Back to Journals » Infection and Drug Resistance » Volume 12
High incidence of multidrug-resistant Escherichia coli coharboring mcr-1 and blaCTX-M-15 recovered from pigs
Authors Shafiq M, Huang J, Ur Rahman S, Shah JM, Chen L, Gao Y, Wang M, Wang L
Received 21 March 2019
Accepted for publication 18 June 2019
Published 16 July 2019 Volume 2019:12 Pages 2135—2149
DOI https://doi.org/10.2147/IDR.S209473
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Joachim Wink
Muhammad Shafiq,1 Jinhu Huang,1 Sadeeq Ur Rahman,2 Jan Mohammad Shah,1 Li Chen,1 Yi Gao,1 Mengli Wang,1 Liping Wang1
1Moe Joint International Research Laboratory of Animal Health and Food Safety, Laboratory of Veterinary Pharmacology and Toxicology, College of Veterinary Medicine, Nanjing Agricultural University, Nanjing, 210095, People’s Republic of China; 2College of Veterinary Sciences and Animal Husbandry, Section Microbiology, Abdul Wali Khan University, Mardan, KP, Pakistan
Purpose: The coexistence of mobile colistin (COL)-resistant gene mcr-1 with extended-spectrum beta-lactamase (ESBL) gene in Escherichia coli has become a serious threat globally. The aim of this study was to investigate the increasing resistance to COL and in particular its coexistence with ESBL-producing E. coli recovered from pig farms in China.
Materials and methods: E. coli were isolated from 14 pig farms in Jiangsu China. Susceptibility testing was identified by micro-dilution method. PCR assay and nucleotide sequencing were used to detect COL-resistant genes, mcr-1 to −5, as well as ESBL genes, blaCTX-M, blaSHV and blaTEM. Conjugation experiment, plasmid replicon typing of the multidrug resistance (MDR), S1-PFGE and DNA southern hybridization were performed to study the transferability of these genes.
Results: Overall, 275 E. coli isolates were recovered from a total of 432 cloacal and nasal swabs. More than 90% of the isolates were MDR, of which 70.18% were resistant to COL. Of these 275 isolates, mcr-1 was identified as the most predominant gene carried by 71.63% (197/275) of isolates, 39.59% (78/197) of the isolates were harboring both mcr-1 and ESBL genes (blaCTX-M, blaSHV and blaTEM). ESBL genotyping showed that blaCTX-M was the most predominant ESBL (68.49%) followed by blaSHV (16.4%) and blaTEM (15%). Sequencing revealed that the most common variants of blaCTX-M identified were, blaCTX-M-15 (69%), blaCTX-M-55 (29%) and blaCTX-M-1 (1.8%). IncHI2, IncFIB, IncFIC, IncN and IncX4 were found to be the most common Inc-types found both in donors and in transconjugants and were associated with the transfer of the mcr-1 and ESBL encoding genes. Six strains carried a total of five different plasmids: approximately 97-, 130-, 160-, 227- and 242-kb plasmids.
Conclusion: The coexistence of the mcr-1- and blaCTX-M-15-carrying isolates displaying high MDR, recovered from E. coli of pig origin, is a major concern for both humans and veterinary medicine.
Keywords: E. coli, colistin, mcr-1, ESBL, coexistence
Introduction
Antimicrobial resistance (AMR) has now been widely recognized as a crucial threat to human and animal health as the extensive use of antimicrobials in humans as well as in food-producing animals.1 Global consumption of antimicrobials in animal settings may rise up to 67% by 2030, determined predominantly by BRICS (Brazil, Russia, India, China and South Africa) countries, as large-scale and intensive farming operations are greatly in demand with the upsurge in revenue and animal protein consumption.2,3 This heavy antimicrobial practice creates a selective pressure that contributes to the emergence and spread of bacterial resistance. One of the major concerns is the rapid increase of the multidrug-resistant (MDR) Escherichia coli in animal settings and clinical medicine.4–6 This is not only because of the lessened number of useful antimicrobials for curing MDR E. coli infections, but also due to the potential transfer of MDR E. coli strains from animals to humans, especially which producing extended-spectrum β-lactamases (ESBLs) and carbapenemases, and display resistance to colistin (COL).7
ESBLs are β-lactamases that confer resistance to oxyimino “second- and third-generation” cephalosporin’s (eg, cefotaxime (CTX), ceftriaxone and ceftazidime) and aztreonam.8,9 ESBL-producing bacteria were first reported in 1980, soon after the introduction of the third-generation cephalosporin’s (CTX and ceftiofur (CEF) into clinical settings.10 Currently, there are more than 350 ESBL genes that have been reported, and these genes are commonly developed through point mutations of the classical SHV-1 and TEM-1 β-lactamases and more increasingly prominent the CTX-M types.11–13 Among the CTX-M enzymes, blaCTX-M-55 has become the leading CTX-M type in ESBL-producing E. coli isolates of animal origin during the last decade.14,15 In contrast, blaCTX-M-15 seems to be the most extensive types in isolates of human origin.16 ESBL-producing E. coli are highly linked with multiple plasmids and studies have reported that ESBL genes are often carried on IncF, IncI1, IncN, IncHI1 and IncHI2 in food-producing animals worldwide.13,17–19 There is potential for ESBL genes/plasmid spreading between E. coli from animals, food and humans.20,21
The co-occurrence of ESBL genes and mcr-1 in E. coli was reported from China in 2016.22 Rhouma and Letellier assumed that a historic relation existed between ESBL genes, carbapenemase genes and mcr-1.23 A recent study proposed that cephalosporin resistance is commonly spread in animals and humans through distinct plasmids.24 It is highly expected that food-producing animals have become the most significant reservoirs in disseminating these resistance genes in the community through horizontal gene transfer. To assess the co-occurrence and emergence of mcr and ESBL genes in E. coli of pig source, we examined 14 pig farms of Jiangsu province in China to evaluate the current scenario of these resistant genes in pigs and further clarified the predominant genotype and plasmids diversity of mcr and ESBL genes.
