Back to Journals » Cancer Management and Research » Volume 13
Giant Intraparenchymal Meningioma in a Female Child: Case Report and Literature Review
Authors Guo H , Liang H, Wang J, Wen S, Wang Y, Wang Y, Ma Z
Received 27 November 2020
Accepted for publication 29 January 2021
Published 25 February 2021 Volume 2021:13 Pages 1989—1997
DOI https://doi.org/10.2147/CMAR.S294224
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Seema Singh
Huachao Guo, Hao Liang, Jiaguang Wang, Shuo Wen, Yong Wang, Yushe Wang, Zhen Ma
Department of Neurosurgery, Henan University People’s Hospital, Henan Provincial People’s Hospital, Zhengzhou, People’s Republic of China
Correspondence: Yushe Wang; Zhen Ma
Department of Neurosurgery, Henan University People’s Hospital, Henan Provincial People’s Hospital, Zhengzhou, 450000, People’s Republic of China
Email [email protected]; [email protected]
Background: Intraparenchymal meningiomas without dural attachment are extremely rare, especially in female children. To our knowledge, fibrous intraparenchymal meningioma located in the temporal lobe has never been reported in female children. The significance in the differential diagnosis of lesions in the temporal lobe should be emphasized.
Case Presentation: A 12-year-old girl was admitted to our hospital, complaining of recurrent generalized seizures for 2 months. Magnetic resonance imaging demonstrated a solid lesion located in the temporal lobe. The lesion underwent gross total resection. Histopathological examination indicated that the lesion was a fibrous meningioma. Postoperative rehabilitation was uneventful.
Conclusion: This case report presents an extremely unusual intraparenchymal fibrous meningioma of the temporal lobe with peritumoral edema and reviewed 21 intraparenchymal meningioma cases in children and to discuss the clinical presentation and treatment, differential diagnosis, and radiological features.
Keywords: meningioma, fibrous, temporal lobe, intraparenchymal
Background
Intraparenchymal meningiomas, considered the same type of subcortical meningiomas, are defined as meningiomas located in the brain parenchyma without dural attachment, even reaching the brain surface.1–3 Intraparenchymal meningiomas are rare, in contrast to ordinary meningiomas, and are more frequent in children and adolescents than in adults.3 To date, only 22 patients aged 0 to 18 years, including ours, have been reported in the English-language literature, which are summarized in Table 1.1,2,4–22 The gender distribution showed a male dominance in children and adolescents. To date, there are only 7 intraparenchymal meningiomas in female children, including ours, reported in the English-language literature, with most of them in the cerebral lobes.
 |
Table 1 Summary of Cases Involving Pediatric Primary Intraparenchymal Meningiomas in the Literature |
Case Presentation
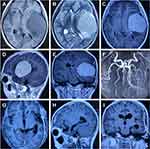
This 12-year-old girl presented with a history of generalized seizures that recurred six times in 2 months. MRI (Magnetic resonance imaging) revealed a solid mass, sized 5.0*6.5*6.0 cm, located in the temporal lobe. The tumor viewed with MRI showed isointensity on T1-weighted images (Figure 1A) and high intensity on T2-weighted images (Figure 1B) and homogeneous enhancement on T1-weighted gadolinium enhancement, but no dural tail sign was noted (Figure 1C–E).
The tumor was located in the left temporal lobe, medium texture, grayish red, intact capsule, abundant blood flow, no definite base, branches of the temporal branch of the middle cerebral artery m2 segment were involved in the blood supply, and the boundary between the lesion and the brain tissue was clear. The lateral fissure vein shifted outward, the middle cerebral artery shifted medially, and the surrounding brain tissue was compressed. The tumor capsule was first cut and then resected with cusa. When the tumor volume was reduced and gradually freed along the envelope and the blood supply artery was broken, the tumor was then excised in blocks and ultimately underwent gross total resection under a microscope. M2 segments of the vein and middle cerebral artery were well protected. Hematoxylin-eosin staining exhibiting a mass composed of interlacing collagen fiber bundles and spindle-shaped cells with oval nuclei that were arranged in streaming or whorl-like patterns. The tumor was diagnosed as a fibrous meningioma (Figure 2). Immunohistochemical features: CD34 (foci+), EMA(+), PR(-), SSTR2(+), STAT6(-), Vimentin(+), GFAP(-), Ki67 (about1%+), (Figure 3). It is worth noting that this girl does not have Neurofibroma. The patient’s postoperative course was uneventful, and there was no evidence of recurrence on an MRI performed 3 months after the operation (Figure 1G–I).
