Back to Journals » ClinicoEconomics and Outcomes Research » Volume 11
Health care utilization and costs following amplified versus non-amplified molecular probe testing for symptomatic patients with suspected vulvovaginitis: a US commercial payer population
Authors Ackerman SJ, Knight T, Wahl PM, Cartwright CP
Received 25 October 2018
Accepted for publication 6 January 2019
Published 20 February 2019 Volume 2019:11 Pages 179—189
DOI https://doi.org/10.2147/CEOR.S191831
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Samer Hamidi
Stacey J Ackerman,1 Tyler Knight,2 Peter M Wahl,2 Charles P Cartwright3
1Covance Market Access Services Inc., San Diego, CA, USA; 2Covance Market Access Services Inc., Gaithersburg, MD, USA; 3Center for Esoteric Testing, Laboratory Corporation of America Holdings, Burlington, NC, USA
Background: Vulvovaginitis (VV) is a common reason women seek medical attention in the USA. Both the non-specific clinical presentation and risk of preterm labor or delivery necessitate accurate identification of the causative agents to guide appropriate therapy. The diagnostic accuracy of amplified molecular probe testing (AMP) has been shown to exceed that of non-amplified molecular probe (NAMP) by 20%–25%.
Objective: To evaluate the impact of diagnosis with AMP testing on health care utilization, direct costs, and health outcomes, compared with NAMP, for symptomatic patients with suspected VV from a commercial payer perspective.
Methods: Symptomatic women (aged 18–64 years) who underwent VV testing with AMP or NAMP from January 1, 2012–December 31, 2016 were identified using the Truven Health Analytics MarketScan Database; those with continuous medical and pharmacy benefit enrollment 6 months pre/post AMP or NAMP testing were included. Patients were propensity score (PS) matched and 6-month all-cause health care resource utilization, all-cause direct costs (2017 USD), risk of all-cause hospitalization, and risk of preterm labor or delivery were compared between cohorts.
Results: After PS match (N=46,810 per group, mean age 34.2 years), AMP had significantly (all P<0.0001) fewer mean hospital outpatient visits (AMP 0.9 vs NAMP 1.0), primary care physician office visits (AMP 1.1 vs NAMP 1.2), and prescription medications (AMP 7.3 vs NAMP 8.0), and a 21% reduction in risk of hospitalization (risk ratio [RR]=0.79, 95% CI= 0.75–0.83, P<0.0001). Total medical expenditures per patient were lower for AMP than NAMP (mean AMP $3,287 vs NAMP $3,555, P<0.0001). Among pregnant women (N=2,175 per group), AMP had a 12% reduction in risk of preterm labor or delivery (RR =0.88, 95% CI=0.77–0.99, P=0.041).
Conclusion: This real-world study offers evidence on the clinical utility for symptomatic patients with suspected VV diagnosed with AMP compared to NAMP – demonstrating an opportunity to improve the patient journey while delivering value-based care.
Keywords: vulvovaginitis, preterm labor, preterm delivery, amplified molecular probe testing, propensity-score matching, health care utilization, costs, NuSwab, clinical utility
Plain language summary
Vulvovaginitis (VV) is characterized by itching and vaginal discharge and is a common reason women seek medical attention in the USA. The condition poses risks, particularly among pregnant women, who face increased risk of preterm labor and delivery. The varied nature of VV symptoms makes the need to accurately diagnose and identify the underlying cause key to determining the appropriate treatment. Because clinical examination and laboratory diagnosis are imprecise, approximately half of symptomatic women evaluated for VV are misdiagnosed when using conventional testing approaches. A previous study has shown that an additional 20%–25% of symptomatic patients with suspected VV are correctly diagnosed with amplified molecular probe testing (AMP) compared to non-amplified molecular probe testing (NAMP). This study is the first to examine – for women aged 18–64 years who are insured by payers in the USA – whether using AMP testing leads to better patient management and improved health, compared with NAMP, for symptomatic women with suspected VV. Our results demonstrated that women tested with AMP had significantly fewer hospital outpatient visits, primary care physician office visits, prescription medications, medical expenditures, and hospitalizations compared with NAMP in the 6 months after testing. Among symptomatic pregnant women, those tested using AMP had significantly lower occurrence of preterm labor or delivery compared with NAMP.
