Back to Journals » Journal of Multidisciplinary Healthcare » Volume 7
Gastrointestinal ulcers, role of aspirin, and clinical outcomes: pathobiology, diagnosis, and treatment
Authors Cryer B, Mahaffey K
Received 11 September 2013
Accepted for publication 17 October 2013
Published 3 March 2014 Volume 2014:7 Pages 137—146
DOI https://doi.org/10.2147/JMDH.S54324
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Byron Cryer,1 Kenneth W Mahaffey2
1University of Texas Southwestern Medical School, Dallas, TX, 2Department of Medicine, Stanford University, Stanford, CA, USA
Abstract: Peptic ulcer disease is a major cause of morbidity and mortality in the US with more than six million diagnoses annually. Ulcers are reported as the most common cause of hospitalization for upper gastrointestinal (GI) bleeding and are often a clinical concern due to the widespread use of aspirin and nonsteroidal anti-inflammatory drugs, both of which have been shown to induce ulcer formation. The finding that Helicobacter pylori infection (independent of aspirin use) is associated with the development of ulcers led to a more thorough understanding of the causes and pathogenesis of ulcers and an improvement in therapeutic options. However, many patients infected with H. pylori are asymptomatic and remain undiagnosed. Complicating matters is a current lack of understanding of the association between aspirin use and asymptomatic ulcer formation. Low-dose aspirin prescriptions have increased, particularly for cardioprotection. Unfortunately, the GI side effects associated with aspirin therapy continue to be a major complication in both symptomatic and asymptomatic patients. These safety concerns should be important considerations in the decision to use aspirin and warrant further education. The medical community needs to continue to improve awareness of aspirin-induced GI bleeding to better equip physicians and improve care for patients requiring aspirin therapy.
Keywords: low-dose aspirin, cardioprotection, ulcers, Helicobacter pylori, gastrointestinal bleeding, cardiovascular disease
Introduction
Peptic ulcer disease (PUD) has evolved as a major cause of morbidity and mortality throughout the 20th and 21st centuries,1 with more than six million people affected each year in the US alone.2 Ulcers are reported as the most common cause of hospitalization for upper gastrointestinal (GI) bleeding3 and remain an important clinical problem due to the increasingly widespread use of nonsteroidal anti-inflammatory drugs (NSAIDs) and aspirin, which are known to induce ulcers.1 The most common ulcers associated with PUD arise in the stomach, duodenum, and jejunum, with gastric ulcers presenting most frequently (Figure 1).2 Although ulcers are widely diagnosed and recognized, asymptomatic ulcers remain a pressing problem and may lead to long-term damage to the GI tract. While the risk of symptomatic ulcers with aspirin is well defined, the association between aspirin and asymptomatic ulcer formation remains less clear.
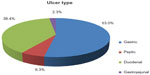  | Figure 1 Hospitalizations based on ulcer type from 1998–2005. |
The general understanding of ulcer formation greatly changed with the discovery of Helicobacter pylori in 1982. Since the discovery of H. pylori, the causes and pathogenesis of ulcers are better understood and the treatment protocols have evolved. The belief that ulcers develop due to an acid-driven mechanism was transformed to an understanding that H. pylori and aspirin are both important contributors to ulcer development. Now ulcer treatment efforts should focus on H. pylori eradication by antibiotics1 and reduction of aspirin’s contribution to ulcer pathogenesis. Strikingly, more than 50% of the population worldwide has chronic H. pylori infection, and an estimated 5%–10% of these persons will develop symptomatic ulcers.1 Although infection with H. pylori is commonly associated with PUD, many of those infected are asymptomatic and, therefore, remain undiagnosed.4 Undiagnosed peptic ulcer perforation is recognized as a cause of severe complications, such as perforations, and can result in death.5,6 Although symptomatic ulcers may be masked or delayed in some high-risk patients by using appropriate GI protection therapy, any delay in making the correct diagnosis may result in increased risk of complications and mortality. Among patients with symptomatic ulcers, nearly 30% of associated upper GI events result in hospitalization or death,7–9 and the total direct and indirect costs for PUD have been reported to be approximately $3.4 billion.10 Thus, evaluation of patients with ulcers for H. pylori infection is an important step in reducing the progression of uncomplicated ulcers to ulcers with complications.
