Back to Journals » Drug Design, Development and Therapy » Volume 9
The effect of curcumin and its nanoformulation on adjuvant-induced arthritis in rats
Authors Zheng Z, Sun Y, Liu Z, Zhang M, Li C, Cai H
Received 9 June 2015
Accepted for publication 27 July 2015
Published 27 August 2015 Volume 2015:9 Pages 4931—4942
DOI https://doi.org/10.2147/DDDT.S90147
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Wei Duan
Zhaoling Zheng,1,* YanHua Sun,2,* Ziliang Liu,1 Mingqin Zhang,1 Chunqing Li,1 Hui Cai3
1Department of Traditional Chinese Medicine, Dongying People’s Hospital, Dongying, 2Shandong Provincial Key Laboratory of Microparticles Drug Delivery Technology, Jinan, 3Department of Integrated Traditional Chinese Medicine and Western Medicine, Nanjing Jinling Hospital, Nanjing, People’s Republic of China
*These authors contributed equally to this work
Background: Rheumatoid arthritis (RA), induced by the prolonged inappropriate inflammatory responses, is one of the most prevalent of all chronic inflammatory joint diseases. Curcumin (CM), a yellow hydrophobic polyphenol derived from the herb turmeric, has various pharmacological activities against many chronic diseases and acts by inhibiting cell proliferation and metastasis and downregulating various factors, including nuclear factor kappa B, interleukin-1β and TNF-α. Given the pathogenesis of RA, we hypothesized that the drug also has antiarthritic effects. The aims of the present study included the following: 1) examining the therapeutic effect of CM administered via intravenous (iv) injection on RA and 2) formulating the drug into oil–water nanoemulsions (Ns) to overcome the low oral bioavailability of CM and achieve oral delivery of the drug.
Methods: The effect of CM administered through iv injection on adjuvant-induced arthritis in rats was studied in terms of paw swelling, weight indices of the thymus and spleen, and pathological changes in nuclear factor kappa B expression and inflammatory cytokines. Methotrexate was used as a positive control. The CM-Ns were prepared using a high-pressure homogenizing method and characterized with respect to the particle size and morphology. The stability of the CM-Ns in simulated gastrointestinal (GI) fluids and in vitro release were also investigated. A pharmacokinetic study of the CM-Ns and suspensions in which the plasma levels were determined using an high performance liquid chromatography method and the pharmacokinetic parameters were calculated based on a statistical moment theory was also performed in rats.
Results: CM administered via iv injection had a therapeutic effect on RA similar to methotrexate. CM-Ns with a diameter of approximately 150 nm were successfully prepared, and the drug was well encapsulated into the Ns without degradation in simulated GI conditions. The area under the curve (AUC) and Cmax for the CM-Ns were more than threefold greater than those for the suspensions; moreover, similar decreases in the levels of TNF-α and interleukin-1β in both synovial fluid and blood serum were obtained from oral administration of CM-Ns and iv injection.
Conclusion: CM was an effective antiarthritic agent, and the present N formulation appeared to be a promising system that allowed RA therapy with CM to be converted from iv to oral administration.
Keywords: curcumin, nanoemulsions, inflammatory cytokines, adjuvant-induced arthritis, drug release, oral bioavailability
Introduction
Rheumatoid arthritis (RA), which is characterized by pain, swelling, and the destruction of joints, is one of most prevalent of all chronic inflammatory joint diseases.1 Approximately 1% of the population in the world suffers from the disease, with more than 3,000,000 new cases being diagnosed each year.2 RA not only induces disability that affects physical function and mobility but also results in premature death; moreover, patients with RA have a poorer life prognosis and a substantially shorter life expectancy than does the general population.3 Thus, the development of an effective therapeutic strategy for RA focusing on the prevention and treatment of the disease, its complications, and associated disorders, is urgent.
Curcumin (CM) is a yellow hydrophobic polyphenol derived from the herb turmeric, the structure of which is presented in Figure 1. CM possesses various pharmacological activities against many chronic diseases, including cancer, type II diabetes, multiple sclerosis, Alzheimer’s disease, and atherosclerosis. The molecular mechanisms for these effects of the drug involve not only the suppression of cell proliferation and metastasis but also the downregulation of various factors, including nuclear factor kappa B (NF-κB), interleukin (IL)-1β, tumor necrosis factor (TNF)-α, hypoxia-inducible factor-1α, and activator protein.4,5 In general, numerous cytokines are expressed and are functionally active in synovial tissues, and multiple factors, including TNF-induced NF-κB, IL-1β and TNF-α, are associated with the pathogenesis of RA.6 In particular, TNF is now targeted in the standard treatment of RA patients.7 Given the activity of CM and the pathogenesis of RA, we hypothesized that the drug could also exert a therapeutic effect on RA.