Materials and methods
Collection of samples
A total of 432 samples (400 from healthy and 32 from dead pigs) were collected from 14 commercial pig farms in Jiangsu, China (Figure S1), during the period of August 2016 until December 2017. From each farm, samples were randomly collected. The anal swabs were collected by inserting the swab into the rectum and being rotated. To collect nasal swabs from swine, the nose was wiped with a piece of paper and a sterile swab was inserted into the nasal cavity and rotated for 3 s at 90°. From 32 dead pigs, all samples were aseptically obtained from different organs. All collected samples were immediately transported at 4°C to the laboratory for microbial examination and processed within 4 hrs.
Isolation and screening of ESBL-producing and COL-resistant E. coli
All samples were directly streaked onto MacConkey agar (Binhe Microorganism Reagent Co. Ltd., Hangzhou, China) supplemented with CTX (1 µg/mL) and COL (2 µg/mL) for the screening of possible ESBL-producing and COL nonsensitive E. coli as previously described.25 Plates were incubated at 37°C for 18–24 hrs. Presumptive E. coli colonies with dark pink to red colors were confirmed microscopically and further verified by species-specific PCR as described previously.26 Confirmed E. coli strains were stored in Luria–Bertani medium (Oxoid, United Kingdom) containing 40% (vol/vol) glycerol in aliquots at −80°C until further use.
ESBL-producing E. coli were further confirmed by double-disk synergy (DDS) testing as recommended by the Clinical and Laboratory Standards Institute (CLSI) guideline, using antibiotic discs of ceftazidime (30 µg), ceftazidime plus clavulanic acid (30/10 µg), CTX (30 µg) and CTX plus clavulanic acid (30/10 µg). DDS test was performed for phenotypic detection of ESBLs. The test result is considered as positive if the zone of inhibition is ≥5 mm larger with clavulanic acid than without.27
Antimicrobial susceptibility testing
Antimicrobial susceptibility was performed by minimum inhibitory concentration (MIC) determination using broth micro-dilution method against 11 antibiotics for all 275 isolates and 17 antibiotics for transconjugants. The MIC data was interpreted according to the CLSI recommendations.27 Antibiotics used in this study, comprised of 5 β-lactams – ampicillin (AMP), CTX, cefoxitin (CFX), CEF and meropenem (MEM) – and 12 non-β-lactams – COL, ciprofloxacin (CIP), chloramphenicol (CHL), enrofloxacin (ENR), gentamycin (GEN), kanamycin, nalidixic acid, polymyxin-B (POL-B), tetracycline (TET), trimethoprim, streptomycin and sulfamethoxazole. The MIC of COL was determined by broth micro-dilution method recommended by the joint CLSI-EUCAST polymyxin breakpoints working group (www.EUCAST.org), CLSI VET01-A4 is used for CEF and ENR which are missing in the human CLSI M100-S27. E. coli ATCC 25922 was used as a quality control in antimicrobial susceptibility testing. Isolates that exhibited resistance to more than 3 antimicrobial agents were classified as MDR.28
PCR assays for detection of mcr and ESBL genes
PCR assay was used to detect COL-resistant genes mcr-1 to mcr-5 as well as ESBL genes (blaCTX-M, blaSHV and blaTEM). Total DNA was isolated by conventional boiling method. All these resistant genes were screened via PCR-based diagnostics with specific primers, as previously described. All the primers and PCR conditions used in this study are listed in Table 1. All PCR positive amplicons of these targeted genes were sequenced by Sanger sequencing in TSINGKE Corporation (Nanjing, PR China).
 |
Table 1 PCR primers and conditions used in this study |
Conjugation experiment
To determine the transferability of resistance genes, 15 COL-resistant E. coli isolates were selected as donors for conjugation. E. coli EC-600 (NalR, RifR) was used as recipient bacteria. Conjugation experiments were performed as previously described.29 These putative transconjugants were further confirmed using antibiotic susceptibility testing, PCR detection and plasmid incompatibility (Inc) groups typing carried by the transconjugants.
Plasmid replicon typing
Plasmid DNA was extracted from both donors and transconjugants using the Wizard Genomic DNA Purification kit (Promega) and was characterized by PCR-based replicon typing method (PBRT). Eighteen pairs of primers were designed to perform 5 multiplex and 3 simples PCRs targeting the FIA, FIB, FIC, HI1, HI2, IncI1, L/M, N, P, W, T, A/C, K, B/O, X, Y, F and FIIA replicons as previously mentioned.30 While primers for two other plasmids IncI2 and IncX4 were designed separately which were missing in the previous replicon typing. All the PBRT primers and PCR conditions used in this study are listed in the Supplementary Table S1. PCR amplicons of plasmids were sequenced by Sanger sequencing (TSINGKE Corporation, Nanjing, PR China) and retrieved sequences were used to confirm replicon types by using BLAST tool available at NCBI web (https://blast.ncbi.nlm.nih.gov/Blast.cgi).
