Back to Journals » Infection and Drug Resistance » Volume 15
Genital HPV Prevalence, Follow-Up and Persistence in Males and HPV Concordance Between Heterosexual Couples in Wenzhou, China
Authors Hu J , Ji L, Li P, Ni X, Huang Y, Tao J, Zhu H
Received 9 September 2022
Accepted for publication 8 November 2022
Published 2 December 2022 Volume 2022:15 Pages 7053—7066
DOI https://doi.org/10.2147/IDR.S387226
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Héctor Mora-Montes
Jiaming Hu,1 Lili Ji,2 Ping Li,3 Xinyu Ni,1 Ying Huang,1 Jinxin Tao,1 Hua Zhu1
1Department of Gynecology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, People’s Republic of China; 2Department of Clinical Laboratory Medicine, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, People’s Republic of China; 3Department of Out-Patient, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, People’s Republic of China
Correspondence: Hua Zhu, Department of Gynecology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, People’s Republic of China, Tel +86-13505777090, Fax +86-577-55578033, Email [email protected]
Purpose: This study aimed to investigate male HPV infection, re-examination rate, clearance rate and relevant influencing factors as well as HPV infection between heterosexual partners in Wenzhou, China.
Methods: The study enrolled 2359 men who accepted ≥ 1 HPV detection in the First Affiliated Hospital of Wenzhou Medical University between June 2014 and June 2020. An outpatient follow-up was carried out for collecting HPV re-test results among males who were tested HPV positive. In addition, we collected female sexual partners’ HPV infection state through outpatient information system or telephone call. For males who had not re-tested HPV more than half a year after the first HPV positive result, a telephone follow-up would be provided.
Results: Male HPV prevalence was 39.39% (928/2359) at baseline, of which the median age was 38.00 years. The percentage of co-infection and HR HPV infection was 35.24% (327/928) and 61.08% (874/1431). Genotype-specific concordance of HPV infection among heterosexual partners was 61.19% (123/201). The most common types were HPV6, HPV52, HPV16, HPV53, HPV11, HPV43 and HPV58. Percentage of males finishing HPV re-examination was low (40.09%). Infection with HR HPV, discomfort or HPV-related diseases and sexual partners with HPV infection were possible motivator for male HPV re-test. The medium time to male HPV clearance was 300.000 (95% CI=274.845– 325.155) days. Infection with HR HPV, sexual partners with HPV infection and growth of age might prolong HPV persistence time included.
Conclusion: Males are susceptible population for genital HPV infection as well as females. Whereas, males are generally less aware of HPV and its influence on their couples. The study showed that male HPV testing as well as the role of males in the diagnosis and treatment of HPV-associated cervical diseases for females should be popularized and co-treatment of couples was necessary.
Keywords: human papillomavirus, males, heterosexual couples, prevalence, persistence, re-test rate
Introduction
HPV (human papillomavirus) infection is closely associated with genital malignant tumors, especially with carcinoma of uterine cervix in females. HPV infection and genital condyloma acuminata in males and females were also highly correlated. Statistics have shown that incidence of penile cancer in males only takes a small percentage (about 0.2%),1 however, nearly 20% of penile cancer might be caused by HPV infection. The most common genotype related with penile cancer are HPV16 and HPV18, especially HPV16, which is similar with uterine cervix cancer in females.2 Genital condyloma acuminata in males is closely associated with HPV6 and HPV11.3 For the past few years, a lot of research reported that frequent HPV infection existed between heterosexual partners during sexual behaviors including genital intercourse, oral sex, anal sex and so on.4–8 Previous studies suggested that HPV prevalence in males was 2.9–65.5% in general population.3,9–13 HPV infection in males (including genital sites, semen and so on) is responsible for repeated and persistent HPV infection in female sexual partners, which can cause cervical lesions. What’s more, HPV infection may be responsible for infertility.14,15
According to relevant guidelines, currently, HPV testing and vaccination are the main methods for cervical cancer screening and prevention in women.16,17 While male HPV vaccination has been popularized and implemented in many countries,18 except for China. In China, some hospitals have introduced male external genital HPV testing. Males with HPV positive female sexual partners and condyloma acuminatum are more likely to be infected with HPV.11,19 Thus HPV testing is needed and meaningful for males with high-risk factors. By now, many males have received the testing. As for whether men with HPV positive need treatment and regular re-examination or not, it is still controversial. In order to further understand the role of males in the prevention and treatment of cervical malignancy in females, and provide new strategies for the prevention and treatment of HPV in females, this study investigated male HPV infection, re-examination rate, clearance rate and relevant influencing factors, and HPV infection between heterosexual partners as well.
