Back to Journals » Drug Design, Development and Therapy » Volume 9
Soy polysaccharide as a novel superdisintegrant in sildenafil citrate sublingual tablets: preparation, characterization, and in vivo evaluation
Authors Ibrahim KMH , Mosli H, Hassan A
Received 23 October 2014
Accepted for publication 24 November 2014
Published 12 January 2015 Volume 2015:9 Pages 465—472
DOI https://doi.org/10.2147/DDDT.S76314
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 6
Editor who approved publication: Professor Shu-Feng Zhou
Khaled Mohamed Hosny,1,2 Hisham Ahmed Mosli,3 Ali Habiballah Hassan4
1Department of Pharmaceutics and Industrial Pharmacy, Faculty of Pharmacy, King Abdulaziz University, Jeddah, Saudi Arabia; 2Department of Pharmaceutics and Industrial Pharmacy, Faculty of Pharmacy, Beni Suef University, Beni Suef, Egypt; 3Department of Urology, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia; 4Department of Orthodontics, Faculty of Dentistry, King Abdulaziz University, Jeddah, Saudi Arabia
Abstract: Sildenafil citrate (SC), a drug used to treat erectile dysfunction, is available in tablet form but has three major problems. First, the drug displays inadequate aqueous solubility, which delays the onset of its action. Second, the drug undergoes extensive first-pass metabolism, resulting in a low (40%) bioavailability. Third, the gastrointestinal effects of SC include dyspepsia and a burning sensation. The aim of this research was to prepare SC as a sublingual tablet utilizing soy polysaccharide as novel superdisintegrant to mitigate the abovementioned problems. The solubility of SC in various hydrophilic carrier solutions was estimated in order to prepare the drug as a coprecipitate. Sublingual tablets were prepared and evaluated for hardness, friability, drug content, wetting time, water absorption ratio, in vitro dispersion time, dissolution rate, and stability study. The pharmacokinetic study of the tablets was carried out on healthy volunteers. The results indicated that the co-precipitation of SC with polyvinylpyrollidone K30 enhanced the solubility of SC by more than eight folds. The tablet contained 8% soy polysaccharide as a superdisintegrant and provided a wetting time of 25 seconds, and in vitro dispersion times of 55 seconds. The drug release was found to be 95.6%. The prepared SC sublingual tablet also exhibited a rapid onset of action, and its bioavailability was enhanced 1.68-fold compared with that of the marketed tablets. It can be concluded that SC sublingual tablet is a promising formulation that results in higher solubility, faster dispersion and onset of action, higher release rate, and higher systemic bioavailability.
Keywords: bioavailability, erectile dysfunction, onset of action, polyvinylpyrrolidone, Emcosoy
Introduction
Sildenafil citrate (SC) is a selective phosphodiesterase-5 inhibitor indicated in the treatment of erectile dysfunction.1 Its solubility is 3.5 mg/mL in 0.1N HCl.2 SC is absorbed following oral administration, with bioavailability of approximately 38%–42%. Peak plasma concentrations are attained within 45–90 minutes (median 60 minutes). The drug has a duration of action lasting up to 4 hours, with less response than that seen at 2 hours.3 SC is cleared predominantly by the hepatic microsomal isoenzymes.4 The absorption of SC is hindered by fatty meals,5 with a mean delay in Tmax of 60 minutes and a mean reduction in Cmax of 29%. The main adverse effects of SC are the gastrointestinal effects resulting in dyspepsia, or burning sensation from reflux due to relaxation of the lower esophageal sphincter.6
A vast variety of pharmaceutical research is directed at developing new dosage forms for oral absorption. Most of these efforts have focused on either formulating novel drug delivery systems or increasing patient compliance. Oral cavity is highly acceptable by patients, and the mucosa is relatively permeable with rich blood supply. The virtual lack of Langerhans cells makes oral mucosa tolerant to potential allergens.7
During the last decade, sublingual tablet technologies have drawn a great deal of attention. Recently, the European Pharmacopeia adopted the term sublingual tablet for a tablet that generally disperses or disintegrates in less than three minutes in the mouth.8
Coprecipitation (C-ppt) is a technique that depends on melting or dissolution of a drug in a soluble carrier that leads to increase the dissolution.9,10 Several hydrophilic carriers can be utilized in the preparation of coprecipitates such as polyvinylpyrolidone (PVP), polyethylene glycol (PEG), cyclodextrin (CD), mannitol, and urea.
