Back to Journals » Drug Design, Development and Therapy » Volume 13
Genetic variations in the transcription factors GATA4 and GATA6 and bleeding complications in patients receiving warfarin therapy
Authors Yee J, Kim W, Chang BC, Chung JE , Lee KE , Gwak HS
Received 12 December 2018
Accepted for publication 19 March 2019
Published 17 May 2019 Volume 2019:13 Pages 1717—1727
DOI https://doi.org/10.2147/DDDT.S198018
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Qiongyu Guo
Jeong Yee,1,* Woorim Kim,2,* Byung Chul Chang,3,4 Jee Eun Chung,5 Kyung Eun Lee,2 Hye Sun Gwak1
1College of Pharmacy & Division of Life and Pharmaceutical Sciences, Ewha Womans University, Seoul 03760, Korea; 2College of Pharmacy, Chungbuk National University, Cheongju-si 28160, Korea; 3Department of Thoracic and Cardiovascular Surgery, Bundang CHA Medical Center, CHA University, Seongnam, Gyeonggi-do, Korea; 4Department of Thoracic & Cardiovascular Surgery, Yonsei University Medical Center, Seoul 03722, Korea; 5College of Pharmacy, Hanyang University, Ansan 15588, Korea
*These authors contributed equally to this work
Purpose: GATA4 and GATA6 are known to have potential roles in vascular regulation by affecting vascular smooth muscle cell differentiation and atrial natriuretic peptide levels. The aim of this retrospective study was to investigate the associations between GATA4 and GATA6 polymorphisms and bleeding complication risk at a therapeutic international normalized ratio (INR) in patients with mechanical heart valves.
Patients and methods: Study patients were included from the Ewha-Severance Treatment (EAST) Group of Warfarin. It consisted of 229 patients who received warfarin therapy after undergoing mechanical heart valve replacement and maintained a stable INR (INR of 2.0–3.0 for at least three consecutive times). Twenty single-nucleotide polymorphisms including VKORC1, CYP2C9, GATA4, and GATA6 were analyzed. Multivariate logistic regression analysis was employed to investigate the independent risk factors for bleeding complications. To evaluate the potential clinical value of genotyping for preventing bleeding complications in patients with high-risk genotype, the number needed to genotype (NNG) was also calculated.
Results: One hundred forty-two patients were included in this study, 21 of whom had bleeding complications. After adjusting covariates, TT genotype carriers of rs13273672 in GATA4 and CC genotype carriers of rs10454095 in GATA6 showed 5.0- (95% CI, 1.6–15.7) and 3.1-fold (95% CI, 1.1–8.7) higher bleeding complications than carriers of C allele and T allele, respectively. NNG for preventing one patient from experiencing bleeding complications in patients with TT genotype of rs13273672 and CC genotype of rs10454095 was 22.2 and 17.5, respectively. Patients with both TT genotype in rs13273672 and CC genotype in rs10454095 showed 8.7-fold (95% CI, 1.7–46.1) higher bleeding complications than those with other genotypes. NNG in patients having both TT genotype in rs13273672 and CC genotype in rs10454095 was calculated to be 40.0.
Conclusions: This study showed that GATA4 and GATA6 gene polymorphisms could affect bleeding complications during warfarin treatment in patients with mechanical heart valves.
Keywords: GATA4, GATA6, warfarin, bleeding, polymorphism
Introduction
Warfarin is a widely used oral anticoagulant for atrial fibrillation, ischemic stroke, deep-vein thrombosis, and pulmonary embolism.1 Despite the introduction of direct oral anticoagulants, it still remains the first-line anticoagulant therapy for patients with heart valve prosthesis.2 Because of the narrow therapeutic range and wide inter- and intraindividual variability of warfarin, careful monitoring of international normalized ratio (INR) is required while using it for anticoagulation therapy.3
Bleeding complication is one of the major concerns with warfarin usage.4 Although an elevated INR level is the most important factor for increasing warfarin-associated bleeding risk, it has been reported that some patients also experienced bleeding complications at therapeutic INR. A study showed that 15.7% of patients suffered from bleeding complications during therapeutic INR control.5 Some studies showed that in addition to high INR, age, hypertension, and concomitant aspirin use were patient-related risk factors for bleeding complications.6 However, the genetic effects on bleeding complications during anticoagulation therapy have rarely been investigated.