Materials and Methods
Study Population
The study enrolled 2359 men who accepted at least one detection of HPV in Wenzhou between June 2014 and June 2020. All of the patients attended at least one of the clinics in the First Affiliated Hospital of Wenzhou Medical University, which included: dermatology department, STD and gynecology clinics. The reasons of HPV testing were as follows: (1) with symptoms of anogenital wart, condyloma acuminata and penis neoplasm. (2) with clinical signs of balanitis, urethritis, genital rash and pruritus. (3) with HPV positive sexual partners. (4) health examination without discomfort. The criteria of the participants were as follows: (1) males with current or past sexual activity; (2) agreement to participant in our research; (3) males aged ≥15 years old; (4) agreement to accept an HPV test. (5) males had not been diagnosed with HPV-related cancer, such as penile cancer, laryngeal cancer and so on. (6) males had not accepted an HPV vaccination before.
Specimen Collection
Participants were asked to abstained from sexual activity 1 day before HPV detection. Exfoliated cells were collected by a single cytobrush from different genital areas including: distal urethral, glans penis, coronal sulcus. The swabs were placed in elution tubes containing cell protection solution and were obtained at 4 degree until processing. The specimens were sent to the affiliated hospital for analysis within 2 weeks.
Testing Principle and HPV Genotyping
Twenty-seven HPV DNAs were detected by multiplex PCR amplification with universal primers and flow fluorescence hybridization typing.20 The detailed experimental operation process was described in previous studies.21,22 For the kit, there were 2 types of probes and 28 classified microspheres. The probes hybridized with HPV DNAs were coated on 27 classified microspheres, and once the signal specific to the corresponding type of probe was positive, this type of HPV was considered as positive. The quality control microsphere was the probe hybridized with human β-globin gene, and the corresponding results indicated whether the whole process of sampling, extraction, PCR and hybridization met relevant requirements. During the testing, the DNA of test sample was amplified by PCR first, and then, the PCR products were hybridized with the probes linked to the microspheres according to the principle of base complementation pairing, and added with fluorescent indicators. At last, the fluorescence signals were detected by a multifunction flow type dot matrix instrument. Specifically, if the PCR products were fully paired with the probes, a microsphere- probe-PCR product-biotin-SA-PE complex was formed by the PCR products labeled with biotin captured by the corresponding probes on the microspheres after added with StrepAvidin (SA) labeled R-phycoerythrin (PE), and thus, the fluorescence signals of classified microspheres could be detected by the instrument, and if the PCR products were unpaired with the probes, the fluorescence signals of the classified microspheres became the background signals, based on which relevant data could be directly used for determining the results after analyzed.
This method can be used for qualification and typing of human papillomavirus (HPV) infection, so as to detect 27 HPV subtypes, suggesting HPV infected gene subtypes directly including 17 high-risk subtypes: HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 26, 53, 68, 82, and 10 low-risk subtypes: HPV 6, 11, 40, 42, 43, 44, 55, 61, 81, 83.
Independent Variables and Results Expected
Baseline Results
The first HPV test result of males and HPV infection state of female sexual partners: The baseline data means the results of the first HPV test and all of the patients had never been admitted as inpatients before. All HPV test data of female sexual partner, including HPV infection rate and HPV concordance between sexual partners, were obtained by telephone follow-up and outpatient visit.
Follow-Up Results
Male HPV re-examination rate, HPV clearance rate and relevant influencing factors.
Age: Age referred to the exact time of life when HPV was first tested positive.
Grouping according to HPV type: Various HPV records were divided into different groups in two ways. 1) High-risk and low risk. B) Single infection and co-infection (including double, triple and above).
HPV-related diseases and clinical symptoms: HPV-related diseases included anogenital wart, condyloma acuminata and penis neoplasm. Clinical symptoms included balanitis, urethritis, genital rash and pruritus.
HPV infection state of female sexual partners was divided into 3 different cases: 1) HPV positive: HPV infection detected, without negative conversion; 2) HPV negative conversion: HPV infection detected, with negative conversion; 3) HPV negative: Always negative for HPV test. The 2) and 3) were combined as female sexual partner with HPV negative.
Data Processing and Follow-Up
From June 2014 to June 2020, a total of 928 males received one or more external genital HPV tests, which indicated HPV positive. We further obtained HPV re-test results by enquiring follow-up data in out-patient information system. An outpatient follow-up was carried out among them for collecting relevant data, including whether HPV was re-examined or not, time interval of re-examination, and re-examination results. For those who had not been re-examined for HPV more than half a year after HPV infection was detected for the first time, a telephone follow-up would be provided. At the same time, they would be reminded of HPV re-examination, and asked about the possible reasons why they were not re-examined, and their knowledge on HPV as well. For males other than those who received HPV tests because their female sexual partners were infected with HPV, whether their female sexual partners were tested for HPV or test results were not known, and so, to maintain the completeness of relevant data, telephone follow-up would be performed to know whether their female sexual partners were infected with HPV or not.