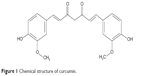  | Figure 1 Chemical structure of curcumin. |
It is well known that oral administration is the most widely used and most readily accepted route for patients, owing to its high patient compliance and flexibility. However, the oral bioavailability of CM is very low, thus greatly limiting its clinical use. The reasons for the low bioavailability include the following:4,8 1) the drug has very low water-solubility (0.0004 mg/mL); 2) oral administration of CM is subject to rapid intestinal metabolism, with approximately 75% of the oral dose being excreted in the feces; and 3) the absorbed CM undergoes rapid first-pass metabolism. Thus, a new dosage form that can improve CM’s bioavailability and stability is highly desirable.
Nanoemulsions (Ns) 100–500 nm in diameter are heterogeneous systems that are composed of water and oil phases. The oil–water-based Ns have various advantages over other nanocarriers for delivering insoluble drugs.9 Generally, a single oral dose for most of insoluble drugs must be high: more than 100 mg/patient. Thus the drug-loading capacity of the nanoformulations is critically important for the delivery of insoluble drugs. Of the current nanocarriers, which include liposomes, Ns, drug nanocrystals, and polymeric micelles, Ns have the highest drug-loading capacity, achieved by solubilizing the drug in the oil phase.10,11 Second, because of the core–shell structure of Ns, the drug within the oil cores can be protected from degradation by gastrointestinal (GI) enzymes and physiological pH.11 Third, an increase in the surface-to-volume ratio of Ns enhances the drug transport across cell membranes, obtaining targeted delivery and prolonging drug release.12 Fourth, Ns are easily fabricated and scaled-up; therefore, this is a promising technique for industrialization and application in the clinic.13,14 Overall, Ns, with their ability to enhance solubility, stability, and bioavailability of drugs, are potent nanocarriers for oral delivery of insoluble drugs.
In the current study, we first studied the therapeutic effect of CM on RA via intravenous (iv) injection to answer the question as to whether CM has antiarthritic effects. Second, to overcome the low oral bioavailability of CM and to achieve oral delivery of the drug, we incorporated the drug into the Ns by solubilizing CM into the oil droplets and then investigated the stability in simulated GI conditions and the drug release, pharmacokinetics and antiarthritic effects of CM-loaded Ns (CM-Ns) via oral administration. To our knowledge, few reports have indicated that CM could produce a strong therapeutic effect on RA. The present study suggested that when administered as an iv injection, CM had antiarthritic effects similar to those of methotrexate (MTX), which is the most frequently chosen disease-modifying antirheumatic therapy for RA.3 Moreover, by formulating the drug into Ns, its oral bioavailability was increased by more than threefold compared with the suspensions, thus resulting in better antiarthritic effects. Overall, we found that CM was an effective antiarthritic agent, and the present Ns appeared to be promising systems, allowing RA therapy with CM to be converted from iv to oral administration.
Materials and methods
Materials and animals
CM and Freund’s complete adjuvant (FCA) were obtained from Sigma-Aldrich Co. (St Louis, MO, USA). MTX was purchased from Jiangsu Hengrui Medicine Co., Ltd. (Lianyungang, People’s Republic of China). Solutol-HS 15 was supplied by BASF Co., Ltd. (Shanghai, People’s Republic of China). Pepsin, pancreatin, and sodium lauryl sulfate were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, People’s Republic of China). Hydroxypropyl methyl cellulose E5 was obtained from Colorcon Co., Ltd. (Shanghai, People’s Republic of China). A MaxVision™ HRP-Polymer anti-Rabbit IHC Kit was from MXB Co., Ltd. (Fuzhou, People’s Republic of China). Soybean oil was from Jinlongyu Co., Ltd. (Taiyuan, People’s Republic of China). TNF-α and IL-1β enzyme-linked immunosorbent assay kits were supplied by Boster Co., Ltd. (Wuhan, People’s Republic of China). Other reagents were of analytical grade.
Male Sprague Dawley rats with body weights of 150±20 g were from the Laboratory Animal Center, General Hospital of Nanjing Military Region (SYXK (su) 2003-0032). All of the animal procedures were reviewed and approved by the Ethics Committee and the Institutional Animal Care and Use Committee of the Anhui University of Traditional Chinese Medicine (Hefei, People’s Republic of China).
Preparation of CM solution, suspensions, and nanoemulsions
The CM-Ns were prepared using a high-pressure homogenization method as described in a previous report.15 Briefly, 100 mg of CM and 300 mg of Solutol-HS 15 were dissolved in 200 mL of soybean oil. The oil phase was then mixed with 20 mL of water using a high speed electric disperser (FS-2, Huier Co. Ltd., Hangzhou, People’s Republic of China) to form a coarse emulsion. Subsequently, the emulsions were homogenized for 20 cycles at 500 bars using a high-pressure homogenizer (AH-2010, ATS Engineering Inc., Brampton, ON, Canada).