Discussion
Primary intraparenchymal meningiomas are rare but are more frequent in males than in females in children. This difference may be associated with increased estrogen levels in adult women and increased susceptibility to meningiomas.23 Female cases are extremely rare, to date, there are only 7 cases, including ours, and most of them have occurred in the cerebral lobes. The most common location was the frontal lobe (n=2) and temporal lobe (n=2), followed by the parietal lobe (n=1), occipital lobe (n=1) and brainstem (n=1). Presenting symptoms depend on tumor location, and seizures (n=4) were most frequent. One case had hemiplegia because the tumor was located in the brainstem.6 Another case was characterized by vomiting and microcephaly because the tumor was giant. The CT (Computerized Tomography) scan of the head showed a 9.5-cm calcified cystic mass in the left frontal region.8
The most common genetic alteration in meningiomas was biallelic mutation or loss of the NF2 (neurofibromatosis type 2) suppressor gene at chromosome 22, which occurs in 50 ~ 60% of sporadic meningiomas. The remaining 40% of meningiomas show complex and diverse genetic mutations including TRAF7, KLF4, AKT1, POLR2A, PIK3CA, SMO, TERT, and SMARCE1 among others, or even combinations of them.24 Some studies have shown that specific gene mutations seem to be predisposed to certain tumor subtypes and locations.25 NF2 mutant meningiomas generally occur in lateral and posterior skull base or parasagittal sinus, falcate, annular and intraventricular meningiomas. KLF4 mutations predispose to the sphenoid wing and midline skull base of tumors. Secretory meningiomas always contain KLF4 mutations, usually coexist with TRAF7, and tend to occur in the anterior skull base. SMO mutation is prone to meningeal epithelial meningiomas in the midline anterior cranial fossa. Meningiomas with PIK3CA and POLR2 mutations predispose to in the anterior and middle skull base. The AKT1 mutant predispose to anterior midline skull base meningiomas are enriched with the meningothelial subtype. At present, no research has revealed the genetic alteration of intraparenchymal meningiomas.
In general, most meningiomas are grade I, accounting for 80%–90%, whereas 5%–15% are grade II and 1%–3% are grade III.26,27 The meningothelial type is the most common, followed by the fibrous type.26 Among intraparenchymal meningiomas in children, the most common meningiomas are grade I (n=12, 63.2%), and the most common type is fibrous (including fibrous components) (n=8). There were 7 grade II and grade III cases, accounting for 36.8%. At present, the cases of female children are extremely rare. Only 5 cases had definite pathological results, which included fibrous (n=2), anaplastic (n=1), clear cell (n=1), and psammomatous(n=1). The present case is the first case of fibrous intraparenchymal meningiomas located in the temporal lobe in female children. Among 12 cases of children, homogeneous enhancement (n=6), fibrous (n=3), and meningothelial (n=1) were noted; none of the remaining cases had clear pathological reports. Heterogeneous enhancement (n=6), atypical (n=2), anaplastic (n=1), fibrous (n=1), clear cell (n=1), and transitional (n=1) were also reported. The present cases were fibrous and showed homogeneous enhancement, including ours; 4/5 fibrous intraparenchymal meningiomas showed homogenous enhancement.