Males who have accomplished at least one regular re-test (the interval between two visits were limited to 1.5 years) were included in the follow-up analysis (Data A), while the rest were rejected. Data A included both single infection and co-infection (including double-infection and multi-infection) records. We split co-infection records into HPV-specific records for separate analysis (Data B) (Figure 1).
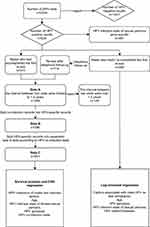 |
Figure 1 Flow chat of data processing and analysis. |
Statistical Analysis
Data were collected and analyzed by Excel 2016 and SPSS version 25.0 (IBM Corp, Armonk, NY, USA). GraphPad Prism 8.0.2 was used to draw the figures.
Multivariate Log-Binomial Regression
Chi-square test was used to filter the possible influential factors associated with male HPV re-test compliance (p value<0.10). We described the OR value (odds ratio) and the impact of each factor by multivariate log-binomial regression. When P value<0.05, the influence of independent variable is significant.
Survival Analysis
HPV clearance referred to one or more negative HPV results after a positive result, and those with recurrence or other types of infection after HPV negative results were not included in the survival analysis.
Cox regression analysis was used to screen out the factors that might affect the male HPV clearance rate. The assignment of every variable is shown in Table 1. The endpoint event, ie, dependent variable (Y), was defined as a certain type of HPV clearance. Y=1 indicates that a certain type of HPV had been cleared or tested negative during follow-up, ie, conventional outcome of death. For censored data during follow-up (including lost to follow-up, persistent positive HPV, and non-negative HPV as endpoint of follow-up), Y=0. Exp (B)(HR) >1 indicated lower risk of persistent HPV infection or condition more conducive to HPV clearance. Log-rank analysis (Kaplan–Meier survival curve analysis) was introduced to compare the survival curves and survival rates, and thus, the median survival time for HPV infection (HPV clearance time) was obtained. HPV co-infection state may change with follow-up time and splitting co-infection records into single HPV infection records might ignore the effect of co-infection status on HPV clearance. To address this, we divided the whole HPV infection period of the same subject into multiple sets of data according to co-infection status for further analysis. The HPV status in each set of data was assigned a value as follows: Single infection was coded as 1, double-infection as 2, triple infection as 3 and so on. Those with the same type of recurrence after HPV negative conversation or a new type of infection during follow-up were not included in the study, for example, a single infection at baseline or a new type of infection found during follow-up resulting in co-infection, change in status of HPV infection (from single infection to multiple infection) with new type of infection excluded from the model.
 |
Table 1 Table of Cox Regression Analysis Assignments and Results of Cox Regression Analysis |
 |
Table 2 HPV Prevalence in Males of Different Age Distribution (Median of Age=38.00 Years) |
Results
HPV Prevalence of Males and Female Sexual Partners
Prevalence of any HPV was 39.39% (928/2359) at baseline during June 2014 to June 2020 among 928 males aged 15–82. The median age of HPV infective males in the study was 38.00 years (age observed abnormal distribution by Kolmogorov–Smirnov test, P<0.05.) The infection rate of the ≤25 year group was 46.38%, which exhibited a peak, but declined rapidly as the age increased. The HPV infection rate increased with age in patients over 36 years old (Table 2). As shown in Table 3 and Figure 2, the percentage of co-infection was 35.24% (327/928), including double-infection (214/327) and multi-infection (113/327) among HPV infected patients. After splitting co-infection records into multiple HPV-specific records, a total of 1431 HPV genotype records were included into further analysis. The amount of detected genotype covered by 9v vaccines are shown in Figure 3. The rate of HR-HPV infection was 61.08% (874/1431), which was higher than the rate of LR-HPV infection (38.92%). Over all, the 7 genotype with the most common detection rate were HPV6 (10.55%), HPV52 (9.01%), HPV16 (7.83%), HPV53 (7.83%), HPV11 (5.31%), HPV43 (5.10%) and HPV58 (4.69%) (Figure 4). After telephone interview, 667 patients were available with HPV infection information of their female sexual partners. In total, 408 female sexual partners were detected HPV positive (408/928, 43.97%, which was less than the actual data), 53 females were HPV negative, while the remaining (527 females in total) were categorized as “the female sexual partners’ HPV infection status was not sure or had never accepted at least one HPV test yet”. Among the HPV positive females, 201 provided specific genotype of HPV they infected. Genotype-specific concordance of HPV infection (at least one HPV genotype in concordant) among heterosexual partners was 61.19% (123/201) by calculation. Among these, 64 partners shared exactly the same HPV types (52.03%,64/123). The most frequent genotype in concordant were HPV16 (16.67%,23/138), HPV52 (10.87%,15/138), HPV53 (10.14%,14/138), HPV6 (7.97%,11/138), HPV58 (7.25%,10/138). which were listed in the attached figure (Figure 3).
