Back to Journals » Cancer Management and Research » Volume 11
Impact of pelvic MRI in routine clinical practice on staging of IB1–IIA2 cervical cancer
Authors Zhang W , Chen C, Liu P, Li W, Hao M, Zhao W, Lu A, Ni Y
Received 8 December 2018
Accepted for publication 26 March 2019
Published 26 April 2019 Volume 2019:11 Pages 3603—3609
DOI https://doi.org/10.2147/CMAR.S197496
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lu-Zhe Sun
Weifeng Zhang,1 Chunlin Chen,1 Ping Liu,1 Weili Li,1 Min Hao,2 Weidong Zhao,3 Anwei Lu,4 Yan Ni5
1Department of Obstetrics and Gynecology, Nanfang Hospital, Southern Medical University, Guangzhou, People’s Republic of China; 2Department of Obstetrics and Gynecology, Shanxi Medical University Second Hospital, Taiyuan, People’s Republic of China; 3Department of Gynecological Oncology, Anhui Provincial Cancer Hospital, Hefei, People’s Republic of China; 4Department of Obstetrics and Gynecology, Guizhou Provincial Maternal and Child Health Care Hospital, Guiyang, People’s Republic of China; 5Department of Obstetrics and Gynecology, Yuncheng Municipal Central Hospital, Yuncheng Municipal Central Hospital, Yuncheng, People’s Republic of China
Purpose: To evaluate the impact of pelvic magnetic resonance imaging (MRI) on staging of IB1–IIA2 cervical cancer in routine clinical practice.
Patients and Methods: A total of 1,016 patients with IB1-IIA2 cervical cancer who underwent primary surgery and preoperative pelvic MRI between January 2009 and December 2015 were identified in a retrospective multicentre study. Data on clinical stage, MRI reports and surgicopathologic findings were extracted from medical records. The impact of MRI on clinical staging was evaluated by comparison before and after combination of MRI. Using surgicopathologic findings as the reference standard, the impact of pelvic MRI on the accuracy of clinical staging was evaluated. Furthermore, the impact on the accuracy of individual staging parameters such as maximal tumor diameter, vaginal involvement or parametrial infiltration were also evaluated.
Results: After combination of pelvic MRI, clinical stage remained unchanged in 59.7%, upstaged in 17.2%, and downstaged in 23.0% of the patients. The overall accuracy of clinical staging increased from 61.0% to 81.4% in our study (P<0.05). As for individual staging parameters, the area under the curve (AUC) for maximal tumor diameter increased from 0.58 to 0.81 (P<0.05). However, the AUC for vaginal involvement decreased from 0.61 to 0.57 (P>0.05). The AUC for parametrial infiltration was also suboptimal (AUC=0.56, P<0.05).
Conclusion: In routine clinical practice, MRI could increase the overall accuracy of clinical staging in IB1–IIA2 cervical cancer. For staging parameters, it only significantly increased the accuracy of maximal tumor diameter.
Keywords: cervical cancer, staging, magnetic resonance imaging, MRI, accuracy, routine clinical practice, surgery
Introduction
Even with the wide adoption of cervical cancer screening and the advent of human papillomavirus (HPV) vaccination, cervical cancer still poses a major risk for women worldwide, especially in developing countries.1 In China, about 87,982 new cases were diagnosed annually, and 23,375 patients died annually from this disease.2 The optimal treatment of cervical cancer depends in large part on the accuracy of clinical staging at presentation. In addition, clinical staging plays a fundamental role in cancer research, such as design of clinical trials or evaluation of treatment outcomes.3
Previously, clinical staging of cervical cancer relied on pelvic examination and several other simple radiologic examinations according to the International Federation of Gynecology and Obstetrics (FIGO).4 The accuracy of clinical staging was reported to range between 47% and 83.2% in cervical cancer.5,6 In 2018, clinical staging of cervical cancer underwent a major revision by FIGO. Radiologic examinations such as magnetic resonance imaging (MRI) are allowed to be combined into clinical staging where available.7 Because of the high soft tissue resolution and increasing availability, application of MRI in clinical staging is expected to become common practice in future.
However, the impact of MRI on clinical staging in routine practice is still unclear. Although most previous studies have demonstrated that MRI could significantly improve the accuracy of clinical staging, these studies are mainly single institutional studies with limited sample size, or prospective studies with dedicated radiologists.8–11 Hricak et al reported in a multicenter prospective study that the staging accuracy of MRI was significantly lower than prior single center studies.10 Hancke et al retrospectively analyzed 255 cervical cancer patients with primary surgical treatment and reported that the efficacy of MRI was lower than pelvic examination for pretreatment staging.12 Unless the incremental role of MRI can be confirmed in a multicenter study with the setting of routine practice, routine application of MRI in staging cervical cancer cannot be justified.