The GATA family, the zinc finger transcription factor, consists of 6 subtypes in vertebrates. GATA-1/2/3 subfamily is expressed in the hematopoietic cell lineage,7 whereas GATA-4/5/6 subfamily is expressed in various mesoderm- and endoderm-derived tissues including the heart, blood vessel, lung, and gut.8 Among GATA 4/5/6 subgroups, GATA4 and GATA6 have similar protein structure and expression pattern. Moreover, both are known to be involved in the regulation of gene expression in cardiomyocytes and vascular smooth muscles.9 In a study that employed an animal model, GATA4/GATA6 double-heterozygous mouse showed impaired differentiation of vascular smooth muscle cells.10
With respect to the association between polymorphisms of GATA4 and GATA6 genes and cell differentiation, GATA4 mutation p.S335X has been found to pre-terminate its translation, producing a truncated GATA4 lacking a conservative region at C-terminus. Truncated GATA4 delayed the cardiomyocyte differentiation in P19cl6 model and prohibited Bcl2 expression, leading to apoptosis.11 In addition, GATA4 was found to be a key modifier of sex steroidogenic cell differentiation through conditional loss-of-function mutations in GATA4 gene.12
The GATA6 mutation p.E386X was identified in a family with bicuspid aortic valves, being transmitted in an autosomal dominant fashion. Cardiac valvular morphogenesis requires accurate regulation of cell proliferation, differentiation, migration, adhesion, and apoptosis. Biological assays revealed that E386X-mutant GATA6 proteins had no transcriptional activity compared with its wild-type counterpart. Furthermore, the E386X mutation led to disrupted synergistic transcriptional activation between GATA4 and GATA6.13
Hemostasis is a multiphase process involving blood vessels, platelets, and coagulation factors; an imbalance in any of the steps of hemostasis may result in bleeding.14 Impaired vascular smooth muscle cell differentiation is involved in vascular malformations,15 which are known to increase bleeding risks in several organs (eg, gastrointestinal tract, retina, and endometrium).16–18
In addition, atrial natriuretic peptide (ANP), the expression of which is regulated by GATA4 and GATA6, plays an important role in vascular function regulation.19 ANP is also involved in platelet aggregation and lipid metabolism.20 ANP level is also known to be associated with cardiovascular diseases (eg, hypertension and hyperlipidemia).21 Although GATA4 and GATA6 have potential roles in vascular regulation, no study has yet investigated the association between GATA gene polymorphisms and bleeding complications in patients receiving warfarin.
Therefore, this study aimed to investigate the association between GATA4 and GATA6 polymorphisms and the risk of bleeding complications at therapeutic INR during warfarin treatment.
Materials and methods
Study patients and data collection
Study patients were included from the Ewha-Severance Treatment (EAST) Group of Warfarin. It consisted of 229 patients who received warfarin therapy after undergoing mechanical heart valve replacement between January 1982 and December 2009 at Severance Cardiovascular Hospital of Yonsei University College of Medicine. Patients who maintained a stable INR (INR of 2.0–3.0 for at least three consecutive times) were eligible for the study. Patients who had experienced bleeding complications at supra- or subtherapeutic INR were excluded. Patients were also excluded if their complications were not verified by health professionals.
Patients were followed up continuously at the outpatient clinic of Severance Cardiovascular Hospital of Yonsei University Medical Center. Blood samples were collected during the regularly scheduled clinic visit. Patients’ first follow-up visits were within 1–2 months after discharge and patients were followed up in 1- to 3-month intervals in accordance with their therapeutic INR. In the case of bleeding occurrences, patients visited the hospital and showed bruises, gum bleeding, and nose bleeding as evidence of bleeding. During the verification of bleeding events by a doctor, INR levels were measured. Data collection was retrospectively done using scanned medical records and electronic medical records of patients from June 1983 to August 2010. Data on sex, age, body weight, height, position of valve prosthesis, valve type, warfarin therapy duration, INR measurements, concurrent medication, comorbidities, and history of bleeding complications were collected. Bleeding complications were classified as major life-threatening, other major, any major, minor, or minimal using the scheme detailed in Platelet Inhibition and Patient Outcomes trial.22
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All patients gave written informed consent for participation. The protocol and informed consent were reviewed and approved by the Institutional Review Board of the Yonsei University Medical Center (approved number: 4-2009-0283).