Pulsed field gel electrophoresis (PFGE) and Southern hybridization
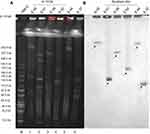
To determine the genetic relatedness and location of transmissible mcr-1-positive elements, the six conjugative E. coli strains were characterized by S1-PFGE and Southern hybridization using a probe specific for mcr-1. Genomic DNA from each of the isolate was digested with S1 nuclease (Thermo scientific) and was examined by PFGE as previously described.31
Southern hybridizations of plasmid DNA were performed with a digoxin-labeled mcr-1-specific probe according to the manufacturer’s instructions (Roche Diagnostics, Mannheim, Germany) as previously described.32
Statistical analysis
Differences in the AMR profiles of E. coli isolates with or without mcr-1 were assessed by a two-tailed Chi-square test or Fisher’s exact test using the Statistical Packages of Social Sciences software for Windows, version 20.0 (IBM Corp., Armonk, NY), with P<0.05 set as the level of significant differences.
Results
Bacterial isolation and antimicrobial susceptibility
Overall, 275 E. coli isolates (243 from healthy and 32 from dead pigs) were recovered from 432 samples of 14 different pig farms. All isolates were observed to be ESBL-producing and COL nonsensitive as determined by phenotypic approaches, giving a carriage rate of 63.6% (275/432). Of these 275 E. coli isolates, 174 (63.27%) were from feces, 69 (25.09%) were from nasal and remaining 32 (11.63%) were from diseased and dead pigs.
The MICs were obtained from the antibiotic susceptibility testing for all isolates. To determine the resistance profiles of 275 representative E. coli strains, susceptibility of 11 antibiotics were used (Table 2). Of the 275 E. coli isolates, the resistant rate to TET was 97.81%, followed by AMP (96.72%), CHL (94.54%), CFX (86.18%), CTX (78.18%), CEF (77.81%), CIP (73.81%), POL-B (71.27%), GEN (70.54%), and COL (70.18%). In contrast, the most effective antibiotic against these isolates was MEM with 99.6% susceptibility. Majority of the E. coli strains showed considerable MDR to β-lactams and several non-β-lactams groups, including polypeptides group, fluoroquinolones, aminoglycosides, amphenicol, quinolone group, sulfonamides and TET.
 |
Table 2 Distribution of MICs of 11 antibiotics for 275 MDR Escherichia coli isolates |
mcr-1 and ESBL genes are prevalent among E. coli strains
Although 70.18% (193 of 275) swine E. coli isolates conferred resistance to COL, the mcr-1 carriage rate was 71.63% (197/275) (Table 3) and only mcr-1 gene was detected in these COL-resistant E. coli isolates. No other colistin-resistant gene (mcr-2 to mcr-5) could be detected in the study population of E. coli isolates. The mcr-1 gene was detected in all farms, and the prevalence rate was enormously high 71.6%, ranging from 47.8% to 100% in different farms, while 40.6% in diseased isolates (Table 3).
 |
Table 3 Prevalence of mcr-1 & or ESBL-producing E. coli in swine samples collected from different farms of Jiangsu China |
We recovered a total of 146 (53.09%) ESBL-producing E. coli strains from 275 samples collected from 14 different farms of pigs (Figure S2 and Table 5). Among the 146 ESBL-producing isolates, 68.49% (100/146) harbored blaCTX-M genes, 22.60% (33/146) harbored blaSHV, while 18.49% (27/146) were carrying blaTEM genes. Among them, 6 isolates carried blaCTX-M and blaSHV, 3 isolates contained blaCTX-M and blaTEM, and one isolate had three genes of blaCTX-M, blaSHV, and blaTEM.
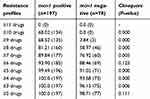 |
Table 4 The resistance patterns of E. coli with or without mcr-1 |
 |
Table 5 Distribution of various resistance genes among 275 MDR Escherichia coli isolates from pigs |
The resistance patterns of E. coli are different in mcr-1-positive and mcr-1-negative isolates
The mcr-1-positive E. coli isolates displayed more resistance to other antimicrobials than those of mcr-1-negative isolates (Table 4). For mcr-1-positive E. coli, all isolates, 100% (197/197), possessed not less than 3 antibiotics resistance pattern, while about 96.15% (75/78) of COL-negative isolates did (P=0.006), with 3 COL-sensitive isolates which displayed resistance to not more than 2 antibiotics. In addition, about two-thirds of mcr-1-positive E. coli isolates, 68.52% (135/197), showed resistance to at least 9 drugs, but only 3.84% (3/78) of mcr-1-negative isolates did (P=0.000). Interestingly, 68.02% (134/197) of mcr-1-positive strains presented resistance profiles to 10 drugs, but no COL-negative E. coli did (P=0.000).