Therefore, the current study was performed and the purpose of this multicenter study was to evaluate the impact of MRI on overall staging of IB1–IIA2 cervical cancer in routine practice. Furthermore, the impact of MRI on the accuracy of individual staging parameters were also evaluated.
Patients and methods
After receiving approvals from the Institutional Review Board of Nanfang hospital and all other participating hospitals, medical records of patients with cervical cancer who were treated between January 2009 and December 2015 in 13 tertiary hospitals around China were retrospectively reviewed. All relevant data on clinical stage, MRI reports, and surgicopathologic findings were extracted from the patients’ original medical records by trained staff in our research team. Patient data were anonymized and de-identified prior to analysis. Therefore, individual consent was not necessary. Inclusion criteria were as follows: (1) pathologically diagnosed cervical cancer; (2) FIGO clinical stage IB1–IIA2; (3) primary surgery including type II or III radical hysterectomy plus bilateral pelvic lymphadenectomy and/or para-aortic lymphadenectomy; (4) MRI was performed within 2 weeks before operation. Patients with preoperative chemotherapy or radiotherapy were excluded to obviate the influence on pathologic examination. Moreover, patients with cone biopsy were also excluded because it is difficult to differentiate small cervical tumors from artifacts appearing on MRI from post-biopsy changes.13 Consequently, 1,016 eligible patients were included in the final analysis.
All patients were clinically staged according to the 2009 FIGO staging criteria.4 The impact of MRI on clinical staging was assessed by comparing original clinical stage with MRI modified clinical stage. The MRI modified clinical stage was formed based on original clinical stage and MRI reports according to the following rules. For maximal tumor diameter, the finding in the MRI report was allowed to revise the original clinical stage. For other staging parameters such as parametrial infiltration or vaginal involvement, positive findings in the MRI report were allowed to revise the original clinical stage, while negative findings in the MRI report were not allowed to restage the patient.
Based on surgicopathologic findings, surgical stage was formed according to the same FIGO staging criteria.4 Maximal tumor diameter was determined by measurements in the surgical record or the pathological report. Other parameters such as vaginal involvement or parametrial infiltration were determined by the pathological report. Using surgicopathologic stage as the reference standard, the impact of MRI on the accuracy of clinical staging was evaluated. Moreover, the accuracy of individual staging parameters such as maximal tumor diameter, vaginal invasion and parametrium infiltration were also evaluated using surgicopathologic findings as reference. Maximal tumor diameter was categorized as ≦4 cm or >4 cm. Parametrial infiltration was defined as direct tumor infiltration or nodal spread through the lymphatic or vascular systems.14
Statistical analyses
Quantitative data were presented as means with standard deviations. Qualitative data were given as absolute and percentages. The sensitivity, specificity, and predictive values for staging parameters were calculated. Receiver operating characteristic (ROC) curve analyses were conducted to assess the performance of clinical staging with and without MRI combination. The area under the curve (AUC) above 0.90 is considered to be of high diagnostic value, 0.70–0.90 is of moderate diagnostic value, and less than 0.70 is of poor diagnostic value.15
All analyses were performed using IBM SPSS Statistics 21.0 (IBM Corporation, Armonk, NY, USA ). A two-sided P-value <0.05 is considered as significant.
Results
Patient characteristics
A summary of the clinicopathological characteristics of the included 1,016 patients with stage IB1–IIA2 cervical cancer is provided in Table 1. The mean age of the patients was 48.68±10.18 years. With regard to pathologic type, squamous cell carcinoma was most prevalent (89.3%, 907), followed by adenocarcinoma (8.7%, 88), adenosquamous carcinoma (1.1%, 11) and others (1.0%, 10). Of the 1,016 patients, 81 (8.0%) patients had highly differentiated carcinoma, 536 (52.8%) patients had moderately differentiated carcinoma, 192 (18.9%) patients had poorly differentiated carcinoma, and 207 (20.4%) patients did not have a specified grade.