H. pylori infection and NSAID (including aspirin) use are independently associated with adverse GI effects ranging from mild dyspepsia to serious GI bleeding, and may additively increase the risk of PUD.11 Other causes of PUD exist, but are much less common (Table 1).12 Aspirin is a common cause of ulcers, even in patients not infected with H. pylori.13 With administration of daily low-dose aspirin (LDA), GI mucosal damage14–16 occurs in approximately 40%–50% of patients; increased risk of GI bleeding17 is also observed. These risks typically peak closer to the beginning of therapy and increase with patient age.17,18 History of GI bleeding is important to consider, as 15% of patients who have bleeding from ulcers report recurrent bleeding within 1 year.19 As such, physicians should be cognizant of the potential for their patients to develop asymptomatic ulcers throughout the course of aspirin administration during cardiovascular disease therapy. In rare cases, patients who are negative for H. pylori and who have no history of aspirin use can develop ulcers.1 Due to the additive nature of these factors, all patients presenting with an ulcer, regardless of whether they are taking aspirin, should be tested for H. pylori infection to determine the best treatment plan.11
  | Table 1 General classifications of peptic ulcers |
For some patients, the clinical consequences beyond GI bleeding may be severe. Patients who continue to exhibit persistent symptoms of PUD should be referred for upper endoscopy. While GI bleeding is a common indication for surgery, the development of complications from PUD, unresponsiveness to therapy or a need for multiple rounds of medical therapy for ulcers, and high-risk factors (eg, history of PUD, dependence upon steroid or NSAID therapy) may increase the need for surgery. Surgical options for these patients include gastroduodenotomy and oversewing of the blood vessel, and excision of the ulcer with vagotomy and drainage/partial gastrectomy in bleeding gastric ulcers.20 If endoscopy is unsuccessful (ie, bleeding continues) or patients are not candidates for surgical intervention, angiography is an option.21
Aspirin-induced ulcers
Aspirin is among the most widely used medications in the US22 and is administered for an extensive variety of indications. Further, it is readily available over the counter.23 Aspirin originated as a medication to treat pain and inflammation, but due to its antiplatelet properties, it has evolved into a drug commonly used to prevent cardiovascular disease. Standard doses of aspirin ranging from 500 to 1,000 mg daily are mostly prescribed for inflammatory conditions and pain relief, whereas doses between 75 and 325 mg daily are usually prescribed as antiplatelet therapy for primary and secondary prevention of cardiovascular and cerebrovascular events.24,25 While the LDA for cardiovascular disease refers to doses <300 mg or <162 mg, it will be categorized as ≤325 mg for the purpose of this review.
All antithrombotic agents increase bleeding; aspirin, in particular, significantly increases the risk of major bleeding by about 60%.26 LDA is associated with intracranial or major extracranial events27 as well as prolonged bleeding;24 however, some of the most common side effects from aspirin are related to the upper GI tract. These range from mild conditions, such as dyspepsia, to more severe side effects, such as PUD and severe GI bleeding.24 Chronic aspirin consumption is associated with lower GI bleeding in more than 50% of aspirin users, indicating that GI injury is not only limited to the upper GI tract.28 Overall, aspirin has emerged as one of the most prominent causes of peptic ulcer bleeding in developed countries over the last 2 decades,29 and is associated with a 2- to 4-fold increased risk of upper GI bleeding and ulcers.30
Aspirin and H. pylori induce ulcers by different mechanisms, and the combination of the two can greatly increase the risk of ulceration (Figure 2).31 Theoretically, aspirin and H. pylori may interact in many ways to influence ulcer formation, yet H. pylori infection is not required for aspirin-associated ulcers to develop.12 Specifically, GI damage attributable to aspirin use is typically caused by a combination of epithelial and microvascular effects with little or no inflammation, while H. pylori ulcers are usually associated with diffuse inflammatory cell infiltration.12 LDA causes GI mucosal and systemic effects from prostaglandin depletion via inhibition of cyclooxygenase-1,30,32,33 but the relative contributions of local versus systemic effects of aspirin-related GI injury remain to be fully elucidated.34 Prostaglandins play a pivotal role in protecting gastric mucosal integrity via increasing local blood flow and promoting synthesis and secretion of mucus and bicarbonate. In the absence of normal prostaglandin synthesis, the gastric environment becomes more vulnerable to exogenous (eg, smoking) or endogenous factors (H. pylori, acid, pepsin, bile salts) and, consequently, more prone to develop peptic ulcer and bleeding complications.35 The acidic environment causes aspirin to remain nonionized, forcing it to accumulate in gastric mucosal cells, which alters the permeability of the cell and causes ulceration.30 Within only minutes of aspirin administration, topical gastric mucosa effects can be visualized by endoscopy.36 Additionally, LDA promotes GI bleeding via its antiplatelet effect.30
Major risk factors for upper GI events associated with aspirin use include a history of peptic ulcer or bleeding ulcer,37–42 concomitant use of other NSAIDs or antithrombotics,38,40,43–46 and H. pylori infection (Table 2).39,47–49 Other factors that may increase the risk of peptic ulcers are smoking, excessive alcohol consumption, drug use, and emotional stress. These factors are considered to be environmental, as they contribute to ulcer formation by increasing gastric acid secretion, ultimately resulting in a weakened mucosal barrier.1 Among the risk factors for LDA-associated ulcer bleeding, H. pylori infection is one of the few that are treatable and curable.29 Combination therapy of aspirin with other antiplatelet or anticoagulant therapy is increasingly common and includes dual antiplatelet therapy in patients treated with coronary stents or after acute coronary syndrome,50,51 in patients with cardiovascular disease who are on anticoagulant therapy for atrial fibrillation,52 or other indications. Dual antiplatelet therapy has clearly been shown to increase bleeding risk.53 Meta-analyses and randomized clinical trials have demonstrated that, while effective in management of cardiovascular disease,54 dual therapy can increase the risk of major bleeding events.55–58
  | Table 2 Risk factors for aspirin-induced ulcers |
Patients who require triple therapy (dual antiplatelet therapy and anticoagulant therapy) face unique clinical challenges and an increased risk of bleeding.59 Many studies have demonstrated a higher risk for bleeding,60–64 with the number of minor and major bleeds reported to be as high as 27.5%.63–66 As a result of these findings, the most critical element when determining the duration of triple therapy appears to be the risk of bleeding.67 US Food and Drug Administration approval of several new oral anticoagulants (dabigatran, apixaban, and rivaroxaban), as well as the ease of use of these agents, may eventually increase the use of anticoagulant therapy compared with current practice patterns with warfarin. It is clear that intracranial hemorrhage is reduced significantly with these agents compared with warfarin. GI side effects and bleeding events may be increased compared with warfarin.68,69 Further work is needed to better understand these observations and strategies to minimize patient risk.
