Back to Journals » Drug, Healthcare and Patient Safety » Volume 11
Inappropriateness of Intravenous Antibiotic Prescriptions at Hospital Discharge at a Tertiary Care hospital in Thailand
Authors Mahatumarat T, Pinmanee N, Injai W , Chaiwarith R
Received 3 July 2019
Accepted for publication 30 November 2019
Published 20 December 2019 Volume 2019:11 Pages 125—129
DOI https://doi.org/10.2147/DHPS.S221430
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Hemalkumar B Mehta
Tuanjai Mahatumarat,1 Napaporn Pinmanee,2 Wichchulada Injai,2 Romanee Chaiwarith1
1Division of Infectious Diseases, Department of Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand; 2Division of Pharmacy, Maharaj Nakorn Chiang Mai Hospital, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
Correspondence: Romanee Chaiwarith
Division of Infectious Diseases, Department of Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai 50200, Thailand
Tel +66-5393-6457
Fax +66-5389-4231
Email [email protected]
Background: Intravenous antibiotics, either as outpatient parenteral antimicrobial therapy (OPAT) or transition of care to community-based management, is a common practice in tertiary care hospitals to minimize hospital stays. However, infectious disease consultation was not mandated for those prescriptions. Therefore, we conducted this study to evaluate the appropriateness of intravenous antibiotic prescriptions at hospital discharge.
Methods: This retrospective cross-sectional study was conducted among patients receiving care at the internal medicine units of the Maharaj Nakorn Chiang Mai Hospital from November 1, 2015, to April 30, 2016. Intravenous antibiotics at hospital discharge were reviewed by an infectious diseases (ID) specialist.
Results: One hundred and twenty-nine prescriptions for 117 patients were reviewed. The most common diagnoses requiring intravenous antibiotics at hospital discharge were upper urinary tract infection (34.2%) and hepatobiliary tract infections (15.4%). The most common intravenous antibiotic was ceftriaxone (36.4%), followed by ertapenem (20.1%). Overall, the inappropriateness of prescriptions was 85.3%. The most common reason for inappropriateness was a failure to switch to oral antibiotics (52.7%), followed by incorrect duration (16.3%).
Conclusion: Antimicrobial stewardship should be considered for intravenous antibiotics at hospital discharge to reduce the inappropriateness of those prescriptions.
Keywords: parenteral antimicrobials, antimicrobial stewardship, outpatient parenteral antimicrobial therapy
Introduction
Nowadays, outpatient parenteral antimicrobial therapy (OPAT) is increasingly utilized as such a modality has led to patient satisfaction without compromising the treatment outcomes.1–5 However, studies have shown that around 30% of parenteral antimicrobials of community-based parenteral anti-infective therapy could be avoided.6,7 Avoiding unnecessary use of antimicrobials may prevent the emergence of drug resistance, decrease C.difficile infections, avoid adverse drug reactions as well as superinfection, and provide cost-savings.8–11 To evaluate whether antimicrobial stewardship should be implemented for parenteral antimicrobials at hospital discharge, the characteristics of those prescriptions should be recognized.
Maharaj Nakorn Chiang Mai Hospital is an affiliated hospital of the Faculty of Medicine, Chiang Mai University. It is a 1400-bed tertiary care referral center in the northern part of Thailand. Infectious disease consultation is not mandated for intravenous antimicrobial prescriptions at hospital discharge, either as OPAT or transition of care to a community hospital. We, therefore, conducted this study aimed to evaluate the appropriateness of those prescriptions.
Materials and Methods
Study Setting, Design, and Population
A retrospective cross-sectional study was conducted among patients receiving care at the internal medicine units of the Maharaj Nakorn Chiang Mai Hospital from November 1, 2015 to April 30, 2016. The medical records of patients aged ≥18 years old who were prescribed intravenous antibiotics at hospital discharge were reviewed by an infectious diseases (ID) specialist. The patients were excluded if they were already evaluated by an ID specialist during hospital admission. The inappropriateness was defined as: 1) no indication for antibiotic use i.e. non-infectious causes of fever, 2) IV antibiotics can be switched to oral antibiotics, 3) ineffective against isolated pathogens, 4) choice of antibiotic can be de-escalated to narrower spectrum, 5) incorrect dosage, and 6) incorrect duration (too short or too long).