 |
Table 3 HPV Infection in Males and Female Sexual Partners and Factors Related to Follow-Up Compliance in HPV-Positive Males During June 2014 to June 2020 |
 |
Table 4 P-value Table of Chi-Square Test and Binary Logistic Regression Analysis of Factors Related to Male Follow-Up Compliance |
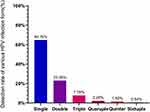 |
Figure 2 HPV prevalence of single and multiple infections. |
 |
Figure 3 (A) proportion of HPV infections covered by 9v HPV vaccine and proportion of non-9v HPV infections. (B) the most frequent types in concordance between sexual couples. |
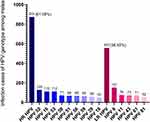 |
Figure 4 Infection cases of the most common HPV genotype of males (Infection rate: HR HPV vs LR HPV, P<0.001). |
Factors Influencing Re-Examination Rate
(1) Percentage of males finishing HPV re-examination over time: Among them, 372 men accomplished the first re-test, the others (number=556) had not appeared in the clinics for a second test. Within 12 months of initial infection, the percentage of males with at least one HPV re-test progressed rapidly, increasing about 2.69% by month. While as time went by, the proportion reached stabilization, which was close to 40.09% (Figure 5).
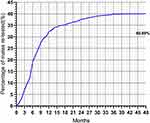 |
Figure 5 HPV re-test proportion varied with time. |
(2) Log-binomial regression: The most likely associated factors which may influence HPV re-test motivation of males were listed (Table 4), including age, HPV infection state of sexual partners, HPV related diseases and HPV genotype. Each factor was tested by Chi-square test. It turned out that HPV-related diseases or clinical symptoms and high-risk types of HPV infection were significant factors that promoted males to visit hospitals for HPV re-examination (p<0.05). While the effect of age, HPV infection state of female partners or male co-infection state were insignificant (p>0.05). All of the possible factors (p<0.1) were tested by binary logistic regression analysis. The results of multi-variable binary regression analysis are shown in Table 4. The odds of finishing a second HPV test was 1.499 times among males with HPV-positive sexual partners when compared to those whose female partners tested HPV negative or HPV infection state were unknown (OR=1.499,95% CI: 1.123–2.001, p=0.006). Males infected with high-risk HPV were more likely to finish a second test, having 1.510 times the odds of being re-tested when compared to those infected by low-risk types (95% CI: 1.081–2.108, p=0.025). Obviously, the re-examination rate in males with HPV-related diseases was 2.098 times higher than in those with latent HPV infection (OR=2.098, 95% CI: 1.463–3.007, p<0.001). Males with HPV co-infection were more likely to finish a second HPV test compared to males with single infection, the effect was not significant (OR=1.088, 95% CI: 0.807–1.466, p=0.580).
Factors for HPV Persistence of Males
After reminding male patients of returning to clinics for HPV re-test by telephone call, a total of 358 males who accomplished at least one re-test within the recommended time frame (the interval between two visits was limited to 1.5 years) were included in the further study (Data A). While the others (n=128) who had not been able to finish a second test within 1.5 years were rejected. After splitting co-infection records into HPV specific records, a total of 596 separated records were included in further analysis (Data B).
Kaplan–Meier method (KM method) was performed to calculate survival time, compare HPV persistence curves by different variables and get a general understanding of their effects on HPV survival rates over time. Relevant data showed that HPV co-infection state was associated with a shorter clearance time (higher clearance rate), and the effect was significant (Figures 6 and 7). Therefore, the whole period of HPV data from the same patient should be split into separated sets of data according to HPV co-infection status, so as to avoid the effect of the factor in survival analysis (Data C, n=651) (Figure 1). The median age of Data C was 40.00 years (Age of Data C observed abnormal distribution by Kolmogorov–Smirnov test, P<0.05). On the whole, the medium time to HPV clearance among males with HPV infection at baseline was 300.000 (95% CI=274.845–325.155) days. When female sexual partners were infected with HPV, the medium time to HPV clearance among males was 330.000 (95% CI=304.962–355.038) days, which was longer than males with negative sexual partners (results were statistically significant, p=0.0429, Figure 6). The medium time to HPV clearance among males with high-risk HPV infection was 330.000 (95% CI=303.043–356.957) months, which was longer than males with low-risk HPV infection (p=0.0036). The effect of age on HPV persistence curves was obvious (Figures 8 and 7). Included each specific HR-HPV type detected at baseline into survival analysis, we found that it was difficult to eliminate HPV39, 51, 52, 53, 58 and 68, while HPV35, 18 and 82 were easy to clear (p=0.1880, Figure 9).