The CM solution was prepared by dissolving 200 mg of CM in 50 mL of phosphate buffered saline (PBS) (pH 7.0) with 1% (w/w) Solutol-HS as the solubilizer.
The CM suspensions were fabricated by dispersing 200 mg of CM in 50 mL of water containing 5% (w/w) hydroxypropyl methyl cellulose E5.
Particle size and morphology determination
The hydrodynamic oil droplet diameter was determined by dynamic light scattering (DLS) using a Mastersizer 3000 (Malvern Instruments Ltd., Shanghai, People’s Republic of China). The determination was carried out at room temperature at an angle of 90°. The samples were diluted 50- to 100-fold before measurement.
The morphology of the oil droplets in CM-Ns was examined using a JEOL 100-X TEM (JEOL, Peabody, MA, USA). After diluting the Ns 100-fold, 10 μL of the CM-Ns was mixed with 20 μL of 2% (w/v) phosphotungstic acid for negative staining. Twenty minutes later, one drop of the mixture was placed onto a copper grid, and the excess sample was removed with a filter paper. The grid surface was finally dried at 25°C for 30 minutes before transmission electron microscope (TEM) examination.
Stability study in simulated GI fluids and drug release
The stability of the CM-Ns and CM suspensions in a simulated gastric fluid (SGF) containing 0.32% (w/v) of pepsin (pH 1.2) and simulated intestinal fluid (SIF) with 1% (w/v) of pancreatin (pH 6.8) were investigated. Briefly, a 5 mL aliquot of the CM formulations was blended with an equal volume of the test fluids, and the combination was incubated for 5 hours at 37°C. At predetermined time intervals, 0.1 mL of the mixture was removed and combined with 0.9 mL of methanol. After centrifugation at 10,000× g for 5 minutes, the drug content in the supernatant was determined using the high performance liquid chromatography (HPLC) method as presented in a previous report.16
The drug release was performed as described later. One milliliter of the CM-Ns or suspension was placed into a dialysis bag (molecular weight cut-off 12,000) that was incubated in 50 mL of water containing 2% (w/w) sodium dodecyl sulfate at 37°C under mild agitation in a water bath. At specific time points, a 1 mL sample was withdrawn from the release medium and replaced with the same volume of fresh medium. The drug content in the release medium was determined using the HPLC method described in a previous report.16
Oral bioavailability study
Fifteen Sprague Dawley rats were randomly divided into A, B and C groups (five rats for each group). Group A was injected iv with the CM solution, and the other two groups were orally administered the CM-Ns or the suspension at a dose of 10 mg/kg CM based on body weight. At predetermined time intervals, 0.5 mL of blood was collected from the orbital plexus into a heparinized micro centrifuge tube. The plasma was obtained by centrifuging the blood at 3,000× g for 10 minutes. The drug levels in the plasma were determined using the HPLC method described in a previous report.17
Two-hundred microliters of plasma was mixed with 50 μL of 5% (w/v) acetic acid and 50 μL of an internal standard (100 ng/mL, 17β-estradiol acetate in methanol). After vortexing for 30 seconds, the mixture was combined with 2 mL of ethyl acetate by vortexing for 5 minutes. After centrifugation at 10,000× g for 10 minutes, the organic layer was collected into 1.5 mL centrifuge tubes and dried under a stream of N2. The dried residual was redispersed with 100 μL of the mobile phase by vortexing for 5 minutes followed by centrifugation at 10,000× g for 10 minutes. Finally, 20 μL of the supernatant was injected into the HPLC system for analysis.
The drug determination was conducted on a Shimadzu HPLC 2010 system (Shimadzu, Kyoto, Japan). The separation was performed on a C18-column (4.6 mm ×250 mm, Diamonsil, Dikma Technologies Inc., Lake Forest, CA, USA) at 30°C. The mobile phase was a mixture of acetonitrile, 20 mM acetate buffer (pH 3) and methanol (60:10:30, v/v) pumped at a rate of 1 mL/min. The fluorescence detector was set at λex =420 nm for CM and λem =280 nm for the internal standard. The recoveries of the low, middle and high quality control levels were greater than 95%, while the intra-day and inter-day precision was less than 10%. The method for the drug detection in plasma was specific, selective, and accurate.
Antiarthritic activity
Adjuvant-induced arthritis animal models and treatment
The adjuvant-induced arthritis (AIA) models were prepared according to a previous report.18 Briefly, the model was induced by a single injection of 0.1 mL of FCA into the plantar skin of the left hind paw.
Six rats without FCA treatment were used as the normal group. The AIA rats were divided into six groups (n=6 per group): CM groups that were iv injected with CM solution or orally administered with CM-Ns or suspensions once a day for 14 days using a CM dose of 50 mg/kg body weight; a positive MTX group that was iv injected with MTX at a dose of 0.5 mg/kg; and a model group and a saline group that were administered with saline via iv injection and oral gavage, respectively.