Genotyping methods
To select single-nucleotide polymorphisms (SNPs) of GATA4 and GATA6 that might be associated with warfarin-related bleeding, genetic information concerning GATA4 and GATA6 was obtained from the PharmGKB database, Haploreg 4.1, and Database of SNP (dbSNP) from NCBI and previous studies.23–26 Sixteen SNPs of GATA4 (rs13273672, rs2645400, rs4841588, rs867858, rs10090884, rs2898292, rs10086064, rs3735814, rs2740434, rs2001470, rs3729849, rs809205, rs2173117, rs62489352, rs2409805, and rs2898293) and 2 SNPs of GATA6 (rs16964670 and rs10454095) were selected. In addition to the selected SNPs, VKORC1 rs9934438 and CYP2C9 rs1057910, which were found to have significant effects on stable doses of warfarin, were also included in the study. Therefore, a total of 20 SNPs were investigated.
Genomic DNA from the patients was isolated from EDTA blood samples using the QIAamp DNA Blood Mini Kit (QIAGEN GmbH, Hilden, Germany) according to the manufacturer’s protocol. Genotyping was performed using a single-base primer extension assay by employing SNaPShot multiplex kits (ABI, Foster City, CA, USA) or TaqMan genotyping assay by employing real-time PCR system (ABI 7300, ABI), according to the manufacturer’s recommendation.
Statistical analysis
Continuous variables in patients with bleeding complications and in those without complications were compared using Student’s t-test. Chi-square test or Fisher’s exact test was used to compare categorical variables between the two groups. Multivariate logistic regression analysis was used to examine independent risk factors for bleeding complications. Factors having a p-value <0.05 in univariate analysis along with clinically relevant confounders were included in multivariate analysis. Odds ratio and adjusted odds ratio were calculated through univariate and multivariate analyses, respectively. The time in therapeutic range (TTR) of INR 2.0–3.0 was measured using Rosendaal method. Attributable risk (%) was calculated by (1–1/adjusted odds ratio) ⨰ 100. To test the model’s goodness of fit, we performed a Hosmer–Lemeshow test. Discrimination of the model was further assessed using an analysis of the area under the receiver operating curve (AUROC), which assesses the ability of the risk factor to predict bleeding. We calculated the number needed to genotype (NNG) for preventing one patient from experiencing a significantly higher incidence of bleeding complications by 1/absolute risk reduction. Absolute risk reduction was achieved by multiplying the relative risk reduction by genotyping and risk of higher incidence of bleeding complications without genotyping. A p-value of <0.05 was considered statistically significant. All statistical analyses were conducted using IBM SPSS statistics, version 20 software (International Business Machines Corp., Armonk, NY, USA).
Results
Of the 229 patients from the EAST Group of Warfarin, 87 patients were excluded due to the following reasons: 28 patients did not reach a stable INR, 4 patients had bleeding complications at supratherapeutic INR, and 55 patients reported minimal bleeding complications which were not verified by health professionals. Accordingly, data from 142 patients who underwent heart valve replacement were used for the analysis.
The median age of the included patients was 60 years (range, 34–81 years), and there were 52 (36.6%) males. The follow-up periods ranged from 1.0 to 29.7 years (mean 14.3 years). During the follow-up period, one thromboembolic event was observed, and there were no deaths. The mean INR monitoring interval was 2.9 months, and the average number of INR measurements per patient was 23. The TTR of INR 2.0–3.0 was 55.2±12.7%. As shown in Table 1, 21 patients (14.8%) experienced bleeding complications at therapeutic INR. Among them, 11 and 10 patients experienced minor and minimal bleeding complications, respectively. One patient experienced bleeding four times, seven patients twice, and 13 patients once. There was no significant difference between the two groups except for atrial fibrillation. Patients with atrial fibrillation had more bleeding complications in therapeutic INR than those without atrial fibrillation (p=0.045).