The highly fibrous soy polysaccharide material generally comprises high molecular weight carbohydrate polymers primarily contained in soybeans, such as arabinose, galactose, mannose, and xylose. It is normally employed as a collective term to describe the fibrous carbohydrate material obtained from the cell wall structural components of soybeans that include soy flakes, flour, or meal.11
The objective of this work was to utilize the sublingual tablet technology using Emcosoy (soy polysaccharide) as novel superdisintegrant in order to provide a solution to the major problems associated with the marketed SC oral tablets.
Materials and methods
Materials
SC was obtained from EIPICO pharmaceutical company (Cairo, Egypt) as a gift, whereas β-cyclodextrins (β-CD) was kindly supplied by Nihon Shukohin Kako Co., Ltd. (Tokyo, Japan). Emcosoy was a gift from RS PHARMA GmbH & Co. KG (Rosenberg, Germany). Explotab and PVP K30 were purchased from Fluka Chemical Company (Buchs, Switzerland). Mannitol, citric acid, and xylitol were purchased from (Merck, Darmstadt, Germany). Other chemicals and reagents were purchased from Sigma-Aldrich (St Louis, MO, USA).
Methodology
Preparation of the SC coprecipitates
The C-ppts were prepared using coevaporation technique,12 in which SC and hydrophilic carriers (PVP K30, PEG 6000, and β-CD) were mixed in 1:1, 1:2, and 1:3 drug:polymer weight ratios, and methanol was added under constant stirring until dissolution. The solvent was then evaporated, and the resultant C-ppt was triturated and passed through an 80 mesh screen sieve before being stored in a dry place until use.
Solubility analysis of the prepared coprecipitates
Excess C-ppt was dispersed in 5 mL of distilled water to obtain a supersaturated solution. These dispersions were shaken for 24 hours at 25°C until equilibrated. Two milliliters of the supersaturated solution was filtered through a 0.22 μm nylon filter, and 1 mL of the filtrate was diluted with 0.1N HCl. The absorbance was measured at 291 nm. Solubility studies were also performed with pure SC.
Differential scanning calorimetry (DSC) studies
DSC thermograms for samples of plain SC, PVP K30, their physical mixture, and coprecipitate were recorded and analyzed.
Fourier Transform Infrared Spectroscopy (FTIR) studies
Samples of plain SC, PVP K30, their physical mixture, and coprecipitate were mixed with about 400 mg of dry potassium bromide powder compressed into a transparent disc under a pressure of 10.000–15.000 psi. The IR spectra were recorded and analyzed.
Drug content
Ten milligrams of the prepared C-ppt was dissolved in 0.1N HCl and filtered (0.22 mm membrane filter). The drug content was determined spectrophotometrically at 291 nm.
Preparation of SC sublingual tablet
SC sublingual tablets were prepared by direct compression using Emcosoy or Explotab as superdisintegrants, citric acid or xylitol as saliva stimulants, avicel as diluents, sodium saccharine as sweetener, and talc and magnesium stearate as lubricants. According to the formula composition listed in Table 1, 12 sublingual formulations of SC were prepared by direct compression using a tableting machine (Erweka Tablet Press-Type EK0).
  | Table 1 Formulae composition of sildenafil citrate (SC) sublingual tablets |
Evaluation of the prepared SC sublingual tablets
Physicochemical characterization
Weight variation, thickness, hardness, drug content, and friability of the prepared tablet formulation were characterized according to Khaled et al13 and the average value for each test was calculated.
In vitro dispersion time
Ten tablets were separately placed in 10 mL of phosphate buffer (pH =6.8) at 37°C±0.5°C and the time required for complete dispersion was determined.
Wetting time and water absorption ratio
According to Cheng et al14 the time (in seconds) for tablet to be completely wet was recorded as the wetting time through observation with the naked eye. The water absorption ratio (R) through the tablet was also determined.
In vitro drug release studies
The test was done by using dissolution testing apparatus II (the paddle method), and the cumulative percentage of drug release was calculated using the equation obtained from a standard calibration curve. The mean of six determinations was considered. For comparison, T90% were calculated and compared.
Stability studies
An accelerated stability test was conducted on the best tablet formulation (F2) at 40°C and 60%±5% RH (relative humidity) for 3 months and tablet properties including hardness, disintegration time, friability, drug content, wetting time, in vitro dispersion time, and dissolution were determined at the end of each month.
In vivo studies on human volunteers
To compare the pharmacokinetics of SC in the sublingual tablet (treatment A) with the conventional Viagra® tablet (Pfizer, Egypt; treatment B), a single dose of SC (50 mg) was given to the volunteers using a double blind, randomized, cross-over design. Fasting subjects were administered a single 50 mg oral dose of SC sublingual tablet or the reference product during each period of the study.