 |
Figure 6 (A) Male HPV co-infection state-specific HPV infection persistence curves. (B) HPV infection state of female sexual partners-specific HPV infection persistence curves. |
 |
Figure 7 (A) Number of HPV co-infections-specific HPV infection persistence curves. (B) Age group-specific HPV infection persistence curves. |
 |
Figure 8 (A) Risk of HPV genotype-specific HPV infection persistence curves. (B) Age-specific HPV infection persistence curves. |
 |
Figure 9 (A) HR HPV common type-specific HPV infection persistence curves. (B) LR HPV common type-specific HPV infection persistence curves. |
Statistics showed that HPV infection state in female sexual partners might affect HPV persistence of males in multi-variate COX regression (Table 1). Compared with males with HPV-positive sexual partners, males with negative sexual partners were more likely to clear HPV (HR of HPV clearance=1.189, 95% CI=0.982–1.440, p=0.076), the effect was not significant. The HPV persistence curve showed that HPV negative state of female sexual partners only promoted male HPV clearance before 400 days of infection, while the effect was not obvious afterwards. Low-risk HPV infection was significantly associated with an decreasing HPV clearance time (HR of HPV clearance=1.245, 95% CI=1.025–1.511, p=0.027). Males younger than 40 years old were more likely to clear HPV than the elders, the effect was significant (HR of HPV clearance=1.245, 95% CI=1.030–1.504, p=0.023). Single infection was associated with longer HPV duration than co-infection (HR of HPV clearance=0.804, 95% CI=0.605–0.981, p=0.032) and the effect was obvious after 400 days of infection.
Discussion
Most of the males receiving HPV testing in the hospital aged 36–45 years, and in all types of HPV that infected them, some were covered by 9-valent vaccines, suggesting the potential effect of HPV vaccines in the prevention and control of HPV in males. Previous research have suggested that the overall HPV prevalence in males was 2.9%–65.5%.3,9–13 Males who are single or divorced, with 3+ lifetime sex partners, with warts, smoking and drinking are more likely to be infected with HPV.9–11 Male sexual partners of women with high grade cervical lesions are a significant reservoir and vector of HPV infection.10 In our study, prevalence of any HPV in males was 39.39% at baseline. Sampling methods, processing, nationalities and races may cause the difference in HPV prevalence among studies around the world. We found that prevalence of HPV6 and HPV16 was 10.55% and 7.83%, respectively, which was similar to previous data. The research performed by Staci L. Sudenga reported that prevalence of HPV6 and HPV16 was 6.0% to 6.7% and 5.6% to 9.2%, respectively, in males from three countries including Brazil, Mexico, and the United States.10 Prevalence of HPV6 was relatively higher in our study than previous results. The probable reason might be that a significant percentage of males presented in the clinic due to genital warts or genital discomfort, which were related to HPV6 infection.
Concordance of specific HPV genotype between sexual partners was various among different research, ranged from 0% to 87.55%. The most common HPV types in concordance were HPV16, HPV18 and HPV6.5,6,19,23–34 In our study, HPV concordance rate between sexual partners was 61.19%, which was consistent with previous data. The most common types in concordance were HPV16 (16.67%,23/138), HPV52 (10.87%,15/138), HPV53 (10.14%,14/138), HPV6 (7.97%,11/138), HPV58 (7.25%,10/138). HPV16 and HPV6 were both highly pathogenic not only to females but also to males. Although incidence rate of HPV related penile cancer was low around the world, long-term and persistent infection of high-risk HPV in females were closely associated with cervical dysplasia and cervical cancer, especially HPV16 and HPV18.35
The number of males who presented in the clinic for HPV-testing was relatively higher in the 36–45 age group. HPV prevalence was relatively higher in the age group of younger than 25 years old, while infection rate rapidly decreased in the very young age group less than 20 years old (15.22%,7/46). In the groups over 36 years old, HPV prevalence increased with age. In the study about age-specific prevalence of HPV in males, peak prevalence of HPV varied a lot with age and was not concentrated in the younger age groups.36 While another study reported that HPV prevalence showed a rapid increase between age 18 and 35.9 It was suggested that peak HPV prevalence was mostly distributed in the age groups of sexually active youths and the elderly.