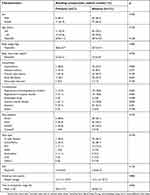 | Table 1 Characteristics of study patients |
As shown in Table 2, statistically significant associations between genotypes and bleeding complications were found for rs13273672, rs4841588, and rs2173117 of GATA4. For GATA6, rs10454095 showed a significant association with bleeding complications. For rs13273672, 8 of 26 patients (30.8%) with TT genotype had bleeding complications, whereas 13 of 116 patients (11.2%) with C allele had bleeding complications (p=0.027). For rs4841588, patients with wild-type homozygote showed a higher bleeding risk than those with variant-allele carriers (31.8% vs 11.7%, p=0.023). A allele carriers of rs2173117 showed approximately 70% lower bleeding complications than CC genotype carriers (p=0.040). For rs10454095 of GATA6, CC genotype carriers experienced more bleeding complications than T allele carriers (23.5% vs 9.9%, p=0.028). The allele frequencies of SNPs used in this study for Koreans and other populations are described in Tables 2 and S1, respectively.
 | Table 2 Factors associated with bleeding complications at therapeutic INR |
Two models were constructed for conducting multivariate analysis (Table 3). Model I included sex, age, and factors having a p-value <0.05 in the univariate analysis, including atrial fibrillation, rs13273672, rs4841588, and rs2173117 of GATA4, as well as rs10454095 of GATA6. Model II included sex, age, atrial fibrillation, rs4841588, rs2173117, and a combination of rs13273672 and rs10454095.
 | Table 3 Multivariate analysis to identify predictors of bleeding complications at therapeutic INR |
As shown in Model I of Table 3, rs13273672 of GATA4 and rs10454095 of GATA6 were significantly associated with bleeding complications (p=0.006 and p=0.031, respectively). After adjusting for related covariates, TT genotype carriers in rs13273672 showed approximately 5.0-fold higher bleeding complications than C allele carriers. For rs10454095 of GATA6, CC genotype carriers showed approximately 3.1-fold higher bleeding complications than T allele carriers after adjusting for covariates. NNG for preventing one patient with TT genotype in rs13273672 from suffering a higher incidence of bleeding complications was calculated to be 22.2. NNG of rs10454095 in GATA6 was 17.5. In Model II, patients with both TT genotype in rs13273672 and CC genotype in rs10454095 showed 8.7-fold higher bleeding complications than those with the other genotypes. NNG in patients having both TT genotype in rs13273672 and CC genotype in rs10454095 was calculated to be 40.0.
The AUROC values of Model I and Model II were 0.770 and 0.724, respectively (Figure 1). The Hosmer–Lemeshow test showed that the fitness of the Model I was satisfactory (χ2=2.396, 7 degrees of freedom, p=0.935) as well as for Model II (χ2=0.618, 3 degrees of freedom, p=0.892).
Discussion
The main finding of this study is that rs13273672 of GATA4 and rs10454095 of GATA6 were associated with bleeding complications at a therapeutic INR during warfarin treatment in mechanical heart valve patients. TT genotype carriers of rs13273672 in GATA4 and CC genotype carriers of rs10454095 in GATA6 had 5.0- and 3.1-fold increased risk of bleeding complications compared with the carriers of C allele and T allele, respectively. Patients having a combination of TT and CC genotypes of rs13273672 and rs10454095 experienced 8.7 times higher bleeding complications than those having the other genotypes. The AUROC value of the models constructed for predicting bleeding complications was approximately 0.75.