Introduction
Vulvovaginitis (VV) is a common condition in women in the USA, and is characterized by pruritus and vaginal discharge.1,2 The majority of cases are infectious in nature, with the preponderance of these cases being attributable to bacterial vaginosis (BV), followed by vulvovaginal candidiasis (VVC), and trichomoniasis (TV).3–6 The prevalence of each etiology among symptomatic women with suspected VV is estimated to be 37.3% BV, 16.5% VVC, and 1.5% TV.7
VV is generally diagnosed based on symptoms, clinical and microscopic examination, and/or laboratory tests. The Centers for Disease Control and Prevention guidelines note the common use of clinical criteria (that is, Amsel’s Diagnostic Criteria8 or Gram stain to diagnose BV, and non-amplified molecular probe (NAMP) testing for diagnosis of BV, VVC, and TV in clinical practice.9 However, clinical diagnosis using Amsel criteria and laboratory diagnosis using Nugent criteria involve subjective components, and approximately half of symptomatic women evaluated for VV are not accurately diagnosed when using conventional testing approaches (Amsel plus wet mount).10,11 Both the non-specific clinical presentation of VV and risk of preterm labor or delivery12–14 necessitate accurate identification of the causative agents to guide appropriate therapy.
Amplified molecular probe (AMP) testing for diagnosis of VV in symptomatic women (NuSwab® VG, Laboratory Corporation of America Holdings, Burlington, NC, USA) has recently been developed to diagnose all three etiologies of VV (BV, VVC, and TV) from a single vaginal swab.15 Compared to NAMP, the primary drivers of improved diagnostic accuracy with AMP are three-fold: the use of microbiome-guided selection of multiple bacterial targets for BV that improves test specificity;3,6,16 the increase in sensitivity for TV afforded by amplification of nucleic-acid prior to detection;17,18 and the ability to identify as well as detect the presence of Candida species.3,6 In a previously published study3 using an Amsel/Nugent reference standard, the diagnostic accuracy of AMP was shown to exceed NAMP by 20%–25% in symptomatic patients with suspected VV, with higher sensitivity (96.9% vs 90.1%) and specificity (92.6% vs 67.6%) for diagnosis of BV, higher sensitivity (98.1% vs 46.3%) for diagnosis of TV, and higher sensitivity (97.7% vs 58.1%) for diagnosis of VVC.
VV has been estimated to account for ~10 million physician office visits annually in the USA, with direct medical costs of preterm labor, preterm delivery, attendant low birth weight (LBW), and other perinatal complications associated with BV in pregnant women estimated at nearly $1.4 billion.10,19 There is, therefore, the potential to improve patient outcomes and decrease cost of management of VV and its complications, given the improved diagnostic accuracy of AMP over NAMP.
In order to evaluate – from a US payer perspective – whether more accurate diagnosis using AMP testing leads to better patient management decisions and improved health outcomes, compared with NAMP, for symptomatic patients with suspected VV, we compared 6-month all-cause healthcare resource utilization (HRU) and medical expenditures and the risk of all-cause hospitalization, in a large commercially insured population. In addition, we assessed the risk of preterm labor or delivery among symptomatic women who were pregnant at the time of the testing.
Materials and methods
Study design and data source
This retrospective cohort study was conducted using administrative claims data from the Truven Health Analytics MarketScan Commercial Claims and Encounters Database. The MarketScan database includes medical claims and outpatient prescription drug claims for over 26.3 million privately insured individuals in the USA annually. An institutional review board (IRB) exemption was granted for this study by the New England IRB because this research involving the study of existing data cannot identify patients directly or through identifiers linked to patients.
