Back to Journals » Journal of Experimental Pharmacology » Volume 13
In vitro Antileishmanial Activity of Some Ethiopian Medicinal Plants
Authors Nigatu H, Belay A, Ayalew H, Abebe B, Tadesse A , Tewabe Y, Degu A
Received 2 October 2020
Accepted for publication 12 December 2020
Published 15 January 2021 Volume 2021:13 Pages 15—22
DOI https://doi.org/10.2147/JEP.S285079
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Bal Lokeshwar
Hulubanchi Nigatu,1 Alemnesh Belay,1 Hiwot Ayalew,1 Besufekad Abebe,1 Alemu Tadesse,1 Yitagesu Tewabe,1 Abel Degu2
1Department of Pharmaceutical Chemistry and Pharmacognosy, School of Pharmacy, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 2Department of Pharmacology and Clinical Pharmacy, School of Pharmacy, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia
Correspondence: Hiwot Ayalew
Department of Pharmaceutical Chemistry and Pharmacognosy, School of Pharmacy, Addis Ababa University, P.O. Box 1176, Addis Ababa Ethiopia
Email [email protected]
Introduction: Leishmaniasis is a group of diseases caused by protozoan parasites, which remains a burden for developing countries. The lack of a vaccine as well as the emergence of resistance toward the recommended drugs pose a challenge for the control of the disease. This urges the demand for new antileishmanial agents to prevent and treat this disease. Consequently, four Ethiopian plants were selected and tested for their antileishmanial activity against two Leishmanial parasites.
Methods: Methanol (80%) was used to macerate the plant materials. In vitro antipromastigote activity of the crude extracts was then tested against promastigotes and axenically cultured amastigotes of Leishmania aethiopica and Leishmania donovani clinical isolates using Alamar Blue assay, and cell viability was measured fluorometrically. 1% DMSO and the media were used as a negative control while amphotericin B was used as a positive control. Furthermore, preliminary phytochemical analysis of the extracts was performed.
Results: From the four plants’ extracts, Ferula communis and Otostegia integrifolia showed better activity with IC50 value of 11.38± 0.55 and 13.03± 0.87 μg/mL against L. aethiopica, respectively. However, the same plant extracts exhibited lower activity against L. donovani with IC50 values of 23.41± 2.32 and 17.24± 1.29 μg/mL, respectively. O. integrifolia exhibited highest effect against amastigotes of L. aethiopica (IC50: 16.84± 0.65) and L. donovani (IC50:14.55± 0.38). F. communis resulted second highest in growth inhibition against amastigotes of L. aethiopica and L. donovani with IC50 value of 14.32± 0.54 and 31.12± 0.19, respectively. The phytochemical analysis of the extracts indicated the presence of phenol, flavonoids, tannins, saponins, terpenoids, and alkaloids.
Conclusion: The findings from this study demonstrate that crude extracts of F. communis and O. integrifolia showed promising antileishmanial activity against L. aethiopica and L. donovani that may be attributed to the presence of different secondary metabolites.