Coexistence of mcr-1 and ESBL genes screened among the strains
Based on the results of this study, among 197 mcr-1 positive isolates, 39.59% (78/197) were identified as carrying both mcr-1 and ESBL genes. Distribution of all mcr-1 positive E. coli isolates (n=78) harboring ESBL genes are analyzed and presented in Figure 1 and Table 5. Our findings indicate that the combination of mcr-1 with blaCTX-M was the most prevalent with the rate of 70.51% (55/78) followed by the combination of mcr-1 and blaSHV (14.10%, 11/78). Finally, 7.69% (6/78) isolates were identified carrying both mcr-1 and blaTEM. Furthermore, combination of mcr-1 with two or more than two ESBL genes was also identified. Results showed that a total of 5.12% (4/78) of mcr-1-positive isolates also carried blaCTX-M and blaSHV, while 1.28% (1/78) of mcr-1-positive isolates carried blaCTX-M and blaTEM. Interestingly, a single isolate was carrying (mcr-1+ blaCTX-M + blaSHV+ blaTEM). In this study, mcr-1 and blaCTX-M were identified as the dominant genes (Table 5).
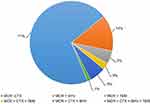 |
Figure 1 Distribution of various resistance genes in combination. |
As mcr-1 in combination with blaCTX-M were identified as the most prevalent (70.51%, 55/78), we further sequenced the blaCTX-M genes to explore the subtypes. Sequencing analysis of these 55 blaCTX-M isolates showed that all these mcr-1-positive isolates were harboring blaCTX-M-1 group. The most prevalent variants identified in these 55 isolates belonged to this group were blaCTX-M-15 in 38/55 (69%) isolates, followed by blaCTX-M-55 in 16/55 (29%) isolates and blaCTX-M-1 in one isolate (1.8%).
mcr-1 and ESBL genes could be conjugative transfer by plasmids with different replicon type
Conjugation experiments were performed on random 15 mcr-1 positive isolates. Of the 15 mcr-1 resistant isolates, 10 isolates were carrying additional bla-CTX-M, while a single isolate was harboring blaTEM. Of these 15 isolates, 12 were successfully transferred to E. coli EC-600. The resistance profiles of the 12 transconjugants were identical to those of the mcr-1 and blaCTX-M carrying E. coli donor isolates, indicating the transfer of antibiotic resistance. In addition, resistant to several non-β-lactam antibiotics, such as aminoglycosides, fluoroquinolones, TET, macrolides and sulfonamides, were also co-transferred along with COL and β-lactam resistance. MICs of COL of these transconjugants revealed 4- to 8-fold increase as compared with the recipient EC-600 (0.125 µg/mL).
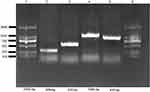
PCR-based replicon typing (PBRT) showed that in the E. coli isolates carrying mcr-1 and blaCTX-M, the plasmids with different replicons, including IncHI2 (n=7), IncFIB (n=7), IncFIC (n=4), IncP (n=4), IncFrepB (n=4), IncN (n=3), IncX4 (n=2) IncY (n=2) and IncI1 (n=1), were detected in the donor strains (Figure 2 and Table 6). However, PBRT of the transconjugants confirmed only five replicons, IncHI2, IncFIB, IncFIC, IncN and IncX4, which were present in both donors and transconjugants and were associated with the transfer of the mcr-1 and ESBL genes.
 |
Table 6 Conjugation experiments and plasmid replicon type detection for 15 mcr-1-positive E. coli |
 |
Figure 2 Detection of plasmid replicon types in multidrug-resistant E. coli using PCR assay. |
S1-PFGE analysis demonstrated that these six strains carried multiple plasmids varying in sizes ranging from ~97 kb to 242 kb (Figure 3A). Southern hybridization assay confirmed that the mcr-1 gene recovered from these six strains was positioned on the following five different types of plasmids: with the size of approximately 97, 130, 160, 227 and 242 kb, respectively (Figure 3B).
Discussion
China alone produces and consumes roughly half the planet’s pigs, about 500 million annually, and has been the leading consumption of antibiotics in the world.33 The increased usage of antibiotics may trigger the emergence of AMR. Reports on emergence of AMR particularly resistance of β-lactam and COL are increasing all over the world.34–37 The prevalence of ESBL in animal origin has been rising since 2003, with slight variances amongst terrestrial regions and different animal species.4,14,38 We report on the high incidence of mcr-1-carrying ESBL-producing E. coli recovered from pigs in Jiangsu, China. Our results indicated that all the mcr-1 and ESBL-producing E. coli isolates showed MDR. The majority of these isolates (77–86%) showed resistance to cephalosporin (Table 2). In addition, high resistance was also observed to common β-lactam and non-β-lactam antimicrobials such as AMP, fluoroquinolones, aminoglycosides, amphenicol, quinolones, sulfonamides and TET which are commonly using in human as well in veterinary practice. Many recent studies have reported MDR ESBL-producing E. coli isolated from poultry,3 pigs,39 cattle36 and humans.40
Recently, plasmid-mediated COL-resistant genes mcr-1 to mcr-8 have been widely discovered around the world.34,41–47 Herein, we screened 275 MDR E. coli isolated from 14 pig farms from Jiangsu province for the presence of mcr-1 to mcr-5 genes. Only mcr-1 gene was detected in isolates from every farm, and the carriage rate was extremely high in 71.6% (197/275). The high prevalence rate of the mcr-1 found in this study from pigs in Jiangsu is consistent with very recent reports from China.48,49 These recent studies in pigs reported similarly high mcr-1-positive carriage (79.2% and 76.2%), ranging from 45% to 100% in different provinces, while the mcr-1 rate in Jiangsu province reported by48 was 71.9% which is very similar to our findings 71.6%. The present study and, together with the previous studies, confirmed a surprisingly high rate of mcr-1 in swine farms and is likely associated with the prolonged and extensive practice of COL as a growth promoter in pigs.