Patient selection
We identified symptomatic women aged 18–64 years in the MarketScan claims database who underwent VV testing with AMP (Current Procedural Terminology [CPT] codes: 87798, 87481, and 87661, all on the same date; coding details can be viewed in Table S1) or NAMP (CPT codes: 87480, 87510, and 87660, all on the same date; coding details in Table S1). The index date was defined as the date of the first recorded VV diagnostic test with either AMP or NAMP occurring between July 1, 2012 and June 30, 2016. Patients were required to have at least 182 days of continuous medical and pharmacy eligibility both before and after the index date. Patients were excluded if they had any of the following during the 182-day pre-index period: International Classification of Disease Version 9/10 (ICD-9/10) diagnosis code of VV, pharmacy claim for a VV medication, previous diagnostic test for VV, or a recorded diagnosis of malignancy (coding details are provided in Tables S2, S3, and S4). Patients with negative medical expenditures were also excluded.
Outcome measures
We examined all-cause HRU in the 182 days following the index date, including the mean per patient number of primary care physician office visits, specialist office visits (gynecology or infectious disease), hospital outpatient visits, emergency department (ED) visits, and prescription medications. In addition, the percent of patients with at least one inpatient stay and the risk of hospitalization were calculated among all propensity score (PS)-matched patients. Associated medical expenditures for each type of HRU were defined as reimbursement amounts and were inflated to 2017 US dollars (USD) using the medical care component of consumer price index.20
The occurrence of preterm labor or delivery was defined as at least one medical claim with a relevant diagnosis code during the 182-day follow-up period among symptomatic women who were pregnant at the time of the VV diagnostic test (coding details in Tables S5 and S6).
Statistical analyses
PS matching is a causal inference method that minimizes bias in order to infer a causal relationship.21–23 To control for potential imbalances in patient characteristics between cohorts, we estimated propensity scores as the predicted probability of receiving the diagnostic test of interest (that is, AMP vs NAMP) conditional upon baseline covariates, which included patient age, geographic region, non-dependent abuse of drugs, drug dependence, high-risk sexual behavior, infections with a predominantly sexual mode of transmission, HIV, and use of hormonal contraception (coding details in Table S7); and number of primary care physician office visits, total hospital length of stay, and number of prescription medications. We then matched patients in the AMP cohort 1:1 with NAMP patients on the PS using a nearest neighbor–matching algorithm.24 PS matching was performed separately among women with and without pregnancy on the index date, and the two sets of matched patients were then combined into one analysis dataset. Balance in pre-index patient characteristics were compared between the cohorts before and after PS matching using standardized differences.
Within all PS-matched patients, we evaluated the relationship of diagnostic test choice with each of the HRU-specific study outcomes using statistical tests that account for the nature of the PS matching.25 For counts of all-cause HRU, we performed paired t-tests to compare the mean number between PS-matched cohorts. For all-cause medical expenditures, we performed non-parametric Wilcoxon signed-rank tests. For models assessing the risk of an event (all-cause hospitalization among all PS-matched patients; preterm labor or delivery among PS-matched pregnant patients), we fit Poisson regression models to estimate risk ratios (RR).
All analyses were performed using SAS version 9.3 or higher statistical software (SAS Institute Inc., Cary, NC, USA).