Keywords: antileishmanial activity, in vitro, Leishmania aethiopica, Leishmania donovani, medicinal plants
Introduction
Leishmaniasis is a protozoan parasitic disease caused by the genus Leishmania. There are more than 20 Leishmania species1 which are mainly transmitted to humans via the vector of infected female Phlebotomus and Lutzomyia sand-flies.2 More than 70 animal species including mammals serve as a host or reservoir, including humans, in the transmission of the parasite.3
The disease is endemic in 98 countries, most of which are developing countries.4 According to World Health Organization’s fact sheet on leishmaniasis, approximately 20,000–30,000 deaths and 0.7–1 million new cases of leishmaniasis occur per annum.1 From the three main forms of the disease, an estimated 50, 000 to 90, 000 new cases of visceral leishmaniasis (VL) and 600, 000 to 1 million new cases of cutaneous leishmaniasis (CL) occur worldwide annually. Furthermore, more than 90% of mucocutaneous leishmaniasis (MCL) cases occur in Bolivia, Brazil, Ethiopia and Peru. In Ethiopia, the disease burden is huge, where the country is highly affected by both VL and MCL, with an estimated 4500 to 5000 new cases of VL per year.5 Besides, the four Leishmania species L. aethiopica, L. donovani, L. major, and L. tropica are common in the country.6
Given that there is still no vaccine available for use in humans, the treatment of leishmaniasis mainly depends on chemotherapy. However, there has been development of resistance toward first line drugs. In addition, the second line drugs have severe adverse effects and are costly in resource-limited areas.7 This urges the demand for new antileishmanial agents to prevent and treat this disease. Moreover, researchers’ attention is being drawn toward naturally derived compounds used to treat parasitic diseases including leishmaniasis for identification of new lead compounds and/or new drugs against the disease which warrant investigation.8
The use of plants as alternative medicine in Ethiopia has been a long-standing practice for centuries. In fact in Ethiopia, for 70%-80% of its people and around 90% of domestic animals, traditional medicines are used as a primary source of treatment.9 Consequently, the following plants of interest were selected; Discopodium peninervium Hochst (Solanaceae) also called “Ameraro” in Amharic,8 Ferula communis L. (Apiaceae) identified colloquially as “Doge”,10 Otostegia integrifolia Benth (Lamiaceae) locally recognized as “Tinjuit”,11 and Urtica simensis Hochst. ex. A. Rich. (Urticaceae) also called “Samma” in Amharic.12
The aforementioned herbs were selected for their traditional use as treatment for skin and wound infection which is ostensibly similar to the manifestations of leishmaniasis.10,12,13 Besides, similar antileishmanial activities were reported within the genus of these plants.8,14–16 Therefore, in this study, in vitro antileishmanial activity and phytochemical analysis of 80% methanolic extracts of each plant were investigated.
Materials and Methods
Collection and Authentication of Plant Materials
Fresh leaves of D. peninervium, O. integrifolia, U. simensis and roots of F. communis were collected from Semien Shewa Ensaro woreda (Latitude: 9° 49ʹ 59.99” N Longitude: 39° 00ʹ 0.00” E) 139 km away from Addis Ababa, in September, 2019. The identification and authenticity of the plant materials was confirmed at the National Herbarium, Department of Biology, Addis Ababa University. The voucher specimen; D. peninervium (HA001), O. integrifolia (AD001), U. simensis (HA003) and F. communis (HA002) was deposited for future reference. The collected parts of plants were separated, washed with tap water, and left to dry in shade. Dried parts of plants were grounded to a coarse powder and stored in a desiccator at room temperature until further use.
Preparation of Plant Extracts
The powdered plant materials (250 g) were macerated using 80% methanol (3 × 2 L, 72 h each). Then the plant extracts were filtered and the methanol in the filtrate was removed using a rotary evaporator (Buchi, Switzerland). The concentrated extract was dried with a lyophilizer (Wagtech, Denmark). Finally, the crude extracts of D. peninervium, O. integrifolia, U. simensis and F. communis yielded 12.5%, 16.6%, 24.4%, and 10.15% respectively.
In vitro Antileishmanial Activity Test
Cell Culture
In vitro antipromastigote activity test was carried out against the promastigote stage of clinical isolates of L. aethiopica and L. donovani. The two clinical isolates were grown in tissue culture flasks containing RPMI 1640 medium supplemented with 10% heat-inactivated fetal calf serum, 100 IU penicillin/mL (Sigma-Aldrich, Germany) and 100 μg/mL streptomycin (Sigma-Aldrich, Germany) solution at 22°C for L. aethiopica and 24°C for L. donovani.17 A cell-free medium was used to grow parasites in vitro and to set up the test system for determination of the IC50 values.