Ulcer diagnosis
Several symptoms are associated with the diagnosis of ulcers, including epigastric pain, fullness, bloating, premature satiety, weight loss, and nausea.1 In some cases, these symptoms are associated with other illnesses, such as gastroesophageal reflux disease, general dyspepsia, or gastritis, which makes evaluation more challenging. Asymptomatic ulcers may go undetected until clinical presentation of a GI bleed.1 Physicians, including cardiologists, may be unaware of the occurrence of asymptomatic ulcers in patients taking LDA for cardioprotection, as these patients do not present with clinical signs or concerns after beginning the aspirin regimen. Awareness of PUD risk factors may help clinicians to identify patients at risk for ulcer formation and initiate appropriate gastric protection therapy.
All patients with an ulcer, regardless of whether they are taking aspirin, should be tested for H. pylori infection to determine the best treatment.11 Surprisingly, up to 35% of patients at high-risk for GI bleeding are neither tested for H. pylori infection nor given proton pump inhibitor (PPI) gastroprotection during the course of their NSAID therapy.49 Several invasive (eg, histology, rapid urease testing) and noninvasive (eg, antibody detection, nonendoscopic urease testing, fecal antigen testing) methods are used to diagnose ulcers and detect H. pylori infection.11 Once a clinician has decided to test a patient for infection, the next step is to determine if endoscopy is necessary for the procedure, based on the patient’s clinical presentation of symptoms.
Treatment, management, and prevention of ulcers
Upon diagnosis of an ulcer, patients’ need for aspirin therapy should be reviewed on a case-by-case basis to evaluate their risk-to-benefit ratio. Patients using LDA for primary prevention have a lower benefit of cardiovascular risk reduction than those using it for secondary prophylaxis. In cases of secondary prevention, failure to resume LDA after ulcer bleeding is associated with an increased mortality rate.70,71 Alternative methods of antiplatelet therapy may be considered in patients with a high risk of GI bleeding who are using aspirin for primary prevention. Previously, clopidogrel was a substitute therapy for LDA in patients with major upper GI intolerance. In 2008, however, guidelines were revised by the American College of Cardiology Foundation, American College of Gastroenterology, and American Heart Association to state that the use of clopidogrel is inferior to the combination of LDA and PPI for gastroprotection and is not recommended.25
Aspirin modifications have been made in attempts to make aspirin more tolerable in the GI tract. For example, enteric-coated aspirin with cellulose or silicon resists disintegration in the stomach, allowing aspirin to dissolve specifically in the duodenum where the pH is more alkaline.72 In other cases, buffering agents, such as calcium carbonate, magnesium oxide, or magnesium carbonate, are used to lower the hydrogen ion concentration in the GI tract, resulting in an increased solubility of aspirin in the stomach, which increases the rate of absorption and reduces the contact time aspirin has with the gastric mucosa, thereby reducing GI irritation.73 Although these modifications may reduce stomach upset in some people, they may not reduce the risk of GI bleeding.19,72 Enteric-coated aspirin has even been found to induce significant injury to the lower gut.28
Many different types of drugs have been used over the years to treat ulcers; those aimed at inhibiting gastric acid secretion have been the most successful (Table 3). Originally, H2-receptor antagonists were used to treat ulcers and were often used in maintenance therapy. Over time, they were gradually replaced with PPIs, which offer more potent acid inhibition and more rapid ulcer healing.1 Although other classes of drugs can be used, including misoprostol (a prostaglandin analog) and bismuth salts, PPIs have become the hallmark in ulcer therapy. Fixed-dose combination medication consisting of aspirin plus a PPI offers an alternative to prescribing patients two separate medications and provides more convenient dosing. Several studies have shown that fixed-dose combination therapy with aspirin and the PPI esomeprazole reduced the incidence of peptic ulcers and dyspeptic symptoms when compared with aspirin with placebo.74–76 Although PPIs are known to reduce gastric symptoms, patient adherence to an aspirin and PPI cotherapy regimen for gastroprotection is poor.34 A fixed-dose combination regimen, therefore, would likely increase patient adherence to an LDA treatment schedule. An additional class of drugs designed to treat ulcers is a noncovalent complex of aspirin and phosphatidylcholine.34 This approach, currently in clinical development, is targeted at reducing aspirin-induced GI toxicity in patients with H. pylori-negative ulcers and may have potential as an alternative to PPIs in certain patient populations.