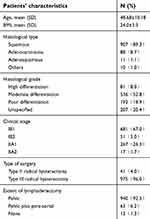 | Table 1 Clinicopathological characteristics of study population (N=1,016) |
Type III radical hysterectomy was the main type of surgery (96.0%, 975). Type II radical hysterectomy was performed on 41 patients (4.0%). As for lymphadenectomy, 940 (92.5%) patients underwent pelvic lymphadenectomy, 63 (6.2%) patients underwent both pelvic and para-aortal lymphadenectomy, and 13 (1.3%) patients did not receive lymphadenectomy.
The findings of MRI and its impact on clinical staging
Table 2 demonstrates the findings of pelvic MRI. No positive findings were found in 59 patients (5.8%). Meanwhile, the positive findings can be categorized as visible lesion in cervical stroma, vaginal invasion, parametrium infiltration or pelvic lymph node metastases. The incidence were 94.2%, 12.1%, 5.0% and 7.4%, respectively.
 | Table 2 The findings of MRI in 1,016 patients with cervical cancer |
After combination of MRI, 607 (59.7%) cases remained unchanged, 175 (17.2%) cases were upstaged, while 234 (23.0%) cases were downstaged (Table 3).
 | Table 3 The impact of MRI on clinical stage |
Impact of MRI combination on the accuracy of clinical staging
Using surgicopathologic stage as the reference standard, the overall accuracy of clinical staging increased from 61.0% to 81.4%, the rate of upstaging decreased from 27.7% to 11.9%, while the rate of downstaging decreased from 11.3% to 6.8% after combination of MRI (Table 4). Except in stage IIA2, the combination of MRI increased the accuracy of clinical staging in all other stages.
 | Table 4 The impact of MRI on the overall accuracy of clinical staging |
Table 5 shows the impact of MRI combination on the accuracy of staging parameters. As for maximal tumor diameter, the AUC of clinical staging increased from 0.58 to 0.81 (P<0.05). However, for vaginal invasion, the AUC of clinical staging decreased from 0.61 to 0.57 (P>0.05). For parametrial infiltration, the AUC were only 0.56, while sensitivity and specificity were 16.7% and 95.3%, respectively.
 | Table 5 The impact of MRI on the individual staging parameters (95%CI) |
Discussion
As a safe, repeatable, and non-radiating imaging technique, MRI is becoming preferred in the pretreatment staging of early stage cervical cancer. Our multicenter study demonstrated that MRI could significantly increase the accuracy of clinical staging even in routine clinical practice. The overall accuracy of clinical staging increased from 61.0% to 81.4% after combination of MRI in our study.
Our results are in accordance with most of the previous studies. A prospective study by Bourgioti et al reported that the AUC of clinical staging increased significantly from 0.59 to 0.84 after combination of MRI8 Another study by Kraljevic et al showed that MRI was better than clinical examination in staging of cervical carcinoma with 90.9% versus 79.0% accuracy rate.16 Contrary to these studies, Hancke et al reported that the accuracy of pelvic examination is higher than MRI (75% vs 58%). However, the accuracy of clinical staging was solely based on parametrial infiltration in their study. Therefore, instead of the overall accuracy of clinical staging, their results only reflect the accuracy of clinical staging for parametrial infiltration.
Although MRI could improve the overall accuracy of clinical staging, the improvement is not equal in each clinical stage. In our study, the accuracy of stage IB1 and IB2 increase from 85.9% and 41.2% to 92.6% and 79.2%, respectively. However, in stage IIA1, the accuracy only increased from 4.1% to 5.7%, and in stage IIA2, the accuracy even decreased from 17.6% to 9.5%. The unequal impact of MRI on each clinical stage stems from the heterogeneous effect of MRI on individual staging parameters. Therefore, the impact of MRI on each staging parameter was further discussed.
Maximal tumor diameter is a major staging parameter with significant impact on treatment planning and prognosis. Traditionally it is determined by pelvic examination with accuracy ranging between 64% and 68%.17,18 In exotic growth type, tumor diameter can be easily determined by pelvic examination. However, it is difficult to determine in endophytic growth type. Yang et al reported that the accuracy of stage IB2 by pelvic examination was 22% and 88% in endogenous type and exogenous type, respectively.6
Because of its high soft tissue resolution, MRI can delineate the contour of tumor clearly in a three-dimensional way.6,19 Measurement of tumor size using MRI has proven to be more accurate than pelvic examination.20,21 Epstein et al evaluated 128 cases of stage IA2–IIA cervical cancer patients with surgical treatment, and found that the accuracy of MRI in determining tumor diameter (>4 cm) is 93%.22 In our study, we found that the AUC of clinical staging for maximal tumor diameter increased from 0.58 to 0.81 after combination of MRI. Therefore MRI could significantly improve the accuracy of clinical staging in determining maximal tumor diameter.