From late stationary phase promastigotes (3 × 106 cells/mL) axenically cultured amastigotes were acquired. Using medium 199 with Hank’s salts supplemented with 20% FBS, 2 mM L-glutamine, 50 IU/mL penicillin, and 50 μg/mL streptomycin, the cells were centrifuged and then resuspended. After that, by using 1 N HCl, the pH was made to 5.5. Following incubation of cells at 31°C for L. aethiopica and 37°C for L. donovani at 5% CO2, amastigote-like rounded morphology together with loss of flagella and cell clumping started appearing within 24 h. The parasites were kept for a week as some motile parasites with intermediate forms and short flagella were detected. The human monocytic leukemia cell line THP-1 cells were incubated in RPMI 1640 medium plus 10% hi-FCS and 20 ng/mL phorbol 12-myristate 13-acetate (PMA; Sigma) at 37°C and 5% CO2 for 72 h. A cell-free medium was used to grow parasites in vitro and to set up the test system for determination of the IC50 values of the extracts.
In vitro Antileishmanial Assay
Each of the plant extracts was added to a separate 96-well microtiter plate containing 100 µLcomplete culture medium to achieve a final concentration of 100 µg/mL. Then, a suspension of 100 µL of the parasites (3.5 × 106 promastigotes of L. aethiopica or L. donovani) obtained from the previous culture was added to each well. After that, parasites were incubated for 72 hrs at room temperature for promastigotes of both strains, at 31°C and 37°C for axenically cultured amastigotes of L. aethiopica or L. donovani respectively, in the presence of various concentrations of extract. Next, resazurin (0.125 mg/mL) was added to 20 µL suspension (10% of the total volume of each well). The mixture was covered with aluminum foil, and left at the previously stated temperature. Fluorescence intensity was measured using Victor 3 Multilabel Counter (PerkinElmer, MA, USA) at excitation wavelength of 544 nm and emission wavelength of 590 nm. The assay was conducted in triplicate and compared to negative controls (1% DMSO and Media alone) and reference drug (Amphotericin B, Sigma-Aldrich, Germany). During the assay, cell viability was monitored by measuring fluorescent signal. The fluorescence intensities produced are proportional to the number of viable cells.17
Cytotoxicity Study in THP-1 Monocyte
Onto 96-well plates, THP-1 monocytes were plated at a density of 4 × 104 cells per well (in 200 volume) in the presence or absence of plant extracts, and plates were incubated at 37°C, 5% CO2 for 72 h. Then Alamar Blue was added, during the last 3 h of incubation, cell viability was measured fluorometrically as described previously.17
Phytochemical Screening
The 80% methanol extracts of each plant were investigated for the presence or absence of secondary metabolites such as alkaloids, flavonoids, phenols, saponins, tannins, and terpenoids following standard procedures.18
Determination of Total Phenols
Folin–Ciocalteu’s method was used to determine the total phenolic content of the extracts. Serial dilutions of the standard (gallic acid) were prepared in distilled water with concentrations 100, 50, 25, 12.5, 6.75 and 3.375 µg/mL, to establish calibration curve. 1 mL of the standard was then transferred into test tubes. After that, 5 mL of distilled water and 0.5 mL of Folin–Ciocalteu’s reagent were added into the test tubes. 5 minutes later, 1.5 mL of Na2CO3 (20%) was added and the volume made up to 10 mL with distilled water. It was allowed to stand for 90 minutes at ambient temperature. The absorbance of the solution was measured at 760 nm using a UV spectrophotometer (Jenway Model 6500, England). Every experiment was conducted in triplicate. Similar procedure was also followed for the extracts (100 µg/mL) and the blank solutions. The total phenolic content was determined using a standard curve of gallic acid (y = 0.0075x - 0.0862, R2 = 0.9665). The calculated total phenolic results were expressed as mg of gallic acid equivalent per 100 g of extracts.19
Determination of Total Flavonoid Content (TFC)
Aluminum chloride complex forming assay was employed to determine the total flavonoid content of the extracts. Serial dilutions of the standard (Quercetin) were prepared in methanol with concentrations 1, 0.50, 0.25, 0.125 and 0.065 mg/mL, to establish calibration curve. 1 mL of the standard was then transferred into test tubes. 0.3 mL 5% NaNO2 was added and left for 5 minutes. Another 0.3 mL of 10% AlCl3 was mixed with the solution and allowed to stand for 5 minutes. After that, 2 mL solution of 1M NaOH was added into the solution followed by quantity sufficient distilled water to make up the volume to 10 mL. Finally the solution was incubated for 30 minutes at ambient temperature. The absorbance of the solution was recorded at 510 nm on UV spectrophotometer (Jenway Model 6500, England). The same procedure was repeated with the extract (1 mg/mL) and the blank solutions. The total flavonoid content was determined using a standard curve of quercetin (y=0.5957x - 0.0055, R2 = 0.9982). The calculated total flavonoid results were expressed as mg of quercetin equivalent per 100 g of extracts. All the procedures were performed in triplicate.20
Data Analysis
Antileishmanial activity (IC50) values were calculated from sigmoidal dose-response curves of percent inhibition using the computer software GraphPad Prism 8.4.3 (GraphPad Sofware, Inc., CA, USA), and Microsoft excel; values were expressed as mean ± SD of triplicate experiments.