  | Table 3 Classification of treatments for peptic ulcers |
For the treatment and long-term prevention of recurrent bleeding ulcers, recommendations differ based on the nature of the ulcer. Patients with H. pylori-associated ulcers should receive therapy specific to H. pylori eradication, whereas patients taking aspirin are recommended to take gastroprotective measures, such as a daily PPI or fixed-dose combination therapy (Figure 3).3 If untreated, long-term infection with H. pylori could potentially lead to asymptomatic chronic gastritis, chronic dyspepsia, duodenal ulcer disease, gastric ulcer disease, or gastric malignancy.77 The gold standard in the eradication of H. pylori is a 1-week regimen of triple therapy, consisting of PPI treatment with two antibiotics.78 In general, H. pylori-positive LDA users with bleeding ulcers showed a reduction in the rate of recurrent bleeding when H. pylori was eradicated or when a PPI was administered in conjunction with LDA.79,80 Another study found that eradicating H. pylori alone in patients undergoing LDA therapy, with a history of ulcer bleeding, was sufficient to reduce the long-term risk of recurrent bleeding.81 These data suggest a possible risk-reduction strategy by employing a test-and-treat tactic for the eradication of H. pylori; however, whether this method of reducing ulcer bleeding is cost-effective remains to be evaluated.
  | Figure 3 Flow chart of recommended management to prevent recurrent ulcer bleeding based on type. Reprinted by permission from Macmillan Publishers Ltd: American Journal of Gastroenterology. Laine L, Jensen DM. Management of patients with ulcer bleeding. Am J Gastroenterol. 2012;107(3):345–360. Copyright 2012.3 |
Raising awareness of possible asymptomatic ulcers
From 2007 to 2008, physicians prescribed aspirin and other antiplatelet medications in 46.9% of patient visits for patients with ischemic vascular disease.82 These data also showed that cardiologists prescribed antiplatelet drugs more frequently than primary care physicians (68% versus 35%, respectively),82 for unclear reasons.
Awareness of the GI adverse events related to aspirin use should rise in tandem with aspirin prescriptions. In fact, the American College of Cardiology Foundation/American College of Gastroenterology/American Heart Association Task Force recommends that aspirin doses >81 mg should not be routinely prescribed, since the risk of GI adverse events increases with dose escalation.25 Because the use of enteric-coated or buffered formulations of aspirin for cardioprotection is not sufficient to reduce the risk of GI bleeding, patients at risk of GI adverse events should be prescribed gastroprotection at the initiation of LDA therapy.
Proper review of patient history and follow-up are essential in the cardiology setting to appropriately risk stratify, manage, and educate patients about GI adverse events. One suggested strategy is to administer a patient history questionnaire with a special focus on the risk factors for GI bleeds at the time of prescription. Cardiologists will become aware of the patient’s need for gastroprotection or H. pylori screening if the patient has a previous history of infection. This approach may help prevent future complications and ulcers. Overall, for patients with an increased ulcer risk, H. pylori screening is recommended to avoid the additive effect of aspirin use with H. pylori infection. A dialogue about the symptoms of peptic ulcers may help both the patient and physician to recognize a potential peptic ulcer before it worsens, and potentially avoid any other serious complications.
Proper patient evaluation and physician awareness provide more than the obvious health benefits. Preventive and follow-up screening have the potential to both decrease adverse events and generate cost-savings to patients who fall within the high-risk population for GI bleeds and are prescribed an antibiotic eradication regimen.12 Economic analysis has demonstrated the benefit of initial noninvasive H. pylori testing for patients with suspected PUD.83 Cost-effectiveness data suggest that H. pylori testing and treatment are appropriate for all patients with suspected PUD, even if the majority of patients will not benefit.83 Although clinicians are faced with several alternatives to eradicate H. pylori, few regimens are able to meet the criteria of being simple, inexpensive, and effective.84 Moreover, even though the early detection of asymptomatic H. pylori infection may help to reduce the risk of ulcer formation, it is more difficult to diagnose an H. pylori-negative asymptomatic ulcer due to aspirin use alone. Endoscopic methods are available and sensitive to this detection; however, endoscopy is not always accessible or covered by health insurance for asymptomatic patients. Although early detection is not always possible, it can prevent serious complications and provide a cost benefit to patients.
Conclusion and future directions
Cardiovascular disease is well recognized as the leading cause of death in the US, as well as in many developing countries around the world.85 Although the benefits of LDA use for cardioprotection are clear, GI side effects result in substantial morbidity for both symptomatic and asymptomatic patients. Gastroduodenal ulcers and erosions are observed in approximately one third of asymptomatic patients taking LDA and gastroprotective agents,30 leading guidelines to recommend aspirin use only in patients with a risk of cardiovascular disease over a particular threshold.26,34,86–89 However, results from the National Ambulatory Medical Care Survey and the National Hospital Ambulatory Medical Care Survey demonstrate that aspirin is prescribed in <50% of patients deemed appropriate for LDA therapy.82
Although the GI effects of aspirin may result in substantial morbidity and mortality, a continued downward trend is observed for the rate of hospitalizations due to PUD over the past decade. The decline in PUD hospitalization rates is attributed in part to a decline in H. pylori infections due to awareness, improvements in antibiotic treatment regimens, and possibly trends in safer use of NSAIDs and gastroprotective agents.2 Data such as these suggest an optimistic future where physician and patient awareness may reduce aspirin-induced GI bleeding.