Subject population
Twelve healthy Egyptian male volunteers aged between 30 years and 40 years (median weight =74±6 kg and median height =180±8 cm) were chosen. The health status of the volunteers was confirmed using a complete medical history and laboratory analyses performed at the beginning of the study. No medication was allowed during the study. The blood pressure of the volunteers was measured; no significant difference in blood pressure was observed before and after the treatment.
Sample collection
The study was conducted according to the agreement protocol of the Declaration of Helsinki and the requirements of the ethical committee of the Faculty of Medicine at Beni Suef University in Egypt. The drug was administered during each period of the study with a one-week washout period. Blood samples (5 mL) were collected at the following time intervals: 0.25 hours, 0.5 hours, 0.75 hours, 1 hour, 2 hours, 4 hours, 6 hours, 8 hours, 10 hours, 12 hours, 18 hours, and 24 hours after ingesting the GC. The blood samples were centrifuged, and the plasma was collected and stored at −20°C until assayed.
Chromatographic conditions
The pharmacokinetic parameters of SC were determined using a method based on that developed by Sheu et al15 involving the extraction of SC with dichloromethane. The DCM was then allowed to evaporate under nitrogen, and the residue was dissolved in 0.2 mL of the mobile phase (acetonitrile and potassium dihydrogen phosphate [30 mM] 56:44, v/v). The column was an RPC18.
Pharmacokinetic analysis
The pharmacokinetic parameters were estimated using WinNonlin® [version 1.5, Scientific consulting, Inc., Cary, NC, USA]. Cmax (ng/mL) and Tmax (hour) were the observed maximum drug concentration and the time needed to reach the maximum concentration, respectively. Area under the curve (AUC)[0–24] (ng h/mL) was calculated using the relative bioavailability (AUC test/AUC standard) ×100.
Statistical analysis
The statistical analyses were performed using a t-test for paired data with SPSS® 7.5 for Windows. The level of statistical significance was P≤0.05.
Results
Results of solubility studies
Table 2 showed that the drug solubility was increased in all prepared C-ppt mixtures compared to pure sildenafil powder, and the solubility was increased by increasing the polymer ratio in C-ppt.
  | Table 2 Characterization of the prepared solid dispersions |
Based on these results, SC-PVP K30 coprecipitate prepared in 1:2 drug:polymer ratio by the coevaporation method was selected and subjected to further evaluation using DSC and FTIR studies, and used for preparation of the sublingual tablet.
DSC studies
Figure 1 shows the DSC thermograms of SC, PVP K30, their physical mixture, and the prepared coprecipitate. SC shows a sharp exothermic melting peak at 195°C. This peak was retained in the thermogram of the physical mixture with no appearance of new peaks that excludes any incompatibility. In C-ppt thermogram, the peak completely disappeared.
  | Figure 1 DSC thermograms of (A) SC, (B) PVP K30, (C) physical mixture, and (D) coprecipitate. |
Infrared spectroscopy (FTIR) studies
Figure 2 shows the FTIR spectrum of SC, PVP K30, and their prepared coprecipitate. The main characteristic peaks at 3,613 cm−1 (free OH stretching vibrations); 3,293 cm−1 (NH stretching), 3,028 cm−1, 3,000 cm−1, and 2,270 cm−1 (CH stretching vibration); and 1,700 cm−1 (C=O stretching vibration), and 1,358 cm−1 and 1,174 cm−1 (SO2 stretching vibration), and 1,240 cm−1 (CN stretching vibration). They were all retained in the case of C-ppt with PVP K30.
  | Figure 2 FTIR spectra of (A) SC, (B) PVP K30, and (C) coprecipitate. |
Physicochemical characterization of the prepared sublingual tablets
All the prepared tablets were characterized by a uniform thickness, diameter, and weight, thus indicating efficient mixing. The formulated tablets exhibited low weight variation, drug content was between 98.24% and 102.18%, and the thickness of tablets varied from 2.25 mm to 2.32 mm. All formulations showed good mechanical resistance and breaking strength according to friability test results; the hardness values were in the range of 5.78–6.81 kg/cm2 that lies within the acceptable pharmacopeia limits as per USP (United State Pharmacopoeia) 27. Results are summarized in Table 3, and in vitro release is illustrated in Figure 3.
  | Table 3 Characterization of hydrophilic properties of SC sublingual tablets |
Stability of the prepared sublingual tablets
Physical properties of the formula F2 after keeping it in accelerated stability conditions are shown in Table 4. The results revealed that formulation F2 could be considered stable even after 3 months of keeping it in accelerated conditions.