Results
Antileishmanial Assay
The methanolic extracts of all the plants showed activity against promastigotes and axenically cultured amastigotes of L. aethiopica and L. donovani with varying IC50 values (Tables 1 and 2, Figures 1 and 2).
 |
Table 1 Antipromastigote Activity IC50 Values and Percent Inhibition of the Plant Extracts Against L. aethiopica and L. donovani |
 |
Table 2 IC50 Values and Percent Inhibition of the Plant Extracts Against Axenically Cultured Amastigotes of L. aethiopica and L. donovani |
Of all the extracts, O. integrifolia exhibited highest antipromastigote activity with percentage inhibition of 71.78% and 71.40% (Table 1) with maximum tested concentration (ie, 100 µg/mL) against L. aethiopica and L. donovani respectively. At this concentration, O. integrifolia exerted similar effect against the amastigotes of L. aethiopica and L. donovani with percentage inhibition of 67.31% and 70.58% respectively (Table 2). F. communis also exerted comparable antipromastigote activity with percentage inhibition of 68.47% and 63.57% against L. aethiopica and L. donovani respectively. While the same plant exerted slightly lower activity against the amastigotes of both Leishmanial species (Table 2).
However, all the extracts showed lower activity as compared to amphotericin B which exhibited IC50 of 1.21±0.11µg/mL and 1.31±0.065 µg/mL (Table 1) against L. aethiopica and L. donovani correspondingly.
Preliminary Phytochemical Analysis
The result found from the investigations revealed the presence of different secondary metabolites such as alkaloids, flavonoids, phenols, saponins, tannins and terpenoids in the plant extracts as summarized in Table 3.
 |
Table 3 Phytochemical Screening of 80% Methanol Extracts of Tested Plants |
Determination of Total Phenols and Flavonoid Content
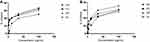
The highest total phenol content was found from D. peninervium (0.43 mg of GAE/g extract) followed by O. integrifolia (0.39 mg of GAE/g extract) as indicated in Table 4 and Figure 3 A. Similarly, the highest amount of flavonoids was measured with D. peninervium (0.38 mg of QE/g extract) while F. communis and O. integrifolia contained comparable amounts of flavonoids, 0.2 and 0.18 mg of QE/g extract respectively as depicted in Table 4 and Figure 3B.
 |
Table 4 Total Phenolic and Flavonoid Content of 80% Methanol Extracts of Tested Plants |
 |
Figure 3 Total phenol (A) and flavonoid (B) contents of 80% methanol plant extracts. |
Discussion
Leishmaniasis is a widespread health burden in developing countries like Ethiopia. This is partly attributed to undernourishment as a result of poverty, compromised immunity, as well as unresponsiveness of the disease to recommended drugs.6,21 Therefore, it is vital to search for alternative drugs from medicinal plants. Accordingly, the present study was carried out to search for the antipromastigote potential of some Ethiopian medicinal plants that are used in folk medicine.
The four crude extracts showed antipromastigote activity against L. aethiopica and L. donovani. However, all the plant extracts exerted lower activity than the reference drug; amphotericin B (Table 1, Figure 1A and B).