Disclosure
Pozen Inc. (Chapel Hill, NC, USA) provided funding for coordination and editorial support of a review on the general topic of aspirin and ulcers. Content and direction were the sole discretion of the authors without review by Pozen Inc. employees. Scientific editorial support was provided by Courtney Mezzacappa Zeni, PhD (QSci Communications, King of Prussia, PA, USA). BC discloses that he has consulting relationships with AstraZeneca Inc., Horizon Therapeutics, Inc., Pozen Inc., Ritter Pharmaceuticals, and Sucampo, Inc. He has received research grants from PLx Pharma and Pfizer Inc. KWM discloses that he has served as a consultant for Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Eli Lilly and Company, GlaxoSmithKline, Johnson & Johnson, Merck, Ortho-McNeil, Pfizer, Polymedix Inc., and Sanofi. He also discloses that he has received institution grant support from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, Eli Lilly and Company, GlaxoSmithKline, Johnson & Johnson, Merck, Portola Pharmaceuticals, Pozen Inc., Regado Biosciences, Sanofi, Schering-Plough (now Merck), and The Medicines Company.
References
Malfertheiner P, Chan FK, McColl KE. Peptic ulcer disease. Lancet. 2009;374(9699):1449–1461. | |
Feinstein LB, Holman RC, Yorita Christensen KL, Steiner CA, Swerdlow DL. Trends in hospitalizations for peptic ulcer disease, United States, 1998–2005. Emerg Infect Dis. 2010;16(9):1410–1418. | |
Laine L, Jensen DM. Management of patients with ulcer bleeding. Am J Gastroenterol. 2012;107(3):345–360. | |
Hasni S, Ippolito A, Illei GG. Helicobacter pylori and autoimmune diseases. Oral Dis. 2011;17(7):621–627. | |
Cina SJ, Mims WW 3rd, Nichols CA, Conradi SE. From emergency room to morgue: deaths due to undiagnosed perforated peptic ulcers. Report of four cases with review of the literature. Am J Forensic Med Pathol. 1994;15(1):21–27. | |
Armstrong CP, Whitelaw S. Death from undiagnosed peptic ulcer complications: a continuing challenge. Br J Surg. 1988;75(11):1112–1114. | |
Griffin MR, Piper JM, Daugherty JR, Snowden M, Ray WA. Nonsteroidal anti-inflammatory drug use and increased risk for peptic ulcer disease in elderly persons. Ann Intern Med. 1991;114(4):257–263. | |
Griffin MR, Ray WA, Schaffner W. Nonsteroidal anti-inflammatory drug use and death from peptic ulcer in elderly persons. Ann Intern Med. 1988;109(5):359–363. | |
Smalley WE, Ray WA, Daugherty JR, Griffin MR. Nonsteroidal anti-inflammatory drugs and the incidence of hospitalizations for peptic ulcer disease in elderly persons. Am J Epidemiol. 1995;141(6):539–545. | |
Sandler RS, Everhart JE, Donowitz M, et al. The burden of selected digestive diseases in the United States. Gastroenterology. 2002;122(5):1500–1511. | |
Saad R, Chey WD. A clinician’s guide to managing Helicobacter pylori infection. Cleve Clin J Med. 2005;72(2):109–110, 112–113, 117–118 passim. | |
Fendrick AM, Scheiman JM. Helicobacter pylori and NSAID gastropathy: an ambiguous association. Curr Rheumatol Rep. 2001;3(2):107–111. | |
Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs. A meta-analysis. Ann Intern Med. 1991;115(10):787–796. | |
Goldstein JL, Lowry SC, Lanza FL, Schwartz HI, Dodge WE. The impact of low-dose aspirin on endoscopic gastric and duodenal ulcer rates in users of a non-selective non-steroidal anti-inflammatory drug or a cyclo-oxygenase-2-selective inhibitor. Aliment Pharmacol Ther. 2006;23(10):1489–1498. | |
Niv Y, Battler A, Abuksis G, Gal E, Sapoznikov B, Vilkin A. Endoscopy in asymptomatic minidose aspirin consumers. Dig Dis Sci. 2005;50(1):78–80. | |
Yeomans ND, Lanas AI, Talley NJ, et al. Prevalence and incidence of gastroduodenal ulcers during treatment with vascular protective doses of aspirin. Aliment Pharmacol Ther. 2005;22(9):795–801. | |
Hernandez-Diaz S, Garcia Rodriguez LA. Cardioprotective aspirin users and their excess risk of upper gastrointestinal complications. BMC Med. 2006;4:22. | |
Moukarbel GV, Signorovitch JE, Pfeffer MA, et al. Gastrointestinal bleeding in high risk survivors of myocardial infarction: the VALIANT Trial. Eur Heart J. 2009;30(18):2226–2232. | |
Hirata Y, Kataoka H, Shimura T, et al. Incidence of gastrointestinal bleeding in patients with cardiovascular disease: buffered aspirin versus enteric-coated aspirin. Scand J Gastroenterol. 2011;46(7–8):803–809. | |
Ramakrishnan K, Salinas RC. Peptic ulcer disease. Am Fam Physician. 2007;76(7):1005–1012. | |
Eisen GM, Dominitz JA, Faigel DO, et al. An annotated algorithmic approach to upper gastrointestinal bleeding. Gastrointest Endosc. 2001;53(7):853–858. | |
Abraham NS, El-Serag HB, Johnson ML, et al. National adherence to evidence-based guidelines for the prescription of nonsteroidal anti-inflammatory drugs. Gastroenterology. 2005;129(4):1171–1178. | |