Results
Patient characteristics
Of 313,145 eligible patients, 46,820 women (15%) had AMP and 266,325 women (85%) had NAMP (Figure 1). PS matching retained the vast majority of AMP patients (46,810 patients per cohort; see Table 1). The mean age in the PS-matched symptomatic population was 34.2 years and pre-index patient characteristics appeared to be adequately balanced overall between the comparison groups. Approximately 25% of each cohort had evidence of hormonal contraception use in the pre-index period, with other comorbid conditions occurring in fewer than 2%.
  | Figure 1 Patient selection criteria. Abbreviations: AMP, amplified molecular probe test; NAMP, non-amplified molecular probe test; PS, propensity score; VV, vulvovaginitis. |
Outcome measures
During the 182-day follow-up period, the proportion of patients having at least one occurrence of health care utilization was similar between AMP and NAMP (prescription medications: 88% vs 90%; physician office visit: 52% vs 52%; specialist visits: 44% vs 43%; hospital outpatient visits: 37% vs 40%; ED visits: 14% vs 14%). The AMP cohort had fewer (all, P<0.0001) all-cause mean hospital outpatient visits (AMP 0.9 vs NAMP 1.0), primary care physician office visits (AMP 1.1 vs NAMP 1.2), and prescription medications (AMP 7.3 vs NAMP 8.0), and a 21% reduction in risk of all-cause hospitalization (RR=0.79, 95% CI=0.75–0.83, P<0.0001; AMP 5.4% vs NAMP 6.9%). ED visits (AMP 0.2 vs NAMP 0.2, P=0.547) and specialist visits (AMP 1.1 vs NAMP 1.1, P=0.156) were similar between groups. Total medical expenditures during the follow-up period were $268 lower per patient for AMP than NAMP (mean AMP $3,287 vs NAMP $3,555, P<0.0001; Figure 2A). Hospital inpatient and outpatient costs were the main drivers of health care costs, followed by prescription medications (Figure 2B). While the upfront cost of the AMP index test exceeds the NAMP index test, cost offsets accrued over time resulting in cost-savings to the commercial payer.
Among PS-matched symptomatic pregnant women (N=2,175 per group), the AMP cohort had a 12% reduction in risk of preterm labor or delivery (RR =0.88; 95% CI=0.77–0.99, P=0.041; AMP 16.9% vs NAMP 19.3%).
Discussion
The present study evaluated whether more accurate diagnosis using AMP testing leads to better patient management decisions, improved health outcomes, and lower medical expenditures, compared with NAMP, for symptomatic patients with suspected VV. In this large, population-based retrospective cohort study, we observed fewer all-cause physician office visits, hospital outpatient visits, prescription medication use, and medical expenditures in symptomatic women undergoing VV diagnostic testing with AMP compared to NAMP. In addition, we observed a lower risk of all-cause hospitalization among all patients and of preterm labor or delivery among women who were pregnant at the time of the test.
The recent research conducted by Cartwright et al3,6 suggests that BV is the fundamental driver of improved performance observed with AMP testing, in part due to the high prevalence of BV relative to VVC and TV. Additionally, previous research has identified increased risks among women with BV for the acquisition of some sexually transmitted diseases (for example, HIV, Neisseria gonorrhoeae, Chlamydia trachomatis, and HSV-2),26,27 cervicitis,28 complications after gynecologic surgery,29 and complications of pregnancy.30 Misidentification, misdiagnosis and mismanagement could, therefore, increase the risk of complications from VV during pregnancy (miscarriage, preterm delivery, uterine infection after pregnancy), after gynecological surgery (serious infection or inflammation such as pelvic inflammatory disease or endometritis), or when exposed to HIV or other sexually transmitted infections, which may have contributed to the observed increased risk of hospitalization and higher medical expenditures in symptomatic women undergoing VV diagnostic testing with NAMP compared to AMP.
In the clinical setting, misdiagnosis of the causative agent also could result in inappropriate drug treatment leading to return office visits and additional prescriptions. Empiric treatment can be prescribed for symptomatic patients with a moderate-to-high probability of BV, usually a course of oral antibiotics, either metronidazole or clindamycin. Treatment of VVC (commonly called a vaginal yeast infection) consists of intravaginal antifungal agents (clotrimazole, miconazole, or tioconazole) or oral fluconazole; whereas TV is treated with oral antibiotics (metronidazole or tinidazole).9 A review of clindamycin trials in women with abnormal vaginal microflora due to BV in early pregnancy has shown a significant reduction in preterm birth and late miscarriage,31 further emphasizing the need for accurate diagnosis and treatment.