Data collection
Demographics (e.g. age, sex, underlying diseases), sites of infection, microbiological test results, intravenous antibiotic prescriptions and their appropriateness were collected.
Statistical Analysis
Data were presented as mean ± SD, median and IQR, and number (%) as appropriate. Comparisons between the appropriate and inappropriate groups were compared using Student’s t-test or Mann Whitney-U test for continuous data, and Chi-2 test or Fisher’s exact test for categorical data. All statistical analyses were performed using Stata statistical software version 10.0 (Stata Statistical Software: Release 10.0, Stata Corporation, College Station, TX, 2007). A two-sided test was used to indicate statistical significance at a p-value of < 0.05.
The study was approved by the Research Ethics Committee 1, Faculty of Medicine, Chiang Mai University; certificate of approval number 173/2016. The informed consent from participants was exempt as the study retrieved the data retrospectively from medical records and no procedure was performed in addition to the standard of care. The personal information were kept confidentiality and only authorized person was able to access to the document. The study was complied with the Declaration of Helsinki.
Results
Baseline Characteristics
One hundred and forty-seven patients were prescribed intravenous antibiotics at hospital discharge, 30 were excluded as an ID specialist was consulted during hospital admission. One hundred and twenty-nine prescriptions for 117 patients were reviewed. Forty-four patients (37.6%) were males and the median age was 60 years (IQR 50, 74). Common underlying diseases were hypertension (36 patients, 30.8%), solid malignancy (33 patients, 28.2%), heart diseases (25 patients, 21.3%), diabetes mellitus (24 patients, 20.5%), and chronic kidney disease (24 patients, 20.5%). The three most common infections were the upper urinary tract infection (40 patients, 34.2%), hepatobiliary tract infections (18 patients, 15.4%), and pneumonia (14 patients, 12.0%). (Table 1)
 |
Table 1 Characteristics of 117 Patients Receiving Intravenous Antibiotics at Hospital Discharge |
Microbiology
Overall, the most common pathogens isolated from urine were Escherichia coli (26/37 patients, 70.2%), followed by Klebsiella pneumoniae (4/37 patients, 10.8%). The most common pathogens isolated from blood were Escherichia coli (16/33 patients, 48.5%), followed by K.pneumoniae (5/33 patient, 15.2%) and Streptococcus spp. (5/33 patient, 15.2%). (Table 2)
 |
Table 2 Pathogens Isolated from Urine and Blood of 117 Patients |
Intravenous Antibiotics Prescriptions
Twelve patients received more than one intravenous antibiotic. The most common intravenous antibiotic prescription was ceftriaxone (47 prescriptions, 36.4%), followed by ertapenem (26 prescriptions, 20.1%), and meropenem (14 prescriptions, 10.8%). (Table 3)
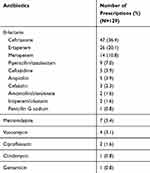 |
Table 3 Intravenous Antibiotics at Hospital Discharge |
Appropriateness of Intravenous Antibiotics Prescriptions
One hundred and ten prescriptions (85.3%) were judged as inappropriate by an ID specialist. The reason of inappropriateness was due to failure to switch to oral antibiotics (68 prescriptions, 52.7%), and incorrect duration (21 prescriptions, 16.3%). (Table 4) No factor was associated with the appropriateness of prescriptions.