Hennekens CH, Sechenova O, Hollar D, Serebruany VL. Dose of aspirin in the treatment and prevention of cardiovascular disease: current and future directions. J Cardiovasc Pharmacol Ther. 2006;11(3):170–176. | |
Valkhoff VE, Sturkenboom MC, Kuipers EJ. Risk factors for gastrointestinal bleeding associated with low-dose aspirin. Best Pract Res Clin Gastroenterol. 2012;26(2):125–140. | |
Bhatt DL, Scheiman J, Abraham NS, et al. ACCF/ACG/AHA 2008 expert consensus document on reducing the gastrointestinal risks of antiplatelet therapy and NSAID use: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. Circulation. 2008;118(18):1894–1909. | |
Antithrombotic Trialists Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002;324(7329):71–86. | |
Pignone M, Alberts MJ, Colwell JA, et al. Aspirin for primary prevention of cardiovascular events in people with diabetes: a position statement of the American Diabetes Association, a scientific statement of the American Heart Association, and an expert consensus document of the American College of Cardiology Foundation. Circulation. 2010;121(24):2694–2701. | |
Lichtenberger LM, Phan T, Okabe S. Aspirin’s ability to induce intestinal injury in rats is dependent on bile and can be reversed if pre-associated with phosphatidylcholine. J Physiol Pharmacol. 2011;62(4):491–496. | |
Chan FK. Anti-platelet therapy and managing ulcer risk. J Gastroenterol Hepatol. 2012;27(2):195–199. | |
Tamura A, Murakami K, Kadota J; OITA-GF Study Investigators. Prevalence and independent factors for gastroduodenal ulcers/erosions in asymptomatic patients taking low-dose aspirin and gastroprotective agents: the OITA-GF study. QJM. 2011;104(2):133–139. | |
Huang JQ, Sridhar S, Hunt RH. Role of Helicobacter pylori infection and non-steroidal anti-inflammatory drugs in peptic-ulcer disease: a meta-analysis. Lancet. 2002;359(9300):14–22. | |
Hawthorne AB, Mahida YR, Cole AT, Hawkey CJ. Aspirin-induced gastric mucosal damage: prevention by enteric-coating and relation to prostaglandin synthesis. Br J Clin Pharmacol. 1991;32(1):77–83. | |
Hawkey CJ. Review article: aspirin and gastrointestinal bleeding. Aliment Pharmacol Ther. 1994;8(2):141–146. | |
Cryer B, Bhatt DL, Lanza FL, Dong JF, Lichtenberger LM, Marathi UK. Low-dose aspirin-induced ulceration is attenuated by aspirin-phosphatidylcholine: a randomized clinical trial. Am J Gastroenterol. 2011;106(2):272–277. | |
Lettino M. Inhibition of the antithrombotic effects of clopidogrel by proton pump inhibitors: facts or fancies? Eur J Intern Med. 2010;21(6):484–489. | |
Lichtenberger LM. Where is the evidence that cyclooxygenase inhibition is the primary cause of nonsteroidal anti-inflammatory drug (NSAID)-induced gastrointestinal injury? Topical injury revisited. Biochem Pharmacol. 2001;61(6):631–637. | |
Lanas A, Scheiman J. Low-dose aspirin and upper gastrointestinal damage: epidemiology, prevention and treatment. Curr Med Res Opin. 2007;23(1):163–173. | |
Cea Soriano L, Rodriguez LA. Risk of upper gastrointestinal bleeding in a cohort of new users of low-dose ASA for secondary prevention of cardiovascular outcomes. Front Pharmacol. 2010;1:126. | |
Lanas A, Fuentes J, Benito R, Serrano P, Bajador E, Sainz R. Helicobacter pylori increases the risk of upper gastrointestinal bleeding in patients taking low-dose aspirin. Aliment Pharmacol Ther. 2002;16(4):779–786. | |
Ng W, Wong WM, Chen WH, et al. Incidence and predictors of upper gastrointestinal bleeding in patients receiving low-dose aspirin for secondary prevention of cardiovascular events in patients with coronary artery disease. World J Gastroenterol. 2006;12(18):2923–2927. | |
Okada K, Inamori M, Imajo K, et al. Clinical study of upper gastrointestinal bleeding associated with low-dose aspirin in Japanese patients. Hepatogastroenterology. 2009;56(96):1665–1669. | |
Serrano P, Lanas A, Arroyo MT, Ferreira IJ. Risk of upper gastrointestinal bleeding in patients taking low-dose aspirin for the prevention of cardiovascular diseases. Aliment Pharmacol Ther. 2002;16(11):1945–1953. | |
Lanas A, Bajador E, Serrano P, et al. Nitrovasodilators, low-dose aspirin, other nonsteroidal antiinflammatory drugs, and the risk of upper gastrointestinal bleeding. N Engl J Med. 2000;343(12):834–839. | |
de Abajo FJ, Garcia Rodriguez LA. Risk of upper gastrointestinal bleeding and perforation associated with low-dose aspirin as plain and enteric-coated formulations. BMC Clin Pharmacol. 2001;1:1. | |
Lanas A, Garcia-Rodriguez LA, Arroyo MT, et al. Risk of upper gastrointestinal ulcer bleeding associated with selective cyclo-oxygenase-2 inhibitors, traditional non-aspirin non-steroidal anti-inflammatory drugs, aspirin and combinations. Gut. 2006;55(12):1731–1738. | |
Weil J, Colin-Jones D, Langman M, et al. Prophylactic aspirin and risk of peptic ulcer bleeding. BMJ. 1995;310(6983):827–830. | |