Since intraparenchymal meningiomas lack dural attachment, characteristics such as cyst formation and the presence of peritumoral edema are relatively common in childhood. Moreover, it is often difficult to preoperatively distinguish it from gliomas, cavernous angiomas, metastatic tumors, malignant lymphomas, or sarcomatous lesions.28 Among 22 cases, cysts were found in 6 cases, and peritumoral edema was found in 11 cases. Especially when cystic components are present, it is more difficult to correctly diagnose these lesions because cysts are often associated with gliomas or metastatic tumors and are rarer among meningiomas. Peritumoral edema of intraparenchymal meningioma surrounding the lesion develops strongly in the region where the lesion is attached to the surrounding brain tissue, whereas peritumoral edema of most gliomas or metastatic tumors tends to surround the entire tumor surface. Wada et al believe that the feature of any peritumoral edema of intraparenchymal meningiomas can help differentiate between intraparenchymal meningiomas and high-grade gliomas or metastatic tumors.29 However, its characterization is not applicable to all intraparenchymal meningiomas. Peritumoral edema surrounding the entire tumor surface was found in 3 of 11 cases.12,14,22 Hence, the radiological features of most pediatric primary intraparenchymal meningiomas, such as cystic components, peritumoral edema surrounding the entire tumor, and no dural tail sign, make accurate preoperative diagnosis extremely difficult.
After reviewing the literature, there are only two patients with intraparenchymal meningiomas underwent MRS (MR spectroscopy) examination, and the date of the two cases was not similar.4,10 Results obtained from MRS of one patient show a high Ala peak similar to those of extracerebral meningiomas. However, unlike typical meningiomas without NAA peak, the peak of NAA was present.4 The results of another patient’s spectral analysis are similar to and compatible with those for typical meningiomas, an elevated peak in the Cho region and decreases in the Cr and NAA regions. There was no obvious evidence of alanine, in short TE. Especially, the Cho peak was very high and the Cho/NAA ratio was 92.53 in the central zone of the lesion.10 At present, the data of spectrum analysis of cerebral parenchymal meningioma are very few, only two cases. Hence, further investigation into the presence of the MRS is needed.
Only one case’s perfusion imaging of meningiomas is reported, a perfusion study was performed using an ASL (Arterial spin labeling) technique, the lesion showed higher CBF (cerebral blood flow) values compared to a corresponding region in the contralateral parietal lobe.10 Perfusion imaging of meningiomas usually shows high rCBF (relative cerebral blood flow) and rCBV (relative cerebral blood volume). The ASL perfusion has shown increased rCBF in meningiomas, especially the angiomatous histological subtype.30 In addition to correlation with vascularity, maximum rCBV has been shown to positively correlate with the Ki-67 proliferative index in meningiomas.31 We believe that the perfusion imaging of intraparenchymal meningiomas is an important way to distinguish meningiomas from other tumors.
SSTR II (Somatostatin receptor II) expression increased in almost all meningiomas.32 The high specific binding to SSTR in meningiomas and extremely low uptake in adjacent structures such as bone or brain tissue, which leads to very high tumor-to-background contrast, because these tracers cannot cross the blood-brain barrier. Recent studies have shown that the sensitivity of PET (positron emission Tomography) with SSTR ligands labeled with Ga-68 (gallium-68) is higher than that of contrast-enhanced MRI, which detected only 171 out of 190 (92%) of the meningiomas that were revealed by PET/CT.33 Although there are no reports of PET images of intraparenchymal meningiomas, it cannot be analyzed and compared, SSTR-PET is useful for differential diagnosis from other tumors.
Almost all intraparenchymal meningiomas were located in the cerebrum and lacked dural attachment and no association with the dura of the skull base or the sinuses. Only 3 of 22 cases in children underwent subtotal resection. For lesions located in the hemisphere, one patient underwent STR (subtotal resection), but the reason was not stated.19 In one case, STR was performed because of the rich blood supply, tight adhesion with the ACA (anterior cerebral artery) and lower tolerance of blood loss in children. There was a 70% remnant in one patient because of the lesion in the brainstem.6 No recurrences occurred in any patients who underwent total tumor resection during the follow-up period (15 weeks-5 years). Postoperative radiotherapy is controversial for children. Some reports have shown the development of meningiomas after high-dose irradiation with long latency periods.34,35 Gosztonyi et al36 believe that low-dose irradiation also causes the development of meningiomas with long latency periods in children. Some people treated patients with malignant meningiomas, and radiotherapy was used in the postoperative course. But, Ghim, TT et al35 believe that high-dose radiotherapy should be avoided in the treatment of meningiomas in children. Some studies showed that the extent of initial surgical resection was the strongest independent prognostic factor for pediatric meningiomas and that upfront radiotherapy achieved no benefit.37,38 Hence, surgery is the best modality of treatment, and resection is recommended to achieve maximum extirpation.