Stable INR was defined as the INR of 2.0–3.0 for three or more consecutive visits. Although American College of Chest Physicians guidelines 2012 suggest INR of 2.5–3.5 in patients with mitral valve replacements,27 many studies of Asian populations suggested considerably lower intensities of warfarin therapy after mechanical valve prostheses including mitral valves.28–30
GATA4, a gene located on chromosome 8, is expressed in cardiomyocytes, smooth muscles, and endothelial cells in the heart and blood vessels.8 It regulates the expression of cardiovascular-related genes, which encode α- and β-myosin heavy chain, cardiac troponin-C, ANP, and brain natriuretic peptide.8,9
Among proteins which are regulated by GATA4, the roles of ANP in vasculature is well-established. It exerts vasodilatory effects through the ANP receptor, which is guanylyl cyclase-A.19 Additionally, it modulates vascular smooth muscle cell proliferation31 as well as endothelial cell growth and permeability.32 It is also known that it is reportedly involved in platelet aggregation and energy metabolism processes such as lipolysis.33,34 It was shown that the ANP gene (NPPA) mutation increased platelet aggregation in vitro as well as in clinical settings.35 In another study, NPPA mutation increased residual platelet reactivity in patients with diabetes mellitus who underwent elective percutaneous coronary intervention.
Accordingly, GATA4 is considered to modulate vessel function and platelet aggregation via ANP regulation. In addition to ANP, vascular endothelial growth factor (VEGF) is also known to be regulated by GATA4. VEGF, an angiogenic cytokine, is related to vessel formation and vascular density.36 Therefore, the effect of GATA4 polymorphisms on bleeding complications was considered to be partially attributable to an altered vessel formation and function.
Rs13273672 is an introgenic SNP of GATA4. In several studies, this SNP was associated with alcohol dependence,37,38 relapse to alcohol drinking, and treatment response to acamprosate.39 In Kiefer et al study, patients with G allele of rs13273672 showed low ANP expression; therefore, the underlying mechanism of the rs13273672 effect on alcohol dependence and treatment was explained by the altered ANP expression. We found that rs13273672 of GATA4 had a significant association with bleeding complications at a therapeutic INR, with homozygous wild-type carriers having increased bleeding risk in both univariate and multivariate analyses. This was also thought to be caused by the alteration in ANP level.
GATA6, which is highly expressed in vascular smooth muscle cells, is known to regulate vascular smooth muscle cell proliferation and its reversible differentiation in vascular injury.40,41 Similar to GATA4, GATA6 is also an upstream regulator of multiple genes expressed during embryogenesis and cardiac morphogenesis, including the gene that encodes the ANP.42 Various studies showed that the GATA6 mutant demonstrated a significantly decreased transcriptional activity on the ANP promoter.43–45 Several studies have shown that GATA4 and GATA6 act cooperatively and synergistically to regulate smooth muscle cells, with similar structure and expression patterns.10,46
In our study, one SNP of GATA6 (rs10454095) exhibited a significant association with bleeding complication. Although rs10454095 is rarely studied, polymorphisms of GATA6 might cause an alteration in transcriptional activity and blood vessel regulation, thereby increasing bleeding risk. When rs10454095 in the GATA6 gene was combined with rs13273672 in the GATA4 gene, it showed an additive effect of each SNP on bleeding complications.
A recent study investigating the association between GATA6 polymorphisms and congenital malformations like bicuspid aortic valve included rs10454095. Although rs10454095 was not associated with bicuspid aortic valve in this study, the authors revealed that three GATA6 gene variants were associated with bicuspid aortic valve. Since the effects of gene polymorphisms may vary according to target organs or diseases, different SNPs could be found to be associated with different outcomes. Meanwhile, the results indicated the role of GATA6 polymorphisms on organ formation.47
GATA4 showed to play an important role on the regulation of CYP2C9 gene expression.48 However, patients with polymorphisms of VKORC1 or CYP2C9 received dose adjustment according to INR measurement, and increased risk of bleeding complication was not found in our study.
To evaluate the potential clinical value of SNP genotyping, which was found to be significant in this study, we calculated NNG for preventing bleeding complications in patients with high-risk genotypes. Using the equations, 22.2, 17.5, and 40.0 were determined to be NNG values in patients with high-risk genotypes of rs13273672, rs10454095, and both SNPs, respectively, indicating that prospective SNP genotyping could be cost-effective in clinical practice.
The limitations of our study are its small sample size and retrospective design. Another shortcoming is a lack of detailed mechanisms. Nevertheless, to our knowledge, this is the first study to investigate the effects of genetic variations in GATA4 and GATA6 genes on warfarin-associated bleeding complications at a therapeutic INR. In addition, this study provides the prediction models for bleeding risk using various statistical tools (eg, attributable risk, AUROC, and NNG), which can be applied for developing individualized drug therapy with warfarin.