The coexistence of mcr-1 with other resistance genes in an E. coli was reported in China.50 One recent study also assumed that a historic bridge existed between mcr-1 and ESBL.23 However, there is scarcity in the incidence of the coexistence of mcr-1 and ESBL in the pig origin. Herein, we screened 275 MDR E. coli–resistant strains from pig farms in Jiangsu during 2016–2017 and found a high occurrence of mcr-1-positive strains with ESBL in the swine 39.5% (78/197), which was very high from the previous report.51 A very recent longitudinal study from China investigated the co-rising of mcr-1 and ESBL in chicken isolates.14 Among ESBL-positive strains, we found that blaCTX-M is the most predominant.
In this study, 39.5% mcr-1-positive E. coli strains were detected which coexist in different ESBL genes. Among them, blaCTX-M was the predominant one which was found in 55 mcr-1-positive E. coli 70.5% (Figure 1). On sequence-based analysis of these 55 blaCTX-M isolates, interestingly the blaCTX-M-15 gene was found to be the most prevalent blaCTX-M gene (69%) followed by blaCTX-M-55 (29%). The spread of the blaCTX-M-15 gene is a common blaCTX-M enzyme and detected widely in Enterobacteriaceae of human origin.19,52 Very few studies have reported the co-occurrence of mcr-1 and blaCTX-M-15 of human origin.22,53 From China, one recent study in dairy cows also found blaCTX-M-15 as the second prevalent ESBL gene 21.4% (62/275), but there was no mcr-1 gene detected in those blaCTX-M-15 isolates.54 Another study also reported the coexistence of mcr-1 and blaCTX-M-15 in Turkey hen meat.55 Herein, this is the first investigation that reveals the coexistence of mcr-1 and blaCTX-M-15 in E. coli strains of pig origin in a very high proportion.
A significant increase in the blaCTX-M-55 was found in E. coli by over a period of ten years.4 Many previous studies reported that blaCTX-M-55 in human strains in China has become the second dominant blaCTX-M type and even the occurrence of blaCTX-M-55 was higher than blaCTX-M-15.51 A high rate of mcr-1 and blaCTX-M-55 was recently detected from the chicken origin in China,14 which was consistent with our results. Thus, the concurrent dissemination of the mcr-1 harboring blaCTX-M-15 and blaCTX-M-55 mediated by a single bacterial clone is existing which suggests that mcr-1 is found in the diverse reservoirs.
In addition, blaCTX-M-15 and blaCTX-M-55 were previously reported on conjugative plasmids, ie, FIB, IncI1, IncHI2, IncK, IncP and IncN.35,56 Therefore, we also detected these incompatibility types by PCR typing. While mcr-1 gene was often found on conjugative plasmids like IncF, IncI2, IncHI2, IncN, IncP and X4, which exhibit an unexpected diversity.57 In conjugation experiment, IncHI2, IncFIB, IncFIC, IncN and IncX4, were found in both donors and transconjugants and were associated with the transfer of the mcr-1 and ESBL encoding genes.
Genetic representation of mcr-1-carrying plasmids demonstrated that this gene is located on different conjugative elements of ~97 kb and 242 kb in size. The fact that mcr-1-carrying E. coli isolates display divergent PFGE profiles suggests that these elements may play a vital role in mcr-1 transmission. The incidence of closely related plasmids that carry mcr-1 and ESBL resistance genes among genotypically varied E. coli strains from various origins is a threat for alarm as it indicates that plasmids can easily disseminate from animals to humans and the spread of these plasmids may be remarkably challenging to control.
Considering that blaCTX-M has become the most prevalent ESBL type of animal origin in the last few years, this situation may suggest that mcr-1 and blaCTX-M emerged and arose due to the extensive use of antimicrobial practice in animal farming in the last decade. Our results also suggested that blaCTX-M-15 and blaCTX-M-55 and other β-lactamase genes coharboring with mcr-1 positive isolates is a potential threat to public health as the pig carrying these genes may enter the food chain. It is recommended that we should pay high consideration in monitoring the incidence of ESBL-producing & COL-resistant E. coli in both clinical and food-producing animals.
Conclusion
Our study reported a high incidence of the mcr-1-carrying ESBL-producing E. coli recovered from pigs in Jiangsu, China. The coexistence of the mcr-1- and blaCTX-M-15-carrying isolates displaying MDR, recovered from pig origin is a major concern for both humans and veterinary medicine. The presence of these genes on the conjugative plasmids with the ability to transfer between similar strains which contain other drug resistance genes emphasizes on urgent intervention.
Acknowledgments
This work was supported by the National Key Research and Development Program of China (2018YFD0500300), the National Natural Science Foundation of China (31702292, 31872517), the Natural Science Foundation of Jiangsu Province (BK20170710), the China Postdoctoral Science Foundation (2017M611841, 2018T110515) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Global Action Plan On Antimicrobial Resistance. World Health Organization; 2015. Available from: http://www.wpro.who.int/entity/drug_resistance/resources/global_action_plan_eng.pdf. Accessed July 2, 2019.
