevalence.
For the common HNF1A variant, we observed the presence of a dominant genotype in HNF1A rs1169288 (p.Ile27Leu), which affected 97 (5.9%) individuals, while 12 (0.9%) individuals were affected with GCK rs4607517, and only one participant carried both, resulting in MODY-associated variants amounting to 6.7%. Another 554 (34.3%) and 28 (2.1%) participants were carriers of HNF1A rs1169288 and GCK rs4607517, respectively. No carriers of HNF1A rs140491072, rs142318174, and rs137853245 were observed, however, 12 (0.7%) participants carried the MODY-associated HNF1A rs115080759 (p.L389V) SNP.20 Although the distribution of genotypes according to glycaemic status were not significantly different, a third of HNF1A rs1169288- or GCK rs4607517-positive individuals exhibited hyperglycaemia. In individuals with diabetes, the HNF1A SNP was observed in 7.1%, whilst the GCK SNP was observed in 0.6% of individuals. C-reactive protein (CRP) levels were significantly lower in participants expressing the HNF1A (rs1169288) common SNP (p = 0.0068), and this remained after linear regression model analysis, which showed that the presence of the C allele significantly reduced CRP and HDL-cholesterol, but increased LDL-cholesterol. This result agrees with previous research reporting on low CRP levels in MODY3 patients.21 Interestingly, participants expressing the rare allele for HNF1A mutation (rs115080759; p.Leu389Val) exhibited significantly increased fasting blood glucose and HbA1c levels, as well as increased glucose to insulin ratios (p = 0.0061, p = 0.0116, and p = 0.0284, respectively), indicating that the presence of the rare allele influences glycaemic measurements, although participants were not diagnosed with MODY. This is particularly important when considering the extra-hepatic complications associated with MODY3, such as microvascular and macrovascular issues.22 Although twelve participants expressed genotype variations of the GCK SNP, no anthropometric or biochemical differences were observed during comparisons between genotypes. Nonetheless, participants expressing the rare MODY allele associated with a GCK mutation (rs4607517) exhibited slightly increased glycaemic measurements as compared to those without the rare allele; these results were non-significant.
The presence of the GCK rs4607517 A-allele non-significantly increased glycaemic indices, but significantly increased triglyceride levels. The reported incidence correlates with MODY research conducted globally.23–26 However, GCK variants in our study population disagrees with the current prevalence estimate of approximately 1 in 1000 people.12 Our results report a 0.9% presence of the GCK rs4607517 AA-genotype in a population of 1361 people, demonstrating an incidence of 0.88%, as opposed to 0.1% cited in literature.12 HNF1A, a human gene located on chromosome 12, comprising of 10 exons that span 23kb of genomic DNA (gDNA), contains mutations scattered throughout the entire gene, with the greatest numbers found in HNF1A exons 2 and 4.27 Although we did not screen the entire HNF1A gene, we observed a high frequency of common variant HNF1A rs1169288 (I27L) and less than 1% of one single low-frequency variant in the HNF1A gene. Interestingly, one participant exhibited both GCK rs4607517 and HNF1A rs1169288 common variants. However, the anthropometric measurements did not exhibit extreme diabetic symptoms. Further research regarding this participant is warranted, including an in-depth investigation into the family history of diabetes.
This study did not follow strict clinical criteria before genetic testing was performed. The models for pre-emptive MODY testing have been developed in primarily European Caucasian populations, as such, our study deviated from this model. Previous research highlighted the prevalence of MODY in Europe,25,26,28 therefore our study provides a foundation for MODY research in Africa. Accurate diagnosis of MODY is crucial for appropriate treatment protocols and management of associated complications. DM misdiagnosis is common in MODY patients, which could result in inappropriate treatment and exacerbation of related illnesses. We hope that this research draws awareness to the subject of MODY and the number of people potentially affected by an undiagnosed illness. Our results further support the need for full genetic testing of DM patients and paediatric cases to accurately diagnose and appropriately treat MODY patients. Limitations to our study include the small study population and the investigation of only a few HNF1A and GCK SNPs. Further, the number of female participants was greater than that of male participants in this study, which is a common limitation in community-based studies in Africa, where women tend to be more interested in participating than men. Other MODY types were not screened, and our testing included six SNPs, demonstrating that numerous mutations could be overlooked. Future studies should address these limitations and include an investigation into family history of DM incidence.
Acknowledgments
We thank the community of Bellville South.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This research project was funded by the South African Medical Research Council with funds from the National Treasury under its Economic Competitiveness and Support Package (MRC-RFA-UFSP-01-2013/VMH Study) and strategic funds from the South African Medical Research Council received from the South African National Department of Health, the South African National Research Foundation (Grant no. 115450). Any opinion, finding, and conclusion or recommendation expressed in this material is that of the authors and the Medical Research Council does not accept any liability in this regard.
Disclosure
The authors declare no conflicts of interest.
References
1. Hoffman LS, Fox TJ, Anastasopoulou C, et al. Maturity Onset Diabetes in the Young. [Updated 2020 Sep 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532900/.
2. Molven A, Njolstad PR. Role of molecular genetics in transforming diagnosis of diabetes mellitus. Expert Rev Mol Diagn. 2011;11(3):313–320. doi:10.1586/erm.10.123
3. Stride A, Vaxillaire M, Tuomi T, et al. The genetic abnormality in the beta cell determines the response to an oral glucose load. Diabetologia. 2002;45(3):427–435. doi:10.1007/s00125-001-0770-9
4. Shepherd M, Sparkes AC, Hattersley AT. Genetic testing in maturity onset diabetes of the young (MODY): a new challenge for the diabetic clinic. Pract Diabetes Int. 2001;18(1):16–21. doi:10.1002/pdi.108
5. Shepherd M, Shields B, Hammersley S, et al. Systematic population screening, using biomarkers and genetic testing, identifies 2.5% of the U.K. pediatric diabetes population with monogenic diabetes. Diabetes Care. 2016;39(11):1879–1888. doi:10.2337/dc16-0645
6. Heuvel-Borsboom H, de Valk HW, Losekoot M, Westerink J. Maturity onset diabetes of the young: seek and you will find. Neth J Med. 2016;74(5):193–200.