The HPV clearance time in our study was longer than in previous studies. Although relevant data varied with country or region there were still interesting points for discussion. Among participants included in this study, quite a few males accepted HPV detection for the reason of HPV infection in sexual partners. Another part of the participants presented in the clinic due to genital warts or genital discomfort. Males with high-risk factors might be easier to get infection or recurrence which might extend HPV clearance time. During survival analysis and COX regression analysis, we excluded data with intervals more than 1.5 year between two consecutive reviews (Previous data suggested that the medium time of HPV duration in males was no more than 12 months10), meanwhile, data with intervals between 12 and 18 months and less than 12 months was included. When the follow-up time was approximate 1.5 years, the recurrence after HPV clearance might be ignored or the HPV clearance time might be overestimated. When HPV clearance occurred in re-examination interval, it was difficult to estimate the exact clearance time.
The advantage of our research was that we studied the HPV follow-up compliance of males who were infected with HPV. We attempted to overview accomplishment of follow-up for a second HPV test and figure out the possible factors. In our study, HPV re-testing accomplishment of the whole HPV infected males were close to 40.09%. Males had HPV related diseases tended to accomplish a second HPV test more than males without any discomfort. What is more, when the lesions were visually cleared or genital discomfort disappeared, males were less likely to present to clinics for HPV re-testing. Surprisingly, among all HPV positive cases, the percentage of HPV latent infection was up to 79.41%. The problem, however, is that HPV re-examination accomplishment of patients was relatively low (40.09%, 372/928) with latent infection or cleared lesions. Males infected with high-risk HPV types tended to review more than males infected with low-risk types. HPV-reexamination of males with high-risk types was 42.42% (277/653).
At present, male HPV screening and specific time for male HPV review has not been recommended in any guidelines. In our study, we also inquired about the possible reason why males had not finished a second HPV test by phone. Among males who failed to re-test HPV, 26.98% (150/556) believed that HPV infection only happened to women or hardly knew nothing about HPV at all. In addition, 53 males refused to re-test after clearing the lesions or genital discomfort disappeared. Propaganda and education of HPV in males were still inadequate. Nearly a half males claimed that the process of going to the hospitals for HPV testing was tiring and inconvenient. HPV self-collecting is a new technique invented in recent years and has been widely supported because of the positive effect on the patients’ preference in sampling procedure and compliance to follow up on positive test results.37 What’s more, male HPV self-collecting in genital area is much easier than females in vagina and cervix.
Conclusion
Combined with previous research, our statistics indicate that males are also susceptible population for genital HPV infection as well as females. Females with HPV-positive male sexual partners are high-risk population for HPV infection. Furthermore, the HPV concordance between sexual partners was relatively high. However, in the patients infected with HPV, the HPV re-examination rate was low, for which the influencing factors included follow-up time, infection with high-risk types of HPV, discomfort or HPV-related diseases and partner with HPV infection. Through follow-up, the study found that males were generally less aware of HPV and its influence on their couples. Hospital examination was one of the reasons why males were unwilling to receive re-examination. The possible factors influencing male HPV clearance time include HPV type, whether spouse has been infected with HPV or not, age and HPV co-infection state. There was difference in male HPV clearance between individuals, therefore, an individualized recommendation for the follow-up time is essential. The study has shown that male HPV testing as well as the role of males in the diagnosis and treatment of HPV-associated cervical diseases for females should be popularized, and women may benefit from HPV vaccination in men and co-treatment of couples.
Ethical Statement
This study was conducted with approval from the ethic committee in Clinical Research (ECCR) of the First Affiliated Hospital of Wenzhou Medical University for clinical research. The process of this study complies with the declaration of Helsinki. Written informed consent for this study was obtained from all participants. For all participants under the age of 18 years, parents/legal guardian consent to participate in the research was obtained.
Acknowledgment
We appreciate every participant of this study and each investigator who contributed to this paper.
Funding
The project was supported by the Scientific and Technological Project of Wenzhou (No. Y20180019) and Center for Uterine Cancer Diagnosis & Therapy Research of Zhejiang Province(No. JBZX −201803).
Disclosure
The authors declared no conflict of interest in this work.