The highest antipromastigote activity was recorded for F. communis (IC50=11.38±0.55 µg/mL). The extract also halted the growth of L. donovani (IC50=23.41±2.32 µg/mL) (Table 1). There are studies that stated similar activities of members of the genus Ferula. The hydroalcoholic extract of F. asafetida and essential oil from F. galbaniflua arrested the growth of L. major (IC50=11.8 µg/mL)22 and L. amazonensis (IC50/24hr=95.70 µg/mL) respectively.23
O. integrifolia exhibited highest effect against L. donovani (IC50=17.24±0.87 µg/mL) as compared to other extracts. In addition, the extract displayed activity against L. aethiopica (IC50=13.03±1.29 µg/mL) (Table 1). Mothana et al reported growth inhibition of hydroalcoholic extract of O. fruticosa against L. infantum (IC50 >64.0 µg/mL).24
D. peninervium showed comparable activity against L. aethiopica (IC50=13.38±1.20 µg/mL) as that of O. integrifolia, while it exhibited lower activity against L. donovani (IC50=63.62±1.84 µg/mL) (Table 1). In another study, the essential oil from the same plant exhibited better activity against L. aethiopica (IC50=12.62 μg/mL) and L. donovani (IC50=19.93 μg/mL).25
On the other hand, U. simensis showed lowest result with IC50 value of 63.78±0.89 μg/mL against L. aethiopica and 44.37±1.61 μg/mL against L. donovani (Table 1). The aqueous extract of U. dioica halted the growth of L. major promastigotes with IC50 of 4500 μg/mL.15
However, no study has reported on the effect of the studied plant extracts against L. aethiopica and L. donovani axenic amastigotes.
The phytochemical screening of the four extracts revealed the presence of various phytochemical constituents, ie, phenols, flavonoids and terpenoids (Table 3). Phenols were reported to possess antipromastigote activity. For instance, rosmarinic acid tested against L. donovani promastigotes and intracellular amastigotes resulted in altered membrane integrity of the mitochondria and the cells.26 Flavonoids were also found to exert significant antileishmanial activity against L. donovani.27 In an attempt to decipher the mechanism of antileishmanial activity of flavonoids, catechins; type of flavonoid were found to form complexes with the parasite cell wall to influence processes requiring cell linking, and hence inhibit the parasite growth.28 Meanwhile, intracellular activities were reported for quercetin that inhibited enzyme topoisomerase II and chelated iron which is used in the replication of the parasite within the macrophage phagolysosomes.29
Nerolidol, a terpenoid commonly found in many plants exerts antileishmanial activity against L. amazonensis via inhibition of various biochemical pathways such as biosynthesis of dolichol, ergosterol, and ubiquinones.30 In addition, a triterpenoid dihydrobetulinic acid inhibited the growth of L. donovani amastigotes by targeting DNA topoisomerase I and II preventing DNA cleavage via forming enzyme–DNA complex, leading to apoptosis. Thus, the antileishmanial activity of the plant extracts, which are rich in phenols, flavonoids and terpenoids, may possibly be due to the specific components and/or synergistic interaction among various principles.31
Conclusion
The present study showed 80% methanol extracts from O. integrifolia and F. communis exhibited promising in vitro antileishmanial activity. This not only corroborates the traditional claim of the plants, it also provides clues for further examination of active principles of these plants for the development of effective and safe antileishmanial drugs.
Abbreviations
CL, cutaneous leishmaniasis; DMSO, dimethyl sulfoxide; IC50, the half maximal inhibitory concentration; MCL, mucocutaneous leishmaniasis; VL, visceral leishmaniasis.
Data Sharing Statement
The data used to support the findings of this study will be available from the corresponding author on reasonable request.
Acknowledgments
The authors are thankful to Mr. Melaku Wondafrash, Addis Ababa University, for identification of the plant material. The authors are thankful to department of microbiology, immunology and parasitology, faculty of medicine, college of health sciences for providing the laboratory setting.
Author Contributions
All authors contributed to data analysis, drafting, or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
No specific funding was granted for this project.
Disclosure
The authors report no conflicts of interest for this work and declare that there is no conflict of interest regarding the publication of this article.