Evaluation of paw swelling
The volumes of the injected paw of all animals were determined using an YLS-7A plethysmometer (Yi Yan Technology Development Co., Ltd., Jinan, People’s Republic of China) on the 7th and 14th day after FCA injection. The paw swelling rate (PSR) was calculated by the formula: PSR = (final paw volume − initial paw volume)/initial volumes.
Determination of the weight index of thymus and spleen
After the last administration on day 14, the rats were killed by anesthesia with an intraperitoneal injection of ketamine (0.1 mL g/100 g) after collecting the blood. The thymus and spleen were immediately removed from the rat and weighed. The weight indexes of the thymus and spleen were obtained by calculating the ratio (g/kg) between the weight of thymus or spleen and body weight.
Histopathologic examination
On the 14th day of the experiment, the fresh synovial tissues from the rats were obtained, fixed in 10% neutral-buffered formalin, decalcified in 5% formic acid and embedded in paraffin. Sections (5 μm) were stained with hematoxylin and eosin to examine the extent of inflammation. The sections were photographed for the analysis of synovial pathology by a histopathologist.
The histological changes were determined in terms of the infiltration of inflammatory cells (IIC), macrophage proliferation (MP), synovial tissue hyperplasia and fibrous tissue hyperplasia. Based on these indicators, the gradation of arthritis was scored from 0 to 3:0, no positive findings; 1, focal to mild findings; 2, pronounced findings; and 3, severe findings.19
Determination of NF-κB
The detection of the expression of NF-κB in synovial tissue was performed by immunohistochemical analysis as described in previous reports.20,21 Briefly, tissue sections of 5 μm thickness were prepared via a routine procedure. After deparaffinization, antigen retrieval was achieved by heating the samples to 97°C for 20 minutes in sodium citrate buffer (10 mM, pH 6.0). After being washed with PBS three times, the sections were subjected to blocking with PBS containing normal goat serum at room temperature for 10 minutes. After removing the serum, the sections were incubated with the primary antibody, an anti-NF-κB monoclonal antibody (1:100 dilution), for 1 hour at 37°C in PBS. Finally, a MaxVision™ kit was used to develop the antibody signal. Images were obtained using a DMLS2 microscope (Leica Microsystems, Wetzlar, Germany).
Immunoreactivity for NF-κB was assessed via a score scale on the basis of intensity and positivity. The staining intensity was scored as four categories: 0, 1, 2, and 3 represent none, weak, moderate, and intense, respectively. If there was heterogeneity in the staining intensity in a section, the staining intensity was scored based on that which was predominantly observed. The positivity percentage, positively stained cells against all cells, was scored as five categories: 0, less than 5%; 1, 5%–15%; 2, 15%–30%; 3, 30%–50%; and greater than 50%. The immunoreactivity scores were calculated through a combination of the two indicators, intensity, and positivity. It was set as: 0 (0, 0) or (1, 0) or (0, 1); 1 (1, 1); 2 (1, 2) or (2, 1); 3 (1, 3) or (3, 1); 4 (1, 4) or (2, 2); 5 (2, 3) or (3, 2); 6 (2, 4); 7 (3, 3); and 8 (3, 4).
Determination of cytokine levels in synovial fluid and blood serum
Samples of the synovial fluid and blood serum were collected from the animals after the last administration. The levels of the cytokines TNF-α and IL-1β were analyzed using enzyme-linked immunosorbent assay kit (Boster Co., Ltd) according to the manufacturer’s instructions.22
Data analysis and statistics
The pharmacokinetic parameters, including area under the curve (AUC), T1/2, Cmax and Tmax, were calculated based on a statistical moment theory using the DAS 2.0 program (Pharmacology Institute of People’s Republic of China). The data are presented as the means ± standard deviation. The comparisons between two groups were performed using one-way analysis of variance with SPSS 11.0 software (SPSS Inc., Chicago, IL, USA). P<0.05 indicated the difference was significant.
Results
Effect of CM on AIA via iv injection
To obtain a proof-of-concept that CM had a therapeutic effect on RA, we first studied the effect of CM administered by iv injection on AIA rats, using paw swelling, the weight indices of the thymus and spleen, pathological changes, NF-κB expression, and inflammatory cytokines as the endpoints.
The effect of CM on the AIA rats is displayed in Figure 2. Throughout the experiment, MTX, one of most prevalent choices for disease-modifying antirheumatic therapy for RA, was used as the positive control (MTX group), with saline as the negative control (model group). In the model group, the PSR increased remarkably following saline treatment for 14 consecutive days. In contrast, the PSRs for the rats from the MTX and CM groups were significantly less than that of model group, and the PSRs from the MTX and CM groups were similar (Figure 2A).