We acknowledge that while “clinical failure” could be due to misdiagnosis (resulting in treatment of the wrong etiology), some patients who are accurately diagnosed will nevertheless fail treatment, thereby contributing to persistent or recurrent VV. While the present study was not designed to assess the impact of treatment failure on subsequent HRU, there is no reason to expect that treatment failure would be substantially different in the AMP vs NAMP cohorts, and thus should not have biased the observed differences between groups.
Multiple investigators have reported that pregnant women with BV are at increased risk for peripartum infections and postpartum complications, including spontaneous abortion, preterm labor, premature birth or LBW babies, preterm premature rupture of the membranes, amniotic fluid infection, postpartum endometritis, and post-cesarean wound infections.19,32 Current clinical practice guidelines emphasize the diagnosis and treatment of BV for symptomatic pregnant individuals;9 published estimates suggest BV is detected in 10% to 30% of pregnant women.32 Of note, lower risk of preterm labor or delivery was observed with AMP compared to NAMP in the current study, which did not consider the costly sequelae of prematurity. As such, the reported cost savings with use of AMP are likely conservative. Further evaluation of other adverse pregnancy outcomes is warranted.
Limitations
The following limitations should be considered when interpreting the results. First, comparisons between observational cohorts in which diagnostic tests are selected based on clinical judgement and patient characteristics may be subject to systematic bias (that is, confounding) due to baseline differences between patients in each cohort. However, PS matching is a well-known method that adjusts for this form of bias by balancing baseline factors between cohorts; the observed effect estimates can then be interpreted causally.23 As with other claims data analyses, we cannot rule out the potential for residual confounding due to unmeasured factors not captured in the data such as socio-economic status and race.
In addition, it is important to note that the current study results apply only to commercially insured women aged 18–64 years without malignancy as of the index date in order to provide evidence consistent with clinical studies that established the performance of the AMP assay in symptomatic patients aged 18 years and older. Similarly, patients aged 65 years and over were excluded due to the fact that the MarketScan commercial claims database only includes Medicare supplemental reimbursement and may miss payments made by Medicare as a primary payer.
Further, while used primarily for reimbursement purposes, VV diagnosis codes may not be recorded on a health insurance claim. Thus, we may have included women whose claims data failed to document the VV diagnosis prior to the index date. In addition, as the focus of this research was on symptomatic individuals suspected of having VV, and to our knowledge there are no published guidelines recommending wellness screening for VV, we excluded asymptomatic women undergoing VV testing during wellness screening (identified with a modifier 33 in the medical claims indicating a preventive service). There is no reason to suspect differential under-reporting of VV diagnosis codes or modifier 33 between the AMP and NAMP cohorts and this is, therefore, unlikely to have affected our findings.
Finally, while PS matching is a balancing procedure intended to mitigate potential confounding, balance in certain pre-index patient characteristics can sometimes worsen after matching, depending on the relative importance of the characteristic in the PS model. While some factors appeared to have less balance after matching (for example, hormonal contraception use), the standardized difference was still within an acceptable range to indicate overall balance,25 and additional exploratory PS models that achieved improved balance in those factors by sacrificing others did not change the results or inferences (data not shown).
Conclusion
This study is the first to document real-world evidence – from a US commercial payer perspective – that using the AMP test to make treatment management decisions improves patient-centered net health outcome in clinical practice. Of note, the propensity score matched study design produced evidence that demonstrates clinical usefulness for AMP testing by establishing the chain of evidence from clinical validity3,6 to clinical utility (better patient management decisions and improved health outcomes). The assumptions for the evidence chain are transparent and derived from commercial health insurance claims data; the performance characteristics of the AMP test have been demonstrated in scientifically rigorous (well-designed and well-conducted) studies among symptomatic women suspected of having VV.3,6 These studies together demonstrate that AMP testing improves existing clinical decision-making by guiding drug selection, which then translates into improvement in patient outcomes. This analysis offers evidence on the clinical utility among symptomatic patients with suspected VV diagnosed with AMP compared to NAMP – demonstrating an opportunity to improve the patient journey while delivering value-based care.