Since this study dealt with patients with INR 2–3, only minimal or minor bleeding events were observed. While there is no doubt that fatal and major hemorrhages are of essential importance, minor bleedings are also important, because they serve as an alert for subsequent major bleedings and may increase the number of visits to clinics and sometimes the emergency room, which results in additional expenditures. They also can result in permanent withdrawal of warfarin therapy, thus depriving patients of the effective therapy available.
In this study, to avoid the possible loss of the true positives, multiple test correction was not performed. It is based on a rigorous follow-up of a cohort of patients with cardiac valve replacements to detect bleeding complications based on objective measurements, followed by a systematic and thorough exploration of polymorphisms in four genes of potential interest to the etiology of bleedings. Bonferroni correction was not applied, as this is considered overly conservative for a hypothesis-generating study. We found possible associations between GATA genes and bleeding risks; however, it should be implemented with a caution with the risk of false-positive results and it is needed to be verified by further replication studies.
Conclusion
This study showed that rs13273672 of GATA4 and rs10454095 of GATA6 were associated with bleeding complications at a therapeutic INR during warfarin treatment for mechanical heart valve patients. Given the retrospective study design and the relatively small sample size, our hypothesis requires further independent validation using a prospective study design with a large sample size.
Acknowledgments
This study was supported by Basic Science Research Program through the National Research Foundation (NRF) of Korea funded by the Ministry of Education (No. 2017R1D1A1B03034033) and the research grant of the Chungbuk National University in 2014.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Roden DM, Johnson JA, Kimmel SE, et al. Cardiovascular pharmacogenomics. Circ Res. 2011;109:807–820. doi:10.1161/CIRCRESAHA.110.230995
2. Nishimura RA, Otto CM, Bonow RO, et al. AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the american college of Cardiology/American heart association task force on practice guidelines. Circulation. 2014;129:2440–2492. doi:10.1161/CIR.0000000000000029
3. Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G;
4. Wysowski DK, Nourjah P, Swartz L. Bleeding complications with warfarin use: a prevalent adverse effect resulting in regulatory action. Arch Intern Med. 2007;167:1414–1419. doi:10.1001/archinte.167.13.1414
5. Pourgholi L, Goodarzynejad H, Mandegary A, et al. Gene polymorphisms and the risk of warfarin-induced bleeding complications at therapeutic international normalized ratio (INR). Toxicol Appl Pharmacol. 2016;309:37–43. doi:10.1016/j.taap.2016.08.026
6. Marie I, Leprince P, Menard JF, Tharasse C, Levesque H. Risk factors of vitamin K antagonist overcoagulation. Qjm. 2012;105:53–62. doi:10.1093/qjmed/hcr136
7. Weiss MJ, Orkin SH. GATA transcription factors: key regulators of hematopoiesis. Exp Hematol. 1995;23:99–107.
8. Molkentin JD. The zinc finger-containing transcription factors GATA-4, -5, and -6. Ubiquitously expressed regulators of tissue-specific gene expression. J Biol Chem. 2000;275:38949–38952. doi:10.1074/jbc.R000029200
9. Charron F, Paradis P, Bronchain O, Nemer G, Nemer M. Cooperative interaction between GATA-4 and GATA-6 regulates myocardial gene expression. Mol Cell Biol. 1999;19:4355–4365.
10. Xin M, Davis CA, Molkentin JD, et al. A threshold of GATA4 and GATA6 expression is required for cardiovascular development. Proc Natl Acad Sci U S A. 2006;103:11189–11194. doi:10.1073/pnas.0604604103
11. Yang F, Wu M, Li Y, et al. Mutation p.S335X in GATA4 reduces its DNA binding affinity and enhances cell apoptosis associated with ventricular septal defect. Curr Mol Med. 2013;13:993–999.