The thymus and spleen are two important organs for immune responses, and their weight changes are vital indicators reflecting immunological function. The weight indices of the two organs were examined on day 14. In comparison to the normal group, the weight indices for the two organs increased significantly (Figure 2B), thus suggesting that the immune organs played an important role in the induction of AIA. The index for the thymus for both the MTX and CM groups was reduced significantly compared to the model group. However, there was little influence of MTX on the spleen index, whereas CM treatment decreased this index significantly. These results indicated that CM produced immune regulation via inhibition of cellular immunity.
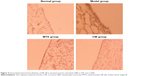
The effects of CM on the histopathological changes are presented in Figures 2C and 3. The rats were killed on day 14 after induction of AIA and subjected to histopathological examination. As shown in Figure 3, intense inflammatory cell infiltration consisting of lymphocyte plasma cells, macrophages and neutrophils; synovial thickening; and significant proliferation of synovial tissue and fibrous tissue were observed in the model group, whereas the normal group displayed no inflammatory cell infiltration. Treatment with CM or MTX resulted in a significant reduction in these symptoms, including inflammatory cell infiltration and MP. To confirm the histopathological examination, the changes in the IIC, MP, synovial tissue hyperplasia and fibrous tissue hyperplasia were scored. As indicated in Figure 2C, the scores for the four indicators in CM and MTX groups were less than those of the model group. In particular, the IIC and MP scores for the CM-treated group were less than those of the MTX group, thus indicating a better therapeutic effect of CM.
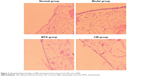  | Figure 3 Histopathological changes in H&E-stained synovial sections in the AIA rats (×200). |
It is well known that NF-κB regulates the expression of pro-inflammatory cytokines and osteoclast differentiation in RA.23 We therefore determined the expression of NF-κB in the synovial tissue using immunohistochemical analysis. As depicted in Figure 4, the positive areas of NF-κB in the model group were significantly higher than those of the normal group. In the animals treated with CM or MTX, the intensity and positivity were significantly reduced or eliminated. The expression level of NF-κB was further analyzed using a score scale according to the intensity and positivity in synovial tissue sections. As displayed in Figure 2D, the scores for NF-κB expression from both the CM and MTX groups were lower than that of the model group.
Cytokines, especially TNF-α and IL-1β, regulate a broad range of inflammatory processes that are implicated in the pathogenesis of RA.7 As shown in Figure 2E and F, the TNF-α and IL-1β levels in both synovial fluid and blood serum from the model group were markedly higher than those of the normal group. Treatment with CM or MTX significantly reduced the TNF-α and IL-1β levels compared with the model group, with approximately fourfold and threefold decreases observed for TNF-α and IL-1β in synovial fluid, respectively, and approximately twofold for both cytokines in blood serum.
Overall, these results indicated that CM possessed a therapeutic effect on RA similar to that of iv-injected MTX, which is one of most common therapies used to treat RA. However, it is well known that oral delivery is by far the preferred and most convenient administration route on the basis of the benefits, including reduced inconvenience, pain, and risks of associated complications. Oral administration also facilitates the development of chronic treatment schedules and reduces the cost of the therapy.24,25 On the other hand, because CM is an insoluble drug, to improve its oral absorption, in the next part, we formulated it into oil–water-based Ns, aiming to achieve a RA treatment with CM via the oral administration route.
Preparation and characterization of CM-Ns
CM had good solubility in the oil phase: approximately 100 mg/mL. The CM-Ns were prepared via a high-pressure homogenizing method with Solutol-HS 15 as an emulsifier, and no drug precipitation was observed during the preparation procedure. The drug could be successfully formulated into Ns with an entrapment efficiency of more than 90%. The particle size of the CM-Ns was approximately 150 nm, with a polydispersity index of 0.21 (less than 0.3) (Figure 5A), thus indicating a homogeneous dispersion.26 To confirm the hydrodynamic diameter of the Ns obtained by DLS and determine the morphology of Ns, TEM analysis was performed. As shown in Figure 5B, uniform and spherical particles with a diameter of approximately 150 nm were observed in the TEM image, which was consistent with the DLS results.
  | Figure 5 Characterization of CM-Ns. |
Stability in simulated GI fluids and drug release
CM is unstable in physiological media, where it undergoes rapid degradation that compromises the therapeutic effect.17 We therefore performed a stability study in SGF and SIF for CM-Ns, using CM suspensions as a control. As shown in Figure 6A, during the incubation of the suspensions with SGF or SIF, the drug was rapidly degraded: approximately 80% of the drug was hydrolyzed within 0.5 hour, and after 3 hours, less than 5% of the drug remained. In contrast, the drug remaining after the CM-Ns incubation was greater than 90%, independent of the incubation time and types of media. This result indicated that by formulating CM into the Ns, the drug stability was significantly improved, preventing the drug from being degraded in the GI fluids.