References
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi:10.3322/caac.21660
2. Kidd LC, Chaing S, Chipollini J, Giuliano AR, Spiess PE, Sharma P. Relationship between human papillomavirus and penile cancer-implications for prevention and treatment. Transl Androl Urol. 2017;6(5):791–802. doi:10.21037/tau.2017.06.27
3. Han JJ, Beltran TH, Song JW, Klaric J, Choi YS. Prevalence of genital human papillomavirus infection and human papillomavirus vaccination rates among US adult men: National Health and Nutrition Examination Survey (NHANES) 2013–2014. JAMA Oncol. 2017;3(6):810–816. doi:10.1001/jamaoncol.2016.6192
4. Widdice LE, Breland DJ, Jonte J, et al. Human papillomavirus concordance in heterosexual couples. J Adolesc Health. 2010;47(2):151–159. doi:10.1016/j.jadohealth.2010.01.006
5. Kero K, Rautava J. HPV infections in heterosexual couples: mechanisms and covariates of virus transmission. Acta Cytol. 2019;63(2):143–147. doi:10.1159/000494710
6. Su Y, Wei F, Huang X, et al. Prevalence, concordance, and transmission of human papillomavirus infection among heterosexual couples in Liuzhou, China: an observational perspective study. J Infect Dis. 2019;220(6):980–989. doi:10.1093/infdis/jiz224
7. Cossellu G, Fedele L, Badaoui B, et al. Prevalence and concordance of oral and genital HPV in women positive for cervical HPV infection and in their sexual stable partners: an Italian screening study. PLoS One. 2018;13(10):e0205574. doi:10.1371/journal.pone.0205574
8. Malagón T, Louvanto K, Wissing M, et al. Hand-to-genital and genital-to-genital transmission of human papillomaviruses between male and female sexual partners (HITCH): a prospective cohort study. Lancet Infect Dis. 2019;19(3):317–326. doi:10.1016/S1473-3099(18)30655-8
9. Berenson AB, Hirth JM, Chang M. Prevalence of genital human papillomavirus by age and race/ ethnicity among males. Clin Infect Dis. 2021;73(9):1625–1633. doi:10.1093/cid/ciab429
10. Sudenga SL, Torres BN, Silva R, et al. Comparison of the natural history of genital HPV infection among men by Country: brazil, Mexico, and the United States. Cancer Epidemiol Biomarkers Prev. 2017;26(7):1043–1052. doi:10.1158/1055-9965.EPI-17-0040
11. Li X, Xiang F, Chen Z, et al. Genital human papillomavirus prevalence and genotyping among males in Putuo District of Shanghai, China 2015–2019. Med Sci Monit. 2021;27:e932093. doi:10.12659/MSM.932093
12. Daugherty M, Byler T. Genital wart and human papillomavirus prevalence in men in the United States From Penile Swabs: results From National Health and Nutrition Examination Surveys. Sex Transm Dis. 2018;45(6):412–416. doi:10.1097/OLQ.0000000000000761
13. Deshmukh AA, Tanner RJ, Luetke MC, Hong YR, Sonawane Deshmukh K, Mainous AG. Prevalence and risk of penile human papillomavirus infection: evidence from the National Health and Nutrition Examination Survey 2013–2014. Clin Infect Dis. 2017;64(10):1360–1366. doi:10.1093/cid/cix159
14. Olivera C, Mosmann JP, Paira DA, et al. Association between Human Papillomavirus and Chlamydia trachomatis genital infections in male partners of infertile couples. Sci Rep. 2021;11(1):19924. doi:10.1038/s41598-021-99279-9
15. Jeršovienė V, Gudlevičienė Ž, Rimienė J, Butkauskas D. Human papillomavirus and infertility. Medicina. 2019;55(7):377. doi:10.3390/medicina55070377
16. Fontham E, Wolf A, Church TR, et al. Cervical cancer screening for individuals at average risk: 2020 guideline update from the American Cancer Society. CA Cancer J Clin. 2020;70(5):321–346. doi:10.3322/caac.21628
17. Saslow D, Andrews KS, Manassaram-Baptiste D, Smith RA, Fontham E. Human papillomavirus vaccination 2020 guideline update: American Cancer Society guideline adaptation. CA Cancer J Clin. 2020;70(4):274–280. doi:10.3322/caac.21616
18. Gerber S, Porsch L, Markowitz J. Human papillomavirus vaccination: ACOG committee opinion summary, number 809. Obstet Gynecol. 2020;136(2):435–436. doi:10.1097/AOG.0000000000004001
19. López Diez E, Pérez S, Iñarrea A, et al. Prevalence and concordance of high-risk papillomavirus infection in male sexual partners of women diagnosed with high grade cervical lesions. Enferm Infecc Microbiol Clin. 2017;35(5):273–277. doi:10.1016/j.eimc.2016.02.014