12. Krachulec J, Vetter M, Schrade A, et al. GATA4 is a critical regulator of gonadectomy-induced adrenocortical tumorigenesis in mice. Endocrinology. 2012;153:2599–2611. doi:10.1210/en.2011-2135
13. Xu YJ, Di RM, Qiao Q, et al. GATA6 loss-of-function mutation contributes to congenital bicuspid aortic valve. Gene. 2018;663:115–120. doi:10.1016/j.gene.2018.04.018
14. Bashawri LA, Ahmed MA. The approach to a patient with a bleeding disorder: for the primary care physician. J Family Community Med. 2007;14:53–58.
15. Uranishi R, Baev NI, Kim JH, Awad IA. Vascular smooth muscle cell differentiation in human cerebral vascular malformations. Neurosurgery. 2001;49:671–679.
16. Bauditz J, Lochs H. Angiogenesis and vascular malformations: antiangiogenic drugs for treatment of gastrointestinal bleeding. World J Gastroenterol. 2007;13:5979–5984.
17. Penn JS, Madan A, Caldwell RB, Bartoli M, Caldwell RW, Hartnett ME. Vascular endothelial growth factor in eye disease. Prog Retin Eye Res. 2008;27:331–371. doi:10.1016/j.preteyeres.2008.05.001
18. Lockwood CJ. Mechanisms of normal and abnormal endometrial bleeding. Menopause. 2011;18:408–411. doi:10.1097/GME.0b013e31820bf288
19. Song W, Wang H, Wu Q. Atrial natriuretic peptide in cardiovascular biology and disease (NPPA). Gene. 2015;569:1–6. doi:10.1016/j.gene.2015.06.029
20. Casco VH, Veinot JP, Kuroski de Bold ML, Rg M, Mm S, de Bold AJ. Natriuretic peptide system gene expression in human coronary arteries. J Histochem Cytochem. 2002;50:799–809. doi:10.1177/002215540205000606
21. Rubattu S, Sciarretta S, Valenti V, Stanzione R, Volpe M. Natriuretic peptides: an update on bioactivity, potential therapeutic use, and implication in cardiovascular diseases. Am J Hypertens. 2008;21:733–741. doi:10.1038/ajh.2008.174
22. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123:2736–2747. doi:10.1161/CIRCULATIONAHA.110.009449
23. Whirl-Carrillo M, McDonagh EM, Hebert JM, et al. Pharmacogenomics knowledge for personalized medicine. Clin Pharmacol Ther. 2012;92:414–417. doi:10.1038/clpt.2012.96
24. Ward LD, Kellis M. HaploReg v4: systematic mining of putative causal variants, cell types, regulators and target genes for human complex traits and disease. Nucleic Acids Res. 2016;44:D877–D881. doi:10.1093/nar/gkv1340
25. Sherry ST, Ward MH, Kholodov M, et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 2001;29:308–311.
26. van Schie RM, Wessels JA, Verhoef TI, et al. Evaluation of the effect of genetic variations in GATA-4 on the phenprocoumon and acenocoumarol maintenance dose. Pharmacogenomics. 2012;13:1917–1923. doi:10.2217/pgs.12.174
27. Whitlock RP, Sun JC, Fremes SE, Rubens FD, Teoh KH. Antithrombotic and thrombolytic therapy for valvular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141:e576S–600S. doi:10.1378/chest.11-2305
28. Zhou XM, Zhuang W, Hu JG, Li JM, Yu JF, Jiang L. Low-dose anticoagulation in Chinese patients with mechanical heart valves. Asian Cardiovasc Thorac Ann. 2005;13:341–344. doi:10.1177/021849230501300410
29. Matsuyama K, Matsumoto M, Sugita T, et al. Anticoagulant therapy in Japanese patients with mechanical mitral valves. Circ J. 2002;66:668–670.
30. Yoon IK, Lee KE, Lee JK, Chang BC, Gwak HS. Adequate intensity of warfarin therapy for Korean patients with mechanical cardiac valves. J Heart Valve Dis. 2013;22:102–109.
31. Hutchinson HG, Trindade PT, Cunanan DB, Wu CF, Pratt RE. Mechanisms of natriuretic-peptide-induced growth inhibition of vascular smooth muscle cells. Cardiovasc Res. 1997;35:158–167.