The release profiles of CM from the CM formulations are presented in Figure 6B. More than 90% of the CM was released from the suspensions within 30 minutes, while a release of less than 25% of CM from the CM-Ns was observed at 2 hours and only approximately 30% of CM was released by 24 hours. The initial burst release during the first 2 hours was attributed to the fact that some drug was present within the emulsifier layers absorbed onto the oil droplets.9 A considerable amount of drug, more than 70% was not released from CM-Ns after 24 hours, indicating that the drug was well encapsulated into the oil core of the Ns.
Oral bioavailability study
The plasma concentration–time curves and pharmacokinetic parameters of CM after oral administration of CM-Ns or suspensions or after an iv injection of CM solution in rats are displayed in Figure 7 and Table 1. Following iv administration, the CM plasma concentration declined rapidly, with a terminal half-life of 0.19 hour (Figure 7A). The plasma concentrations following the oral administration of the CM-Ns or suspensions increased quickly and reached maximum concentrations at 1.5 and 0.75 hours, respectively. The Cmax values for the CM-Ns and suspensions were 146.38±56.88 and 40.20±10.52 μg/mL, respectively. Thus, a greater than threefold increase from CM-Ns was obtained compared with the suspension formulation. The AUC/absolute bioavailability (F %) values were 533.89±250.23 μg·h/mL/86.39% for the CM-Ns and 166.49±38.09 μg·h/mL/26.86% for the suspensions, with the values from the former being more than threefold higher than the latter. These results indicated that the Ns formulation significantly enhanced the absorption of CM.
Therapeutic effect of CM-Ns on AIA via oral administration route
Following the oral administration of the two CM formulations (CM-Ns and suspensions) and the iv injection of the CM solution, the expression of the two most important cytokines, TNF-α and IL-1β, was examined. The therapeutic effect of the CM formulations on AIA is displayed in Figure 8. Compared with the saline group, the CM injection resulted in a significant reduction in the levels of TNF-α and IL-1β in both synovial fluid and blood serum, which the reduction was similar to that MTX group. These results confirmed the better effect of CM on AIA. After the oral administration of the CM-Ns or suspensions, as expected, the levels of the two indicators in the synovial fluid of the CM-Ns-treated group were more than twofold lower than those of the suspension group (Figure 8A). A similar threefold decrease was observed in the blood serum (Figure 8B). These results indicate the better therapeutic effect of CM-Ns than CM suspensions on RA. Importantly, the levels of the two indicators from the animals that were orally administered the CM-Ns and those injected with the CM solution were similar, suggesting the feasibility of oral administration of CM-Ns for RA therapy.
Discussion
We have demonstrated that by decreasing NF-κB expression and downregulating TNF-α and IL-1β, CM administered by iv injection produced a therapeutic effect on RA. Inflammatory cytokines, including IL-6, IL-1, IL-1β, and TNF-α, are involved in the pathogenesis of RA by inducing the infiltration of immune cells, stimulating the release of matrix metalloproteinase and other proteinases to degrade cartilage, and upregulating the expression of pro-inflammatory genes.6,27 Of the cytokines, TNF-α and IL-1β (especially TNF-α) play vital roles in the communication among the inflammatory cells in the rheumatoid joint.27 TNF-α is a key to the cytokine cascade in RA because it can promote the production of many other inflammatory cytokines, including IL-1, IL-6, and granulocyte–monocyte colony-stimulating factor. Moreover, TNF-α is also believed to stimulate fibroblasts to express adhesion molecules, including intracellular adhesion molecule-1.28 In contrast, IL-1β more potently causes the degradation of cartilage and bone than TNF-α, and the whole IL-1 super-family is involved in the development and progression of RA.29 It is well known that NF-κB is one of the most important regulators of pro-inflammatory gene expression on the basis that it can mediate the production of cytokines, including TNF-α, IL-1β, and IL-6.21 As displayed in Figure 2D–F, the expression of NF-κB was significantly inhibited, and the levels of both TNF-α and IL-1β were markedly downregulated by iv administration of CM. Thus, it was concluded that CM exerted its antiarthritic activity by inhibiting the NF-κB signaling pathway and suppressing the pro-inflammatory cytokine release.