References
1. WHO (2020) Leishmaniasis, Available at: https://www.who.int/news-room/factsheets/detail/leishmaniasis.
2. Alemayehu B, Alemayehu M. Leishmaniasis: a review on parasite, vector and reservoir host. Health Science Journal. 2017;11(4):1. doi:10.21767/1791-809X.1000519
3. McGwire BS, Satoskar AR. Leishmaniasis: clinical syndromes and treatment. QJM. Int J Med. 2014;107(1):7–14.
4. Didwania N, Shadab M, Sabur A, Ali N. Shadab, M., Sabur, A. and Ali, N. Alternative to Chemotherapy—The Unmet Demand against Leishmaniasis. Front Immunol. 2017;8:1779. doi:10.3389/fimmu.2017.01779
5. Leta S, Dao THT, Mesele F, Alemayehu G, Ghedin E. Visceral Leishmaniasis in Ethiopia: an Evolving Disease. PLoS Negl Trop Dis. 2014;8(9):e3131. doi:10.1371/journal.pntd.0003131
6. Assefa A. Leishmaniasis in Ethiopia: A systematic review and meta-analysis of prevalence in animals and humans. Heliyon. 2018;4(8):e00723. doi:10.1016/j.heliyon.2018.e00723
7. Ghorbani M, Farhoudi R. Leishmaniasis in humans: drug or vaccine therapy? Drug Des Devel Ther. 2017;12:25. doi:10.2147/DDDT.S146521
8. Ayalew H, Solomon Tadesse DB, Lindemann P, Tewabe Y, Hailu A, Chemical Composition AK. In Vitro Antipromastigotes Activity of Essential Oil of the Leaves of Discopodium pennnervium Hochst. EPJ. 2018;34(2):75–80.
9. Chekole G. Ethnobotanical study of medicinal plants used against human ailments in Gubalafto District, Northern Ethiopia. J Ethnobiol Ethnomed. 2017;13(1):55. doi:10.1186/s13002-017-0182-7
10. Geyid A, Abebe D, Debella A, et al. Screening of some medicinal plants of Ethiopia for their anti-microbial properties and chemical profiles. J Ethnopharmacol. 2005;97(3):421–427. doi:10.1016/j.jep.2004.08.021
11. Getahun A. Some Common Medicinal and Poisonous Plants Used in Ethiopian Folk Medicine. Addis Ababa: Faculty of Science, Addis Ababa University; 1976.
12. Enyew A, Asfaw Z, Kelbessa E, Nagappan R. Ethnobotanical study of traditional medicinal plants in and around Fiche District, Central Ethiopia. Res J Pharm Biol Chem Sci. 2014;6(4):154–167.
13. Yineger H, Kelbessa E, Bekele T, Lulekal E. Plants used in traditional management of human ailments at Bale Mountains National Park, Southeastern Ethiopia. J Med Plants Res. 2008;2(6):132–153.
14. Ramzi MA, Nawal A-M-M, Mohamed A-AF, Paul C, Louis M. Evaluation of the in vitro antiplasmodial, antipromastigotes, and antitrypanosomal activity of medicinal plants used in saudi and yemeni traditional medicine. Evid Based Complement Alternat Med. 2014;905639. doi:10.1155/2014/905639
15. Badirzadeh A, Heidari-Kharaji M, Fallah-Omrani V, Dabiri H, Araghi A, Salimi Chirani A. Antileishmanial activity of Urtica dioica extract against zoonotic cutaneous leishmaniasis. PLoS Negl Trop Dis. 2020;14(1):e0007843. doi:10.1371/journal.pntd.0007843
16. Essid R, Rahali FZ, Msaada K, et al. Antileishmanial and cytotoxic potential of essential oils from medicinal plants in Northern Tunisia. Ind Crop Prod. 2015;77:795–802. doi:10.1016/j.indcrop.2015.09.049
17. Tewabe Y, Kefarge B, Belay H, Bisrat D, Hailu A, Asres K. Antipromastigotes Evaluation of the Leaf Latex of Aloe macrocarpa, Aloin A/B, and Its Semisynthetic Derivatives against Two Leishmania Species. Evid Based Complement Alternat Med. 2019;2019.