 |
Table 4 Appropriateness of Intravenous Antibiotics for 129 Prescriptions |
Discussion
This study demonstrated that 85% of intravenous antimicrobials at hospital discharge were inappropriate and at least 64% of them could be avoided (i.e. no indication for antibiotic use, can switch to oral antibiotics, and if duration was too long), comparable to previous reports.1,12–14 The most common reason of inappropriateness was a failure to switch to oral antibiotics (53%). As the three most common diagnoses were upper urinary tract infection, hepatobiliary tract infection, and pneumonia, all had no contraindication for switching to oral antibiotics.15 Although ESBL-producing enterobacteriaceae may be responsible for those diagnoses, these pathogens accounted for only 18% of the total patients. In addition, some antibiotics with high bioavailability i.e. ciprofloxacin and metronidazole can be switched to oral antibiotics, but there was a failure to do so. The second most common reason for inappropriateness was incorrect duration (16.3%). For example, the duration of treatment for complicated urinary tract infections was too short, or antibiotics can be discontinued due to complete course of treatment. The third and fourth most common reasons for inappropriateness were no indication for antibiotic use as the causes of fever were non-infectious etiology and failure to de-escalate antibiotics to a narrower spectrum (10.9% each). Maintaining “a unique value” in our hospital environment i.e. 7 days, 10 days, or 14 days of treatment may have an influence on the duration of antimicrobials. Even if the clinical picture was great enough to discontinue antibiotics, they were prescribed until that unique value was reached. Antibiotics, sometimes, could be de-escalated to a narrower spectrum, but the physicians were reluctant to narrow down as the patients were clinically stable with those particular antibiotics. Other study reported that 1–11% and 9–39% of planned OPAT can be discontinued and de-escalated to oral antimicrobials, respectively.1,12–14 In addition, incorrect choices, doses, and duration of OPAT were reported in 30%, 11%, and 7%, respectively.14
The most common intravenous antibiotic prescription was ceftriaxone, followed by ertapenem due to ease of administration. These two antibiotics are also effective in the treatment of the most common pathogens responsible for the two most common sites of infection i.e. upper urinary tract and hepatobiliary infections. On the other hand, the most common antibiotics for community-acquired skin and soft tissue infections that required intravenous antibiotics as OPAT were cefazolin and ceftriaxone.16 Frequently used antibiotics depended on the clinical diagnoses of that particular study.17
This study revealed room for improvement in antimicrobial stewardship for antibiotics at hospital discharge either to community-based management or OPAT. Although we have not conducted a well-designed study to demonstrate the value of ID consultation for this kind of prescription, the previous studies have existed.6,7,14 An ID specialist, by theoretically, prescribed antimicrobials more appropriately than non-ID physicians. Therefore, ID consultation should be considered for intravenous antibiotics at hospital discharge as one of the strategies for antimicrobial stewardship.
This study has several limitations. First, due to the nature of the retrospective study, some information may be missing and may lead to misinterpretation. Second, only patients in the internal medicine department were evaluated. It would be interesting to evaluate the appropriateness of intravenous antibiotics prescribed at hospital discharge in another department, as a study from Thailand reported that the inappropriateness of antibiotics prescription was higher in the Department of Surgery, and Department of Obstetrics and Gynecology.18 Knowing the extent of the problem in the hospital may help to design and implement a strategy of antimicrobial stewardship program for particular units.
Conclusions
The inappropriateness of intravenous antibiotics at hospital discharge was high during the study period. ID consultation or a well-designed antimicrobial stewardship program should be considered as an effective way to reduce the inappropriateness of those prescriptions.
Data Sharing Statement
Data will not be shared as the local IRB has no policy to share the data without prior permission.