32. Kuhn M. Endothelial actions of atrial and B-type natriuretic peptides. Br J Pharmacol. 2012;166:522–531. doi:10.1111/j.1476-5381.2012.01827.x
33. Ulker S, Akgür S, Evinç A, Soykan N, Koşay S. Platelet aggregation and atrial natriuretic peptide. Gen Pharmacol. 1995;26:1409–1412.
34. Lafontan M, Moro C, Berlan M, Crampes F, Sengenes C, Galitzky J. Control of lipolysis by natriuretic peptides and cyclic GMP. Trends Endocrinol Metab. 2008;19:130–137. doi:10.1016/j.tem.2007.11.006
35. Carnevale R, Pignatelli P, Frati G, et al. C2238 ANP gene variant promotes increased platelet aggregation through the activation of Nox2 and the reduction of cAMP. Sci Rep. 2017;7:3797. doi:10.1038/s41598-017-03679-9
36. Heineke J, Auger-Messier M, Xu J, et al. Cardiomyocyte GATA4 functions as a stress-responsive regulator of angiogenesis in the murine heart. J Clin Invest. 2007;117:3198–3210. doi:10.1172/JCI32573
37. Reamon-Buettner SM, Cho SH, Borlak J. Mutations in the 3′-untranslated region of GATA4 as molecular hotspots for congenital heart disease (CHD). BMC Med Genet. 2007;8:38. doi:10.1038/jhg.2010.84
38. Azakie A, Fineman JR, He Y. Myocardial transcription factors are modulated during pathologic cardiac hypertrophy in vivo. J Thorac Cardiovasc Surg. 2006;132:1262–1271. doi:10.1016/j.jtcvs.2006.08.005
39. Kiefer F, Witt SH, Frank J, et al. Involvement of the atrial natriuretic peptide transcription factor GATA4 in alcohol dependence, relapse risk and treatment response to acamprosate. Pharmacogenomics J. 2011;11:368–374. doi:10.1038/tpj.2010.51
40. Yin F, Herring BP. GATA-6 can act as a positive or negative regulator of smooth muscle-specific gene expression. J Biol Chem. 2005;280:4745–4752. doi:10.1074/jbc.M411585200
41. Mano T, Luo Z, Malendowicz SL, Evans T, Walsh K. Reversal of GATA-6 downregulation promotes smooth muscle differentiation and inhibits intimal hyperplasia in balloon-injured rat carotid artery. Circ Res. 1999;84:647–654.
42. Pikkarainen S, Tokola H, Kerkelä R, Ruskoaho H. GATA transcription factors in the developing and adult heart. Cardiovasc Res. 2004;63:196–207. doi:10.1016/j.cardiores.2004.03.025
43. Li J, Liu W, Yang Z, Yang Y. Novel GATA6 loss-of-function mutation responsible for familial atrial fibrillation. Int J Mol Med. 2012;30:783–790. doi:10.3892/ijmm.2012.1068
44. Lin X, Huo Z, Liu X, et al. A novel GATA6 mutation in patients with tetralogy of Fallot or atrial septal defect. J Hum Genet. 2010;55:662–667. doi:10.1038/jhg.2010.84
45. Zheng GF, Wei D, Zhao H, Zhou N, Yang YQ, Liu XY. A novel GATA6 mutation associated with congenital ventricular septal defect. Int J Mol Med. 2012;29:1065–1071. doi:10.3892/ijmm.2012.930
46. Molkentin JD, Lin Q, Duncan SA, Olson EN. Requirement of the transcription factor GATA4 for heart tube formation and ventral morphogenesis. Genes Dev. 1997;11:1061–1072.
47. Gharibeh L, Komati H, Bossé Y, et al. GATA6 regulates aortic valve remodeling, and its haploinsufficiency leads to right-left type bicuspid aortic valve circulation. Circulation. 2018;138:1025–1038. doi:10.1161/CIRCULATIONAHA.117.029506
48. Mwinyi J, Nekvindová J, Cavaco I, et al. New insights into the regulation of CYP2C9 gene expression: the role of the transcription factor GATA-4. Drug Metab Dispos. 2010;38:415–421. doi:10.1124/dmd.109.029405
Supplementary material
 | Table S1 Allele frequencies of 20 single-nucleotide polymorphisms (SNPs) analyzed in the study |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