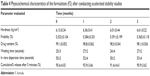  | Table 4 Physicochemical characteristics of the formulation (F2) after conducting accelerated stability studies |
In vivo absorption studies
The mean plasma concentration–time curves of the prepared SC sublingual tablet and the reference tablet are illustrated in Figure 4. The pharmacokinetic parameters were evaluated using WinNonLin® Professional software. The total drug amount absorbed over a period of 24 hours is shown in Table 5; this measurement was determined using the differences in Tmax between the two treatments. The AUCs were statistically significant (P<0.05).
Moreover, the relative bioavailability of the prepared sublingual tablet was 168% compared with Viagra® tablets as the reference standard.
Discussion
SC C-ppt with PVP K30 showed better solubility results than PEG 6000 and β-CD. This could be because of the fact that PVP K30 is a polymer with a large number of monomers, each of which possesses a –C=O group, which shows the inhibitory effect on the crystallization of drugs, resulting in a higher dissolution rate of drug.15
In the DSC thermograms of C-ppt, the characteristic peak of SC completely disappeared, indicating the formation of solid dispersion and conversion of drug from the crystalline to amorphous state.13 The main characteristic FTIR spectrum of SC were retained in physical mixtures and C-ppt, clearly indicating that no chemical interaction exists between the pure drug and the polymer in C-ppt.
The disintegration time of sublingual tablets is generally less than 1 minute and the actual disintegration time that a patient can experience ranges from 30 seconds to 60 seconds.16,17
SC sublingual formulae containing the superdisintegrant component (Emcosoy or Explotab) were acceptable and their behavior in contact with water was excellent as indicated by the short wetting time that ranged between 25 seconds and 40 seconds, high water absorption ratio that ranged between 41 and 60, and short in vitro dispersion time that ranged between 55 seconds and 74 seconds. While the formulae prepared without superdisintegrant (F11, F12) showed long wetting and dispersion time, formulae F2 and F4 that contained superdisintegrant in a large amount (20 mg) were excellent in their wetting and dispersion behavior when compared with same formulae prepared with a small amount of (10 mg) superdisintegrant. This indicated that the presence of superdisintegrant in the sublingual tablet formula with optimum concentration is an essential factor for achieving an acceptable level of dispersion of the tablets.
The dissolution profile of SC sublingual tablets prepared with different formulae indicated a higher, faster, and maximum drug release in the case of formulae F2 and F1 followed by F6 and F5; more than 90% of the drug was released in 5 minutes in the case of F2. Although F4 showed an acceptable wetting and dispersion behavior, T90% for this formula was prolonged (14.7 minutes). These results indicate that the presence of citric acid is essential for the rapid release of SC from the tablet; this could be because SC is highly soluble in acidic media compared to neutral and alkaline media. The presence of citric acid in the tablet created an acidic environment around SC molecules and improved the solubility and, in turn, the release of the drug from the tablet.
The results also revealed that formulation F2 can be considered stable as after 3 months of being kept in accelerated conditions, no significant changes were observed in all tested parameters.
The pharmacokinetic study revealed that the administration of SC as a sublingual tablet (Formula F2) can modify its pharmacokinetic profile, increasing its bioavailability by more than 1.68-fold relative to that of the marketed oral tablet. Compared with the marketed tablet, the Tmax for the sublingual tablet was significantly lower (P<0.05) because SC is a lipophilic drug with poor aqueous solubility. The preparation of this drug as a C-ppt enhanced its solubility and tissue permeability, thereby shortening the time of onset of its action. The onset of action for the reference tablet begins after 1 hour because the plasma level reaches 215 ng/mL at this point; this concentration was attained after just 15 minutes using the sublingual tablet, as shown in Figure 4.
These results demonstrate that the sublingual tablets containing SC as C-ppt with PVP K30 were easily absorbed, indicating that the sublingual tablet circumvented the delayed onset observed with the commercial tablets. Additionally, when a sublingual tablet is administered orally, it is instantly wet by saliva, resulting in its rapid dissolution to release the drug.18,19 In addition, the bioavailability of SC exhibited an increase of more than 1.68-fold; this increase could be attributed to the decreased amount of hepatic first-pass metabolism associated with orally administered tablets that improved the in-vivo bioavailability.