Author Contributions
All authors contributed to the trial design, data collection, data analysis, data interpretation, and drafted the manuscript. All authors have read and approved the final manuscript, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Paladino JA, Poretz D. Outpatient parenteral antimicrobial therapy today. Clin Infect Dis. 2010;51(Suppl 2):S198–S208. doi:10.1086/653520
2. MacKenzie M, Rae N, Nathwani D. Outcomes from global adult outpatient parenteral antimicrobial therapy programmes: a review of the last decade. Int J Antimicrob Agents. 2014;43(1):7–16. doi:10.1016/j.ijantimicag.2013.09.006
3. Gilchrist M, Seaton RA. Outpatient parenteral antimicrobial therapy and antimicrobial stewardship: challenges and checklists. J Antimicrob Chemother. 2015;70(4):965–970. doi:10.1093/jac/dku517
4. Tice AD, Rehm SJ, Dalovisio JR, et al. Practice guidelines for outpatient parenteral antimicrobial therapy. IDSA guidelines. Clin Infect Dis. 2004;38(12):1651–1672. doi:10.1086/420939
5. Corwin P, Toop L, McGeoch G, et al. Randomised controlled trial of intravenous antibiotic treatment for cellulitis at home compared with hospital. BMJ. 2005;330(7483):129. doi:10.1136/bmj.38309.447975.EB
6. Shrestha NK, Bhaskaran A, Scalera NM, Schmitt SK, Rehm SJ, Gordon SM. Antimicrobial stewardship at transition of care from hospital to community. Infect Control Hosp Epidemiol. 2012;33(4):401–404. doi:10.1086/664758
7. Shrestha NK, Bhaskaran A, Scalera NM, Schmitt SK, Rehm SJ, Gordon SM. Contribution of infectious disease consultation toward the care of inpatients being considered for community-based parenteral anti-infective therapy. J Hosp Med. 2012;7(5):365–369. doi:10.1002/jhm.1902
8. Fowler S, Webber A, Cooper BS, et al. Successful use of feedback to improve antibiotic prescribing and reduce clostridium difficile infection: a controlled interrupted time series. J Antimicrob Chemother. 2007;59(5):990–995. doi:10.1093/jac/dkm014
9. Hsueh PR, Chen WH, Luh KT. Relationships between antimicrobial use and antimicrobial resistance in gram-negative bacteria causing nosocomial infections from 1991–2003 at a university hospital in Taiwan. Int J Antimicrob Agents. 2005;26(6):463–472. doi:10.1016/j.ijantimicag.2005.08.016
10. Willemsen I, van den Broek R, Bijsterveldt T, et al. A standardized protocol for perioperative antibiotic prophylaxis is associated with improvement of timing and reduction of costs. J Hosp Infect. 2007;67(2):156–160. doi:10.1016/j.jhin.2007.07.025
11. Eljaaly K, Enani MA, Al-Tawfiq JA. Impact of carbapenem versus non-carbapenem treatment on the rates of superinfection: a meta-analysis of randomized controlled trials. J Infect Chemother. 2018;24(11):915–920. doi:10.1016/j.jiac.2018.08.004
12. Babouee Flury B, Elzi L, Kolbe M, et al. Is switching to an oral antibiotic regimen safe after 2 weeks of intravenous treatment for primary bacterial vertebral osteomyelitis? BMC Infect Dis. 2014;14:226. doi:10.1186/1471-2334-14-226
13. Conant MM, Erdman SM, Osterholzer D. Mandatory infectious diseases approval of outpatient parenteral antimicrobial therapy (OPAT): clinical and economic outcomes of averted cases. J Antimicrob Chemother. 2014;69(6):1695–1700. doi:10.1093/jac/dku015
14. Sharma R, Loomis W, Brown RB. Impact of mandatory inpatient infectious disease consultation on outpatient parenteral antibiotic therapy. Am J Med Sci. 2005;330(2):60–64. doi:10.1097/00000441-200508000-00002
15. Beique L, Zvonar R. Addressing concerns about changing the route of antimicrobial administration from intravenous to oral in adult inpatients. Can J Hosp Pharm. 2015;68(4):318–326. doi:10.4212/cjhp.v68i4.1472
16. Chan M, Ooi CK, Wong J, Zhong L, Lye D. Role of outpatient parenteral antibiotic therapy in the treatment of community acquired skin and soft tissue infections in Singapore. BMC Infect Dis. 2017;17(1):474. doi:10.1186/s12879-017-2569-4
17. Barr DA, Seaton RA. Outpatient parenteral antimicrobial therapy (OPAT) and the general physician. Clin Med (Lond). 2013;13(5):495–499. doi:10.7861/clinmedicine.13-5-495
18. Apisarnthanarak A, Danchaivijitr S, Bailey TC, Fraser VJ. Inappropriate antibiotic use in a tertiary care center in Thailand: an incidence study and review of experience in Thailand. Infect Control Hosp Epidemiol. 2006;27(4):416–420. doi:10.1086/503348
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