Acknowledgments
The authors thank Rebecca Baik of Covance Market Access Services Inc. for her contributions to the statistical analysis plan and programming; Rachael Mann of Covance Market Access Services Inc. for medical writing and editorial support in the development of this manuscript; and Tawana McIver of Laboratory Corporation of America Holdings for providing ICD-9-CM and ICD-10-CM codes. This study was supported by Laboratory Corporation of America Holdings (LabCorp; Burlington, NC, USA). LabCorp supported this research by acquiring the claims data extract.
Disclosure
At the time of this study, Dr Ackerman, Mr Knight, and Dr Wahl were employed by Covance Inc., which is wholly owned by LabCorp, and Dr Cartwright was employed by LabCorp. Dr Ackerman and Mr Knight are shareholders in LabCorp. Dr Cartwright is an inventor on issued patents (9,057,111; 9,624,552; 9,970,064) relevant to this work and is a shareholder in LabCorp. The authors report no other conflicts of interest in this work.
References
Kent HL. Epidemiology of vaginitis. Am J Obstet Gynecol. 1991;165(4 Pt 2):1168–1176. | ||
van der Pol B. Diagnosing vaginal infections: it’s time to join the 21st century. Curr Infect Dis Rep. 2010;12(3):225–230. | ||
Cartwright CP, Lembke BD, Ramachandran K, et al. Comparison of nucleic acid amplification assays with BD Affirm VPIII for diagnosis of vaginitis in symptomatic women. J Clin Microbiol. 2013;51(11):3694–3699. | ||
Granato PA. Vaginitis: clinical and laboratory aspects for diagnosis. Clin Microbiol Newsl. 2010;32(15):111–116. | ||
Sobel JD. Vaginal infections in adult women. Med Clin North Am. 1990;74(6):1573–1602. | ||
Cartwright CP, Pherson AJ, Harris AB, Clancey MS, Nye MB. Multicenter study establishing the clinical validity of a nucleic-acid amplification-based assay for the diagnosis of bacterial vaginosis. Diagn Microbiol Infect Dis. 2018;92(3):173–178. | ||
Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130(1):181–189. | ||
Amsel R, Totten PA, Spiegel CA, Chen KC, Eschenbach D, Holmes KK. Nonspecific vaginitis. Diagnostic criteria and microbial and epidemiologic associations. Am J Med. 1983;74(1):14–22. | ||
Workowski KA, Bolan GA, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64(RR-03):1–137. | ||
Carr PL, Rothberg MB, Friedman RH, Felsenstein D, Pliskin JS. “Shotgun” versus sequential testing. Cost-effectiveness of diagnostic strategies for vaginitis. J Gen Intern Med. 2005;20(9):793–799. | ||
Schaaf VM, Perez-Stable EJ, Borchardt K. The limited value of symptoms and signs in the diagnosis of vaginal infections. Arch Intern Med. 1990;150(9):1929–1933. | ||
Holst E, Goffeng AR, Andersch B. Bacterial vaginosis and vaginal microorganisms in idiopathic premature labor and association with pregnancy outcome. J Clin Microbiol. 1994;32(1):176–186. | ||
Kurki T, Sivonen A, Renkonen OV, Savia E, Ylikorkala O. Bacterial vaginosis in early pregnancy and pregnancy outcome. Obstet Gynecol. 1992;80(2):173–177. | ||
Hay PE, Lamont RF, Taylor-Robinson D, Morgan DJ, Ison C, Pearson J. Abnormal bacterial colonisation of the genital tract and subsequent preterm delivery and late miscarriage. BMJ. 1994;308(6924):295–298. | ||
NuSwab product information and indications for use. Available from: https://www.labcorp.com/test-menu/36416/vaginitis-plus-vg-nuswab%C2%AE. Accessed January 22, 2019. | ||