The oral bioavailability of CM was significantly enhanced by formulating the drug into the Solutol-HS 15 stabilized Ns, thereby allowing RA therapy with CM to be converted from iv to oral administration. As shown in Table 1, the AUC for the CM-Ns increased by more than threefold compared with the suspension formulation. Following the oral administration of CM-Ns, the Ns may undergo lipid digestion by enzymes, with a portion remaining intact. The lipid digestion would result in drug precipitation and loss of solubilization in the GI tract, thus compromising the absorption. However, the surfactants, lipid digestion products, and endogenous surfactants, including bile salts and phospholipids present in the GI tract, can form mixed micelles that, to some extent, help to maintain the drug in the solubilized form. These processes facilitate the drug absorption by promoting the translocation of the drug across the unstirred water layer and into systemic circulation.30,31 Moreover, the presence of Solutol® HS15, which is used as an emulsifier in the Ns formulation, can enable the drug to transport across the epithelial cell layer in intestine by maintaining the solubilization of poorly soluble drugs and inhibiting P-glycoprotein and CYP450.32,33 A recent earlier report indicated that a nanoparticulate formulation could permeate the enterocytes, thus behaving as a nanotransporter without markedly affecting the physiologic activity of P-glycoprotein, and reach circulation via the intestinal lymphatic system.34 Moreover, lipid nanocarriers have favored access to the lymphatics.35 As indicated in Figure 6, intact Ns are present in the GI tract after administration because a considerable amount of drug was not degraded or released after 6 hours incubation in SGF or SIF. Thus, the intact lipid Ns that were not destroyed by enzymes might also translocate across the enterocytes into the lymphatic system, achieving better drug absorption.
Conclusion
This study indicated that when administered by the iv route, CM had a therapeutic effect on RA through the decreased expression of NF-κB and inhibition of the release of inflammatory mediators, including TNF-α and IL-1β. To deliver CM via the oral route, CM was formulated into oil–water Ns and CM-Ns with a diameter of approximately 150 nm were successfully prepared. Importantly, both the AUC and Cmax of the N formulation were more than threefold greater than those obtained with the suspension, indicating significantly enhanced drug absorption. Moreover, similar therapeutic effects on RA were obtained for the iv injection and oral administration of the N formulation of CM, thereby accomplishing the conversion from iv to oral administration for RA therapy. In conclusion, CM was an effective antiarthritic agent, and the present N formulation appeared to be promising systems that allowed RA therapy with CM to be converted from iv to oral administration.
Disclosure
The authors report no conflicts of interest in this work.
References
Smolen JS, Aletaha D, Koeller M, Weisman MH, Emery P. New therapies for treatment of rheumatoid arthritis. Lancet. 2007;370(9602):1861–1874. | ||
Scott DL, Wolfe F, Huizinga TWJ. Rheumatoid arthritis. Lancet. 2010;376(9746):1094–1108. | ||
Choi HK, Hernán MA, Seeger JD, Robins JM, Wolfe F. Methotrexate and mortality in patients with rheumatoid arthritis: a prospective study. Lancet. 2002;359(9313):1173–1177. | ||
Naksuriya O, Okonogi S, Schiffelers RM, Hennink WE. Curcumin nanoformulations: A review of pharmaceutical properties and preclinical studies and clinical data related to cancer treatment. Biomaterials. 2014;35(10):3365–3383. | ||
Yallapu MM, Jaggi M, Chauhan SC. Curcumin nanoformulations: a future nanomedicine for cancer. Drug Discov Today. 2012;17(1–2):71–80. | ||
McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365(23):2205–2219. | ||
McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol. 2007;7(6):429–442. | ||
Sharma RA, Gescher AJ, Steward WP. Curcumin: the story so far. Eur J Cancer. 2005;41(13):1955–1968. | ||
He W, Tan Y, Tian Z, Chen L, Hu F, Wu W. Food protein-stabilized nanoemulsions as potential delivery systems for poorly water-soluble drugs: preparation, in vitro characterization, and pharmacokinetics in rats. Int J Nanomedicine. 2011;6:521–533. | ||
Mei L, Zhang Z, Zhao L, et al. Pharmaceutical nanotechnology for oral delivery of anticancer drugs. Adv Drug Deliv Rev. 2013;65(6):880–890. | ||
He W, Lu Y, Qi J, Chen L, Hu F, Wu W. Nanoemulsion-templated shell-crosslinked nanocapsules as drug delivery systems. Int J Pharm. 2013;445(1–2):69–78. | ||