18. Evans WC. Pharmacopoeial and Related Drugs of Biological Origin. Text Book of Pharmacognosy.
19. Shi P, Du W, Wang Y, Teng X, Chen X, Ye L. Total phenolic, flavonoid content, and antioxidant activity of bulbs, leaves, and flowers made from Eleutherine bulbosa (Mill.) Urb. Int J Food Sci Nutr. 2019;7(1):148–154.
20. Patel A, Patel A. Estimation of flavonoid, polyphenolic content and in vitro antioxidant capacity of leaves of Tephrosia purpurea Linn. (Leguminosae). Int J Pharma Sci and Res. 2010;1(1):66–77.
21. Van Griensven J, Gadisa E, Aseffa A, Hailu A, Beshah AM, Diro E. Treatment of cutaneous leishmaniasis caused by Leishmania aethiopica: a systematic review. PLoS Negl Trop Dis. 2016;10(3):3. doi:10.1371/journal.pntd.0004495
22. Iranshahi M, Arfa P, Ramezani M, et al. Sesquiterpene coumarins from Ferula szowitsiana and in vitro antileishmanial activity of 7-prenyloxycoumarins against promastigotes. Phytochemistry. 2007;68(4):554–561. doi:10.1016/j.phytochem.2006.11.002
23. Andrade MA, Azevedo CDS, Motta FN. Essential oils: in vitro activity against Leishmania amazonensis, cytotoxicity and chemical composition. BMC Complement Altern Med. 2016;16(1):444. doi:10.1186/s12906-016-1401-9
24. Mothana RA, Al-Musayeib NM, Al-Ajmi MF, Cos P, Maes L. Evaluation of the in vitro antiplasmodial, antileishmanial, and antitrypanosomal activity of medicinal plants used in Saudi and Yemeni traditional medicine. Evid Based Complement Alternat Med. 2014;2014.
25. Ayalew H, Solomon Tadesse DB, Lindemann P, et al. Chemical composition and in vitro antileishmanial activity of essential oil of the leaves of Discopodium pennnervium Hochst. EPJ. 2018;34(2):75–80.
26. Antwi CA, Amisigo CM, Adjimani JP, Gwira TM, Rafati S. In vitro activity and mode of action of phenolic compounds on Leishmania donovani. PLOS Neglected Tropical Diseases. 2019;13(2):e0007206. doi:10.1371/journal.pntd.0007206
27. Tasdemir D, Kaiser M, Brun R, Yardley V, Schmidt TJ, Tosun F. Antitrypanosomal and Antileishmanial Activities of Flavonoids and Their Analogues: in Vitro, In Vivo, Structure-Activity Relationship, and Quantitative Structure-Activity Relationship Studies. Antimicrob Agents Chemother. 2006;50(4):1352–1364. doi:10.1128/AAC.50.4.1352-1364.2006
28. Ogeto TK, Odhiambo RA, Shivairo RS, et al. Antipromastigotes activity of Aloe secundiflora plant extracts against Leishmania major. Adv Life Sci Tech. 2013;13:9–18.
29. De Arias AR, Pandolfi E, Celeste Vega MC, Rolón M. Selected natural and synthetic phenolic compounds with antileishmanial activity: a five-year review. Curr Bioact Compd. 2013;8(4):307–333. doi:10.2174/1573407211208040002
30. Arruda DC, D’Alexandri FL, Katzin AM, Uliana SRB. Antileishmanial activity of the terpene nerolidol. Antimicrob Agents Chemother. 2005;49(5):1679–1687. doi:10.1128/AAC.49.5.1679-1687.2005
31. Chowdhury AR, Mandal S, Goswami A, et al. Dihydrobetulinic acid induces apoptosis in Leishmania donovani by targeting DNA topoisomerase I and II: implications in antileishmanial therapy. Molecular Medicine. 2003;9(1–2):26–36. doi:10.1007/BF03402104
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.