Fredricks DN, Fiedler TL, Thomas KK, Oakley BB, Marrazzo JM. Targeted PCR for detection of vaginal bacteria associated with bacterial vaginosis. J Clin Microbiol. 2007;45(10):3270–3276. | ||
Hobbs MM, Seña AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infect. 2013;89(6):434–438. | ||
Nye MB, Schwebke JR, Body BA. Comparison of APTIMA Trichomonas vaginalis transcription-mediated amplification to wet mount microscopy, culture, and polymerase chain reaction for diagnosis of trichomoniasis in men and women. Am J Obstet Gynecol. 2009;200(2):188.e1–188.e7. | ||
Oleen-Burkey MA, Hillier SL. Pregnancy complications associated with bacterial vaginosis and their estimated costs. Infect Dis Obstet Gynecol. 1995;3(4):149–157. | ||
Bureau of Labor Statistics, Division of Consumer Prices and Price Indexes. CPI-All urban consumers (current series). Available from: https://data.bls.gov/cgi-bin/surveymost?cu. Accessed January 22, 2019. | ||
Hernán MA, Robins JM. Using big data to emulate a target trial when a randomized trial is not available. Am J Epidemiol. 2016;183(8):758–764. | ||
Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399–424. | ||
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41–55. | ||
Parsons LS. Reducing bias in a propensity score matched-pair sample using greedy matching techniques, 2001 Paper 214–26. In Proceedings of the Twenty-Sixth Annual SAS Users Group International Conference; Cary, NC. | ||
Austin PC. Type I error rates, coverage of confidence intervals, and variance estimation in propensity-score matched analyses. Int J Biostat. 2009;5(1):13. | ||
Martin HL, Richardson BA, Nyange PM, et al. Vaginal lactobacilli, microbial flora, and risk of human immunodeficiency virus type 1 and sexually transmitted disease acquisition. J Infect Dis. 1999;180(6):1863–1868. | ||
Schwebke JR, Desmond R. Risk factors for bacterial vaginosis in women at high risk for sexually transmitted diseases. Sex Transm Dis. 2005;32(11):654–658. | ||
Marrazzo JM, Wiesenfeld HC, Murray PJ, et al. Risk factors for cervicitis among women with bacterial vaginosis. J Infect Dis. 2006;193(5):617–624. | ||
Lin L, Song J, Kimber N, et al. The role of bacterial vaginosis in infection after major gynecologic surgery. Infect Dis Obstet Gynecol. 1999;7(3):169–174. | ||
Watts DH, Krohn MA, Hillier SL, Eschenbach DA. Bacterial vaginosis as a risk factor for post-cesarean endometritis. Obstet Gynecol. 1990;75(1):52–58. | ||
Lamont RF. Advances in the prevention of infection-related preterm birth. Front Immunol. 2015;6(21):eCollection 2015:566. | ||
McGregor JA, French JI. Bacterial vaginosis in pregnancy. Obstet Gynecol Surv. 2000;55(5 Suppl 1):S1-S19. |
Supplementary material
  | Table S3 Current procedure terminology codes for vulvovaginitis diagnostic tests |
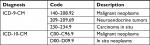  | Table S4 Diagnosis codes for malignancy Abbreviation: ICD-9/10-CM, International Classification of Disease, Ninth or Tenth Revision, Clinical Modification. |
  | Table S5 Diagnosis codes for pregnancy Abbreviation: ICD-9/10-CM, International Classification of Disease, Ninth or Tenth Revision, Clinical Modification. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.