Some authors thought that intraparenchymal meningiomas arise from arachnoid cells located within the pia mater and enter the surface of the brain or sulcus with perforating blood vessels.8,28 Several cases involved the MCA (middle cerebral artery) branches or had feeding arteries of the MCA in children.7,14,20 A patient’s tumor is closely related to the perforating arteries of the ACA and middle cerebral artery and the recurrent artery of Heubner.2 In one case, several small arteries were found to enter into the lesion.15 These cases provide better support for the establishment of this theory. Therefore, for patients suspected to be intraparenchymal meningiomas and for whom the tumor is located in the blood supply range of the ICA (internal carotid artery), MRA (MR Angiography) or CTA (CT angiography) should be performed to evaluate the relationship between the tumor and blood vessels before the operation. During surgery, we should pay attention to the protection of blood vessels to avoid the neurological dysfunction caused by cerebral infarction after surgery, such as hemiplegia and aphasia. Our patient’s preoperative CTA showed that the lesion appeared intimately connected to proximal MCA branches. During the operation, the temporal branch of the m2 segment of the middle cerebral artery was found to be involved in the blood supply of the tumor.
Conclusions
We present an extremely unusual case of intraparenchymal fibrous meningiomas of the temporal lobe with peritumoral and review the pertinent literature. Imaging features such as cystic degeneration, peritumoral edema, and absence of meningeal tail signs make accurate preoperative diagnosis difficult. MRS, perfusion and PET imaging of intraparenchymal meningioma are of great help to the differential diagnosis. Some intraparenchymal meningiomas have rich blood supply, which is closely related and supplied by the small branches of the internal carotid artery. It is extremely important to perform CTA or MRA before operation to clarify the relationship between lesions and intracranial vessels. Total resection of tumor is the most important factor affecting the prognosis of patients. Surgery is the best modality of treatment, and resection is recommended to achieve maximum extirpation. However, for children without total resection, radiotherapy should be carefully considered.
Abbreviations
MRI, Magnetic resonance imaging; CT, Computerized Tomography STR, subtotal resection; GTR, gross total resection; MCA, middle cerebral artery; ACA, anterior cerebral artery; NF2, neurofibromatosis type 2; MRA, MR Angiography; CTA, CT angiography; ICA, internal carotid artery; NF2, neurofibromatosis type 2; MRS: MR spectroscopy; ASL, Arterial spin labeling; CBF, cerebral blood flow; rCBF, relative cerebral blood flow; rCBV, relative cerebral blood volume; SSTR II, Somatostatin receptor II; PET, positron emission Tomography; Ga-68, gallium-68.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of Henan University People’s Hospital, and informed consent was obtained from parents and legal guardians.
Consent for Publication
We have obtained consent for publication from parents and legal guardians.
Acknowledgments
The authors would also like to acknowledge the surgeons, nurses, and pathologists who helped to manage patients and provide pathological images.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. Yushe Wang, and Yong Wang contributed to the conception of the study. Shuo Wen and Hao Liang collected the data. Huachao Guo wrote the manuscript. Jiaguang Wang took part in drafting. Zhen Ma contributed significantly to analysis and manuscript preparation; All authors read and approved the final manuscript.
Funding
This work was supported by grants from provincial or ministry-level project of China (No. SB201902027).
Disclosure
The authors declare that they have no competing interests.