2. Van Boeckel TP, Brower C, Gilbert M, et al. Global trends in antimicrobial use in food animals. Proc Natl Acad Sci U S A. 2015;112(18):5649–5654. doi:10.1073/pnas.1503141112
3. Brower CH, Mandal S, Hayer S, et al. The prevalence of extended-spectrum beta-lactamase-producing multidrug-resistant Escherichia coli in poultry chickens and variation according to farming practices in Punjab, India. Environ Health Perspect. 2017;125(7):077015. doi:10.1289/EHP36
4. Rao L, Lv L, Zeng Z, et al. Increasing prevalence of extended-spectrum cephalosporin-resistant Escherichia coli in food animals and the diversity of CTX-M genotypes during 2003–2012. Vet Microbiol. 2014;172(3–4):534–541. doi:10.1016/j.vetmic.2014.05.029
5. Kong LH, Lei CW, Ma SZ, et al. Various sequence types of escherichia coli isolates coharboring blaNDM-5 and mcr-1 genes from a commercial swine farm in China. Antimicrob Agents Chemother. 2017;61(3). doi:10.1128/AAC.02167-16
6. Wang R, Liu Y, Zhang Q, et al. The prevalence of colistin resistance in Escherichia coli and Klebsiella pneumoniae isolated from food animals in China: coexistence of mcr-1 and blaNDM with low fitness cost. Int J Antimicrob Agents. 2018;51(5):739–744. doi:10.1016/j.ijantimicag.2018.01.027
7. Jackson CR, Davis JA, Frye JG, Barrett JB, Hiott LM. Diversity of plasmids and antimicrobial resistance genes in multidrug-resistant escherichia coli isolated from healthy companion animals. Zoonoses Public Health. 2015;62(6):479–488. doi:10.1111/zph.2015.62.issue-6
8. Bush K, Jacoby GA. Updated functional classification of β-lactamases. Antimicrob Agents Chemother. 2010;54(3):969–976.
9. Moawad AA, Hotzel H, Neubauer H, et al. Antimicrobial resistance in Enterobacteriaceae from healthy broilers in Egypt: emergence of colistin-resistant and extended-spectrum β-lactamase-producing Escherichia coli. Gut Pathog. 2018;10:39. doi:10.1186/s13099-018-0266-5
10. Yamasaki S, Le TD, Vien MQ, Van Dang C, Yamamoto Y. Prevalence of extended-spectrum beta-lactamase-producing Escherichia coli and residual antimicrobials in the environment in Vietnam. Anim Health Res Rev. 2017;18(2):128–135. doi:10.1017/S1466252317000160
11. Pitout JD, Nordmann P, Laupland KB, Poirel L. Emergence of Enterobacteriaceae producing extended-spectrum beta-lactamases (ESBLs) in the community. J Antimicrob Chemother. 2005;56(1):52–59. doi:10.1093/jac/dki166
12. Cheng VC, Wong SC, Ho PL, Yuen KY. Strategic measures for the control of surging antimicrobial resistance in Hong Kong and mainland of China. Emerg Microbes Infect. 2015;4(2):e8. doi:10.1038/emi.2015.8
13. Brolund A, Sandegren L. Characterization of ESBL disseminating plasmids. Infect Dis (Lond). 2016;48(1):18–25. doi:10.3109/23744235.2015.1062536
14. Feng S, Shen C, Chen H, et al. Co-production of MCR-1 and NDM-5 in Escherichia coli isolated from a colonization case of inpatient. Infect Drug Resist. 2018;11:1157–1161. doi:10.2147/IDR.S171164
15. Abbas G, Khan I, Mohsin M, Sajjad Ur R, Younas T, Ali S. High rates of CTX-M group-1 extended-spectrum beta-lactamases producing Escherichia coli from pets and their owners in Faisalabad, Pakistan. Infect Drug Resist. 2019;12:571–578. doi:10.2147/IDR.S189884
16. Woerther PL, Burdet C, Chachaty E, Andremont A. Trends in human fecal carriage of extended-spectrum beta-lactamases in the community: toward the globalization of CTX-M. Clin Microbiol Rev. 2013;26(4):744–758. doi:10.1128/CMR.00023-13
17. Carattoli A. Plasmids in Gram negatives: molecular typing of resistance plasmids. IJMM. 2011;301(8):654–658. doi:10.1016/j.ijmm.2011.09.003
18. Casella T, Nogueira MCL, Saras E, Haenni M, Madec JY. High prevalence of ESBLs in retail chicken meat despite reduced use of antimicrobials in chicken production, France. Int J Food Microbiol. 2017;257:271–275. doi:10.1016/j.ijfoodmicro.2017.07.005
19. Day MJ, Rodriguez I, van Essen-Zandbergen A, et al. Diversity of STs, plasmids and ESBL genes among Escherichia coli from humans, animals and food in Germany, the Netherlands and the UK. J Antimicrob Chemother. 2016;71(5):1178–1182. doi:10.1093/jac/dkv485
20. Dolejska M, Papagiannitsis CC. Plasmid-mediated resistance is going wild. Plasmid. 2018. doi:10.1016/j.plasmid.2018.09.010
21. Bi Z, Berglund B, Sun Q, et al. Prevalence of the mcr-1 colistin resistance gene in extended-spectrum beta-lactamase-producing Escherichia coli from human faecal samples collected in 2012 in rural villages in Shandong Province, China. Int J Antimicrob Agents. 2017;49(4):493–497. doi:10.1016/j.ijantimicag.2016.12.018