Conclusion
Depending on the results of the study it can be concluded that PVP K30 is an efficient polymer to prepare SC as a coprecipitate. The drug-to-polymer ratio is critical and affects the drug solubility. The selection of suitable superdisintegrants for the preparation of the sublingual tablet is highly effective, and Emcosoy (soy polysaccharide) significantly enhanced all behaviors of the tablet when in contact with water. In addition, the presence of citric acid in the tablet is regarded as a key factor that affects the release rate of the drug from the tablet. The bioavailability is enhanced by more than 1.68-fold relative to that of the marketed tablet, and the onset of action is shortened to 15 minutes. The use of this sublingual formula may lead to the elimination of the major drawbacks associated with the conventional tablet and allow patients to tolerate the drug without fear of gastrointestinal side effects.
Disclosure
The authors report no conflicts of interest in this work.
References
Goldenburg MM. Safety and efficacy of sildenafil citrate in the treatment of male erectile dysfunction. Clin Ther. 1998;20(6):1033–1048. | ||
USP. Approved Drug Products and Legal Requirements. Vol. vol. III. 21st ed. USA: MicroMedex Inc; 2001. [4:490]. | ||
Reynolds JEF, editor. Martin Dale, The Extra Pharmacopoeia. 32nd ed. London: The Royal Pharmaceutical Society; 1999:1629. | ||
Hyland R, Roe G, Jones C, Smith A. Identification of the cytochrome P450 enzymes involved in the N-demethylation of sildenafil. Br J Clin Pharmacol. 2001;51(3):239–248. | ||
Jetter A, Kinzig M, Walchner M, et al. Effects of grapefruit juice on the pharmacokinetics of sildenafil. Clin Pharmacol Ther. 2002;71(1): | ||
Boolell M, Gepi S, Gingell C, Allen J. Sildenafil, a novel effective oral therapy for male erectile dysfunction. Br J Urol. 1996;78(2):257. | ||
Priyanka N, Kusum S, Iti C, et al. Orally disintegrating tablets: formulation, preparation techniques and evaluation. J Appl Pharm Sci. 2011;1(04): | ||
Fu Y, Yang S, Jeong SH, Kimura S, Park K. Orally fast disintegrating tablets: developments, technologies, taste-masking and clinical studies. Crit Rev Ther Drug Carrier Syst. 2004;21(6):433–475. | ||
Ansari T, Sunderland B. Solid dispersions of dihydroartemisinin in polyvinylpyrrollidone. Arch Pharm Res. 2008;31(3):390–398. | ||
Rashmi D, Ashish P, Sanjay N. Formulation and evaluation of mouth dissolving tablets containing amlodipine besylate solid dispersion. Int J Chem Tech Res. 2010;2(1):706–715. | ||
Raun P, Yang B, Guang MF, Dan Z. Improving the solubility of amelopsin by solid dispersions and inclusion complexes. J Pharm Biomed Anal. 2005;38:457–464. | ||
Yan B, Gui Y, Hong S, et al. Effect of substituted group of β-cyclodextrin derivatives on the dispersing of carbon nanotubes. J Dispers Sci Technol. 2010;31(3):353–358. | ||
Khaled MH, Ahmed K, Seham SA. Preparation and evaluation of orodispersible tablets containing hydroxylbutyl-β-cyclodextrin-simvastatin solid dispersion. Trop J Pharm Res. 2013;12(4):469–476. | ||
Cheng C, Demirci A, Catchmark M. Pullulan: biosynthesis, production, and applications. Appl Microbiol Biotechnol. 2011;92(1):29–44. | ||
Sheu T, Wu B, Yeh C, Hsia A. Development of a liquid chromatographic method for bioanalytical applications with sildenafil. J Chromatogr B. 2003;791:255–262. | ||
Paramita D, Sabyasachi M. Orodispersible tablets: a new trend in drug delivery. J Nat Sci Biol Med. 2010;1(1):2–5. | ||
Radke S, Jadhav K, Chajeed R. Formulation and evaluation of orodispersible tablets of baclofen. Int J Chem Tech Research. 2009;1: | ||
Alka T, Kiran S, Nitesh SC, Ashu M. Formulation and evaluation of fast dissolving oral film of dicyclomine as potential route of buccaldelivery. Int J Drug Dev Res. 2012;4:408–417. | ||
Watanabe T, Hasegawa S, Wakiyama N, et al. Comparison between polyvinylpyrollidone and silica nanoparticles as carriers for indomethacin in a solid state dispersion. Int J Pharm. 2003;250:283–286. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