References
1. Schroeder BA, Samaraweera RN, Starshak RJ, et al. Intraparenchymal meningioma in a child: CT and MR findings.[J]. J Comput Assist Tomogr. 1987;11(1):192. doi:10.1097/00004728-198701000-00046
2. Liu X, Zhang Y, Zhang S, et al. Intraparenchymal atypical meningioma in basal ganglia region in a child: case report and literature review[J]. J Korean Neurosurg Soc. 2018;61(1):120–126. doi:10.3340/jkns.2015.0609.001
3. Ohba S, Abe M, Hasegawa M, et al. Intraparenchymal meningioma: clinical, radiologic, and histologic review[J]. World Neurosurg. 2016;92:23–30. doi:10.1016/j.wneu.2016.04.098
4. Wenjie L. Rare parenchyma meningioma in an adolescent female with cheek tingling: a case report.[J]. Medicine. 2016;2:254.
5. Pinto PS, Huisman TAGM, Ahn E, et al. Magnetic resonance imaging features of meningiomas in children and young adults: a retrospective analysis.[J]. J Neuroradiology. 2012;39(4):218–226. doi:10.1016/j.neurad.2011.06.003
6. Teo JGC, Goh KYC, Rosenblum MK, Muszynski CA, Epstein FJ. Intraparenchymal clear cell meningioma of the brainstem in a 2-year-old child.[J]. Pediatr Neurosurg. 1998;28(1):27–30. doi:10.1159/000028614
7. Kohama I, Sohma T, Nunomura K, et al. Intraparenchymal meningioma in an infant–case report.[J]. Neurol Med Chir (Tokyo). 1996;36(8):598. doi:10.2176/nmc.36.598
8. Mamourian AC, Lewandowski AE, Towfighi J. Cystic intraparenchymal meningioma in a child: case report.[J]. Ajnr Am J Neuroradiol. 1991;12(2):366–367.
9. Morimoto M, Aoki H, Sadamitsu N, et al. Cystic meningioma–report of two cases (author’s transl)[J]. Neurol Surg. 1976;8:805–809.
10. Vescovi MC, Bagatto D, Capo G, et al. A multimodal approach to the treatment of intraparenchymal meningioma in a 7-year-old boy a case report.[J]. Pediatr Neurosurg. 2018;53(3):175–181. doi:10.1159/000487808
11. Nayil K, Ramzan A, Makhdoomi R, et al. Intraparenchymal anaplastic meningioma in a child: a rare entity[J]. Asian J Neurosurg. 2015;10(2):111–113. doi:10.4103/1793-5482.154980
12. Werbrouck C, Florin D, Holsbeeck BV, et al. Intraparenchymal meningioma in a child[J]. Orgaan Van De Koninklijke Belgische Vereniging Voor Radiologie. 2014;97(1):46.
13. Y S J, Song YJ. Meningioma in a 20-month-old boy[J]. J Korean Neurosurg Soc. 2012;51(4):219–221. doi:10.3340/jkns.2012.51.4.219
14. Shimbo D, Kato T, Takeda M, et al. Intraparenchymal Meningioma in a Child[J]. Neurol Med Chir (Tokyo). 2011;51(11):793. doi:10.2176/nmc.51.793
15. Jian Z, Ling-yi C, Bin M, et al. Meningioma without dural attachment: case report, classification, and review of the literature.[J]. Surg Neurol. 2007;67(5):25.
16. Karadereler S, Aker F, Berkman Z. Intraparenchymal meningioma in a child. Case report and review of the literature[J]. J Neurosurg. 2004;101(2):112. doi:10.3171/ped.2004.101.2.0112
17. Starshak RJ. Cystic meningiomas in children: a diagnostic challenge[J]. Pediatr Radiol. 1996;26(10):711–714. doi:10.1007/BF01383385
18. Perilongo G, Sutton LN, Goldwein JW, et al. Childhood meningiomas. Experience in the modern imaging era.[J]. Pediatr Neurosurg. 1992;18(1):16–23. doi:10.1159/000120637
19. Erdincler P. Intracranial meningiomas in children.[J]. Pediatr Neurosci. 1985;12(3).
20. Sakaki S, Nakagawa K, Kimura H, et al. Intracranial meningiomas in infancy.[J]. Surg Neurol. 1987;28(1):51–57. doi:10.1016/0090-3019(87)90206-0
21. Kimura H, Nakagawa K, Sakaki S, et al. [Intracranial meningioma of an infant: a case report][J]. No Shink Geka Neurol Surg. 1987;15(6):663.