Because of the unique anatomic location, pelvic examination has been thought to be of high accuracy in determining vaginal involvement.23,24 Contrary to this common belief, our study demonstrated that the AUC for vaginal invasion by pelvic examination was only 0.61, while the sensitivity and specificity were 48.7% and 72.9% respectively. Meanwhile, the efficacy of MRI was also suboptimal. The AUC of clinical staging after combination of MRI was only 0.57, while the sensitivity and specificity were 25.6% and 88.4% respectively. Both pelvic examination and MRI have lower accuracy than previous studies.5,8,25–27
The possible explanations for the low accuracy in determining vaginal involvement by pelvic examination are as follows. First, the mean age of the patients in our study was 48.68±10.18, which implies that a significant portion of patients were in menopause or postmenopause. The atrophied cervix in these patients makes the diagnosis of vaginal involvement difficult. Second, in case of suspicion, the treating physicians in China have a tendency to classify it as positive for concern of underestimation. In another retrospective study from China,28 the accuracy of stage IIA was reported to be as low as 35.3%, which also supports this assumption.
Vaginal invasion is detected when the normal low signal intensity of the vaginal wall was replaced by the high signal intensity tumor in MRI images. However, the distinction between vagina and cervix is difficult to discern in early stage cervical cancer. Exophytic tumors may expand the vaginal fornix and stretch the vaginal wall without invading it. The stretched and thin vaginal fornix may be difficult to recognize as intact, even though it is not disrupted by tumor.26,27 The use of vaginal gel in MRI scan was reported to allow a more accurate definition of vaginal fornix on MRI images.29
Parametrial infiltration is not only important in clinical staging, but also the shunting point of treatment modality.30 The radiological criteria for parametrial infiltration is based on the breach or disruption of the hypointense cervical stromal ring with nodular or irregular tumor signal intensity extending to the parametrium on T2-weighted MRI. A wide range of accuracy have been reported for the detection of parametrial infiltration by MRI.31 Bourgioti et al prospectively evaluated 115 patients with cervical cancer, and found that the addition of MRI to the clinical assessment increased the accuracy in determining parametrial infiltration. The AUC increased from 0.64 to 0.88, while the sensitivity and specificity were 73.33% and 92.50% respectively.8 However, Yang et al reported that the accuracy of MRI for parametrial infiltration was not reliable. There were many false-positive (14.9%) or false-negative (7.9%) cases.32
The wide range of detecting parametrial infiltration reflects the fact that high level of both equipment and reading skill are needed in determining this parameter. The diagnostic performance of MRI is reported to be subjected to many factors such as MRI device, scanning protocols and the experience of radiologists. Woo et al reported that using 3-T scanners and diffusion-weighted imaging (DWI) may improve diagnostic performance.33 DWI can provide functional information about water mobility, tissue cellularity, and the integrity of the cellular membranes. Combined with T2-weighted imaging, DWI can improve the diagnostic performance in detecting parametrial infiltration.34 The pooled sensitivity and specificity were higher in studies that used DWI than in those that did not (0.82 vs 0.72 for sensitivity; and 0.97 vs 0.91 for specificity; P<0.010).33
In our study, the AUC for parametrial infiltration was only 0.56, and the sensitivity was 16.7%. One possible explanation for the poorer performance of MRI is that parametrial infiltration has already been excluded from the study population by pelvic examination. Therefore parametrial infiltration in our study was more likely early or even micro-infiltration, which is less likely to be detected by MRI. Second, data were extracted retrospectively from medical records. Without dedicated radiologists with special interest and expertise for gynecological oncology, both implementation of the examination and interpretation of the images may be affected correspondingly. The high accuracy reported by previous studies might be the result of the use of more experienced and highly specialized radiologists, technically more advanced equipment and scanning protocols that are not universally available.
Our study has the following limitations. First, selection bias could be inherent given the study’s retrospective nature. MRI may be done mainly on patients whose pelvic examination was doubtful especially in the early period. Second, the potential impact of various imaging protocols, forms of MRI machine and experience of radiologist had not been evaluated. Third, the MRI report in the medical records was used and central review was not performed. More positive findings may be found if central review were performed.
With the above-mentioned limitations, our study has the merit of better reflecting the real situation in routine clinical practice. We did not reevaluate MRIs or pathologic specimens, and we relied on the original radiologic or pathologic reports for data analysis. Our study revealed the significant difference in the efficacy of MRI between routine practice and clinical trials. Therefore establishing a general protocol and enhancing the quality control of MRIs across different institutions are imperative.