22. Zhang H, Seward CH, Wu Z, Ye H, Feng Y. Genomic insights into the ESBL and MCR-1-producing ST648 Escherichia coli with multi-drug resistance. Sci Bull. 2016;61:875–878. doi:10.1007/s11434-016-1086-y
23. Rhouma M, Letellier A. Extended-spectrum β-lactamases, carbapenemases and the mcr-1 gene: is there a historical link? Int J Antimicrob Agents. 2017;49(3):269–271. doi:10.1016/j.ijantimicag.2016.11.026
24. de Been M, Lanza VF, de Toro M, et al. Dissemination of cephalosporin resistance genes between Escherichia coli strains from farm animals and humans by specific plasmid lineages. PLoS Genet. 2014;10(12):e1004776. doi:10.1371/journal.pgen.1004541
25. Vounba P, Rhouma M, Arsenault J, Bada Alambedji R, Fravalo P, Morris Fairbrother J. Prevalence of colistin resistance and mcr-1/mcr-2 genes in ESBL/AmpC-producing E. coli isolated from chickens in Canada (Quebec), Senegal and Vietnam. J Glob Antimicrob Resist. 2019. doi:10.1016/j.jgar.2019.05.002
26. Tantawiwat S, Tansuphasiri U, Wongwit W, Wongchotigul V, Kitayaporn D. Development of multiplex PCR for the detection of total coliform bacteria for Escherichia coli and Clostridium perfringens in drinking water. Southeast Asian J Trop Med Public Health. 2005;36(1):162–169.
27. CLSI. Performance Standards for Antimicrobial Susceptibility Testing, CLSI Supplement M100S. Wayne (PA): Clinical and Laboratory Standards Institute. 2016.
28. Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
29. Alba P, Leekitcharoenphon P, Franco A, et al. Molecular epidemiology of mcr-encoded colistin resistance in enterobacteriaceae from food-producing animals in Italy revealed through the EU harmonized antimicrobial resistance monitoring. Front Microbiol. 2018;9(1217). doi:10.3389/fmicb.2018.01217
30. Carattoli A, Bertini A, Villa L, Falbo V, Hopkins KL, Threlfall EJ. Identification of plasmids by PCR-based replicon typing. J Microbiol Methods. 2005;63(3):219–228. doi:10.1016/j.mimet.2005.03.018
31. Barton BM, Harding GP, Zuccarelli AJ. A general method for detecting and sizing large plasmids. Anal Biochem. 1995;226(2):235–240. doi:10.1006/abio.1995.1220
32. Cao L, Li X, Xu Y, Shen J. Prevalence and molecular characteristics of mcr-1 colistin resistance in Escherichia coli: isolates of clinical infection from a Chinese University Hospital. Infect Drug Resist. 2018;11:1597–1603. doi:10.2147/IDR.S166726
33. Collignon P, Voss A. China, what antibiotics and what volumes are used in food production animals? Antimicrob Resist Infect Control. 2015;4:16
34. AbuOun M, Stubberfield EJ, Duggett NA, et al. mcr-1 and mcr-2 (mcr-6.1) variant genes identified in Moraxella species isolated from pigs in Great Britain from 2014 to 2015. J Antimicrob Chemother. 2018;73(10):2904. doi:10.1093/jac/dky272
35. Bai L, Hurley D, Li J, et al. Characterisation of multidrug-resistant Shiga toxin-producing Escherichia coli cultured from pigs in China: co-occurrence of extended-spectrum beta-lactamase- and mcr-1-encoding genes on plasmids. Int J Antimicrob Agents. 2016;48(4):445–448. doi:10.1016/j.ijantimicag.2016.06.021
36. Ali T, Rahman SU, Zhang L, et al. Characteristics and genetic diversity of multi-drug resistant extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli isolated from bovine mastitis. Oncotarget. 2017;8(52):90144–90163. doi:10.18632/oncotarget.21496
37. Nakayama T, Kumeda Y, Kawahara R, Yamaguchi T, Yamamoto Y. Carriage of colistin-resistant, extended-spectrum beta-lactamase-producing Escherichia coli harboring the mcr-1 resistance gene after short-term international travel to Vietnam. Infect Drug Resist. 2018;11:391–395. doi:10.2147/IDR.S153178
38. Kraemer JG, Pires J, Kueffer M, et al. Prevalence of extended-spectrum beta-lactamase-producing Enterobacteriaceae and Methicillin-Resistant Staphylococcus aureus in pig farms in Switzerland. Sci Total Environ. 2017;603–604:401–405
39. Liu X, Liu H, Wang L, et al. Molecular characterization of extended-spectrum β-Lactamase-Producing multidrug resistant Escherichia coli from swine in Northwest China. Front Microbiol. 2018;9:1756. doi:10.3389/fmicb.2018.01756
40. Huang IF, Lee WY, Wang JL, et al. Fecal carriage of multidrug-resistant Escherichia coli by community children in southern Taiwan. BMC Gastroenterol. 2018;18(1):86. doi:10.1186/s12876-018-0863-2
41. Liu YY, Wang Y, Walsh TR, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis. 2016;16(2):161–168. doi:10.1016/S1473-3099(16)30197-9
42. Xavier BB, Lammens C, Ruhal R, et al. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, Belgium, June 2016. Euro Surveill. 2016;21(27):30280.