22. Legius E, J S V, Casaer P, et al. Intraparenchymal meningioma in a 14-month-old infant: case report[J]. Brain Dev. 1985;7(6):622–624. doi:10.1016/S0387-7604(85)80011-5
23. Germano IM, Edwards MSB, Davis RL, et al. Intracranial meningiomas of the first two decades of life.[J]. J Neurosurg. 1994;80(3):447–453. doi:10.3171/jns.1994.80.3.0447
24. Delgado-López PD, Cubo-Delgado E, González-Bernal JJ, et al. A Practical Overview on the Molecular Biology of Meningioma[J]. Curr Neurol Neurosci Rep. 2020;20(12):62. doi:10.1007/s11910-020-01084-w
25. Abedalthagafi M, W L B, Aizer AA, et al. Oncogenic PI3K mutations are as common as AKT1 and SMO mutations in meningioma[J]. Neuro Oncol. 2016;v316.
26. Park JS, Sade B, Oya S, et al. The influence of age on the histological grading of meningiomas[J]. Neurosurg Rev. 2014;37(3):425–429. doi:10.1007/s10143-014-0537-7
27. Saraf S, Mccarthy BJ, Villano JL. Update on Meningiomas[J]. Oncologist. 2011;16(11):1604–1613. doi:10.1634/theoncologist.2011-0193
28. Senol J, Alexandru CS, Uwe D, et al. Intraparenchymal meningioma mimicking cavernous malformation: a case report and review of the literature.[J]. J Med Case Rep. 2014;8:5.
29. Wada T, Suzuki M, Beppu T, et al. A Case of Subcortical Meningioma[J]. Acta Neurochirurgica. 2000;142(2):209–213. doi:10.1007/s007010050026
30. Qiao XJ, Kim HG, Wang DJJ, et al. Application of arterial spin labeling perfusion MRI to differentiate benign from malignant intracranial meningiomas[J]. Eur J Radiol. 2017;97:31–36. doi:10.1016/j.ejrad.2017.10.005
31. D T G, Mangla R, Yeaney G, et al. Correlation of diffusion and perfusion MRI with Ki-67 in high-grade meningiomas.[J]. Ajr Am J Roentgenol. 2010;195(6):1391–1395. doi:10.2214/AJR.10.4531
32. Barresi V, Alafaci C, Salpietro F, et al. Sstr2A immunohistochemical expression in human meningiomas: is there a correlation with the histological grade, proliferation or microvessel density?[J]. Oncol Rep. 2008;20(3):485.
33. Afshar-Oromieh A, Giesel FL, Linhart HG, et al. Detection of cranial meningiomas: comparison of 68Ga-DOTATOC PET/CT and contrast-enhanced MRI[J]. Eur J Nucl Med Mol Imaging. 2012;39(9):1409–1415. doi:10.1007/s00259-012-2155-3
34. Strojan P, Popović M, Jereb B. Secondary intracranial meningiomas after high-dose cranial irradiation: report of five cases and review of the literature[J]. Int J Radiat Oncol Biol Phys. 2000;48(1):65–73. doi:10.1016/S0360-3016(00)00609-X
35. Ghim TT, Seo JJ, O’Brien M, et al. Childhood intracranial meningiomas after high‐dose irradiation[J]. Cancer. 1993;71.
36. Gosztonyi G, Slowik F, Pásztor E. Intracranial meningiomas developing at long intervals following low-dose X-ray irradiation of the head.[J]. J Neurooncol. 2004;70(1):59–65. doi:10.1023/B:NEON.0000040812.19235.d1
37. Rishi SK, Elaine MP, Elisabeth JR, et al. Meningiomas in children and adolescents: a meta-analysis of individual patient data.[J]. Lancet Oncol. 2011;12(13):154.
38. Rishi SK, Peter J, Catherine HC, et al. Morbidity in survivors of child and adolescent meningioma.[J]. Cancer. 2013;119(24):12544.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.