Udd M, Miettinen P, Palmu A, et al. Analysis of the risk factors and their combinations in acute gastroduodenal ulcer bleeding: a case-control study. Scand J Gastroenterol. 2007;42(12):1395–1403. | |
Sostres C, Gargallo CJ, Arroyo MT, Lanas A. Adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs, aspirin and coxibs) on upper gastrointestinal tract. Best Pract Res Clin Gastroenterol. 2010;24(2):121–132. | |
Zullo A, Hassan C, Oliveti D, et al. Helicobacter pylori management in non-steroidal anti-inflammatory drug therapy patients in primary care. Intern Emerg Med. 2012;7(4):331–335. | |
Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction – executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation. 2004;110(5):588–636. | |
Writing Committee Members, Jneid H, Anderson JL, Wright WS, et al. 2012 ACCF/AHA focused update of the guideline for the management of patients with unstable angina/Non-ST-elevation myocardial infarction (updating the 2007 guideline and replacing the 2011 focused update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2012;126(7):875–910. | |
Wann LS, Curtis AB, January CT, et al. 2011 ACCF/AHA/HRS focused update on the management of patients with atrial fibrillation (Updating the 2006 Guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2011;57(2):223–242. | |
Bhatt DL, Scheiman J, Abraham NS, et al. ACCF/ACG/AHA 2008 expert consensus document on reducing the gastrointestinal risks of antiplatelet therapy and NSAID use: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2008;52(18):1502–1517. | |
Rubboli A, Halperin JL. Pro: ‘Antithrombotic therapy with warfarin, aspirin and clopidogrel is the recommended regime in anticoagulated patients who present with an acute coronary syndrome and/or undergo percutaneous coronary interventions’. Thromb Haemost. 2008;100(5):752–753. | |
[No authors listed]. Low-molecular-weight heparin during instability in coronary artery disease, Fragmin during Instability in Coronary Artery Disease (FRISC) study group. Lancet. 1996;347(9001):561–568. | |
Andreotti F, Testa L, Biondi-Zoccai GG, Crea F. Aspirin plus warfarin compared to aspirin alone after acute coronary syndromes: an updated and comprehensive meta-analysis of 25,307 patients. Eur Heart J. 2006;27(5):519–526. | |
Collins R, MacMahon S, Flather M, et al. Clinical effects of anticoagulant therapy in suspected acute myocardial infarction: systematic overview of randomised trials. BMJ. 1996;313(7058):652–659. | |
Yusuf S, Mehta SR, Xie C, et al. Effects of reviparin, a low-molecular-weight heparin, on mortality, reinfarction, and strokes in patients with acute myocardial infarction presenting with ST-segment elevation. JAMA. 2005;293(4):427–435. | |
Paikin JS, Wright DS, Crowther MA, Mehta SR, Eikelboom JW. Triple antithrombotic therapy in patients with atrial fibrillation and coronary artery stents. Circulation. 2010;121(18):2067–2070. | |
Buresly K, Eisenberg MJ, Zhang X, Pilote L. Bleeding complications associated with combinations of aspirin, thienopyridine derivatives, and warfarin in elderly patients following acute myocardial infarction. Arch Intern Med. 2005;165(7):784–789. | |
Gilard M, Blanchard D, Helft G, et al. Antiplatelet therapy in patients with anticoagulants undergoing percutaneous coronary stenting (from STENTIng and oral antiCOagulants [STENTICO]). Am J Cardiol. 2009;104(3):338–342. | |
Mattichak SJ, Reed PS, Gallagher MJ, Boura JA, O’Neill WW, Kahn JK. Evaluation of safety of warfarin in combination with antiplatelet therapy for patients treated with coronary stents for acute myocardial infarction. J Interv Cardiol. 2005;18(3):163–166. | |
Orford JL, Fasseas P, Melby S, et al. Safety and efficacy of aspirin, clopidogrel, and warfarin after coronary stent placement in patients with an indication for anticoagulation. Am Heart J. 2004;147(3):463–467. | |
Ruiz-Nodar JM, Marin F, Hurtado JA, et al. Anticoagulant and antiplatelet therapy use in 426 patients with atrial fibrillation undergoing percutaneous coronary intervention and stent implantation implications for bleeding risk and prognosis. J Am Coll Cardiol. 2008;51(8):818–825. | |
Rubboli A, Halperin JL, Airaksinen KE, et al. Antithrombotic therapy in patients treated with oral anticoagulation undergoing coronary artery stenting. An expert consensus document with focus on atrial fibrillation. Ann Med. 2008;40(6):428–436. | |
Schomig A, Sarafoff N, Seyfarth M. Triple antithrombotic management after stent implantation: when and how? Heart. 2009;95(15):1280–1285. | |
Faxon DP, Eikelboom JW, Berger PB, et al. Antithrombotic therapy in patients with atrial fibrillation undergoing coronary stenting: a North American perspective: executive summary. Circ Cardiovasc Interv. 2011;4(5):522–534. | |