Chen Y, Gao Y, Chen H, et al. Engineering inorganic nanoemulsions/nanoliposomes by fluoride-silica chemistry for efficient delivery/co-delivery of hydrophobic agents. Adv Funct Mater. 2012;22(8):1586–1597. | ||
Sun DQ, Wei XB, Xue X, et al. Enhanced oral absorption and therapeutic effect of acetylpuerarin based on D-alpha-tocopheryl polyethylene glycol 1000 succinate nanoemulsions. Int J Nanomedicine. 2014;9:3413–3423. | ||
Kotta S, Khan AW, Ansari SH, Sharma RK, Ali J. Anti HIV nanoemulsion formulation: optimization and in vitro–in vivo evaluation. Int J Pharm. 2014;462(1–2):129–134. | ||
He W, Lv Y, Zhao Y, et al. Core–shell structured gel-nanocarriers for sustained drug release and enhanced antitumor effect. Int J Pharm. 2015;484(1–2):163–171. | ||
Shaikh J, Ankola DD, Beniwal V, Singh D, Kumar MNVR. Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer. Eur J Pharm Sci. 2009;37(3–4):223–230. | ||
Mohanty C, Sahoo SK. The in vitro stability and in vivo pharmacokinetics of curcumin prepared as an aqueous nanoparticulate formulation. Biomaterials. 2010;31(25):6597–6611. | ||
Li M, He J, Jiang LL, et al. The anti-arthritic effects of Aconitum vilmorinianum, a folk herbal medicine in Southwestern China. J Ethnopharmacol. 2013;147(1):122–127. | ||
Wang Y, Wang S, Li Y, et al. Therapeutic effect of Cryptotanshinone on collagen-induced arthritis in rats via inhibiting nuclear factor kappa B signaling pathway. Transl Res. 2014;165(6):704–716. | ||
Yang M, Xiao C, Wu Q, et al. Anti-inflammatory effect of Sanshuibaihu decoction may be associated with nuclear factor-κB and p38 MAPKα in collagen-induced arthritis in rat. J Ethnopharmacol. 2010;127(2):264–273. | ||
Zhang Y, Meng Z, Zhang M, et al. Immunohistochemical evaluation of midkine and nuclear factor-kappa B as diagnostic biomarkers for papillary thyroid cancer and synchronous metastasis. Life Sci. 2014;118(1):39–45. | ||
Ahmad SF, Attia SM, Zoheir KMA, Ashour AE, Bakheet SA. Attenuation of the progression of adjuvant-induced arthritis by 3-aminobenzamide treatment. Int Immunopharmacol. 2014;19(1):52–59. | ||
Naidu VGM, Dinesh Babu KR, Thwin MM, Satish RL, Kumar PV, Gopalakrishnakone P. RANKL targeted peptides inhibit osteoclastogenesis and attenuate adjuvant induced arthritis by inhibiting NF-κB activation and down regulating inflammatory cytokines. Chem Biol Interact. 2013;203(2):467–479. | ||
Patel K, Patil A, Mehta M, Gota V, Vavia P. Oral delivery of paclitaxel nanocrystal (PNC) with a dual Pgp-CYP3A4 inhibitor: preparation, characterization and antitumor activity. Int J Pharm. 2014;472(1–2):214–223. | ||
Andreani T, Souza ALRd, Kiill CP, et al. Preparation and characterization of PEG-coated silica nanoparticles for oral insulin delivery. Int J Pharm. 2014;473(1–2):627–635. | ||
He W, Jin Z, Lv Y, et al. Shell-crosslinked hybrid nanoparticles for direct cytosolic delivery for tumor therapy. Int J Pharm. 2015;478:762–772. | ||
Lin B, Zhao Y, Han P, et al. Anti-arthritic activity of Xanthium strumarium L. extract on complete Freund’s adjuvant induced arthritis in rats. J Ethnopharmacol. 2014;155(1):248–255. | ||
Zhang X, Sun J, Xin W, et al. Anti-inflammation effect of methyl salicylate 2-O-β-D-lactoside on adjuvant induced-arthritis rats and lipopolysaccharide (LPS)-treated murine macrophages RAW264.7 cells. Int Immunopharmacol. 2015;25(1):88–95. | ||
Zuo J, Xia Y, Li X, Chen J-W. Therapeutic effects of dichloromethane fraction of Securidaca inappendiculata on adjuvant-induced arthritis in rat. J Ethnopharmacol. 2014;153(2):352–358. | ||
Simovic S, Heard P, Hui H, et al. Dry hybrid lipid-silica microcapsules engineered from submicron lipid droplets and nanoparticles as a novel delivery system for poorly soluble drugs. Mol Pharm. 2009;6(3):861–872. | ||
Porter CJ, Trevaskis NL, Charman WN. Lipids and lipid-based formulations: optimizing the oral delivery of lipophilic drugs. Nat Rev Drug Discov. 2007;6(3):231–248. | ||
Illum L, Jordan F, Lewis AL. CriticalSorb™: a novel efficient nasal delivery system for human growth hormone based on Solutol HS15. J Control Release. 2012;162(1):194–200. | ||
Buggins TR, Dickinson PA, Taylor G. The effects of pharmaceutical excipients on drug disposition. Adv Drug Deliv Rev. 2007;59(15):1482–1503. | ||
Attili-Qadri S, Karra N, Nemirovski A, et al. Oral delivery system prolongs blood circulation of docetaxel nanocapsules via lymphatic absorption. Proc Natl Acad Sci U S A. 2013;110(43):17498–17503. | ||
Ryan GM, Kaminskas LM, Porter CJH. Nano-chemotherapeutics: maximising lymphatic drug exposure to improve the treatment of lymph-metastatic cancers. J Control Release. 2014;193:241–256. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.