43. Yin W, Li H, Shen Y, et al. Novel plasmid-mediated colistin resistance gene mcr-3 in Escherichia coli. mBio. 2017;8(3):pii:e00543-17. doi:10.1128/mBio.00543-17
44. Carattoli A, Villa L, Feudi C, et al. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Euro Surveill. 2017;22(31):pii:30589. doi:10.2807/1560-7917.ES.2017.22.31.30589
45. Borowiak M, Fischer J, Hammerl JA, Hendriksen RS, Szabo I, Malorny B. Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar Paratyphi B. J Antimicrob Chemother. 2017;72(12):3317–3324. doi:10.1093/jac/dkx066
46. Yang YQ, Li YX, Lei CW, Zhang AY, Wang HN. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J Antimicrob Chemother. 2018. 73 1791–1795. doi:10.1093/jac/dky111
47. Wang X, Wang Y, Zhou Y, et al. Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae. Emerg Microbes Infect. 2018;7(1):122. doi:10.1038/s41426-018-0124-z
48. Zhang J, Chen L, Wang J, et al. Molecular detection of colistin resistance genes (mcr-1, mcr-2 and mcr-3) in nasal/oropharyngeal and anal/cloacal swabs from pigs and poultry. Sci Rep. 2018;8(1):3705. doi:10.1038/s41598-018-22084-4
49. Tong H, Liu J, Yao X, et al. High carriage rate of mcr-1 and antimicrobial resistance profiles of mcr-1-positive Escherichia coli isolates in swine faecal samples collected from eighteen provinces in China. Vet Microbiol. 2018;225:53–57. doi:10.1016/j.vetmic.2018.09.018
50. Li A, Yang Y, Miao M, et al. Complete sequences of mcr-1-harboring plasmids from extended-spectrum-beta-lactamase- and carbapenemase-producing enterobacteriaceae. Antimicrob Agents Chemother. 2016;60(7):4351–4354. doi:10.1128/AAC.00550-16
51. Hu YY, Cai JC, Zhou HW, et al. Molecular typing of CTX-M-producing Escherichia coli isolates from environmental water, swine feces, specimens from healthy humans, and human patients. Appl Environ Microbiol. 2013;79(19):5988–5996. doi:10.1128/AEM.01740-13
52. Fischer J, Rodriguez I, Baumann B, et al. blaCTX-M-(1)(5)-carrying Escherichia coli and Salmonella isolates from livestock and food in Germany. J Antimicrob Chemother. 2014;69(11):2951–2958. doi:10.1093/jac/dku270
53. Sanchez-Benito R, Iglesias MR, Quijada NM, et al. Escherichia coli ST167 carrying plasmid mobilisable mcr-1 and blaCTX-M-15 resistance determinants isolated from a human respiratory infection. Int J Antimicrob Agents. 2017;50(2):285–286. doi:10.1016/j.ijantimicag.2017.05.005
54. Zheng B, Feng C, Xu H, et al. Detection and characterization of ESBL-producing Escherichia coli expressing mcr-1 from dairy cows in China. J Antimicrob Chemother. 2018;74(2):321–325.
55. Falgenhauer L, Waezsada S-E, Gwozdzinski K, et al. Chromosomal locations of mcr-1 and bla CTX-M-15 in fluoroquinolone-resistant Escherichia coli ST410. Emerg Infect Dis. 2016;22(9):1689–1691. doi:10.3201/eid2209.160692
56. Ceccarelli D, Kant A, van Essen-Zandbergen A, et al. Diversity of plasmids and genes encoding resistance to extended spectrum cephalosporins in commensal Escherichia coli from dutch livestock in 2007–2017. Front Microbiol. 2019;10:76. doi:10.3389/fmicb.2019.00076
57. Wang Q, Sun J, Li J, et al. Expanding landscapes of the diversified mcr-1-bearing plasmid reservoirs. Microbiome. 2017;5(1):70. doi:10.1186/s40168-017-0288-0
58. Ali T, Ur Rahman S, Zhang L, et al. ESBL-producing Escherichia coli from cows suffering mastitis in china contain clinical class 1 integrons with CTX-M linked to ISCR1. Front Microbiol. 2016;7:1931. doi:10.3389/fmicb.2016.01931
Supplementary materials
 |
Figure S1 Map of sampling sites in Jiangsu province. |
 |
Table S1 List of primers used for plasmid replicons typing in this study |
References
1. Carattoli A, Bertini A, Villa L, Falbo V, Hopkins KL, Threlfall EJ. Identification of plasmids by PCR-based replicon typing. J Microbiol Methods. 2005;63(3):219–228.
2. Johnson TJ, Bielak EM, Fortini D, et al. Expansion of the IncX plasmid family for improved identification and typing of novel plasmids in drug-resistant Enterobacteriaceae. Plasmid. 2012;68(1):43–50.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.