Bytzer P, Connolly SJ, Yang S, et al. Analysis of upper gastrointestinal adverse events among patients given dabigatran in the RE-LY trial. Clin Gastroenterol Hepatol. 2013;11(3):246–252. e1–e5. | |
Nessel CC, Mahaffey KW, Piccini JP, et al. Incidence and outcomes of gastrointestinal hemorrhage in patients with atrial fibrillation treated with rivaroxaban or warfarin: results from the ROCKET AF trial. Chest. 2012;142(Suppl 4):84A. | |
Antithrombotic Trialists’ (ATT) Collaboration, Baigent C, Blackwell L, Collins R, et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373(9678):1849–1860. | |
Sung JJ, Lau JY, Ching JY, et al. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med. 2010;152(1):1–9. | |
Kelly JP, Kaufman DW, Jurgelon JM, Sheehan J, Koff RS, Shapiro S. Risk of aspirin-associated major upper-gastrointestinal bleeding with enteric-coated or buffered product. Lancet. 1996;348(9039):1413–1416. | |
Lanza FL, Royer GL Jr, Nelson RS. Endoscopic evaluation of the effects of aspirin, buffered aspirin, and enteric-coated aspirin on gastric and duodenal mucosa. N Engl J Med. 1980;303(3):136–138. | |
Burness CB, Scott LJ. Acetylsalicylic acid/esomeprazole fixed-dose combination. Drugs Aging. 2012;29(3):233–242. | |
Yeomans N, Lanas A, Labenz J, et al. Efficacy of esomeprazole (20 mg once daily) for reducing the risk of gastroduodenal ulcers associated with continuous use of low-dose aspirin. Am J Gastroenterol. 2008;103(10):2465–2473. | |
Scheiman JM, Devereaux PJ, Herlitz J, et al. Prevention of peptic ulcers with esomeprazole in patients at risk of ulcer development treated with low-dose acetylsalicylic acid: a randomised, controlled trial (OBERON). Heart. 2011;97(10):797–802. | |
Tytgat GN. Long-term consequences of Helicobacter pylori eradication. Scand J Gastroenterol Suppl. 1994;205:38–44. | |
Sabbi T. Short review about Helicobacter pylori infection in pediatric age: epidemiological and clinical findings, diagnosis, therapy and role of probiotics. Pediatr Med Chir. 2011;33(5–6):221–226. | |
Chan FK, Chung SC, Suen BY, et al. Preventing recurrent upper gastrointestinal bleeding in patients with Helicobacter pylori infection who are taking low-dose aspirin or naproxen. N Engl J Med. 2001;344(13):967–973. | |
Lai KC, Lam SK, Chu KM, et al. Lansoprazole for the prevention of recurrences of ulcer complications from long-term low-dose aspirin use. N Engl J Med. 2002;346(26):2033–2038. | |
Chan FK, Ching J, Sune BY, et al. Effect of H. pylori eradication on the long-term incidence of recurrent ulcer bleeding in high-risk aspirin users: a 10-year prospective cohort study. Gastroenterology. 2011;140:173–174. | |
George MG, Tong X, Sonnenfeld N, et al. Recommended use of aspirin and other antiplatelet medications among adults – National Ambulatory Medical Care Survey and National Hospital Ambulatory Medical Care Survey, United States, 2005–2008. MMWR Surveill Summ. 2012;61(2):11–18. | |
Fendrick A, Forsch R, Harrison R, Scheiman J. Peptic ulcer disease. University of Michigan Health System Guidelines for Clinical Care. 2005:1–7. Available from: http://www.med.umich.edu/1info/fhp/practiceguides/newpud/pud.pdf. | |
Jone G, Morreale A. Cost-benefit computer modeling of Helicobacter pylori testing and treatment in patients on long-term H2-blocker prophylaxis. J Managed Care Pharm. 2000;6(5):383–389. | |
Hennekens CH. Increasing burden of cardiovascular disease: current knowledge and future directions for research on risk factors. Circulation. 1998;97(11):1095–1102. | |
Antiplatelet Trialists’ Collaboration. Collaborative overview of randomised trials of antiplatelet therapy-III: Reduction in venous thrombosis and pulmonary embolism by antiplatelet prophylaxis among surgical and medical patients. BMJ. 1994;308(6923):235–246. | |
US Preventive Services Task Force. Aspirin for the prevention of cardiovascular disease: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150(6):396–404. | |
O’Grady NP, Alexander M, Dellinger EP, et al. Guidelines for the prevention of intravascular catheter-related infections. Am J Infect Control. 2002;30(8):476–489. | |
British Cardiac Society, British Hypertension Society, Diabetes UK, Heart UK, Primary Care Cardiovascular Society, Stroke Association. JBS 2: Joint British Societies’ guidelines on prevention of cardiovascular disease in clinical practice. Heart. 2005;91 Suppl 5:v1–v52. | |
Lanza FL, Chan FK, Quigley EM; Practice Parameters Committee of the American College of Gasreoenterology. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol. 2009;104(3):728–738. | |
Quinlan DJ, Eikelboom JW, Goodman SG, et al. Implications of variability in definition and reporting of major bleeding in randomized trials of oral P2Y12 inhibitors for acute coronary syndromes. Eur Heart J. 2011;32(18):2256–2265. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.