20. Liao Y, Zhou Y, Guo Q, et al. Simultaneous detection, genotyping, and quantification of human papillomaviruses by multicolor real-time PCR and melting curve analysis. J Clin Microbiol. 2013;51(2):429–435. doi:10.1128/JCM.02115-12
21. Li Z, Liu F, Cheng S, et al. Prevalence of HPV infection among 28,457 Chinese women in Yunnan Province, southwest China. Sci Rep. 2016;6:21039. doi:10.1038/srep21039
22. Poynten IM, Machalek D, Templeton D, et al. Comparison of age-specific patterns of sexual behaviour and anal HPV prevalence in homosexual men with patterns in women. Sex Transm Infect. 2016;92(3):228–231. doi:10.1136/sextrans-2015-052032
23. Davarmanesh M, Dezfulian M, Gharavi MJ, et al. Human papilloma virus (HPV) genotypes concordance between Iranian couples referrals. Infect Agent Cancer. 2019;14:22. doi:10.1186/s13027-019-0241-x
24. Rob F, Tachezy R, Pichlík T, et al. High prevalence of genital HPV infection among long-term monogamous partners of women with cervical dysplasia or genital warts-Another reason for HPV vaccination of boys. Dermatol Ther. 2017;30(1). doi:10.1111/dth.12435
25. Rob F, Tachezy R, Pichlík T, et al. Concordance of HPV-DNA in cervical dysplasia or genital warts in women and their monogamous long-term male partners. J Med Virol. 2017;89(9):1662–1670. doi:10.1002/jmv.24824
26. Grabowski MK, Gravitt PE, Gray RH, et al. Trends and determinants of human papillomavirus concordance among human immunodeficiency virus-positive and -negative heterosexual couples in Rakai, Uganda. J Infect Dis. 2017;215(5):772–780. doi:10.1093/infdis/jiw631
27. Grabowski MK, Kong X, Gray RH, et al. Partner human papillomavirus viral load and incident human papillomavirus detection in heterosexual couples. J Infect Dis. 2016;213(6):948–956. doi:10.1093/infdis/jiv541
28. Wissing MD, Louvanto K, Comète E, et al. Human papillomavirus viral load and transmission in young, recently formed heterosexual couples. J Infect Dis. 2019;220(7):1152–1161. doi:10.1093/infdis/jiz238
29. Nyitray AG, Menezes L, Lu B, et al. Genital human papillomavirus (HPV) concordance in heterosexual couples. J Infect Dis. 2012;206(2):202–211. doi:10.1093/infdis/jis327
30. Benevolo M, Mottolese M, Marandino F, et al. HPV prevalence among healthy Italian male sexual partners of women with cervical HPV infection. J Med Virol. 2008;80(7):1275–1281. doi:10.1002/jmv.21189
31. Burchell AN, Coutlée F, Tellier PP, Hanley J, Franco EL. Genital transmission of human papillomavirus in recently formed heterosexual couples. J Infect Dis. 2011;204(11):1723–1729. doi:10.1093/infdis/jir644
32. Widdice L, Ma Y, Jonte J, et al. Concordance and transmission of human papillomavirus within heterosexual couples observed over short intervals. J Infect Dis. 2013;207(8):1286–1294. doi:10.1093/infdis/jit018
33. Lorenzon L, Terrenato I, Donà MG, et al. Prevalence of HPV infection among clinically healthy Italian males and genotype concordance between stable sexual partners. J Clin Virol. 2014;60(3):264–269. doi:10.1016/j.jcv.2014.04.003
34. Reiter PL, Pendergraft WF, Brewer NT. Meta-analysis of human papillomavirus infection concordance. Cancer Epidemiol Biomarkers Prev. 2010;19(11):2916–2931. doi:10.1158/1055-9965.EPI-10-0576
35. Tota JE, Chevarie-Davis M, Richardson LA, Devries M, Franco EL. Epidemiology and burden of HPV infection and related diseases: implications for prevention strategies. Prev Med. 2011;53(Suppl 1):S12–21. doi:10.1016/j.ypmed.2011.08.017
36. Smith JS, Gilbert PA, Melendy A, Rana RK, Pimenta JM. Age-specific prevalence of human papillomavirus infection in males: a global review. J Adolesc Health. 2011;48(6):540–552. doi:10.1016/j.jadohealth.2011.03.010
37. Aranda Flores CE, Gomez Gutierrez G, Ortiz Leon JM, Cruz Rodriguez D, Sørbye SW. Self-collected versus clinician-collected cervical samples for the detection of HPV infections by 14-type DNA and 7-type mRNA tests. BMC Infect Dis. 2021;21(1):504. doi:10.1186/s12879-021-06189-2
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