7. Fajans SS, Bell GI, Polonsky KS. Molecular mechanisms and clinical pathophysiology of maturity-onset diabetes of the young. N Engl J Med. 2001;345(13):971–980. doi:10.1056/NEJMra002168
8. Pontoglio M, Prie D, Cheret C, et al. HNF1alpha controls renal glucose reabsorption in mouse and man. EMBO Rep. 2000;1(4):359–365. doi:10.1093/embo-reports/kvd071
9. Najmi LA, Aukrust I, Flannick J, et al. Functional investigations of HNF1A identify rare variants as risk factors for type 2 diabetes in the general population. Diabetes. 2017;66(2):335–346. doi:10.2337/db16-0460
10. Thanabalasingham G, Owen KR. Diagnosis and management of maturity onset diabetes of the young (MODY). BMJ. 2011;343(oct19 3):d6044. doi:10.1136/bmj.d6044
11. Bacon S, Kyithar MP, Rizvi SR, et al. Successful maintenance on sulphonylurea therapy and low diabetes complication rates in a HNF1A-MODY cohort. Diabet Med. 2016;33(7):976–984. doi:10.1111/dme.12992
12. Chakera AJ, Steele AM, Gloyn AL, et al. Recognition and management of individuals with hyperglycemia because of a heterozygous glucokinase mutation. Diabetes Care. 2015;38(7):1383–1392. doi:10.2337/dc14-2769
13. Shields BM, McDonald TJ, Ellard S, Campbell MJ, Hyde C, Hattersley AT. The development and validation of a clinical prediction model to determine the probability of MODY in patients with young-onset diabetes. Diabetologia. 2012;55(5):1265–1272. doi:10.1007/s00125-011-2418-8
14. Bjorkhaug L, Sagen JV, Thorsby P, Sovik O, Molven A, Njolstad PR. Hepatocyte nuclear factor-1 alpha gene mutations and diabetes in Norway. J Clin Endocrinol Metab. 2003;88(2):920–931. doi:10.1210/jc.2002-020945
15. Census 2011 Statistical release, Statistics South Africa. 2011.
16. Erasmus RT, Soita DJ, Hassan MS, et al. High prevalence of diabetes mellitus and metabolic syndrome in a South African coloured population: baseline data of a study in Bellville, Cape Town. S Afr Med J. 2012;102(11 Pt 1):841–844.
17. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15(7):539–553. doi:10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S
18. World Health Organization. Global Report on Diabetes. Geneva: World Health Organization; 2016.
19. Matsha TE, Hassan MS, Kidd M, Erasmus RT. The 30-year cardiovascular risk profile of South Africans with diagnosed diabetes, undiagnosed diabetes, pre-diabetes or normoglycaemia: the Bellville, South Africa pilot study. Cardiovasc J Afr. 2012;23(1):5–11. doi:10.5830/CVJA-2010-087
20. Bellanne-Chantelot C, Carette C, Riveline JP, et al. The type and the position of HNF1A mutation modulate age at diagnosis of diabetes in patients with maturity-onset diabetes of the young (MODY)-3. Diabetes. 2008;57(2):503–508. doi:10.2337/db07-0859
21. Owen KR, Thanabalasingham G, James TJ, et al. Assessment of high-sensitivity C-reactive protein levels as diagnostic discriminator of maturity-onset diabetes of the young due to HNF1A mutations. Diabetes Care. 2010;33(9):1919–1924. doi:10.2337/dc10-0288
22. Fajans SS, Bell GI. MODY: history, genetics, pathophysiology, and clinical decision making. Diabetes Care. 2011;34(8):1878–1884. doi:10.2337/dc11-0035
23. Pihoker C, Gilliam LK, Ellard S, et al. Prevalence, characteristics and clinical diagnosis of maturity onset diabetes of the young due to mutations in HNF1A, HNF4A, and glucokinase: results from the SEARCH for Diabetes in Youth. J Clin Endocrinol Metab. 2013;98(10):4055–4062. doi:10.1210/jc.2013-1279
24. Kropff J, Selwood MP, McCarthy MI, Farmer AJ, Owen KR. Prevalence of monogenic diabetes in young adults: a community-based, cross-sectional study in Oxfordshire, UK. Diabetologia. 2011;54(5):1261–1263. doi:10.1007/s00125-011-2090-z
25. Eide SA, Raeder H, Johansson S, et al. Prevalence of HNF1A (MODY3) mutations in a Norwegian population (the HUNT2 Study). Diabet Med. 2008;25(7):775–781. doi:10.1111/j.1464-5491.2008.02459.x
26. Pavic T, Juszczak A, Pape Medvidovic E, et al. Maturity onset diabetes of the young due to HNF1A variants in Croatia. Biochem Med. 2018;28(2):020703.
27. Colclough K, Saint-Martin C, Timsit J, Ellard S, Bellanné-Chantelot C. Clinical utility gene card for: maturity-onset diabetes of the young. Eur J Hum Genet. 2014;22(9):1153. doi:10.1038/ejhg.2014.14
28. Lehto M, Wipemo C, Ivarsson SA, et al. High frequency of mutations in MODY and mitochondrial genes in Scandinavian patients with familial early-onset diabetes. Diabetologia. 1999;42(9):1131–1137. doi:10.1007/s001250051281
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.