In conclusion, our study demonstrated that MRI could improve the overall accuracy of clinical staging in early cervical cancer even in routine clinical practice. For staging parameters, it only significantly increases the accuracy of clinical staging for maximal tumor diameter.
Disclosure
Dr Chunlin Chen reports grants from the National Science & Technology Pillar Program of China during the 12th Five-year Plan period (2014BAI05B03), grants from Key Project of Science Program of Guangzhou for collaborative health and medicine innovation (201508020264), and grants from the Natural Science Foundation of Guangdong, China, during the conduct of the study (2015A030311024). The other authors report no conflicts of interest in this work.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi:10.3322/caac.21262
2. Chen W, Zheng R, Zeng H, Zhang S. The updated incidences and mortalities of major cancers in China, 2011. Chin J Cancer. 2015;34:502–507. doi:10.1186/s40880-015-0042-6
3. Benedet JL, Pecorelli S. Why cancer staging? Int J Gynecol Obstet. 2006;95(Supplement 1):S3. doi:10.1016/S0020-7292(06)60026-X
4. Bermudez A, Bhatla N, Leung E. Cancer of the cervix uteri. Int J Gynaecol Obstet. 2015;131(Suppl 2):S88–95. doi:10.1016/j.ijgo.2015.06.004
5. Ozsarlak O, Tjalma W, Schepens E, et al. The correlation of preoperative CT, MR imaging, and clinical staging (FIGO) with histopathology findings in primary cervical carcinoma. Eur Radiol. 2003;13:2338–2345. doi:10.1007/s00330-003-1928-2
6. Yang Z, Xu W, Ma Y, Liu K, Li Y, Wang D. (18)F-FDG PET/CT can correct the clinical stages and predict pathological parameters before operation in cervical cancer. Eur J Radiol. 2016;85:877–884. doi:10.1016/j.ejrad.2016.02.010
7. Bhatla N, Aoki D, Sharma DN, Sankaranarayanan R. Cancer of the cervix uteri. Int J Gynaecol Obstet. 2018;143(Suppl 2):22–36. doi:10.1002/ijgo.12611
8. Bourgioti C, Chatoupis K, Rodolakis A, et al. Incremental prognostic value of MRI in the staging of early cervical cancer: a prospective study and review of the literature. Clin Imaging. 2016;40:72–78. doi:10.1016/j.clinimag.2015.09.012
9. Meva J, Chaudhary RK, Bhaduri D, Bhatia M, Hatti S, Ba R. Lacunae in International Federation of Gynecology and Obstetrics (FIGO) classification for cervical carcinoma observational study using TNM classification as comparator. Int J Gynecol Cancer. 2013;23:1071–1077. doi:10.1097/IGC.0b013e31829783c4
10. Hricak H, Gatsonis C, Chi DS, et al. Role of imaging in pretreatment evaluation of early invasive cervical cancer: results of the intergroup study American College of Radiology imaging network 6651-gynecologic oncology group 183. J Clin Oncol. 2005;23:9329–9337. doi:10.1200/JCO.2005.02.0354
11. Dhoot NM, Kumar V, Shinagare A, Kataki AC, Barmon D, Bhuyan U. Evaluation of carcinoma cervix using magnetic resonance imaging: correlation with clinical FIGO staging and impact on management. J Med Imaging Radiat Oncol. 2012;56:58–65. doi:10.1111/j.1754-9485.2011.02333.x
12. Hancke K, Heilmann V, Straka P, Kreienberg R, Kurzeder C. Pretreatment staging of cervical cancer: is imaging better than palpation?: role of CT and MRI in preoperative staging of cervical cancer: single institution results for 255 patients. Ann Surg Oncol. 2008;15:2856–2861. doi:10.1245/s10434-008-0088-7
13. Fridsten S, Hellstrom AC, Hellman K, Sundin A, Soderen B, Blomqvist L. Preoperative MR staging of cervical carcinoma: are oblique and contrast-enhanced sequences necessary? Acta radiol open. 2016;5:2058460116679460. doi:10.1177/2058460116679460
14. Kong TW, Kim J, Son JH, et al. Preoperative nomogram for prediction of microscopic parametrial infiltration in patients with FIGO stage IB cervical cancer treated with radical hysterectomy. Gynecol Oncol. 2016;142:109–114. doi:10.1016/j.ygyno.2016.05.010
15. Linden A. Measuring diagnostic and predictive accuracy in disease management: an introduction to receiver operating characteristic (ROC) analysis. J Eval Clin Pract. 2006;12:132–139. doi:10.1111/j.1365-2753.2005.00598.x
16. Kraljevic Z, Viskovic K, Ledinsky M, et al. Primary uterine cervical cancer: correlation of preoperative magnetic resonance imaging and clinical staging (FIGO) with histopathology findings. Coll Antropol. 2013;37:561–568.
17. Van Nagell JR
18. Averette HE, Ford JH
19. Kim SH, Lee HJ, Kim YW. Correlation between tumor size and surveillance of lymph node metastasis for IB and IIA cervical cancer by magnetic resonance images. Eur J Radiol. 2012;81:1945–1950. doi:10.1016/j.ejrad.2011.04.053
20. Hricak H, Lacey CG, Sandles LG, Chang YC, Winkler ML, Stern JL. Invasive cervical carcinoma: comparison of MR imaging and surgical findings. Radiology. 1988;166:623–631. doi:10.1148/radiology.166.3.3340756
21. Mitchell DG, Snyder B, Coakley F, et al. Early invasive cervical cancer: tumor delineation by magnetic resonance imaging, computed tomography, and clinical examination, verified by pathologic results, in the ACRIN 6651/GOG 183 intergroup study. J Clin Oncol. 2006;24:5687–5694. doi:10.1200/JCO.2006.07.4799
22. Epstein E, Testa A, Gaurilcikas A, et al. Early-stage cervical cancer: tumor delineation by magnetic resonance imaging and ultrasound - A European multicenter trial. Gynecol Oncol. 2013;128:449–453. doi:10.1016/j.ygyno.2012.09.025
23. Greco A, Mason P, Leung AWL, Dische S, McIndoe GAJ, Anderson MC. Staging of carcinoma of the uterine cervix: MRI-surgical correlation. Clin Radiol. 1989;40:401–405.
24. Sala E, Rockall AG, Freeman SJ, Mitchell DG, Reinhold C. The added role of MR imaging in treatment stratification of patients with gynecologic malignancies: what the radiologist needs to know. Radiology. 2013;266:717–740. doi:10.1148/radiol.12120315
25. Choi SH, Kim SH, Choi HJ, Park BK, Lee HJ. Preoperative magnetic resonance imaging staging of uterine cervical carcinoma: results of prospective study. J Comput Assist Tomogr. 2004;28:620–627.
26. Sheu MH, Chang CY, Wang JH, Yen MS. Preoperative staging of cervical carcinoma with MR imaging: a reappraisal of diagnostic accuracy and pitfalls. Eur Radiol. 2001;11:1828–1833. doi:10.1007/s003300000774
27. Shweel MA, Abdel-Gawad EA, Abdel-Gawad EA, Abdelghany HS, Abdel-Rahman AM, Ibrahim EM. Uterine cervical malignancy: diagnostic accuracy of MRI with histopathologic correlation. J Clin Imaging Sci. 2012;2:42. doi:10.4103/2156-7514.99175
28. Qin Y, Peng Z, Lou J, Liu H, Deng F, Zheng Y. Discrepancies between clinical staging and pathological findings of operable cervical carcinoma with stage IB-IIB: a retrospective analysis of 818 patients. Aust N Z J Obstet Gynaecol. 2009;49:542–544. doi:10.1111/j.1479-828X.2009.01065.x
29. Balleyguier C, Sala E, Cunha TD, et al. Staging of uterine cervical cancer with MRI: guidelines of the European society of urogenital radiology. Eur Radiol. 2011;21:1102–1110. doi:10.1007/s00330-010-1998-x
30.
31. Kim M, Suh DH, Kim K, Lee HJ, Kim YB, No JH. Magnetic resonance imaging as a valuable tool for predicting parametrial invasion in stage IB1 to IIA2 cervical cancer. Int J Gynecol Cancer. 2017;27:332–338. doi:10.1097/IGC.0000000000000878
32. Yang K, Park W, Huh SJ, et al. Parametrial involvement on magnetic resonance imaging has no effect on the survival of early-stage cervical cancer patients. Int J Gynecol Cancer. 2017;27:507–513. doi:10.1097/IGC.0000000000000909
33.
34. Park JJ, Kim CK, Park SY, Park BK. Parametrial invasion in cervical cancer: fused T2-weighted imaging and high-b-value diffusion-weighted imaging with background body signal suppression at 3 T. Radiology. 2015;274:734–741. doi:10.1148/radiol.14140920
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
